Abstract
Fatigue in multiple sclerosis is a key symptom associated with work-related problems and poor quality of life outcomes. The five-item Modified Fatigue Impact Scale is a brief self-assessment tool for measuring the impact of fatigue on cognitive, physical and psychosocial function. A non-interventional, cross-sectional study was conducted to assess dimensionality and item characteristics of the five-item Modified Fatigue Impact Scale in multiple sclerosis. A total of 302 subjects were studied. Mokken analysis found the five-item Modified Fatigue Impact Scale is a strong one-dimensional scale (overall scalability index H = 0.67) with high reliability (Cronbach’s alpha = 0.90). The confirmatory factor analysis model confirmed the one-dimensional structure (comparative fit index = 1.0, root-mean-square error of approximation = 0.035). Samejima’s model fitted well as an unconstrained model with different item difficulties. The five-item Modified Fatigue Impact Scale shows appropriate psychometric characteristics and may constitute a valuable and easy-to-implement addition to measure the impact of fatigue in clinical practice.
Introduction
Fatigue is one of the most frequent and disabling symptom reported by people with multiple sclerosis (pwMS).1,2 Several factors may contribute to its development, requiring a careful workup to search for all possible underlying causes. 1 Fatigue was a significant predictor of a sustained Expanded Disability Status Scale (EDSS) worsening in a longitudinal follow-up of a cohort of pwMS from the New York State Multiple Sclerosis Consortium (hazard ratio (HR) 1.4, 95% confidence interval (CI) 1.2–1.7). 3 High levels of fatigue were associated with impairment in functional mobility, depression, poor physical and mental quality of life, and work-related difficulties.4,5
Fatigue is usually assessed using self-report questionnaires. 1 The Multiple Sclerosis Council for Clinical Practice Guidelines recommended the 21-item Modified Fatigue Impact Scale (MFIS) for use in clinical practice and research. 1 Later on, a shortened version of the questionnaire was developed, the five-item MFIS (MFIS-5). 6 The MFIS-5 includes five of the 21 items from the original scale, selected from those showing higher correlation with the total MFIS score: two items from the physical subscale (#10 and #17), two items from the cognitive subscale (#1 and #19) and one item from the psychosocial subscale (#9). 6 Most previous psychometric research has been done with the 21-item MFIS.7,8 In the few studies conducted so far, the MFIS-5 has shown good psychometric properties regarding internal reliability, test-retest reliability and sensitivity to change.9,10
There is often not enough time during neurology visits to address all of the patient’s concerns and needs. In this context, the MFIS-5 takes only two minutes to administer and may constitute a useful tool to quantify fatigue symptoms in clinical practice. However, no previous study has reported the whole spectrum of its psychometric properties. The aim of this study was to assess dimensionality and item characteristics of the MFIS-5 in the management of multiple sclerosis (MS).
Methods
A non-interventional, cross-sectional study was conducted in 18 MS units throughout Spain (Infoseek-MS Study). Key eligibility criteria included an age of at least 18 years, a diagnosis of MS according to the 2010 revised McDonald criteria and an EDSS score of 0–6.0. Written informed consent was obtained from all subjects. The study was approved by the institutional review board of the Hospital Universitario Gregorio Marañón (Madrid, Spain).
The MFIS-5 scores each item on a five-point Likert scale from 0 (never) to four (almost always). The total score ranges from 0–20, with a higher score indicating more severe fatigue. 6 The validated Spanish version of the instrument was used. 11
Statistical analysis
A non-parametric item response theory (IRT) procedure, Mokken analysis and confirmatory factor analysis were performed to assess the underlying dimensions of the MFIS-5. All items were required to have a scalability coefficient (Hi) ≥ 0.3 and the overall scale a scalability coefficient (H) ≥ 0.3. Mokken analysis is a non-parametric IRT model closely related to the parametric Rasch approach but presenting less restrictive assumptions. It allows the rank order of persons and measures the homogeneity of items and scale with respect to a latent trait.12,13 The scalability coefficient is an index of homogeneity; it ranges from 0–1 with higher values indicating better discrimination among values of the underlying latent trait. As a rule of thumb, scalability coefficient Hi values <0.3 are considered unacceptable for an item to be included in the scale, and hence by implication the scalability coefficient H must also be ≥ 0.3. A scale is considered weak when 0.3 ≤ H < 0.4, medium when 0.4 ≤ H < 0.5 and strong when H ≥ 0.5. 14
Samejima’s graded response model (GRM) was conducted to obtain estimates of the relationship between the latent construct and item characteristics. Finally, a confirmatory factor analysis (CFA) model was fitted to corroborate the dimensionality of the MFIS-5 scale. We considered a comparative fit index (CFI) value >0.95 as an acceptable model fit and root-mean-square error of approximation (RMSEA) values < 0.08 and < 0.05 as adequate and good model fit, respectively.
Results
A total of 302 pwMS were studied. The main socio-demographic and clinical characteristics of the sample are shown in Table 1. The minimum possible score of zero (floor effect) and the maximum possible score of 20 (ceiling effect) for MFIS-5 were achieved by less than 15% of the patients (5.3% and 2.0%, respectively). The median MFIS-5 score was 10 with an interquartile range (IQR) wide enough to cover an appropriate range of fatigue scores (IQR 2–18).
Demographic and clinical characteristics of the sample.
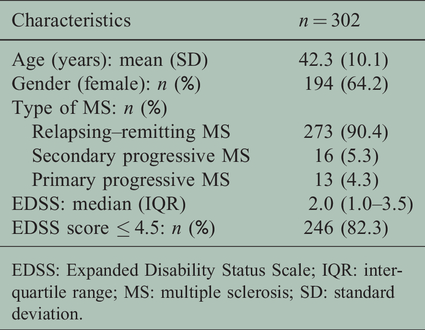
EDSS: Expanded Disability Status Scale; IQR: interquartile range; MS: multiple sclerosis; SD: standard deviation.
Internal reliability
The MFIS-5 showed high reliability (Cronbach’s alpha = 0.90, bootstrap 95% CI = 0.87–0.91).
Non-parametric (Mokken) IRT
The MFIS-5 is a strong one-dimensional scale with an overall scalability index H = 0.67, and all items showing scalability coefficients Hi > 0.50. The monotone homogeneity model fitted the data well, but not the double monotonicity model. Table 2 shows the item endorsement frequencies and Mokken scalability coefficients.
Endorsement frequencies, Mokken scalability, information and confirmatory factor analysis (CFA) loadings for the five-item Modified Fatigue Impact Scale (MFIS-5).

Hi: scalability coefficient; SD: standard deviation.
MFIS-5 response format, 0: never, 1: rarely, 2: sometimes, 3: often, 4: almost always.
Item characteristics: GRM
We compared two Samejima’s GRM, the unconstrained and the constrained model. The constrained model assumes that all items have equal discriminative power, whereas the unconstrained model assumes that items have a unique discriminative power. Our results support an unconstrained model for the MFIS-5 with different item difficulties (likelihood ratio test between both models = 104.26 on 4 df, p < 0.001). The item characteristic curve showed all items presented appropriate shape and difficulty parameters. Items #2 (“limited to do things away from home”), #3 (“had trouble maintaining physical effort for long periods”) and #4 (“less able to complete tasks requiring physical effort”) represent 80% of the total information or accuracy with which the latent trait of fatigue is estimated (Table 2).
Factor structure
The CFA model confirmed the one-dimensional structure suggested by Mokken analysis (CFI = 1.0, RMSEA = 0.035). All freely estimated unstandardized parameters were statistically significant (p < 0.001). All standardized parameters (effect sizes) showed salient loadings (>0.60) (Table 2).
Discussion
Patient‐reported outcomes are designed to capture patient perceptions of their health condition. Unfortunately, their application in MS is still inconsistent. 15 A number of validated questionnaires for assessing fatigue have been used in MS, such as the Chalder Fatigue Questionnaire, Fatigue Assessment Scale, Fatigue Impact Scale, Modified Fatigue Impact Scale, Fatigue Severity Scale, Fatigue Scale for Motor and Cognitive Functions, Fatigue Descriptive Scale, Checklist Individual Strength and Patient-Reported Outcomes Measurement Information System (PROMIS) Fatigue-Short Form. 1 The diversity of assessment tools available highlights the fact that there is still no consensus on the underlying mechanisms of fatigue.1,16,17 In a recent study by Pust et al., none of the calculated CFA and exploratory structural equation modelling could verify the factor structures of the Chalder Fatigue Questionnaire, Modified Fatigue Impact Scale and Fatigue Scale for Motor and Cognitive Functions. 16 Hobart et al. conducted a qualitative evaluation of the 40-item Fatigue Impact Scale (FIS) using the expert opinion of 30 MS health professionals and researchers that did not support its content or face validity. 17 Nevertheless, standard quantitative psychometric evaluations involving 333 pwMS found that FIS subscales were reliable and valid. Therefore, the authors state the need to ensure that patient-reported instruments are both psychometrically robust and qualitatively valid. 17
Our study found that the MFIS-5 is a strong one-dimensional scale that shows good psychometric properties regarding internal consistency and item discrimination and information. Internal consistency was found to be even higher in our study than has been previously reported by D’Souza (Cronbach’s alpha = 0.90 and 0.80, respectively). 9 The MFIS has limitations when interpreting the scores, particularly when trying to understand what a change in score might mean. 1 Smith et al. recently found that MFIS-5 is also a reliable instrument to assess changes in fatigue over time. 10 Four-point differences on the MFIS-5 represent the amount of change required to be statistically significant at the 0.70 confidence level. 10
This study has one main limitation. The study population included a sample of clinically stable people with low physical disability, mainly with a diagnosis of relapsing–remitting MS. The results may thus not be generalizable to less stable subjects and other clinical subtypes. Despite this limitation, the sample of 302 subjects was managed in 18 different MS units on a national level, which allows results to be generalized to community practice.
Conclusions
Fatigue is the most frequent symptom in MS and is reported to affect between 50–80% of subjects. Understanding fatigue in MS may be crucial for carrying out specific intervention strategies. Despite the shortcomings of available self-rated fatigue questionnaires, the MFIS-5 is a quick, easy-to-implement and reliable instrument to assess the impact of fatigue in clinical practice.
Footnotes
Acknowledgments
The authors wish to thank the patients for making the Infoseek-MS study possible. The Infoseek-MS Study Group includes: Eduardo Agüera (Hospital Universitario Reina Sofia, Córdoba), Yolanda Aladro-Benito (Hospital Universitario de Getafe, Getafe), Amaya Álvarez de Arcaya (Hospital Universitario Araba, Vitoria), Rafael Arroyo (Hospital Quirónsalud, Madrid), Antonio Belenguer (Hospital General Universitario de Castellón, Castellón), Laura Borrega (Hospital Universitario Fundación Alcorcón, Alcorcón), Antonio Candeliere-Merlicco (Hospital Rafael Méndez, Lorca), Pilar Casquero (Hospital General Mateu Orfila, Maó), Pedro Serrano-Castro (Hospital Regional Universitario Carlos Haya, Málaga), María del Carmen Fernández-Moreno (Hospital Universitario Nuestra Señora de Valme, Sevilla), Antonio García-Merino (Hospital Universitario Puerta de Hierro, Madrid), Nicolás Herrera-Varo (Hospital Universitario de Burgos, Burgos), Cristina Iñíguez (Hospital Clínico Universitario Lozano Blesa, Zaragoza), Lamberto Landete (Hospital Universitario Dr Peset, Valencia), María L Martínez-Ginés (Hospital Universitario Gregorio Marañon, Madrid), Virginia Meca-Lallana (Hospital Universitario La Princesa, Madrid), Amelia Mendoza-Rodríguez (Complejo Asistencial de Segovia, Segovia) and Cristina Ramo-Tello (Hospital Universitario Germans Trias i Pujol, Badalona).
The abstract of this article was presented at the 34th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) as a poster presentation with interim findings (Berlin, Germany; 10–12 October 2018).
Conflict of interests
MB, ES and JM are employees of Roche Farma Spain. None of the other authors report any conflict of interest.
Funding
The author(s) disclosed receipt of the following financialsupport for the research, authorship, and/or publication of this article: This study was funded by the Medical Department of Roche Farma Spain.
