Abstract
Background:
Comorbidity is often associated with negative outcome of prognosis. The aim of this study was to investigate the prevalence and impact of comorbidities in a retrospective nationwide population-based study of patients with oropharyngeal and hypopharyngeal cancers in Taiwan.
Materials and Methods:
A total of 4733 oropharyngeal and hypopharyngeal cancer patients diagnosed in the period 2007–2010 were identified through the Taiwan Cancer Registry (TCR) database. By linking this data set to the National Health Insurance Research Database (NHIRD) and the Death Registry (DR), information on comorbidity and cause of death was obtained and adapted to the Charlson’s comorbidity index (CCI). The influence of comorbidity on overall survival and cancer-related death was evaluated by Kaplan–Meier estimates and Cox regressions.
Results:
There were 23.5% of patients with comorbidities during the year before cancer diagnosis. Patients with comorbidities are associated with lower survival rates for stage 3 and for oropharynx. Although there was a tendency that patients with comorbidities received less curative treatment within the same cancer stage, the statistical significance was not reached. The data suggest having a co-morbidity condition had a significant effect on all-cause death (age and gender adjusted hazard ratios = 0.68-0.40) but no significance in cancer-related deaths.
Conclusion:
The cancer management was not significantly different between patients’ comorbidity conditions. However, the larger differences between all-cause and cancer-specific survival rates in certain treatment groups emphasize the importance of managing other medical conditions in cancer patients.
Introduction
Smoking, alcohol drinking and areca/betel quid chewing are associated with occurrence of oral and oropharyngeal cancers. 1 These factors also contribute to several systemic diseases, and, therefore, coexistence of oral cancer with other medical conditions is often encountered in clinical practice. Comorbidity is often associated with negative outcome of prognosis. 2 Patients with comorbidity may tend to have less aggressive non-curative treatments 3 or more treatment complications. 4 The effects of these diseases on selection of cancer treatments and effectiveness of treatments become crucial issues in patients’ prognoses. 3
Our previous investigation in patients with oral cavity 5 cancers confirmed that the survival rates of oral cancer patients in early stages are lower in patients with comorbidity conditions. This may be due to less aggressive cancer treatment offered to this group of patients. Comorbidities may affect the decision on cancer treatment modality, and thereby outcomes. 5 Reports from large-scale databases primarily focused on head and neck cancers, 6,7 including oral cavity cancer, pharynx/larynx and thyroid cancer. The corresponding risk factors and treatment modalities are often different for cancers in these different subsites of head and neck. Therefore, it is desirable for these anatomical subsites to be investigated separately in terms of association between mortality rates and comorbidity. The study objectives were to investigate the management of oropharyngeal and hypopharyngeal cancer patients with different comorbidity conditions and further to compare their overall survival and cancer-specific survival rates.
Materials and methods
Data source
The research databases included Taiwan Cancer Registry (TCR), National Health Insurance Research Database (NHIRD) and Death Registry (DR) from the Health and Welfare Data Science Center (HWDC), Department of Statistics, Minister of Health and Welfare in Taiwan. Detailed background of these databases was described by Yang and Warnakulasuriya. 5 The TCR, accounting for the registration of 90% of major cancers, includes the American Joint Committee on Cancer (AJCC) staging, date of initial diagnosis, primary cancer site, histology, clinical Tumour, Node, Metastasis staging (TNM), pathological TNM, metastasis, surgical procedure, chemotherapy and radiotherapy. 8 The NHIRD contains inpatient and outpatient physician claims and prescription drug claims as well as demographic data on all beneficiaries. The coverage rates of NHI program reached 99.6% in 2010 of Taiwan’s total population. The DR database was used for identifying death dates and cause of death for cancer patients.
Study cohort
We identified oropharyngeal and hypopharyngeal cancer patients from 2007 to 2010 in TCR database by using the International Classification of Diseases for Oncology (ICD-O): C09–C10 and C12–C14.The cancer stage and treatment modality were obtained from the TCR. Initially, 4929 oropharyngeal and hypopharyngeal cancer patients newly diagnosed during 2007–2010 with stages 1–4 were identified from TCR. We excluded 23 patients aged less than 18 years old and 196 patients who did not receive any surgery, radiotherapy or chemotherapy treatments. The reason for excluding untreated patients in our study is because that the National Health Insurance Program in Taiwan is practically available to all patients, and any decision of no curative treatments might be made by patients themselves and their families, and not by medical teams. In addition, the palliative management is not considered as a curative treatment modality in this study. These 196 patients accounted for 2.9%, 3.4%, 3.1% and 4.3% of the stages 1–4 at presentation, and with 3.3%, 5.0% and 7.7% in none, mild to moderate and severe comorbidity conditions, respectively. There were, finally, 4733 patients included in the analysis. Death dates and causes of death for cancer patients were determined by DR. This study was approved by the Internal Review Board of Kaohsiung Medical University Hospital (KMUHIRB-EXEMPT(I)-20160031).
The data management and statistical analyses were performed in a designated workplace of HWDC. For the confidentiality of cases during databases processing, personal identifications were first encrypted, and only authorized researchers are permitted to perform data linkage and statistical analyses of these identification encrypted databases. To ensure the de-identification process, researchers can only bring out the final statistical analysis results (tables and graphs), and none of these statistics were computed from cell sizes of 1 or 2.
Analysis of variables
The primary cancer sites were determined by the first reported site; categories included oropharynx (C09–C10), hypopharynx (C12–C13) and pharynx and ill-defined sites in lip, oral cavity and pharynx (C14). Treatment modality was identified by reported information of primary cancer treatments in TCR, which has dates of cancer diagnosis, starting date of chemotherapy, starting date of radiotherapy and surgical procedure of primary site. We identified nine treatment modalities as (1) radiotherapy only, (2) chemotherapy only, (3) radiotherapy and chemotherapy, (4) concomitant chemoradiotherapy, (5) surgery only, (6) surgery with radiotherapy, (7) surgery with chemotherapy, (8) surgery with radiotherapy and chemotherapy and (9) surgery with concomitant chemoradiotherapy. In the analyses of stage/comorbidity subgroups, due to small sample sizes, the first three treatment groups were combined together as radiotherapy and/or chemotherapy; similarly, the sixth, seventh and eighth groups were combined as surgery with radiotherapy and/or chemotherapy. The comorbidity groups were then dichotomized into with or without comorbidities.
Diseases specified in the Charlson’s comorbidity index (CCI) 9 were identified from the diagnosis code of inpatients and outpatients records using the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) 10,11 from NHIRD during the year before cancer diagnosis. To prevent any diagnosis codes given for screening purposes and to assure that a patient had been diagnosed and treated for a disease, we defined that a treated disease should have three or more outpatient records or one or more hospital admissions of a given condition. The CCI was further categorized into three groups (0 = no comorbidity; 1 = mild to moderate comorbidity; 2 = severe comorbidity) in statistical analyses. Age at cancer diagnosis was used for defining age groups.
Statistical analysis
Continuous and categorical variables were presented as mean (±standard deviation, SD) and frequency distribution tables. The Kaplan–Meier estimates of survival rates and Cox regressions were used for comparing outcome between comorbidity groups. The outcomes included events of all-cause deaths and cancer deaths. Dates of deaths were obtained from DR database. For patients who were censored from events, the duration from cancer diagnosis to 31 December 2012 was considered as the follow-up time. We also computed the difference of 2-year survival rates between all-cause and cancer-related rates. A p value of less than 0.05 was considered as statistical significance. The statistical software of SAS V9.4 (SAS Institutes, Inc., Cary, North Carolina, USA) was used to conduct data management and statistical analyses.
Results
The study cohort comprised 4512 males (95.3%) and 221 females (4.7%). Mean (±SD) age of diagnosis was 54.7 (±11.0). Majority of patients were diagnosed at stage 4 (69.6%). The average of CCI was 0.4 (±0.9) and diabetes mellitus was found to be the most common comorbidity (7.0%): 23.5% of patients were recorded to have one or more comorbidities (CCI ≥ 1) during the year before diagnosis, and the percentage increased up to 35.1% when considering all prior disease diagnosis up to 6 years before cancer diagnosis (Table 1).
Characteristics of oropharyngeal and hypopharyngeal cancer patients in Taiwan (2007-2010).

CCD: Charlson’s comorbidity index SD: standard deviation.
Table 2 lists survival rates and univariate hazard ratios (HRs) for all-cause deaths and cancer-related deaths by patients’ comorbidity condition (CCI = 0, =1, ≥2) for each cancer stage. Within each cancer stage, the overall survival and cancer-related death rates were significantly different among comorbidity condition at stages 3 and 4 (p values < 0.05). The corresponding univariate HRs were all significantly higher for the group with severe comorbidity (CCI ≥ 2) versus no comorbidity. The Kaplan–Meier survival curves for comparing patients with comorbidities (CCI > 0) versus no comorbidity were shown in subgroups of oropharyngeal, hypopharyngeal and cancer stages (Figures 1 and 2). Patients with comorbidities showed significantly less survival rates in oropharyngeal site (Log-rank p value = 0.0468) and particularly in stage 3 (Log-rank p value = 0.0002) (Figure 2).
Survival rates and univariate hazard ratios for all-cause death and cancer-related death.

HR: hazard ratio; CI: confidence interval; CCI: Charlson’s comorbidity index.

Kaplan–Meier survival curves for comparing patients without (solid blue) and with (dash red) comorbidities of oropharyngeal (upper) and hypopharyngeal (lower) cancer.

Kaplan–Meier survival curves for comparing patients without (solid blue) and with (dash red) comorbidities of stages 1–4 (Oropharynx and hypopharynx combined).
Furthermore, a comparison of cancer treatment management within each combination of stage/comorbidity condition is shown in Table 3. Although there is tendency that patients with comorbidities have less aggressive treatment, the χ2 tests do not indicate any statistical differences (all p value > 0.05). The different management types may yield 20.7%, 23.3%, 33.0% and 31.4% differences from best versus worse 2-year survival rates in the patient groups of stages 1, 3 and 4 without comorbidity and stage 4 with comorbidity, which the Log-rank tests were significant. In addition, we computed the differences of 2-year survival rates between all-cause and cancer-related rates, and found that groups with differences greater than 10% were in patients with comorbidities and treated by radiotherapy or chemotherapy without surgery in stages 2 and 3.
Comparison of treatment management in stages with comorbidity condition.
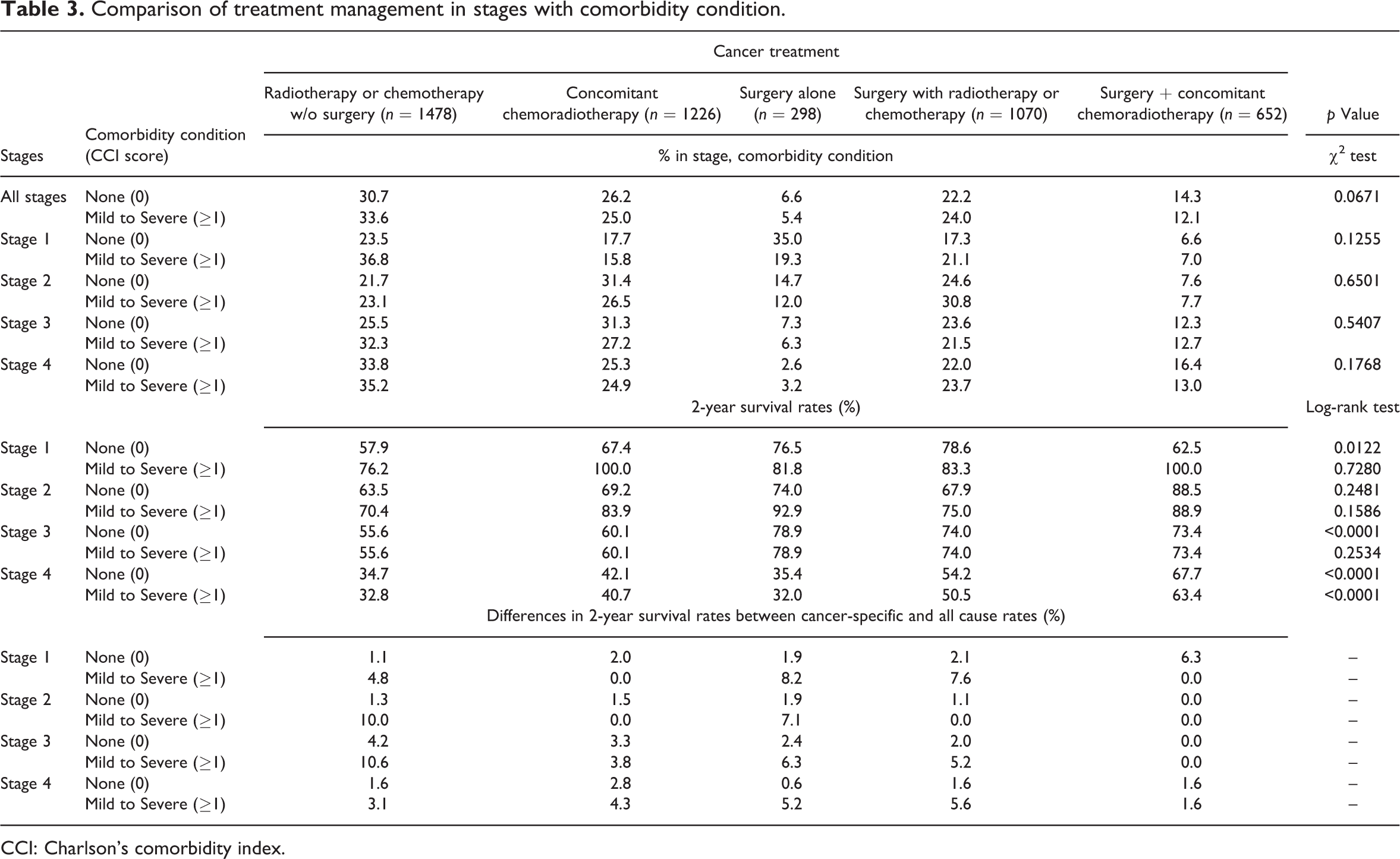
CCI: Charlson’s comorbidity index.
The age and gender adjusted HRs (Table 4) showed majority of the treatments having less mortality risk than the reference treatment of radiotherapy or chemotherapy. For patients of stage 4 with comorbidities, treatments of surgery with radiotherapy or chemotherapy and surgery with concomitant chemoradiotherapy had relatively less risk (HR = 0.69, 95% CI = 0.55–0.86, p value = 0.0013; HR = 0.44, 95% CI = 0.32–0.60, p value <0.0001). After adjusting for age, gender and stages, all treatment groups in patients without comorbidities showed significantly lower mortality risk than the reference treatment in the analysis of all stages combined. For patients with comorbidities, the treatment of surgery alone became non-significant (HR = 0.81, 95% CI = 0.55–1.20, p value = 0.2963). In both of the comorbidity groups, the treatment by surgery with concomitant chemoradiotherapy had the least HR (without comorbidity: HR = 0.40, 95% CI = 0.34–0.47, p value < 0.0001; with comorbidity: HR = 0.48, 95% CI = 0.36–0.64, p value < 0.0001).
Adjusted hazard ratios for treatments in different stage and comorbidity condition.a

HR: hazard ratio; CI: confidence interval; CCI: Charlson’s comorbidity index.
aHRs of all-cause death are adjusted by age, gender and stages.
Discussion
Many studies have suggested the negative impact of comorbidities on the prognosis of head and neck cancer, 12,13 that includes nasopharyngeal, oropharyngeal and laryngeal cancers. 2 Our previous study also found that patients with severe comorbidity conditions were associated with 6–17% lower 3-year survival rates for all stages of oral cavity cancer. 5 In this study, we investigated the impact of comorbidity in oropharyngeal and hypopharyngeal cancer patients, and significant differences were also found in stages 3 and 4 (Table 2). The 3-year survival rates were 15% and 11%, respectively, lower in patients with severe comorbidity condition. While lifestyle is often the major cause of oral cancers, cigarette smoking, excessive alcohol drinking and betel quid chewing contribute not only to oral, oropharyngeal and hypopharyngeal cancer but also other medical conditions. Therefore, a proportion of oral, oropharyngeal and hypopharyngeal cancer patients would also have comorbidities at the time of cancer diagnosis.
Piccirillo 12 used a modified Kaplan–Feinstein comorbidity index to compare the severity of comorbidity among several common cancers. There were 45% of the head and neck cancer patients with comorbidities, being the third most common cancer next to lung cancer (62%) and colorectal cancer (49%), and higher than breast cancer (37%), gynecologic cancer (38%) and prostate cancer (37%). Measurement of comorbidity can be obtained from self-reports of patients, from the review of their medical records and also from claim-based administration database. Each method has its own advantages and disadvantages. 14 Considering CCI is a convenient and validated instrument, 4,15 we derived the information of comorbidity from ICD-9 codes 10,11 by using the criterion of three or more outpatient diagnosis or one hospitalization to avoid any disease code given for screening purpose. Our study revealed that 76.5% of patients were free of any comorbidity, which is similar to our previous findings in oral cavity cancer (78.7%) 5 . If we did not limit diseases within 1 year before cancer diagnosis, then the percentage dropped to 64.9%. A Danish study using the DAHANCA database using up to 10 years of discharge records showed a 64% had no comorbidity, 7 and 30% and 35% were comorbidity-free in laryngeal and pharyngeal cancer patients. 6 Therefore, at least one-fourth of head and neck cancer patients are presenting with comorbidities at the time of diagnosis. Our previous study 5 reported the prevalence rates of diseases and condition indicated by the CCI from the whole NHI samples in 2010 of Taiwan. The proportion with any of the diseases in the CCI was 14.9%. There were 5.4% with diabetes and 1.1% with diabetes with chronic complication. Participants of current study had higher proportion of diabetes than the general population (7.0% vs. 5.4%). Diabetes is associated with increased risk for several cancers, including liver, pancreas, endometrium, colorectal, breast and bladder. 16 A meta-analysis later indicated that individuals with type 2 diabetes have an elevated risk of oral cancer. 17 Our results support this earlier indicated association.
The decision of cancer management primarily depends on the clinical stage and pathological conditions found at the time of cancer diagnosis. In addition, patients’ other medical conditions are also very important to be taken into consideration. A recent clinical cross-sectional study from India investigated changes in therapeutic decision-making. Among 139 patients, there was significant positive correlation between comorbidity and changes in therapeutic decision-making. 3 Our previous study in oral cavity cancer patients showed that patients with comorbidities received less curative treatments for patients with stages 2–4 of cancer. 5 Consequently, the 2-year survival rates within each combination of stage and comorbidity condition were 13–37% lower, indicating a survival benefit from aggressive treatment management. 5 An earlier report also showed that curative-intent aggressive treatment improved survival by 63–67% (HRs = 0.37–0.33), given the same degrees of comorbidity condition for locally advanced (stages 3 and 4) head and neck squamous cell carcinoma patients. 18 Although there was a tendency that patients with comorbidities received less curative treatment within the same cancer stage, the statistical significance was not reached. From our results, curative treatments are associated with 32–60% less in all-cause mortality risk regardless of comorbidity conditions (HRs = 0.68–0.40).
The DAHANCA study showed that comorbidity has a negative prognostic impact on overall survival in head and neck cancer patients, but cancer-specific death was not significantly affected by comorbidity suggesting that patients die from their other medical conditions rather than their cancer. 6 Our previous results from oral cavity cancer supported these findings for patients in the early stages of cancer. 5 The analysis of all stages from current study also reflects a significant difference among comorbidity conditions in all-cause death (p = 0.0001) and no significance in cancer-related death (p = 0.0695). However, for patients with advanced stages, both all-cause and cancer-related deaths were all significantly different (p < 0.05). Unlike our previous report, the crucial challenges to mortality on management of other medical conditions were not limited to early cancer stages. In fact, we found that the greater difference of 2-year survival rates between all-cause and cancer-related survival rates was in patients with comorbidities and treated by radiotherapy or chemotherapy without surgery in stages 2 and 3. Possible interactions between cancer treatment decision and other medical conditions could be investigated in future studies.
Our study has limitations on some confounders of cancer prognosis, including the usage information of betel quid chewing, cigarette smoking and alcohol drinking as well as other lifestyles. These factors were not recorded in any of the three databases used in this study, and might have effects on the estimates of risk if they were included in regression analyses. We suspect that the effects might be minimal since these factors could affect in both directions. We used the ICD-9CM to identify comorbid diseases. Since sometimes disease codes may be given for screening purposes, to assure that a patient had been diagnosed and treated for a comorbidity disease we restricted that a disease had three or more outpatient records or one hospital admission of a given condition to minimize any false positives. Majority of oropharyngeal cancer patients identified from TCR was in stage 4. Consequently, we did not have enough sample sizes for more comorbidity categories in early stages. A further limitation of the study was lack of human papillomavirus (HPV) status in databases. HPV-positive oropharyngeal cancers are known to have a better survival, and ideally this should be entered in the logistic regression. Additionally, the C14 in ICD10 code may contain some lip and oral cavity cancer cases. Therefore, patients in C14 may not all be oropharyngeal cancers. Since the number is very small (n = 16), we suspect the effect might be minimal.
Our study results together with our previous report 5 support an inverse relationship of comorbidity with survival outcome. The importance of curative-intent cancer management as well as managing other medical conditions is pertinent to enhance better patients’ survival outcome.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research is supported by Kaohsiung Medical University Research Fund (KMU-M103014).
