Abstract
Background
The Patient-Determined Disease Steps (PDDS) and Neuro-QoL Lower Extremity Function—Short Form (Neuro-QoL LEF-SF) are patient-reported outcome measures used to monitor mobility symptoms in multiple sclerosis (MS).
Objective
To develop and validate a crosswalk between the PDDS and Neuro-QoL LEF-SF.
Methods
Using equipercentile equating, we created a crosswalk from a training sample of 619 MS patients (mean PDDS = 2.4 ± 2.3; mean Neuro-QoL LEF-SF = 33.0 ± 8.3). Validation was performed in a separate sample of 147 MS patients (mean PDDS = 0.6 ± 1.2; mean Neuro-QoL LEF-SF = 38.9 ± 2.8). Agreement between original and converted scores was evaluated using paired t-tests and concordance correlation coefficients.
Results
PDDS and Neuro-QoL LEF-SF scores were strongly correlated in both training (r = −0.89) and validity (r = −0.76) samples. No significant differences in change scores were observed. Concordance coefficients of original and converted scores supported good to very good agreement.
Conclusion
We developed a crosswalk for the conversion of scores between the PDDS and the Neuro-QoL LEF-SF for patients with MS. Evidence for the preliminary validity of this crosswalk was demonstrated with both the initial training sample and a separate validity sample. This crosswalk facilitates score comparisons and data harmonization across studies and clinical registries using either measure.
Introduction
Multiple sclerosis (MS) is a chronic, immune-mediated disease of the central nervous system that leads to a range of physical, cognitive, and emotional symptoms. Among the most prominent and disabling symptoms experienced by individuals with MS are difficulties with mobility, which often stem from disease-related impairments in lower motor function. 1 These mobility challenges can significantly affect quality of life and functional independence; therefore, highlighting the accurate assessment and monitoring of symptoms is a key priority in both clinical care and research.
One approach to evaluating mobility-related symptoms in MS involves the use of patient-reported outcome measures (PROs). PROs capture a patient's perspective on symptom severity, disease progression, and daily functioning, often providing complementary information to clinician-administered tools.2,3 PROs are especially valuable in chronic neurological conditions like MS, where symptom fluctuations and subjective experiences may not always be fully captured through conversations with providers and objective assessments alone.3,4 Two known PROs in the MS clinical and research spaces for assessing mobility dysfunction are the Patient-Determined Disease Steps (PDDS) and the Neuro-QoL™ Lower Extremity Function—Short Form (Neuro-QoL LEF-SF). 5 The PDDS, a patient-reported measure of overall disability, is a single-item ordinal scale consisting of nine levels that range from 0 (normal) to 8 (bedridden), designed to approximate the Expanded Disability Status Scale (EDSS). 6 It is brief, easy to administer, and has demonstrated strong criterion validity with the EDSS, as well as evidence of convergent and divergent validity across multiple studies.7,8
The Neuro-QoL LEF-SF, on the other hand, is a multi-item PRO developed as part of the National Institutes of Health's Quality of Life in Neurological Disorders initiative. 9 It assesses self-reported difficulty in performing daily activities requiring lower extremity function, such as standing, walking, and climbing stairs. Scores range from 8 to 40, where higher scores indicate better functioning. Moreover, the Neuro-QoL LEF-SF showed acceptable internal consistency (α = 0.95) and found to correlate highly (Spearman r = 0.83) with the PDDS in a sample of MS patients, demonstrating evidence of convergent validity.10,11
Although both the PDDS and Neuro-QoL LEF-SF are used to assess mobility symptoms in patients with MS, they differ in structure, scaling, and item content. These differences can create challenges for comparing or pooling results across studies or clinical settings where one PRO may be preferred over the other in consideration of factors, like ease of administration and time constraints. As a result, there is a growing need for methods that allow for conversion of scores between these measures.
Therefore, in the current study we developed and preliminarily validated a crosswalk between the PDDS and the Neuro-QoL LEF-SF using two samples of individuals with MS. This crosswalk provides a practical solution that enables the opportunity for conversion of scores across different settings using the PDDS or the Neuro-QoL LEF-SF for tracking of mobility symptoms in MS.
Methods
Samples
For development of the crosswalk, we retrospectively examined data collected from MS patients at the University of Colorado's Rocky Mountain Multiple Sclerosis Center between 2014 and 2019. As part of the patients’ visits, patient-reported outcome (PROs) measures were collected to track progression of MS through surveys, either administered in the clinic using iPads or at home with a personal device.
A dataset from a neuroimaging study that included the PDDS and Neuro-QoL LEF-SF was examined to evaluate the validity of the crosswalk in a separate sample. Survey data were collected electronically using iPads from MS patients participating in the separate imaging study at the University of Colorado's Rocky Mountain Multiple Sclerosis Center between 2014 and 2018.
Measures
The PDDS is a single-item patient-reported rating scale of disability associated with MS, ranging from 0 to 8 where 0 = Normal, 1 = Mild Disability, 2 = Moderate Disability, 3 = Gait Disability, 4 = Early Cane, 5 = Late Cane, 6 = Bilateral Support, 7 = Wheelchair/Scooter, and 8 = Bedridden.12,13 For the PDDS, higher scores relate to higher disability. The Neuro-QoL LEF-SF is an 8-item rating scale with response options ranging from 1 to 5 where 1 = Unable to do, 2 = With much difficulty, 3 = With some difficulty, 4 = With a little difficulty, and 5 = Without any difficulty.10,14,15 Higher scores on the Neuro-QoL LEF-SF relate to higher functioning, and total scores range from 8 to 40. To ensure that both measures had the same valence for equipercentile matching, where higher scores related to better functioning, the PDDS was multiplied by −1 such that −8 indicated “Bedridden” and 0 indicated “Normal.” After equipercentile matching, the crosswalk results for the PDDS were multiplied by −1 to return values to the original scale.
Analysis
Analyses were completed in R. 16 To create the crosswalk between the PDDS and Neuro-QoL LEF-SF scores, the equipercentile equating method was used. 17 This method involves identifying scores between both scales that share the same percentile ranks. The use of this method involves some assumptions. It is assumed that the two scales must measure the same construct and possess at least a moderate Spearman correlation (r > 0.3). 17 Using the R Studio “equate” package, 18 we performed equipercentile matching and compared results without presmoothing and with various presmoothing methods (“bump,” “average,” “loglinear”). Presmoothing is used to reduce low or zero counts and to minimize irregularities in the frequency distribution, resulting in a smoother score distribution. Bump adds a small constant to each score frequency to reduce zero counts, average replaces sparse frequencies with averages of adjacent values, and loglinear fits polynomial-based models to generate a smooth expected distribution. Validity of the crosswalk was assessed by comparing the original PDDS and Neuro-QoL LEF-SF scores with the crosswalk converted scores derived from the crosswalked measure. The use of presmoothing versus no presmoothing was based on results from the evaluation of original and converted scores.
When performing the crosswalk, there was a discrepancy after rounding when converting between the highest level of disability on each measure. The maximal disability score on the PDDS conferred a minimal functioning score on the Neuro-QoL LEF-SF of 8; however, the minimal functioning score of 8 on the Neuro-QoL LEF-SF conferred a PDDS score of 7 (Wheelchair/Scooter). This was likely due to the low frequency of PDDS = 8 with only one participant reporting the level of disability noted as “Bedridden.” Therefore, we applied a manual researcher correction to ensure alignment between the two measures, such that the highest level of disability and the lowest level of functioning were equivalent across measures.
Crosswalk converted scores were compared to original scores with multiple methods. Concordance correlation coefficients were used to assess the consistency/agreement between scores, ranging from 0 to 1 with 1 indicating perfect agreement. 19 Interpretation of agreement was based on Altman's (1990) guidelines (Poor = <0.20, Fair = 0.21–0.40, Moderate = 0.41–0.60, Good = 0.61–0.80, Very good = 0.81–1.00). 20 Linear regressions were also conducted to provide common language effect sizes for expected change in the original score as predicted by the converted score and evaluate variance explained of original scores by converted scores; with perfect agreement, both metrics would be equal to 1. Mean scores across employment status on crosswalk converted and original scores were evaluated; higher mobility functioning was expected among those who were employed compared to unemployed. Responsiveness to change was also evaluated by calculating change scores for the original and crosswalk converted scores from the most recent entry and baseline assessments. A paired t-test was conducted to assess whether there were significant differences in change scores between original and crosswalk converted versions.
The same set of comparison procedures were evaluated in the validity sample. This set of validation analyses used baseline data for the primary score comparisons, and most recent entry and baseline for responsiveness to change. Differences between the training and validity samples were evaluated with Welch's two sample t-test and Fisher's exact test for continuous and categorical variables, respectively. Cohen's d and guidelines 21 were used to quantify differences between continuous variables (Small = 0.20–0.49, Medium = 0.50–0.79, Large = ≥0.80).
Results
Sample characteristics
The final analytic training sample consisted of 619 MS patients with an average age of 52.3 ± 12.5 years (17–103), 459 (75%) were female, and with the following breakdown of race/ethnicity: 294 (91%) White, 10 (3.1%) Black/African American, 2 (0.6%) Asian, 15 (4.7%) Other, 1 (0.3%) More Than One Race, 24 (6.3%) Hispanic/Latino (all races), and 297 Unknown (Table 1). The data were from the patients’ most recent assessment. The mean PDDS and Neuro-QoL LEF-SF scores were 2.4 ± 2.3 (range: 0–8) and 33.0 ± 8.3 (range: 8–40), respectively. Median (IQR) PDDS and Neuro-QoL LEF-SF scores were 2 (0–4) and 36 (27–40), respectively.
Sample characteristics.

PDDS = Patient-Determined Disease Steps; Neuro-QoL LEF-SF = Neuro-QoL Lower Extremity Function—Short Form. 1 Mean ± SD (range); n (%).
2 Cohen's standardized mean difference (95% Confidence Interval).
3 Welch Two Sample t-test; Fisher's exact test.
The separate sample used to assess the validity of the crosswalk consisted of 147 patients at baseline with an average age of 41.0 ± 8.5 years (25–55), 120 (83%) were female, and with the following breakdown of race/ethnicity: 123 (92%) White, 6 (4.5%) Black/African American, 4 (3%) More Than One Race, 15 (11%) Hispanic/Latino (all races), and 14 Unknown. In this sample, average PDDS score was 0.6 ± 1.2 (range: 0–6) and mean Neuro-QoL LEF-SF score was 38.9 ± 2.8 (range: 24–40). Median (IQR) PDDS and Neuro-QoL LEF-SF scores were 0 (0–1) and 40 (40–40), respectively.
Compared to the validity sample, the training sample was older (d = 0.92, p < 0.001) and demonstrated higher levels of mobility impairment (PDDS d = 0.86, p < 0.001; Neuro-QoL LEF-SF d = −0.79, p < 0.001). The training sample also had higher male representation (25% compared to 17%, p = 0.029) and levels of unemployment (49% compared to 17%, p < 0.001). Regarding ethnicity, there were no significant differences (p = 0.094) with 6.3% Hispanic/Latino in the training sample compared to 11% in the validity sample. Although both samples were comprised of roughly the same percent of participants that indicated White race (91% training, 92% validity), there were differences in representation across other endorsed groups (p = 0.003). Levels of education were only available in the validity sample, as such no comparison could be evaluated between groups.
Presmoothing
The raw score distributions conferred the same crosswalk conversions between measures as the “average” and “bump” methods. The loglinear presmoothing method resulted in the same Neuro-QoL LEF-SF to PDDS conversions as the raw distributions. However, a slightly different conversion from the PDDS to Neuro-QoL LEF-SF resulted. A PDDS score of 6 mapped to a Neuro-QoL LEF-SF score of 20, and a PDDS score of 7 mapped to a score of 12; after loglinear presmoothing, these mappings shifted to scores of 21 and 13, respectively. All other conversions were identical. Given that minimal differences between the no presmoothing and loglinear presmoothing were detected, the no presmoothing method was retained for subsequent analyses (Supplementary Table 1).
Score agreement
The PDDS and Neuro-QoL LEF-SF scores were highly correlated in both the training (Spearman r = −0.89) and validation (Spearman r = −0.76) samples (Figure 1). The crosswalk between the measures is presented in Table 2. Concordance correlation coefficients of original and converted scores were interpreted as in the very good range for the PDDS (CCC = 0.89) and Neuro-QoL LEF-SF (CCC = 0.89) in the training sample, and good for the PDDS (CCC = 0.79) and Neuro-QoL LEF-SF (CCC = 0.76) in the validity sample. 20 Linear regression models indicated that a one-point increase in converted scores of the PDDS conferred a 0.88- and 0.91-point increase in original scores in the training (R2 = 0.79) and validity (R2 = 0.64) samples, respectively. In the Neuro-QoL LEF-SF, original scores increased by 0.87 and 0.65 points for each one-point increase in the converted scores among the training (R2 = 0.79) and validation (R2 = 0.61) samples, respectively.
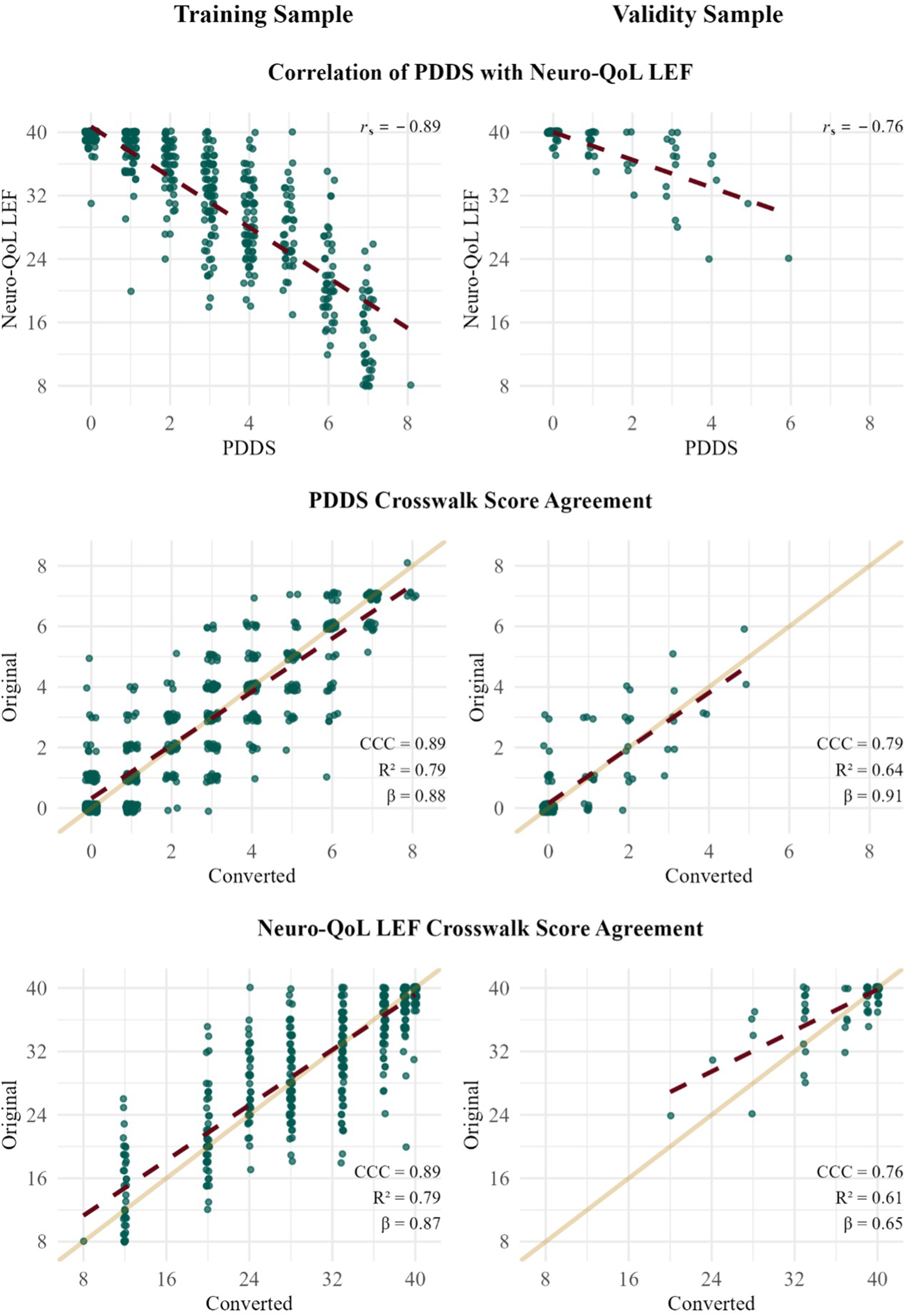
Relationships between PDDS and Neuro-QoL LEF-SF scores.
Crosswalk between the PDDS and Neuro-QoL LEF-SF.

PDDS = Patient-Determined Disease Steps; Neuro-QoL LEF-SF = Neuro-QoL Lower Extremity Function—Short Form.
Employment
Welch's t-tests were conducted to examine average scores across employment status on crosswalk converted and original scores. Employment status was defined as individuals who were employed (either full-time or part-time) versus unemployed. As expected, in the training sample, unemployed individuals had significantly higher PDDS original scores (M = 3.50 ± 2.19) compared to the scores (M = 1.43 ± 1.84) of employed individuals (p < 0.001; Cohen's d = 1.03). Findings were consistent with PDDS-converted scores (Munemployed = 3.49 ± 2.21; Memployed = 1.37 ± 1.83; p < 0.001; Cohen's d = 1.05). A similar pattern was found when comparing scores in the unemployed versus employed for the Neuro-QoL LEF-SF original scores (Munemployed = 29.31 ± 8.66; Memployed = 36.45 ± 6.08; p < 0.001; Cohen's d = -0.96) and converted scores (Munemployed = 29.25 ± 8.85; Memployed = 36.34 ± 6.24; p < 0.001; Cohen's d = -0.93). These differences remained consistent in the validity sample as well, suggesting higher mobility functioning among those who were employed (Supplementary Table 2).
Responsiveness to change
Responsiveness to change was evaluated by calculating change scores from baseline to the most recent assessment for both the original and crosswalk-converted scores. Per available data, median follow-up from baseline to most recent assessment in the training sample was 60 weeks. The validity sample median follow-up time was 104 weeks. For the training sample, paired t-test revealed no significant difference between the change scores of the PDDS original and crosswalk converted scores (difference = 0.03, 95% CI [−0.17, 0.23], t(141) = 0.28, p = 0.782; Cohen's d = 0.02, 95% CI [−0.14, 0.19]). This was similarly found between change scores of the Neuro-QoL LEF-SF original and converted scores (difference = 0.16, 95% CI [−0.50, 0.82], t(141) = 0.49, p = 0.627; Cohen's d = 0.04, 95% CI [−0.12, 0.21]). A similar pattern of results was evident for change scores in the validity sample for both the PDDS (difference = −0.08, 95% CI [−0.21, 0.05], t(137) = −1.21, p = 0.229; Cohen's d = −0.10, 95% CI [−0.27, 0.06]) and the Neuro-QoL LEF-SF (difference = −0.21, 95% CI [−0.63, 0.21], t(137) = −0.99, p = 0.325; Cohen's d = −0.08, 95% CI [−0.25, 0.08]), indicating that the crosswalk-preserved responsiveness to change.
Discussion
The primary aim of the present study was to develop a crosswalk linking the Patient-Determined Disease Steps (PDDS) and the Neuro-QoL Lower Extremity Function—Short Form (Neuro-QoL LEF-SF)—two patient-reported outcome measures. We used the equipercentile method to create the crosswalk across two samples, an initial training sample and a separate validity sample, composed of individuals with MS. The PDDS and Neuro-QoL LEF-SF scores were found to be highly correlated in both the training and validity samples. Concordance correlation coefficients of original and crosswalk converted scores between the PDDS and the Neuro-QoL LEF-SF were interpreted as in the very good range for the training sample and in the good range for the validity sample. When examining employment status across both samples, there was evidence to support higher mobility functioning among individuals with MS who were employed compared to those who were unemployed. We also found no significant differences between change scores from baseline to the most recent assessment from the original scores and those derived from the crosswalk converted scores, across both measures in the training and validity samples.
In consideration of the study's results, evidence for the preliminary validation of this crosswalk was demonstrated with both the initial training sample and a separate validity sample. Concordance coefficients in the good to very good range for the original and crosswalk converted scores highlight that both measures are likely capturing similar underlying constructs of lower extremity mobility. This further reinforces the utility of the crosswalk as a tool for seamless conversion of scores between the PDDS and the Neuro-QoL LEF-SF. In practical terms, the crosswalk can facilitate comparisons across research studies and/or clinical settings using either of these measures.
Several limitations of the present study are worth noting. In both the initial training and validity samples, some of the data included multiple timepoints of assessment. When this occurred, only the most recent assessment was used in the crosswalk development. As a result, changes in lower extremity functioning between earlier and later timepoints of assessment may not be fully captured in the current crosswalk. To address this concern, we evaluated the crosswalk's responsiveness to change from baseline to most recent assessment, which demonstrated preserved sensitivity to change over time. Another limitation pertains to the generalizability of the crosswalk. The validity sample included limited representation at higher levels of disability (PDDS > 4), which restricted score variability. Prior research indicates that that the PDDS is not interchangeable with clinician-rated disability measures, such as the EDSS, in individuals with mild-to-moderate MS, where PDDS-EDSS agreement tends to be weaker. 24 This is particularly relevant given that the present training sample was predominantly composed of individuals with lower levels of disability. Therefore, future validation in samples with broader disability severity is needed to evaluate crosswalk performance across the full PDDS range. Additionally, because the validity sample differed from the initial training cohort on multiple parameters (e.g., age, gender, race), conclusions regarding validity should be considered preliminary, and further validation in independent samples beyond the present study is needed. Although the initial training sample included individuals with a wide range of lower extremity functioning, variability in overall disease progression (e.g., mild vs. moderate vs. severe MS) may not fully be represented as well as limited ethnoracial diversity in the current samples. Moreover, while the association of crosswalk was strong, discrepancies may arise for individuals with significant disability unrelated to lower-extremity functioning (e.g., visual loss, bladder or bowel dysfunction, upper limb impairment), and this should be considered when interpreting or applying the crosswalk. Measurement validation is a continuous process, as such future research may examine the crosswalk's performance in more clinically distinct subgroups, including those with more advanced disease or progressive presentations of MS.
The value of crosswalks for facilitating tracking of PROs in individuals with MS has been well established, particularly for factors such as fatigue and health-related quality of life.22,23 However, despite the substantial impact of MS on mobility, no prior study to our knowledge has developed a crosswalk specifically targeting measurement of lower extremity functioning. A key strength of the present study is the linking of two widely used PROs assessing disability and mobility. Furthermore, the crosswalk was derived using a large and heterogenous training sample, including individuals with MS who varied in age, lower motor functioning severity, and employment status—enhancing the generalizability of the findings and utility of the derived crosswalk.
Overall, the present study provides initial evidence for a novel and clinically relevant approach to tracking MS-related mobility changes by establishing a crosswalk between two highly cited PROs of lower extremity functioning. By enabling score conversions between the PDDS and the Neuro-QoL LEF-SF, this crosswalk can enhance continuity and comparability across studies and clinical contexts that use different assessments of disability and lower motor functioning. Future research may further evaluate its applicability across tracking of clinical course in other neurological conditions impacting mobility, like Parkinson's disease, to increase its utility and generalizability.
Supplemental Material
sj-docx-1-mso-10.1177_20552173261427107 - Supplemental material for Measurement of functional mobility in multiple sclerosis: A crosswalk between the patient-determined disease steps and the Neuro-QoL Lower Extremity Function—Short Form
Supplemental material, sj-docx-1-mso-10.1177_20552173261427107 for Measurement of functional mobility in multiple sclerosis: A crosswalk between the patient-determined disease steps and the Neuro-QoL Lower Extremity Function—Short Form by Alexa S Gonzalez, Joshua M Garcia, Eric Engebretson and Luis D Medina in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgments
This paper is dedicated to the memory of Brooke Valdez. Her dedication, expertise, and insights greatly enriched this work, and we honor her lasting contributions. The authors would like to thank the patients, staff, and clinicians at the Rocky Mountain Multiple Sclerosis Center.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The present study was funded by the National Institute on Aging (NIA; UH2/UH3AG083264, U19AG078109). This work was also supported by the Rocky Mountain Multiple Sclerosis Center.
Supplemental material
Supplemental material for this article is available online.
