Abstract
Background
Patient-reported outcomes (PROs) are widely measured in multiple sclerosis (MS) studies. However, the quality of instrument development processes varies, raising concerns about the meaningfulness of associated data.
Objectives
To review the development of selected PROs commonly used in MS studies, including definitions of the concepts measured, use of conceptual frameworks, and degree of input from people living with MS (PlwMS). To gain insights and recommendations from PlwMS on their experience with these PROs.
Methods
We assessed 6 PROs (FSIQ-RMS, modified-FIS, MSQoL-54, Leeds 8-item MSQoL, MSIS-29 and EQ-5D) for alignment with regulatory and scientific requirements on PRO structure/development. PlwMS evaluated the degree to which the PROs reflect disease aspects they perceive important.
Results
Definitions, clarifications and conceptualisations of the measurement variables were often lacking. PlwMS were variably involved in PRO development. Ethnic diversity was rarely documented. PlwMS identified individualisation, ease of understanding, time burden, and mode of administration as factors affecting PRO usability.
Conclusions
To date, the PRO development process has consistently lacked clear definitions of concepts of interest, use of conceptual frameworks and patient involvement, thereby compromising the validity of data they generate. PRO instrument development must be conducted more robustly to maximise the value of pivotal clinical trials.
Introduction
People living with Multiple sclerosis (PlwMS) have a range of symptoms impacting their life quality.1,2 Some can be measured objectively, others require assessment and quantification of patient's perceptions. 3 Patient-reported outcome (PRO) measures seek to provide these data. 4
PROs play key roles in MS studies. 5 They quantify MS impacts, monitor these longitudinally, determine therapeutic and cost effectiveness, and interpret the clinical meaningfulness of changes in objective measures. Decisions based on PRO interpretations influence the lives of PlwMS, health care utilisation, and public expenditure. It is hard to construct an argument to support compromising PRO quality.
Guidance for PRO development and selection for clinical trials is evolving. Recent guidance highlight the importance of conceptual frameworks, patient involvement, and advanced psychometric methods.4,6,7 Conceptual frameworks are the hypothesised relationships between a measurement variable (clinical concept), its components, their sub-components, proposed scale items, and scores generated. They are not just important, they underpin PRO validity. 8 Our experience is that these recommendations and advances are not yet fully appreciated by MS clinical trialists.
The PROs that matter to PlwMS initiative (PROMPT-MS) aims to highlight the need for a new generation of robustly designed PROs. Specifically, to emphasise the need for clear and specific PRO concept definitions, promote PRO selection strategies, highlight alignment with regulatory and best available science guidance and, most importantly, what really matters to PlwMS.
This study had two aims. First, to review the development of selected commonly used PROs, with respect to PlwMS's involvement, concept definitions, and conceptual frameworks used. Second, to gain insights from PlwMS in relation to their experiences of completing PROs, how effectively they perceive these instruments capture factors relevant to PlwMS, and the strengths and weaknesses of selected PRO instruments.
Methods
Evaluation of PRO development
A published literature review 9 combined with guidance from the PROMPT-MS Steering Committee ( Supplementary Table 1 ) identified 6 PROs for evaluation that were considered to be representative of available PROs. Two measured fatigue: the Fatigue Symptoms and Impacts Questionnaire – Relapsing MS (FSIQ-RMS) 10 and 21-item modified Fatigue Impact Scale (mFIS).11,12 Three measured ‘quality of life’: 54-item MS QoL scale (MSQoL-54); 13 8-item Leeds MS QoL measure (LMSQoL), 14 and EuroQol EQ-5D. 15 The final, 29-item MS Impact Scale (MSIS-29), 16 measured the physical and psychological impact of MS. The selected instruments are not intended to constitute an exhaustive list of PROs. We chose a small set of relevant instruments designed to assess a range of important patient-centric symptoms that are challenging domains to define accurately. The selections of the mFIS and FSIQ-RMS were to contrast older and newer PROs (i.e. instruments developed before and after the publication of regulatory guidelines).
We assessed the PRO development process from the original PRO development papers, extracting information on instrument purpose, concept and domain definitions, conceptual frameworks, patient input, and instrument items and response formats. We developed a profile for each instrument (Figure 1).
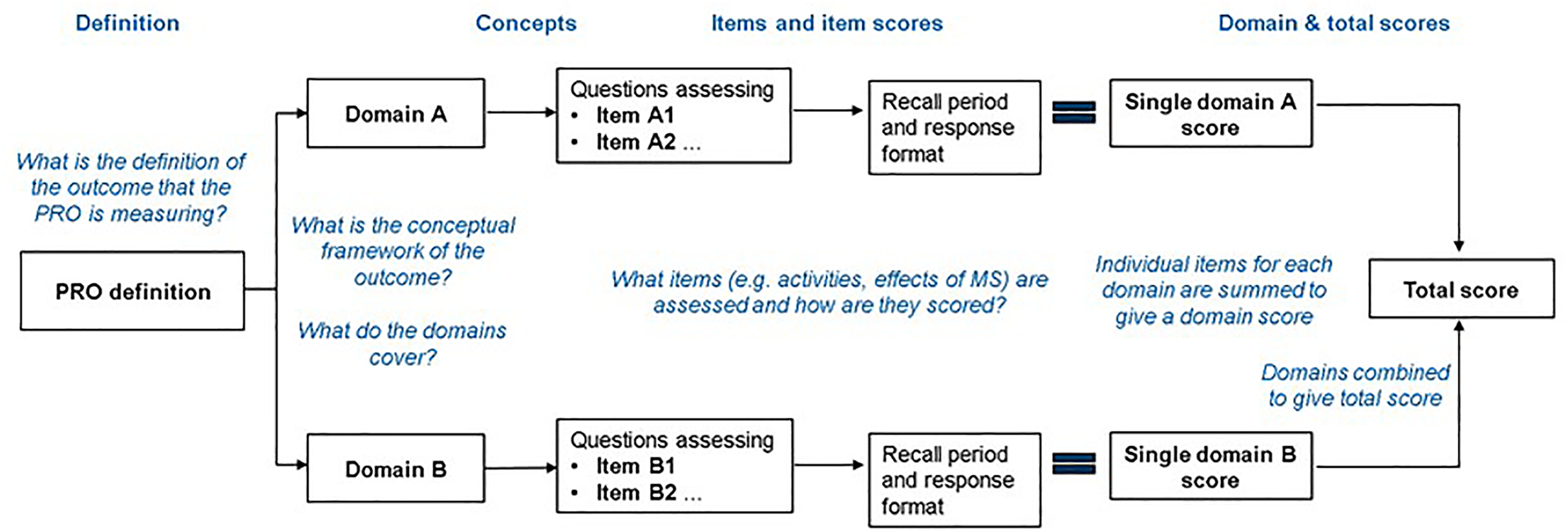
Flow-chart template for profiling the key elements of PRO instruments.* This standardized approach to profiling PROs allowed conclusions to be drawn about the extent to which scores generated by each instrument accurately reflect what, by definition, each PRO was designed to assess. Note: *Populated flow-charts for each of the six PROs are available in Supplementary Material
PROs: qualitative insights from PlwMS
PlwMS (n = 25) were interviewed to gain insights on their prior experience of using PROs, perceived appropriateness of PRO item response options, opinions of suitable recall periods, the degree to which scores accurately represent concepts being measured, and views on alternative approaches to assessing the impact of MS. These insights complemented the profiling exercise described above. The PlwMS who were interviewed were all actively involved and vocal members of the MS community, and many shared their experiences on social media. Table 1 shows their characteristics. Most were females (72%) and had relapsing-remitting MS (92%). A range of nationalities, ethnicities, ages, disease durations, and relationship statuses were represented. Both interviewer and interviewees had MS, to help interviewees feel at ease, to ensure that interviewers were subject matter experts, and to embed the patient perspective throughout the research process. The interviewer was trained under the European Patients’ Academy on Therapeutic Innovation (EUPATI) initiative (
Characteristics of study participants.

Interviewees also completed a post-interview survey on the strengths and weaknesses of the six PROs. The purpose of the post-interview survey was to capture additional information not gathered during the interviews. We recognised the value of a period of reflection following the interviews. The survey enabled participants to have a second opportunity to comment on specific issues after a period of reflection. Specifically, the post-interview survey related to patients’ perceptions of PRO strengths and weaknesses, and whether they felt the tools effectively assessed the impact of MS on aspects of their life quality, whether they covered aspects that are relevant to people living with MS, whether they would create a significant time burden, and whether they would be manageable to complete. The interview schedule and questionnaire are available in the
The aims of our research necessitated a mixed-methods approach. The quantitative investigation of the PROs allowed for a consistent assessment to be conducted across the PROs, while the qualitative work provided important insights from the patient perspective. Combining these methods enabled a more comprehensive examination of the subject than using either method in isolation. 19
Results
Evaluation of PRO development
Table 2 shows data extracted for each PRO.
Supplementary Figure 1
shows each instrument's standardised flow chart. A detailed account of each instrument's development is available in the
Data extraction summary by PRO instrument.

In either the development publication or the instrument itself.
Questions/items have been condensed for brevity; they are not intended to be comprehensive or verbatim.
LMSQoL is a copyright of the University of Leeds, and is available from the University of Leeds fast-licence platform: https://licensing.leeds.ac.uk/product/lms-qol-leeds-multiple-sclerosis-quality-of-life-scale.
MSIS-29 is a copyright of the University of Plymouth and is used under permission/licence.
© EuroQol Research Foundation. EQ-5D™ is a trade mark of the EuroQol Research Foundation.
FSIQ-RMS © 2017 Mapi Research Trust.
FSIQ-RMS: Fatigue Symptoms and Impacts Questionnaire – Relapsing Multiple Sclerosis; HRQoL: health-related QoL; LMSQoL: Leeds MS QoL instrument; mFIS: modified Fatigue Impact Scale; MS: multiple sclerosis; MSIS-29: 29-item MS Impact Scale; PlwMS: people living with MS; PRO: patient-reported outcome; QoL: quality of life; RMS: relapsing multiple sclerosis.
Fatigue PROs
mFIS
The mFIS was developed in 1997 20 from the 40-item Fatigue Impact Scale (FIS). 11 The FIS developers aimed to develop a measure of PlwMS's perceptions of functional limitations attributable to fatigue. FIS items were selected from existing fatigue scales and n = 30 qualitative interviews with PlwMS. No explicit conceptual framework underpins the FIS. It was constructed to have 3 functioning subscales (physical k = 10 items; cognitive k = 10 items; psychosocial k = 20 items), reflecting the interview responses and dimensions from other health status and quality of life measures. 11 The 21-item mFIS was constructed by removing one item from the physical and 18 items from the psychosocial functioning dimensions. Neither the FIS nor mFIS developers define fatigue.
All FIS/mFIS items have five response categories from 0 (Never) to 4 (Almost always). The recall period is 1 month. mFIS/FIS items are summed to generate four scores: three subscale scores and a total score.
FSIQ-RMS
The FSIQ-RMS, developed in 2019, 10 aims to assess fatigue symptoms and their impacts on people with RMS. It has 20-items (7 symptoms, 13 impacts). The developers do not define explicitly what they mean by fatigue.
The FSIQ-RMS was developed in stages. A literature review led to preliminary conceptual frameworks for fatigue symptoms and impacts in RMS. Details are not given. These informed interview guides for n = 17 concept elicitation interviews with PlwMS, resulting in the generation of 84 fatigue-related symptom and impact concepts. Concepts reported by
Rasch analysis of the n = 20 completions of the preliminary 30-item scale led to one excluded item being re-introduced (k = 23; k = 9 symptoms; k = 14 impacts). This version was administered to PlwMS (n = 164) and controls (n = 74). Response data were analysed (floor/ceiling effects, item-item correlations, exploratory factor, Rasch analyses). The symptoms items were reduced from 9 to 7 due to redundancy. One impacts item was removed because of high ceiling effects. The exploratory factor analysis implied the remaining 13 impacts items exist in three 5-item subdomains (physical, cognitive and emotional, and coping).
FSIQ-RMS generates four scores: 1 symptom and 3 impacts subdomain scores. 10
Life quality PROs
LMSQoL
The LMSQoL, developed in 2001, 14 is a patient-completed MS-specific quality of life measure. It has 8 items, each has four response categories (0 = Not at all to 3 = Most of the time). 21 The recall period is the past month. Item scores are summed to generate a total score. There was no a priori explicit definition of quality of life nor conceptual framework for measurement. It measures ‘a variable related to well-being’.
Instrument development had multiple stages, including two focus groups with PlwMS (n = 30). The first identified ‘the main areas of concern’ and the second generated 25 potential items for instrument inclusion. 14 These were completed by n = 24 people. Analyses of response data (Cronbach's alpha and Rasch analyses) and consideration of item relevance led to 8 items being removed. The 17 remaining items had floor and ceiling effects, so 3 new items were identified and added to reflect the extremes of the QoL variable. The resultant 20-item scale was examined in two samples, one for test-retest reproducibility (n = 27) the other for construct validity (n = 43). Rasch analyses of the n = 43 completions identified 4 misfitting items, which were removed. The revised 16-item scale was administered to a stratified community sample. Rasch analysis of response data from n = 180 completions identified 8 misfitting items that were removed, leaving the final 8 item scale.
MSQoL-54
The MSQoL-54, developed in 1995, was created by adding 18 MS-specific items to the 36 generic items of RAND's 36-item health survey (SF-36). 22 The aim was to develop a measure of health-related quality of life (HRQoL) for MS combining the ability to compare across diseases and provide sensitive within-disease comparisons.
The SF-36 has 8 multi-item subscales (physical and social functioning, physical and emotional role limitations, general health perceptions, energy/vitality, emotional well-being, pain) and one single item assessing change in health over the last year. Of the 18 MS-specific items: 3 items are added to three existing SF-36 subscales (Pain, Energy, Social Functioning); 14 items are added as 4 new multi-item subscales (Health distress, cognitive and sexual functioning, QoL); one item assesses satisfaction with sex. 13
The 18 MS items were generated by a literature review and input from specialist MS healthcare providers (n = 3), covering aspects understood to be particularly relevant to PlwMS. Recall periods and item response formats vary between subscales. No explicit conceptual framework or PlwMS involvement guided MSQoL-54 development. Measurement performance testing was conducted in 179 PlwMS. 13
EQ-5D
The EQ-5D, first published in 1990, 23 was developed as a standardised non-disease-specific instrument for describing and valuing HRQoL. There are three-level and five level versions (EQ-5D-3L; EQ-5D-5L), differing only in the number of response categories of the five items (dimensions). Both versions have two parts. First, 5 items grading severity of problems with mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Second, a visual analogue scale (VAS) rating health state from 0 = worst to 100 = best imaginable. In the EQ-5D-3L, each item has 3 severity levels (no problems; some/moderate; extreme problems/unable to do); the 5-level version has 5 severity (none; slight, moderate, severe, unable/extreme).
The developers do not give definitions of health status or quality of life, and there is no explicit underpinning conceptual framework. Whilst the items were selected from a ‘detailed examination of the descriptive content of existing health status measures’, 23 and there was ‘agreement among scientists and clinicians’, specific detail is not documented. Details of patient involvement have not been published.
EQ-5Ds generate three scores: an unweighted health status profile across the five dimensions (e.g. 1 2 1 2 3), a weighted sum score (index) derived from the unweighted profile, and a VAS health state score.15,24,25 The recall period is ‘today’.
MS impact
MSIS-29
The MSIS-29 was developed in 1999 to measure the physical (20-items) and psychological (9-items) impact of MS. Two scores are generated, one for each domain. All items in MSIS-29v2 have 4 response categories. 26
There was no definition of MS impact reported. No a priori conceptual framework underpinned MSIS-29 development. Development involved input from multiple disciplines, expert opinions, literature review, and PlwMS. A pool of k = 141 items was generated from n = 30 semi-structured interviews with PlwMS. These were administered to a random sample of n = 1530 PlwMS. The k = 129 non-walking-related items were analysed using traditional psychometric methods and then reduced using statistical criteria to form the two subscales. 27 The item content of the scales drove the name of their measurement variables and these were refined accordingly. MSIS-29's measurement performance was determined from an independent sample of n = 1250 PlwMS, test-retest reproducibility in a subgroup of these and responsiveness in an independent sample of n = 55. The 12 walking-related items formed the MSWS-12. 28
Interview insights on PROs from PlwMS (n = 22)
Fatigue PROs
mFIS
PlwMS reported that the mFIS provided an accurate description of fatigue, the questions related to cognition and fatigue were relevant, and that the scale was clear. PlwMS commented that the mFIS does not assess the impact of fatigue on emotions or other aspects of everyday life. The scoring was described as confusing as questions are both positively and negatively phrased. Furthermore, PlwMS suggested a 4-week recall period is too short to measure the impact of fatigue on everyday life (Table 3). Respondents did not suggest alternative timeframes.
PlwMS feedback of PROs.

FSIQ-RMS: Fatigue Symptoms and Impacts Questionnaire – Relapsing Multiple Sclerosis; HRQoL: health-related QoL; LMSQoL: Leeds MS QoL instrument; mFIS: modified Fatigue Impact Scale; MS: multiple sclerosis; MSIS-29: 29-item MS Impact Scale; PlwMS: people living with MS; PRO: patient reported outcome; QoL: quality of life.
FSIQ-RMS
PlwMS felt the FSIQ-RMS covered a wide range of domains. The assessment of symptoms and functional impacts was welcomed. Digital administration was deemed convenient. Perceived weaknesses included the limited timeframe (24 h for symptoms, 7 days for impacts), the questionnaire was considered too long (20 items), and psychosocial aspects were not covered comprehensively (Table 3).
Life quality PROs
LMSQoL
PlwMS said the LMSQoL contained thought-provoking questions, reflected the connection between mental health and MS, and was a good instrument to track changes in symptoms. However, interviewees said the LMSQoL did not adequately reflect the relationship between physical and emotional symptoms or the relationship between fatigue, and cognitive and sexual functioning (Table 3).
MSQoL-54
PlwMS said the MSQoL-54 questions provide a holistic view of their MS experience, covered most of the emotional aspects of MS, provided a variety of response formats, and considered the fluctuating nature of their symptoms. However, PlwMS felt the weaknesses were the PRO's length (54 items), focus on disability rather than ability, and limited range of pain questions. They recommended the wording of sex-related questions could be more considered (less direct) (Table 3).
EQ-5D
Interviewees appreciated the EQ-5D was short, simple, quick to complete, and covered relevant topics pertaining to general health. However, they reported that the questions were not detailed enough, overly simple or too generic (while recognising that this is a general non-MS specific measure). They found the five-digit summary score hard to relate to
MS impact PRO
MSIS-29
PlwMS said the MSIS-29 included relatable question wording, covered a diverse range of topics, and explored both physical and psychological impacts of MS. However, interviewees highlighted the unequal focus on the two domains, with fewer items dedicated to psychological aspects. Respondents also reported an insufficient focus on pain.
Table 4 summarises key interview insights. Various themes emerged. Patients reported that there is no one-size fits all PRO, and that it would be helpful for instruments to be tailored to specific relevant characteristics like disease type/stage or cultural background. It was also recognised that different people prefer different modes of instrument completion (e.g. paper and pencil or digital) and that these preferences should be accounted for during administration. With regards to fatigue, respondents stated that instruments often do not adequately capture its impact, especially the fluctuating nature of this symptom. The interviewees also expressed that it is important for them to fully understand the purpose of the PRO instrument and how it will support the delivery of optimal care. Other key themes to emerge included the need for questions to be simple, carefully worded, and relevant, for response scales to be meaningfully related to the symptom in question, and that recall periods account for potential memory impairment.
Summary of key insights from PlwMS on PROs.

MS: multiple sclerosis; PlwMS: people living with MS; PRO: patient-reported outcome.
Survey insights on PROs from PlwMS
Figure 2 shows the results of the post-interview interviewee survey on topics related to the six PROs. For the fatigue-specific PROs, participants generally agreed that the two PROs effectively assessed the impact of MS on their fatigue, covered aspects relevant to PlwMS, weren’t significantly time-demanding, and would be manageable to complete. In relation to the other PROs, a number of respondents said the LMSQoL (n = 7, 39%) and EQ-5D (n = 8, 44%) could not effectively assess the impact of MS on their life quality, or that they covered aspects relevant to them as a PlwMS. Most respondents said all PROs would be manageable to complete. Five respondents (28%) said the MSQoL-54 would create a significant time burden to complete.

PlwMS post-interview survey on fatigue (A) and QoL or physical/psychological (B) PROs using a 5-point Likert scale. Note: Data represents responses from 18/22 interviewees. FSIQ-RMS: Fatigue Symptoms and Impacts Questionnaire – Relapsing Multiple Sclerosis; LMSQoL: Leeds MS QoL instrument; mFIS: modified Fatigue Impact Scale; MS: multiple sclerosis; MSQoL-54: 54-item MS QoL; MSIS-29: 29-item MS Impact Scale; PlwMS: people living with MS; PRO: patient-reported outcome; QoL: quality of life.
Discussion
When PROs are used in MS studies we assume the measured effect adequately approximates the actual effect. More specifically, we assume the PROs provide accurate and precise measurements of the clinical variables they purport to measure, and changes in PRO scores are valid indications of what happens in practice. For example, if fatigue is measured with a PRO in a treatment trial, and data show fatigue is not improved, we assume this reflects what happens in practice. These assumptions are requirements for high stakes MS studies (i.e. pivotal trials of MS disease modifying therapies that ultimately have implications for the care of individuals and the expenditure of public funds). It is noteworthy that suboptimal measurement generates type 2 errors. 7
This study concerns one of these requirements: how we can begin to tell if PROs generate valid measurements. We examined the development of selected PROs rather than their psychometric (statistical) performance, because PRO validity is primarily determined by its structure and item content more than its psychometric performance. The limitations of statistical validity tests were highlighted decades ago, 29 and their potential to mislead demonstrated empirically more recently. 8
We examined selected PROs for definitions of the variables they measured and their conceptual underpinnings. No development paper of any selected PROs reviewed provided a thorough definition of the variables they sought to measure. Most gave no definition. Some determined the variable measured post hoc from the items left by statistically-driven reductions of larger item sets, or from correlations with other PROs. We found similar omissions of conceptual frameworks. Whilst all development papers provided, in differing forms, information on the structure of the final instrument post hoc, none provided conceptual frameworks a priori for the variable for measurement that enabled us to determine the validity of the final instrument. Most notable was the paucity of documented information about these essential aspects of scale development; without definitions and conceptualisation the extent to which scores reflect concepts is unknown.
Approaches to selected PRO development varied, yet all provided scores purporting to be valid measurements of clinical variables. Different criteria were applied. Many appeared arbitrary. Interestingly, a number of the PROs examined generated large amounts of information (concepts and items) from qualitative research, the majority of which was ultimately discarded. On no occasion was this rich information structured into an explicit framework to aid understanding of the variable of interest
We recognise our selection of PROs was limited and open to criticism. However, the issues we highlight are widespread in health measurement. For example, the widely used Fatigue Severity Scale (FSS) suffers from exactly the same weaknesses as the other fatigue PROs we have examined. The original development paper of 1989 does not include any definition of fatigue. 30 There is no conceptual underpinning. The 9 items were selected from a pool of 28 items (that was neither provided, described, nor referenced) based on ‘a factor analysis, item analysis, and theoretic considerations’. As such, based on the original development paper for this instrument, we are left uncertain as to the extent to which FSS scores accurately reflect fatigue in MS.
It is curious that PROs have been developed to measure complex clinic variables, like fatigue and quality of life, without more attention to variable definitions and conceptualisations. There are a number of possible explanations. These include the absence of regulatory requirement until recently, limited guidance on defining and conceptualising variables, and scale development strategies dominated by quantitative methods with scant attention to qualitative techniques. Certainly, the scale development zeitgeist previously was a triad of generation of an item pool, scale formation by statistically driven item reduction, and statistical evaluation of the scales produced. It is hardly surprising that a set of items selected because they are statistically cohesive, are statistically cohesive when examined subsequently.
We also examined the roles of PlwMS in selected PRO development and gained PlwMS's feedback on the PROs. PlwMS's involvement in PRO development was varied from none to quite extensive. For some PROs it was unclear. Their feedback also varied. There were positive and negative comments for each PRO. Most PROs were considered to lack relevant components. We think the absence of variable definitions and conceptual frameworks makes it very difficult for PlwMS to critique PROs meaning. They need this information to set a frame of reference for their input. In addition, there is little guidance as to exactly how PlwMS, or other conditions, should best be involved in PRO development and evaluation, and how to maximise quality control of this process. We identified several key themes from the interviews that may serve to optimise the development of future PRO instruments. Some of these insights may be more challenging than others to incorporate, such as tailoring instruments to specific patient characteristics or using a recall period that is acceptable to everyone. However, many of these important interview insights are relatively straightforward and can be easily accommodated, such as providing a clear explanation of the PROs purpose, offering a choice of administration formats, using clear and simple question wording, and making sure that item wording reflects constructs that are meaningful and relevant to those who are responding. Implementing these insights to inform instrument development will ensure that future PROs are fit for purpose and acceptable to patients.
Findings from our interviews identified geographical and cultural background as important aspects to consider when developing PRO instruments. It is therefore critical that during the development of PRO instruments, PlwMS are not only heavily involved in the process, but that those involved represent a diverse range of characteristics, ensuring that the resultant instruments are fit for purpose across the widely diverse population of PlwMS. If not, then there is a risk that these tools may have limited or imperfect generalizability to under-represented minority groups. Interestingly, only two of the development papers for the six PRO instruments assessed in this study provided details on the ethnicity of the development sample. For the FSIQ-RMS the percentage of non-Caucasian/White participants ranged from 15% to 60% across the three content development stages. 10 For the MSIS-29, the development sample was entirely white. 16
Our findings reiterate the requirement for PRO developers to provide explicit definitions and detailed conceptualisation of the variables they seek to measure, as well as the importance of patient involvement. Whilst these are recognised requirements,4,31,32 it appears that such guidance has generally not been followed to date. Future instrument development aligned to best practiced principles will result in fit-for-purpose PROs, enabling strategic PRO selection to underpin clinical-decisions in the care of PlwMS. Until then the validity of PRO data in MS remains questionable.
One reviewer of this study raised three very relevant and important questions. How should we interpret studies using these instruments? What guidance is there for instrument selection? How do we optimize existing PROs while waiting for better instruments to be developed? Each question warrants a detailed answer beyond the scope of this manuscript and are being addressed actively by us in other studies. In short, it is difficult to quantify accurately the impact of poor measurement especially in the absence of clear definitions and conceptualisations of the clinical variables they seek to measure. Without that information the extent to which an instrument's score reflects the construct of interest, its validity, is unclear. Detailed head-to-head comparisons of PROs, using qualitative and sophisticated quantitative methods are required, in specific contexts of use, to enable clear understandings of the trade-offs associated with competing PROs. Such detailed evaluations enable PRO strengths and limitations to be identified. Importantly, they also act as a platform for PRO modification to maximise their current performance as measures. PRO measurement strategies must be well thought-out, critically appraised, and the associated science conducted robustly.
Supplemental Material
sj-docx-1-mso-10.1177_20552173221105642 - Supplemental material for Patient-reported outcome measures in MS: Do development processes and patient involvement support valid quantification of clinically important variables?
Supplemental material, sj-docx-1-mso-10.1177_20552173221105642 for Patient-reported outcome measures in MS: Do development processes and patient involvement support valid quantification of clinically important variables? by Trishna Bharadia, Jo Vandercappellen, Tanuja Chitnis, Piet Eelen, Birgit Bauer, Giampaolo Brichetto, Andrew Lloyd, Hollie Schmidt, Miriam King, Jennifer Fitzgerald, Thomas Hach and Jeremy Hobart in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
The authors would like to thank the people living with MS, who consented to be interviewed and completed our survey, for contributing their views and experiences to the PROMPT-MS initiative. We would also like to thank Nanette Rombach-Mullan and David McMinn, PhD (both of Novartis CONEXTS, Ireland) for providing medical writing support, which was funded by Novartis Pharma AG.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Birgit Bauer has received compensation for consulting from Novartis, Roche, Merck, Teva and Sanofi. Disclosures do not show a conflict with the work being presented. Trishna Bharadia in the last 3 years has received compensation for serving as a consultant, writer and/or speaker for or has received honoraria from: 67Health, Abbvie, Actelion (Janssen), Admedicum, Blue Latitude Health (Fishawack), Corex Logistics, Curatio, DHL Life Sciences, Envision Pharma, Faculty of Pharmaceutical Medicine, Future Medicine, Gilead Sciences, Greenphire, ISMPP, Kayentis, Medipace, Merck KgA, NIHR, Norgine, Novartis, NovoNordisk, Parexel, Pfizer, Prime Global Roche, Savvy Cooperative, Synchrogenix (Certara), talkHealth, Teva, University College London, University of Central Lancashire, University of Surrey, WEGO Health, Wellcome Trust, and Vitaccess. Disclosures do not show a conflict with the work being presented. Tanuja Chitnis has received compensation for consulting from Biogen, Novartis Pharmaceuticals, Roche Genentech, and Sanofi Genzyme. She has received research support from the National Institutes of Health, National MS Society, US Department of Defense, Sumaira Foundation, Brainstorm Cell Therapeutics, EMD Serono, I-Mab Biopharma, Mallinckrodt ARD, Novartis Pharmaceuticals, Octave Bioscience, Roche Genentech, and Tiziana Life Sciences. Disclosures do not conflict with the work being presented. Piet Eelen has received compensation for consulting, advising and presenting from Merck, Convatec, Novartis and Biogen. Disclosures do not conflict with the actual work being presented. Giampaolo Brichetto has been member of the advisory board of Novartis and Roche. Disclosures do not conflict with the work being presented. Andrew Lloyd works for and holds stock in Acaster Lloyd Consulting Ltd which has received fees from Novartis. Disclosures do not show a conflict with the work being presented. Hollie Schmidt has received compensation for consulting from Celgene, and Accelerated Cure Project has received grants, collaboration funding and consulting payments from Biogen, Bristol Myers Squibb, Celgene, EMD Serono, Genentech, MedDay, Novartis and Sanofi Genzyme. Disclosures do not show a conflict with the work being presented. Jeremy Hobart has received consulting fees, honoraria, support to attend meetings or research support from Acorda, Asubio, Bayer Schering, Biogen Idec, F. Hoffmann-La Roche, Genzyme, Merck Serono, Novartis, Oxford PharmaGenesis and Teva. Disclosures do not show a conflict with the work being presented. Miriam King, Jennifer Fitzgerald, Thomas Hach, and Jo Vandercappellen, are employees of Novartis Pharma AG.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by Novartis Pharma AG.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
