Abstract
The recent introduction of direct oral anticoagulants, including rivaroxaban, dabigatran, apixaban, and edoxaban, for the acute treatment and secondary prevention of venous thromboembolism and in atrial fibrillation has been shown to provide greater clinical benefit than oral vitamin K antagonists. However, direct oral anticoagulants are associated with adverse events, the most common being major bleeding; such events require the reversal of the anticoagulant effects by specific agents. In this case report, we describe an 87-year-old female with atrial fibrillation treated with dabigatran who had massive rectal bleeding. Idarucizumab 5 g (2 × 2.5 g/50 mL) was successfully used to reverse dabigatran effect; subsequent to this, treatment with dabigatran was resumed, and there were no further bleeding events. This suggests that dabigatran can be safely restarted after major bleeding, but this outcome needs to be confirmed in studies involving larger groups of patients.
Introduction
The recent development of direct oral anticoagulants (DOACs), including rivaroxaban, dabigatran, apixaban, and edoxaban, for the acute treatment and secondary prevention of venous thromboembolism (VTE) and in atrial fibrillation (AF) has been associated with greater clinical benefit compared with oral vitamin K antagonists (VKA).1,2 While DOACs have improved stroke management, they can lead to adverse events, the most common of which is bleeding, which requires reversal of the anticoagulant effects by specific agents. Reversing the effects of VKAs is done with vitamin K, as well as with fresh frozen plasma and prothrombin complex concentrates. For DOACs, andexanet is used for the factor Xa inhibitors (including rivaroxaban, apixaban, and edoxaban), aripazine for the Xa inhibitors and possibly dabigatran, while idarucizumab is used for dabigatran; the latter is a monoclonal antibody aimed at immediately and specifically reversing the anticoagulant effect of dabigatran. 3 The utility of idarucizumab as a rapid reversal agent has been demonstrated in a prospective cohort study whereby idarucizumab 5 g given intravenously completely reversed the anticoagulant effect of dabigatran within minutes among patients with serious bleeding or required urgent surgical procedures. 4 This report addresses the ability of idarucizumab to reverse a major gastrointestinal (GI) bleed during treatment with dabigatran and details the outcome of resuming dabigatran treatment.
Case report
The patient was an 87-year-old female who was hospitalized on 19 March 2016 for heart failure in chronic AF and recent GI bleeding for hemorrhoids. In addition, she had comorbid arterial hypertension, asthma, osteoporosis, and allergy to acetylsalicylic acid. During previous anticoagulant treatment with sodium enoxaparin, the patient had experienced rectal bleeding that resulted from a fecaloma with hemorrhoid rupture. At the time of hospital admission, she had been receiving treatment with dabigatran 110 mg twice daily (bid), furosemide 25 mg bid, canrenone 100 mg once daily (od), bisoprolol 1.25 mg bid, pantoprazole 40 mg od, fluticasone/salmeterol 250/25 µg 2 puffs bid, and tiotropium 2.5 µg 2 puffs od.
In the days following hospital admission, the patient developed fever and dyspnea, with high levels of C-reactive protein (CRP). She was diagnosed with pneumonia and treated with piperacillin/tazobactam, azithromycin, and prednisone 25 mg at tapering doses. Despite increasing doses of diuretics, her heart failure worsened; the patient was moved to the cardiology unit, where an echocardiography showed a moderate-to-severe mitral insufficiency, biatrial dilatation, and bilateral pleural effusion. Following clinical improvement from treatment (which also included oxygen therapy and non-invasive ventilation for respiratory failure), the patient was transferred to the subacute care unit.
On 30 April, major hemorrhoidal bleeding occurred; the patient was awake with hypotension (blood pressure 90/50 mm/Hg), tachycardia (heart rate 120 bpm), and SpO2 94% breathing room air. A Foley catheter was inserted into the rectum (inflated at 40 cc) and surrounded by a Spongostan hemostatic sponge, but this only resulted in a minor reduction in bleeding. Bleeding continued, and the patient became drowsy (blood pressure 80/40 mm/Hg, heart rate 130 bpm, hemoglobin 10.3 g/dL, compared with 11.7 g/dL the previous day, and international normalized ratio (INR) 1.2). Subsequently, the patient was taken to the operating theater in hemorrhagic shock caused by massive proctorrhagia; blood pressure rose to 100/40 mm/Hg after infusion of Emagel (polygeline) 500. Rectoscopy detected two bleeding mucosal lesions near the anal sphincter, possibly caused by friction of hard stools. The surgeon found widespread mucosal inflammation, but no indication for surgical repair. Upon rectal examination, the surgeon found hemorrhoid congestion with two bleeding lesions at 3 o’clock, which were sutured by applying a double Spongostan tampon. Following surgery, the patient regained consciousness without complications (blood pressure 104/55 mm/Hg, heart rate 120 bpm, and SaO2 99% in O2 2L via nasal cannula), although some rectal bleeding persisted after the removal of the local compression with Foley catheter in traction. The laboratory data gave a thrombin time (TT) >140 and <20 before and after surgery, respectively, and an activated partial thromboplastin time (aPTT) >50 before surgery. After hemostatic measures were conducted on 30 April, including transfusion of Emagel 500 cc, 3 units of fresh frozen plasma, and 2 units of packed red blood cells, as well as tranexamic acid in 2 doses of 20 mg/kg intravenously, idarucizumab 5 g (2 × 2.5 g/50 mL) was administered in order to reverse the anticoagulant effect of dabigatran. Administration of idarucizumab was not initiated sooner because of practical considerations related to availability. Due to the massive perioperative bleeding, idarucizumab was administered by rapid infusion, resulting in complete cessation of bleeding after approximately 8 h. The dabigatran level, as assessed by the dilute thrombin time (dTT), was189 ng/mL before surgery and 30 mg/mL 12 h after idarucizumab administration. The patient remained under monitoring in the intensive care unit for 4 days without complications. Post-operative thromboprophylaxis was initiated on day 4 with enoxaparin 4000 IU bid.
Complete recovery of the patient took place in about 15 days, with stabilization of clinical parameters and mobilization from bed to chair. No further evidence of rectal bleeding was observed. On 15 May, assessments were made to determine the feasibility of resuming dabigatran treatment. The delay in restarting the treatment was caused by the initial refusal by and the subsequent indecision of the patient and her relatives to consent to the resumption of the drug. During this time, thromboprophylaxis with enoxaparin bid was maintained.
The risks of AF/stroke and major bleeding were determined using the CHA2DS2-VASc (congestive heart failure, hypertension, age ≥ 75 years, diabetes, stroke or transient ischemic attack, vascular disease) and the HAS-BLED (hypertension, abnormal renal or liver function, stroke, bleeding, labile INR, elderly age (>65 years), drugs (acetylsalicylic acid and nonsteroidal anti-inflammatory drugs) or alcohol) scoring scales, respectively. As the cause of bleeding had been effectively treated and eliminated and given that the patient had a CHA2DS2-VASc score of 5 and a HAS-BLED score of 3, it was considered appropriate to resume treatment with dabigatran. On 18 May, echocardiography showed severe dilatation of the left atrium (LA), with a volume of 209 mL, equal to 122 mL/m2 (Figure 1). The finding of a severely enlarged LA, together with the presence of a high CHA2DS2-VASc score, confirmed the decision to resume anticoagulation in this patient, as high LA volume is a risk factor for thromboembolism and hemorrhage. 5 Dabigatran treatment was re-initiated on 20 May at a dose of 110 mg bid. No bleeding occurred in the period leading up to when the patient was discharged on 23 May 2016, nor during the period until 6 February 2017, when the patient was followed-up.
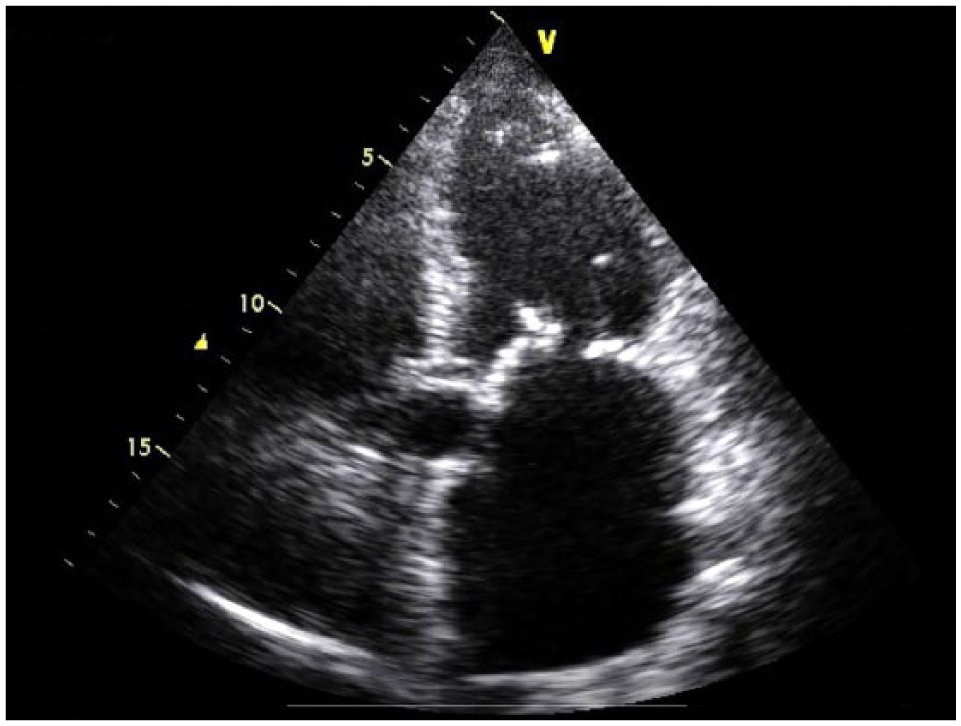
Patient echocardiography showing severe dilatation of the left atrium.
Discussion
Reports have suggested that DOACs are associated with increased risk of GI bleeding compared with VKAs, but a recent meta-analysis of 23 studies found that the risk of major GI bleeding was not different between DOACs and VKAs or low-molecular-weight heparin among patients with AF, patients undergoing orthopedic surgery and those with VTE. 6 With regard to restarting anticoagulation in patients who had GI bleeding, a retrospective cohort study that enrolled 1329 patients with GI bleeding during anticoagulant therapy with VKAs, 653 of whom restarting the treatment, no increased risk of GI bleeding was detected. 7 There does not appear to be any data available as yet for DOACs, but the results of our case presentation suggest that dabigatran is not only a DOAC with a specific reversal agent, idarucizumab, but one that can be resumed after a major bleeding event. A thorough assessment of thromboembolic risk is critical in the management of patients with AF. The thromboembolic risk varies among patients with AF, and oral anticoagulant therapy may not benefit every patient. The expected benefit of anticoagulation (i.e. reduction in thromboembolic risk) must exceed the expected risk of major bleeding; however, the CHA2DS2-VASc and HAS-BLED risk scores do not take into consideration other important parameters (e.g. echocardiography findings) which can also indicate potential thromboembolic events. 5 A positive correlation between LA dimensions and cardiovascular events has been shown in some8–11 but not all12,13 studies, and atrial dimension is increasingly being considered a useful predictive marker of cardiovascular complication in patients with nonvalvular AF in association with CHADS2 and CHA2DS2-VASc risk scores.14,15 In a study of 582 hospitalized patients with AF, transthoracic echocardiography and thromboembolic risk assessment with both CHADS2 and CHA2DS2-VASc scores showed an association between the mean risk scores and increasing LA diameter. 15 LA enlargement is highly prevalent in patients with AF, and CHADS2 and CHA2DS2-VASc risk scores are significantly higher in patients with LA enlargement, 15 and the incorporation of LA size to the current stroke risk scoring symptoms has been proposed in order to better define the risk evaluation and management of AF. 14 Therefore, the evidence that LA volume assessment with echocardiography may be a useful additional parameter in stratification of thromboembolic risk of patients with AF supported our decision to resume DOAC therapy in our patient.
Conclusion
The present case suggests that based on the risk of AF/stroke against the risk of major bleeding as assessed by CHA2DS2-VASc and HAS-BLED scores, it is likely that dabigatran can be safely restarted after major GI bleeding has been effectively managed. Data from case-control studies on large groups of patients are warranted to confirm this outcome.
Footnotes
Acknowledgements
The authors thank Matt Weitz, of Springer Healthcare Communications, for English-language editing of this case report.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Incorvaia reports personal fees from Bayer S.p.A., personal fees from Stallergenes Italy S.r.l., outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Editorial assistance was funded by Boehringer Ingelheim, Italy.
Informed consent
Written informed consent was obtained from the patient for her anonymized information to be published in this article.
