Abstract
Objectives:
Patients on phenprocoumon presenting for surgery are often converted to heparin preoperatively. We assessed the impact of this conversion on outcome in cardiac surgery patients.
Methods:
From November 2007 to September 2013, 115 patients with phenprocoumon therapy presented for surgery and were operated either before or after the international normalized ratio (INR) had normalized (group A, INR > 1.3, n = 71; group B, INR ⩽ 1.3, n = 44 at day of surgery; group C, matched control cohort without preoperative phenprocoumon, INR ⩽ 1.3, n = 60).
Results:
Patients received a mix of cardiac surgical procedures (including valves, coronary artery bypass grafting, ventricular assist devices, and transplantation) without intergroup differences. Surgery was emergent in 11%, 2%, and 13% in groups A, B, and C, respectively (p = 0.2). Preoperative hospital stay was longest in group B (p < 0.01). Hospital mortality was high with 26% in group A (EuroSCORE 22 ± 22), 13% in group B (EuroSCORE 17 ± 16), and 7% in group C (EuroSCORE 10 ± 15) p < 0.01. Mean chest tube drainage (first 24 h) was lowest in group C (mean/min/max, A: 1096/350/4750 mL; B 1086/300/4100 mL; C 757/120/2170 mL, p = 0.03), and preoperative hemoglobin was highest (A: 7.5 ± 1.2, B: 7.5 ± 1.4, C: 8.1 ± 1.2 mmol/L, p = 0.01). If transfusion was required, patients in group A received more red blood cells (mean units, A: 4.5 ± 8; B: 3.5 ± 6; C: 1.5 ± 3, p = 0.02). By multivariate analysis, preoperative INR was not an independent predictor of postoperative bleeding (p = 0.8). Only low preoperative fibrinogen predicted increased chest tube drainage, independent of phenprocoumon (p = 0.01).
Conclusion:
Patients presenting for cardiac surgery with preoperative phenprocoumon represent a high-risk population. Bleeding complications for these patients are higher but do not seem to be influenced by preoperative INR. Our data question the need for conversion to heparin.
Introduction
Long-term oral anticoagulation therapy is frequently prescribed for patients at risk of arterial or venous thromboembolism, such as patients with atrial fibrillation, mechanical heart valves, or prior thromboembolic events. 1 Phenprocoumon is still the main drug currently prescribed for these purposes and is the only drug available for anticoagulation in the presence of mechanical valves. 2 Continuation of phenprocoumon in elective surgery is believed to be associated with increased postoperative bleeding. 3
Excessive postoperative hemorrhage in cardiac surgery is a serious complication which may lead to adverse postoperative events. 4 It is associated with increased risks of re-exploration, transfusion needs, intensive care support, manpower requirements, and it places a high demand on hospital resources. 5 It is common practice to stop phenprocoumon and bridge therapy with heparin until normalization of the international normalized ratio (INR) is achieved. This is done with the goal to minimize the risk of postoperative complications, mainly bleeding. However, the evidence for this strategy has recently been questioned. 6
Current trials have investigated such bridging strategies and demonstrate even more bleeding complication with bridging therapy in patients undergoing cardiovascular implantable electronic device procedures.7,8 Therefore, we assessed whether normalization of the INR leads to changes in outcome of patients undergoing cardiac surgery in our patient population.
Materials and methods
Patient population
We retrospectively evaluated all 115 patients under phenprocoumon therapy admitted to our center for cardiac surgery of any kind (coronary artery bypass grafting (CABG), valves, assist devices, and transplantation or combinations procedures) between November 2007 and September 2013. All patients had an INR > 1.3 on admission. This INR value represents the upper limit of normal in our laboratory. We divided the study population into two groups based on the INR at the day of surgery (group A, without bridging therapy: n = 71, INR > 1.3 and group B, with bridging therapy: n = 44, INR ⩽ 1.3). The main inclusion criterion was long-term intake of oral anticoagulation (phenprocoumon). The cohort was matched for procedure, age, and gender to patients without phenprocoumon treatment on admission during the same period (group C; INR ⩽ 1.3, n = 60). There were no exclusion criteria. We reviewed the patients’ medical charts for postoperative chest tube drainage within the first 24 h, transfusion requirement including all blood products and hemostasis factors as well as re-exploration rates.
Conversion protocol to heparin
In group B, all patients were admitted to the hospital with an INR above 1.3 and the decision of the surgical team was made to delay surgery until INR had normalized. In these patients, phenprocoumon was discontinued and unfractionated heparin was started (dose-adjusted for a partial thromboplastin time (PTT) of 50–60 s) when the INR went below 2.0. There were regular coagulation checks. The mean time of bridging was 5–7 days.
Statistical analysis
Baseline patient characteristics were reported as mean ± standard deviation or standard error of the mean (SEM) for quantitative variables and as frequencies and percentages for categorical variables. Multivariable linear regression analysis was applied to assess the effect of preoperative INR level, thrombocyte count, and fibrinogen level on postoperative bleeding. In addition, we applied chi-square or Fisher’s exact test, Mann–Whitney U-tests, and one-way ANOVA with post hoc test for statistical comparison of groups. Differences were considered significant at p < 0.05. All statistical calculations were performed using SPSS 23.0 software for Windows®.
Results
Table 1 shows the demographic and clinical characteristics of all patients of the three groups either with (A and B) or without (C) preoperative phenprocoumon. Only patients in group A had INR above normal at day of surgery. The mean age ranged between 65 and 70 years and the majority of patients were male; one-third was diabetic. Almost 10% were diagnosed with endocarditis in all groups. The ejection fraction was highest in group C and most of the patients were in New York Heart Association (NYHA) class III categories in all three groups. There were five patients on dialysis, and two patients had signs of acute liver injury preoperatively. They all belonged to group A. Group C had the most patients with aspirin. The reasons for anticoagulation in groups A and B were atrial fibrillation in the majority of patients, followed by patients with mechanical valves. There were two patients with left ventricular thrombus, two with assist devices. All of them belonged to group A. Taken together, perioperative risk was highest in group A (also reflected in the EuroSCORE). Accordingly, mortality was highest in group A (group A 26%, group B 13%, and group C 7%, p ⩽ 0.01). Supplementary Table S1 shows a detailed listing of the perioperative characteristics, risk scores, and causes of death for those patients who died in the hospital.
Demographic characteristics of cardiac surgery patients who did (group A: INR > 1.3 and group B: INR ⩽ 1.3) or did not receive phenprocoumon preoperatively (group C: matched cohort with INR ⩽ 1.3).
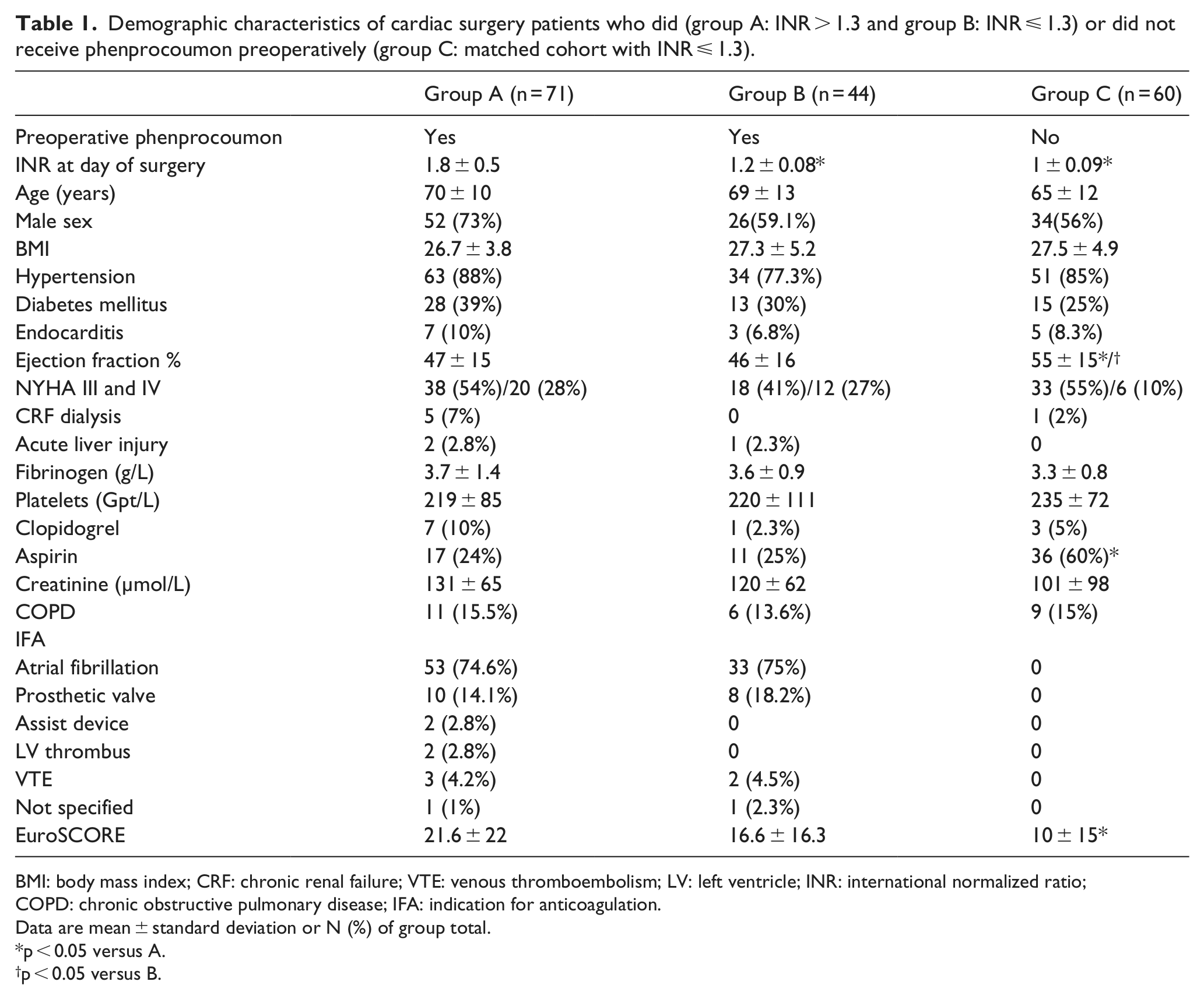
BMI: body mass index; CRF: chronic renal failure; VTE: venous thromboembolism; LV: left ventricle; INR: international normalized ratio; COPD: chronic obstructive pulmonary disease; IFA: indication for anticoagulation.
Data are mean ± standard deviation or N (%) of group total.
p < 0.05 versus A.
p < 0.05 versus B.
Table 2 shows the perioperative features of the three groups. Group B had the longest preoperative hospital stays. The difference was due to the time needed for bridging therapy with heparin. An important preoperative difference between the groups was identified with respect to the urgency of the operation. Group A had the only two patients with surgery started under cardiopulmonary resuscitation (CPR). There was only one patient with an emergency indication in group B. All groups contained patients that underwent heart transplantation. These patients were coded as urgent in this article. There was no difference with respect to cardiopulmonary bypass times, cross clamp times, or other technical details of the operative procedures. The operative procedures performed were the same in all three groups with the majority being valves followed by CABG plus combined procedures and transplantations. Preoperative hemoglobin levels were highest in group C. This difference was no longer present after surgery.
Perioperative data of cardiac surgery patients who did (group A: INR > 1.3 and group B: INR ⩽ 1.3) or did not receive phenprocoumon preoperatively (group C: matched cohort with INR ⩽ 1.3).
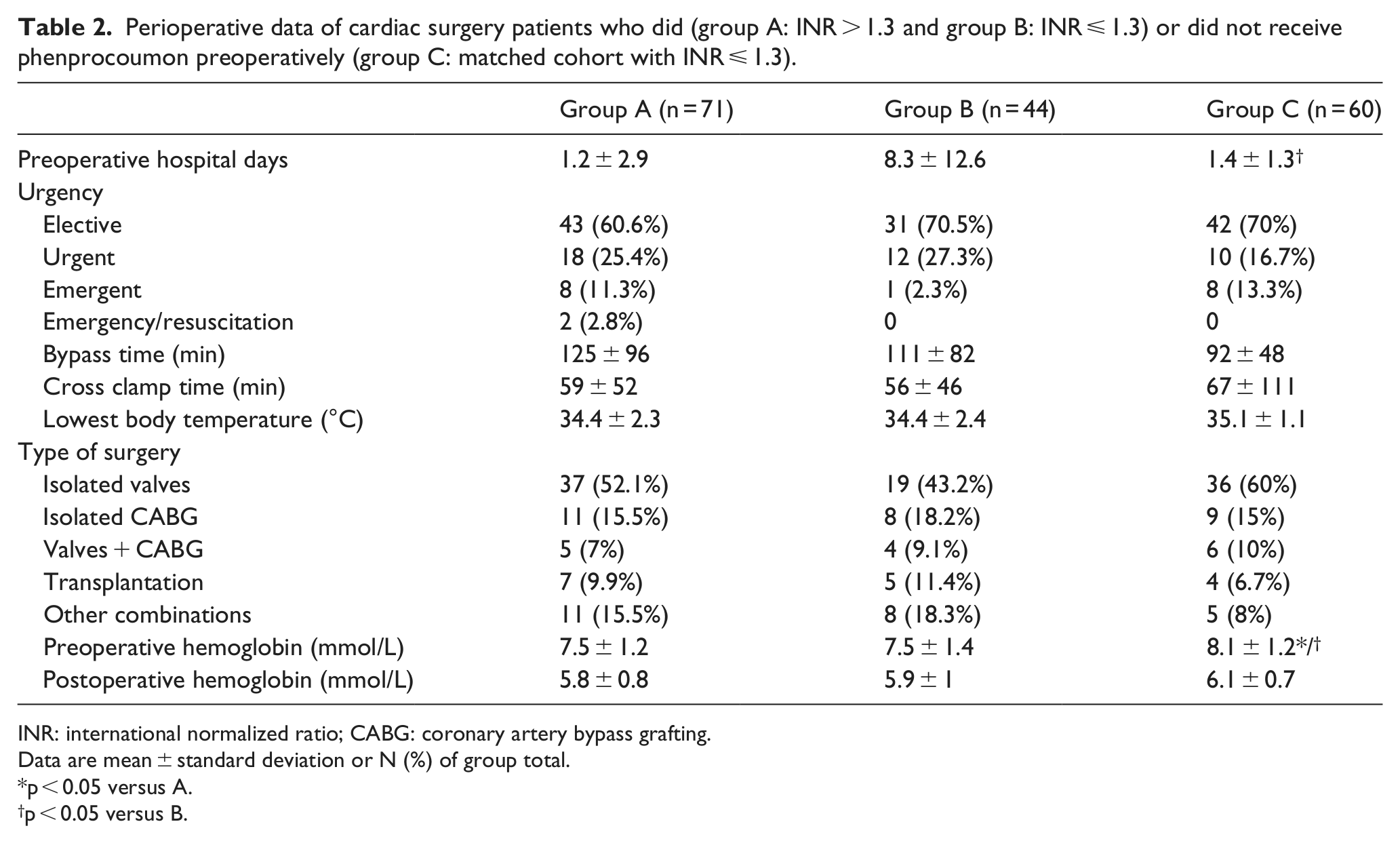
INR: international normalized ratio; CABG: coronary artery bypass grafting.
Data are mean ± standard deviation or N (%) of group total.
p < 0.05 versus A.
p < 0.05 versus B.
Figure 1(a) shows the average chest tube drainage within the first 24 h postoperatively in all three groups. Patients in group C bled the least. However, those patients with phenprocoumon bled identical amounts independent of INR at the day of surgery. The fraction of patients requiring any kind of transfusion was also lowest in group C (66.2%, 56.8%, 48.3%; p = 0.1). Figure 1(b) illustrates the number of packed red blood cells transfused. Again, patients with preoperative phenprocoumon received the highest number of blood transfusions (groups A and B) and those without phenprocoumon received the least (group C). Finally, Figure 1(c) shows the rates of re-operation in all three groups. Re-exploration due to bleeding was higher in groups A and B compared to C, even if this difference was not statistically significant.
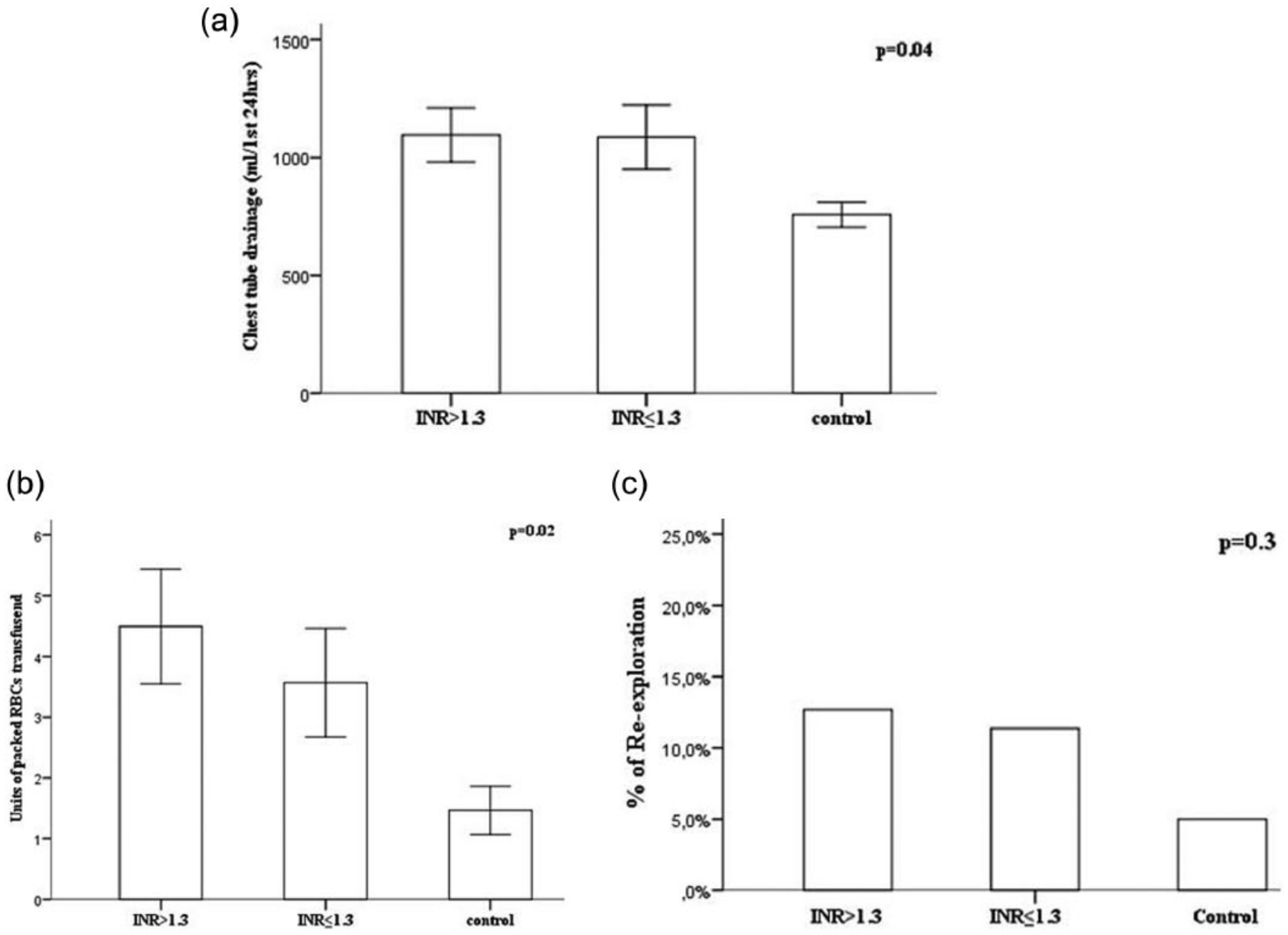
(a) Chest tube drainage within the first 24 h postoperatively, (b) number of packed red blood cell units transfused postoperatively, and (c) rate of re-exploration among patients with phenprocoumon with (group B, INR ⩽ 1.3) or without bridging to heparin (group A: INR > 1.3) and matched patients without phenprocoumon (group C: control, INR ⩽ 1.3). Data are mean ± SEM (a and b) and percentage (c).
Thus far, we presented only mean values, but in surgical procedures, bleeding can vary significantly and may have different causes. Table 3 therefore shows the characteristics of the 15 patients who bled more than 2000 mL within the first 24 h. There was a broad mix of operative procedures performed, most of them being complex. There were more patients with bleeding amounts above 2 L in group A. When re-explorations were performed due to bleeding, a clear bleeding cause was identified in only one case (group A). The mean preoperative INR in 10 patients belonging to group A was 1.8 ± 0.6 and the postoperative INR was 1.5 ± 0.2. The preoperative INR in five patients belonging to group B was 1.2 ± 0.05 and the postoperative INR was 1.5 ± 0.1. If the final outcome was death, sepsis and multiorgan dysfunction was the most common cause. There was no obvious difference in patient characteristics between the groups.
Characteristics of the 15 patients who bled more than 2000 mL within the first 24 h.
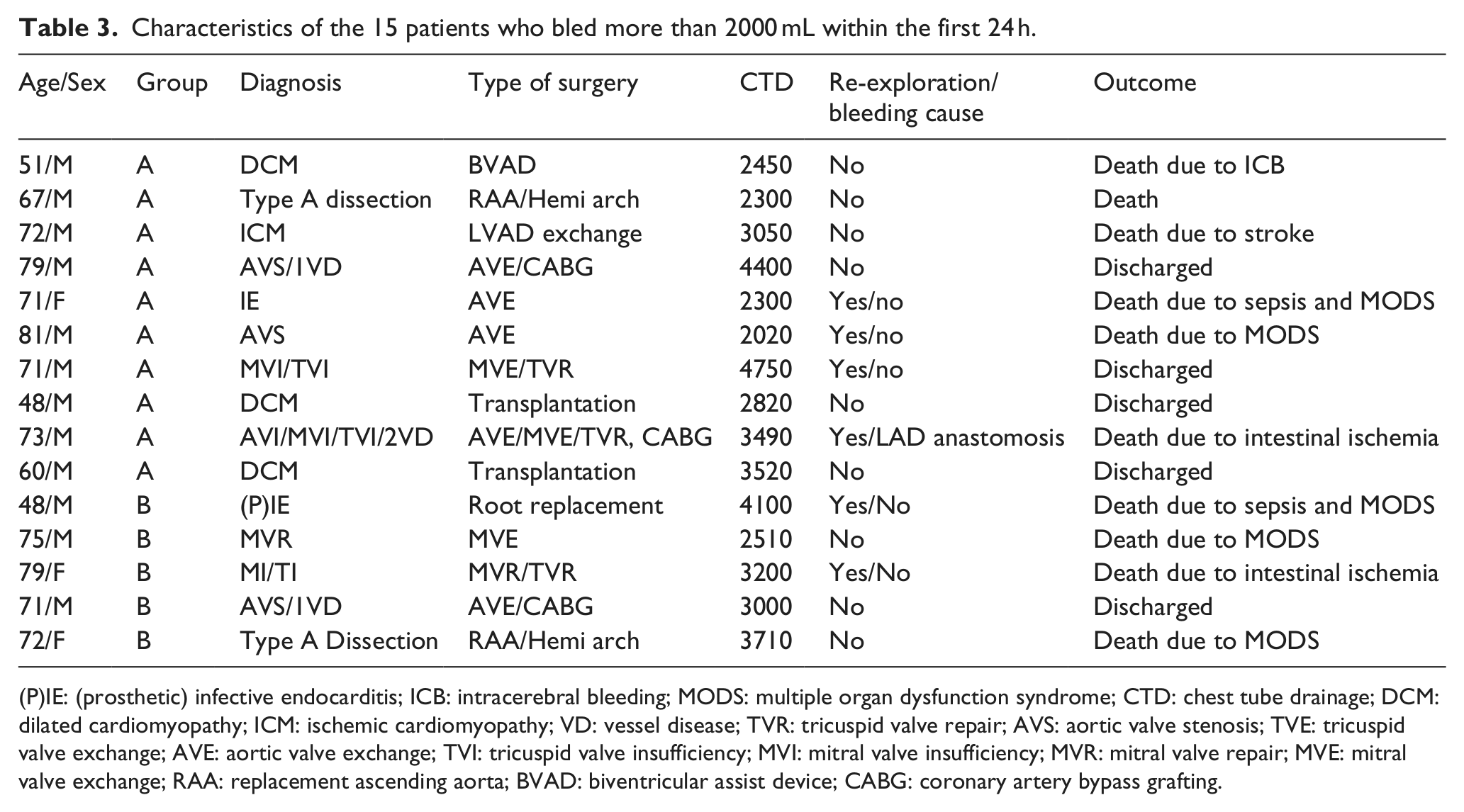
(P)IE: (prosthetic) infective endocarditis; ICB: intracerebral bleeding; MODS: multiple organ dysfunction syndrome; CTD: chest tube drainage; DCM: dilated cardiomyopathy; ICM: ischemic cardiomyopathy; VD: vessel disease; TVR: tricuspid valve repair; AVS: aortic valve stenosis; TVE: tricuspid valve exchange; AVE: aortic valve exchange; TVI: tricuspid valve insufficiency; MVI: mitral valve insufficiency; MVR: mitral valve repair; MVE: mitral valve exchange; RAA: replacement ascending aorta; BVAD: biventricular assist device; CABG: coronary artery bypass grafting.
Table 4 shows the outcome of the multivariable linear regression analysis aimed at identifying independent predictors for chest tube drainage. None of the expected factors including the preoperative INR, the number of preoperative platelets, or lowest body temperature independently predicted postoperative chest tube drainage. Nevertheless, the patients’ preoperative fibrinogen level was an independent predictor of chest tube drainage. In addition, cardiopulmonary bypass time also predicted chest tube drainage.
Outcome of multiple linear regression analysis identifying independent predictors of chest tube drainage.
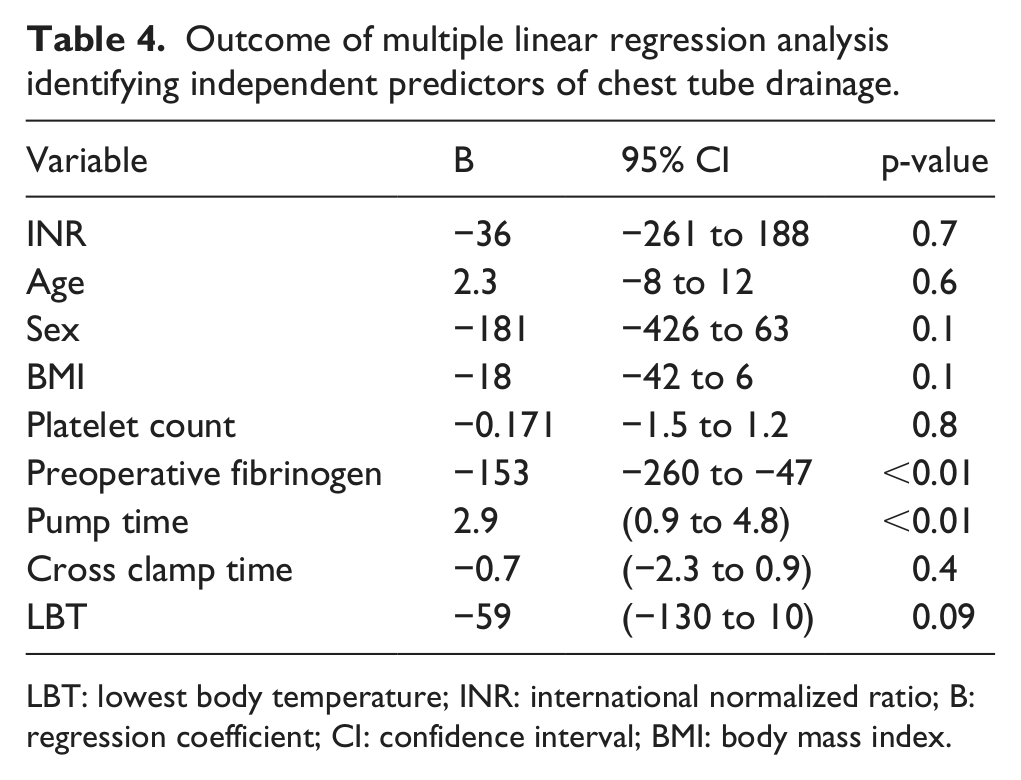
LBT: lowest body temperature; INR: international normalized ratio; B: regression coefficient; CI: confidence interval; BMI: body mass index.
Discussion
We demonstrate in this study that patients presenting for cardiac surgery with preoperative phenprocoumon represent a high-risk population. Bleeding complications for these patients are higher than for those without preoperative phenprocoumon, but they do not seem to be influenced by preoperative INR. Our data question the need for conversion to heparin.
For decades, it has been standard practice for patients to discontinue phenprocoumon intake and convert anticoagulation to heparin before surgery in order to prevent excessive bleeding during or after the procedure. 9 The rationale consisted of the argument that phenprocoumon was not readily antagonizable. Based on this rationale bridging with unfractionated heparin appears reasonable. However, in most cases, anticoagulation is never fully stopped. In addition, the experience in the transplant field, where many phenprocoumon patients have been waiting for an organ and had to be operated “under phenprocoumon” once an organ was available, also led us to question the need for a switch, because the re-exploration or bleeding rates post-transplant are usually not excessively high.10,11 Thus, it was no surprise to see the already cited report where bleeding was not lower when phenprocoumon was converted to heparin in patient populations where implantable cardioverter defibrillators (ICDs) were exchanged.12–14 We therefore undertook this analysis.
We were able to demonstrate that patients with preoperative phenprocoumon therapy bleed more and require more transfusions and re-exploration than patients who are not treated with preoperative phenprocoumon. Importantly, we failed to show any significant impact of bridging. The outcomes with repsect to bleeding within the first 24 h, re-exploration rates, and transfusion requirements were identical in the groups with and without bridging, although the INR at the day of surgery was significantly higher in the unbridged group. Our results are consistent with the recent reports in the ICD population12–14 but also find support from the cardiac surgical field where warfarin did not seem to increase bleeding rates. 15
Thus, despite the similar conclusion that was already reached 20 years ago, it is interesting to note, that there was no change in routine practice or visible action seen with respect to further investigation of this topic. However, we believe that the current level of evidence argues very much in favor of a prospective randomized trial. Such a trial could also address the second important finding of our study, that is, the role of preoperative fibrinogen levels on postoperative chest tube drainage.
When we performed our multivariate analysis, most variables expected to be predictive for bleeding, including INR, platelet count, and cross clamp time failed to be independently associated with increased chest tube drainage postoperatively (Table 4). Only cardiopulmonary bypass time fulfilled its expectations. However, a low fibrinogen level before surgery predicted a high amount of postoperative bleeding. Low levels of fibrinogen have already previously been identified as a predictor of postoperative bleeding after cardiac surgery in patient populations who were not receiving oral anticoagulation.16,17 Thus, our finding provides support for this conclusion in a patient population with oral anticoagulation. Taken together, these results suggest that transfusion or bleeding risk may be more related to fibrinogen levels rather than INR.18,19 Considering that some studies even report that correction of fibrinogen levels my decrease bleeding rates,20,21 this finding requires further investigation.
Assessing bleeding rates also requires taking other major outcomes into account. Mortality in group A (INR > 1.3) was the highest in all three groups. It may be argued that the early time point of operation lead to excessive mortality. Indeed, 10 of the 19 patients who died in group A had chest tube drainage rates above 2 L (Supplementary Table S1) and a clear bleeding cause was only identified in one patient (Table 3, group A). However, four of the six patients in the group B who had normal INR at the beginning of the procedure and had been converted to heparin also bled more than 2 L; one of the four patients in group C bled more than 2 L. In addition, a much larger fraction of patients in group A was operated under emergent conditions, with the only two patients where surgery was started under CPR. Considering the fact that group A was almost twice as large and contained higher rates of emergency indications, it is difficult to pin the difference in mortality to the different INR management.
In addition to the already addressed limitations, the conclusions drawn from this study are limited by the retrospective nature of the study. It is not possible to assure that all physicians universally followed the same transfusion protocols and indications for surgery may have varied among surgeons. In addition to that, we did not include the use of intraoperative transfusion requirements of platelet and other hemostatic factors. A second limitation is the small sample size of the groups.
Conclusion
We show that patients with preoperative phenprocoumon treatment undergoing cardiac surgery represent a high-risk population. We mainly demonstrate that bleeding complications or transfusion requirements for patients with preoperative phenprocoumon do not seem to be influenced by preoperative normalization of the INR, questioning the need for heparin bridging in cardiac surgery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
