Abstract
Background
Cognitive disengagement syndrome (CDS) refers to a set of attentional symptoms comprising daydreaming, mental fogginess, and hypoactivity clearly distinguishable from ADHD-IN and other forms of psychopathological issues.
Objective
The present study: (1) assessed CDS among adults with multiple sclerosis (MS) and healthy controls, and (2) investigated the associations between CDS and depression, insomnia, fatigue, and paresthesia in persons with MS.
Methods
Two-hundred and seven adults with MS (mean age: 36.64 years; 72.5% females) and 213 healthy controls (mean age: 34.19 years; 54.5% females) provided self-ratings of CDS, insomnia, depression, fatigue, and paresthesia.
Results
Compared to healthy controls, adults with MS reported higher scores for CDS (t(418 = 4.29, p < 0.001, d = 0.42), depressive symptoms (t(41) = 2.21, p = 0.014, d = 0.22), and insomnia (t(418) = 1.66, p = 0.049). Among the MS sample, higher levels of CDS were associated with higher depression (r = 0.64), insomnia (r = 0.34), fatigue (r = 0.53), and paresthesia (r = 0.28). Further, higher scores for depression, insomnia, and fatigue were independently associated with higher CDS.
Conclusions
Adults with MS experience greater CDS symptoms than healthy controls. Higher CDS scores were associated with depression, insomnia, fatigue, and paresthesia, which are typical of adults with MS. MS may contribute to or exacerbate CDS symptoms and be a potentially important target for further investigation.
Keywords
Introduction
Multiple sclerosis (MS) is the most prevalent chronic inflammatory disease of the central nervous system. 1 Worldwide, more than two million children, adolescents, but above all adults, suffer from MS, 1 and females are more affected than males. The etiology of MS is still not fully understood, though genetic and environmental factors, 2 including the gene-environment interaction appear to develop and maintain the occurrence of MS. Further, while at a physiological level, persons with MS (PwMS) may typically report impaired vision, limb weakness, sensory numbness, gait-balance issues, and gastro-intestinal problems, 3 at a psychological level, PwMS report symptoms of insomnia, 4 depression, 5 fatigue, 6 and paresthesia. 7 PwMS further have impairments in social functioning8,9 and socio-cognitive processing 10 than the general population.
Examining cognitive disengagement syndrome in MS
While there is sufficient evidence that PwMS report impaired cognitive functions than the general population, 11 studies have yet to examine cognitive disengagement syndrome (CDS) symptoms in the context of MS. CDS (formerly also called sluggish cognitive tempo 12 ) comprises a set of attentional symptoms that include daydreaming, staring, mental fogginess/confusion, and slowed behavior/thinking. 13 Initially identified as a potential symptom set within attention-deficit/hyperactivity disorder (ADHD), over the past two decades a rapidly growing body of research has shown CDS symptoms to be (1) distinct from ADHD and other mental health dimensions, and (2) independently associated with poorer functional outcomes including social and academic difficulties and global functional impairment. However, despite growing recognition of the importance of the CDS construct for understanding psychopathology and functional outcomes, the vast majority of studies have been conducted in school-aged children, leaving our understanding of CDS in adults especially limited. Further, as noted in a recent systematic review, 14 very few studies have examined CDS in the context of chronic or complex medical conditions, despite the potential importance of the CDS symptom set for understanding patterns of psychosocial adjustment.
Although unexamined in MS, there is clear evidence that higher CDS symptoms are associated with significantly greater internalizing psychopathology symptoms and sleep problems in other populations, primarily studies conducted in non-clinical samples of children, adolescents, and adults. To illustrate, children and adolescents scoring high on CDS also reported higher scores for internalizing issues, 15 symptoms of depression,16–18 insomnia and other sleep-related difficulties,18,19 including a possible overlap with alexithymia, emotion regulation difficulties, 20 and mind wandering. 21 The limited studies conducted in adults also find that adults scoring high on CDS also reported higher scores for depression, both in a non-clinical 22 and clinical sample, 23 stress, and insomnia, cross-sectionally, 24 quasi-longitudinally, 25 and also in a clinical sample. 23 However, these associations have yet to be examined in adults with chronic medical conditions such as MS, despite depressive 5 and insomnia4,26 symptoms being highly prevalent in this population.
The present study
The aims of the present study were: (1) to compare CDS scores, symptoms of depression, insomnia, and fatigue between PwMS and healthy controls, and (2) to investigate among PwMS the associations between CDS and MS-related symptoms (i.e. insomnia, depression, fatigue, and paresthesia). The second aim allowed us to specifically examine CDS symptoms in relation to well-established MS-related symptoms to determine whether CDS was associated with poorer functioning in this unique clinical population. We hypothesized that PwMS would have higher scores for CDS than healthy controls. We further hypothesized that among PwMS higher scores for CDS were associated with higher illness-related symptoms (i.e. symptoms of depression and insomnia, fatigue, and paresthesia).
Methods
Participants
The MS sample
The sample included 207 individuals with MS (mean age: 36.50 years SD = 9.08; range: 18–65 years; 72.5% females). Inclusion criteria were: (1) age between 18 and 65 years; (2) diagnosed with relapsing-remitting MS, based on the McDonald's criteria, 27 and ascertained by an experienced neurologist not otherwise involved in the study; (3) EDSS score<6; (4) willing and able to comply with the study conditions; (5) stable MS-related medication (interferon beta, glatiramer acetate, natalizumab, fingolimod, and dimethyl fumarate), and clinical conditions for at least 60 days; (6) signed written informed consent. Exclusion criteria were: (1) other neurological diseases; (2) severe psychiatric conditions such as substance use disorder, severe major depressive disorder, bipolar disorders, generalized anxiety disorders, or PTSD, based on a clinical psychiatric interview 28 ; (3) acute suicidality; (4) any adverse physical condition, such as relapses; (5) for females: being pregnant or breastfeeding, as these may alter mood and sleep.
The healthy control sample
Healthy controls were recruited via advertisements on the webpages of hospitals and universities. A total of 213 individuals of the general population were recruited using convenience sampling method. Inclusion criteria were: (1) Age between 18 and 65 years; (2) psychopathologically healthy, as ascertained by a brief clinical interview for DSM-5 psychiatric disorders 28 conducted by a clinical psychologist not otherwise involved in the study; (3) willing and able to comply with the study conditions; (4) signed written informed consent. Exclusion criteria were: (1) acute suicidality; (2) withdrawal from the study; (3) for females: being pregnant or breastfeeding, as these may alter mood and sleep.
The Ethical Committee of the Kharazmi University (Tehran, Iran; code IR. KHU.REC.1401.014) approved the study, which was performed in accordance with the current edition 29 of the Declaration of Helsinki.
Procedure
PwMS registered at the Iranian Multiple Sclerosis Society (Tehran, Iran) were approached in the MS Association Center of Tehran (Iran) between June and November 2022 to participate in the present cross-sectional study. Participants were fully informed about the aims of the study and the confidential data handling. Thereafter, signed informed consent was obtained. Participants completed a series of self-rating questionnaires covering sociodemographic and MS-related information, symptoms of depression, insomnia, fatigue, paresthesia, and CDS. Participants needed between 20 and 30 min to complete the questionnaires. For healthy controls, they were also informed about the aims of the study and about the secure and confidential data handling, after which they signed the written informed consent. Last, identical to participants with MS, healthy controls completed a series of self-rating questionnaires covering sociodemographic information and symptoms of CDS, depression, insomnia, but not paresthesia. Healthy controls needed between 20 and 30 min to complete the questionnaires.
Measures
Sociodemographic and MS-related information
Participants reported on their age (years), sex at birth (female; male), and civil status (single, married, other), highest educational level (compulsory school, high school, higher educational level), and current job condition (unemployed, employed, student). For participants with MS, disease duration and current EDSS scores were taken from medical records.
Measures
CDS symptoms
To self-assess adult CDS, participants completed the Farsi version of the Adult Concentration Inventory (ACI 30 ). The ACI is a self-report measure of 16 CDS symptoms, although, as in a previous study, one item assessing low motivation has consistently shown poor convergent and discriminant validity 31 and was not used in this study. Example items include: “I am slow at doing things”; “My mind feels like it is in a fog”; or “I get lost in my own thoughts.” Each item is rated on a four-point scale (0 = not at all, 1 = sometimes, 2 = often, and 3 = very often), with higher sum scores reflecting a higher degree of CDS symptoms (Cronbach's alpha: 0.81). The Farsi ACI has been previously psychometrically validated. 32
Symptoms of depression
Participants completed the Farsi version 33 of the Beck Depression Inventory-Fast Screen (BDI-FS). 34 The BDI-FS is a brief self-report inventory designed to evaluate symptoms of depression in patients with medical illness. It consists of seven items, and every item has a set of four possible responses, representing different levels of symptom severity (e.g. sadness: 0 = “I don't feel sad”; 1 = “I feel sad”; 2 = “I'm sad all the time and I can't snap out of it”; 3 = “I'm so sad/unhappy, that I can't stand it”). Higher sum scores reflect a greater severity of depressive symptoms (range: 0–21) (Cronbach's alpha: 0.84).
Insomnia
Participants completed the Farsi version 35 of the Insomnia Severity Index. 36 This questionnaire is a seven-item screening measure for insomnia. The items, answered on a 5-point Likert scale (0 = not at all to 4 = very much), refer in part to the Diagnostic and Statistical Manual of Mental Disorders 37 criteria for insomnia by assessing difficulties in falling asleep, difficulty in remaining asleep, early morning awakenings, impaired daytime functioning, low satisfaction with sleep, and worry about sleep. Higher scores indicate greater insomnia symptoms (Cronbach's alpha: 0.81).
Fatigue
Participants completed the Farsi version 38 of the Fatigue Severity Scale (FSS 39 ). The FSS consists of nine items, and answers are given on 7-point Likert scales (1 = not at all to 7 = definitively/almost always), with higher scores reflecting higher levels of fatigue (Cronbach's alpha: 0.79).
Paresthesia
Participants with MS rated their degree of paresthesia on a 10-point visual analogue scale ranging from 0 (no sensations at all) to 10 (severe sensations).
Statistical analysis
Preliminary calculations: With a series of Pearson's correlations, we explored whether age was associated with symptoms of depression, insomnia, fatigue, paresthesia, and CDS. Correlation coefficients were between r = −.04 and −.09; the decision was not to introduce age as a confounder. Similarly, we performed a series of t-tests to compare symptoms of depression, insomnia, fatigue, paresthesia, and CDS between female and male participants. T-values were between 1.17 and 1.76; accordingly, gender was not introduced as a possible confounder.
To compare sociodemographic information between participants with and without MS, a t-test was performed for age, and a series of X2-tests was performed for gender, civil status, highest educational level, and current job position.
To compare scores for CDS, depression, insomnia, and fatigue between participants with MS and healthy controls, a series of single t-tests was performed. Effect sizes for t-tests were reported as Cohen's d with the following ranges: d = 0 to 0.19: trivial effect sizes [T]; d = 0.20 to 0.49: small effect sizes [S]; d = 0.50 to 0.79: medium effect sizes [M]; d ≥ 0.80: large effect sizes [L].
To investigate whether CDS scores were associated with MS-related symptoms among the MS sample, we undertook a series of Pearson's correlations. For correlation coefficients, the following cut-off values were reported: r < 0.29 = small effect size; r = 0.30 to 0.49 = medium effect size; r ≥ 0.5 = large effect size.
Last, with a multiple regression analysis, we explored whether symptoms of depression, insomnia, fatigue, and paresthesia were independently associated with CDS scores among the MS sample. Preliminary conditions to run a multiple regression model were met: N = 207 > 100; predictors explained the dependent variable (R = 0.715, R2 = 0.511), and the Durbin–Watson coefficient was 1.906, indicating that the residuals of the predictors were independent. The number of predictors × 10 was lower than the sample size: 4 × 10 < 207. Further, the variances inflation factors (VIF) were between 1.194 and 1.296; while there are no strict cut-off points to report the risk of multicollinearity, VIF < 1 and VIF > 10 indicate multicollinearity.
The nominal significance level was set at alpha < 0.05. All statistical calculations were performed in SPSS® version 29.0 (IBM Corporation, Armonk, NY, USA) for Apple Mac®.
Results
Sociodemographic information between participants with and without MS
Table 1 provides the descriptive and inferential statistical comparisons of sociodemographic information between participants with and without MS. Compared to healthy controls, PwMS were more likely to be older, female, married, unemployed, and with a descriptively lower educational level.
Sociodemographic information, separately for individuals with MS and healthy controls.

Comparisons of cognitive disengagement syndrome, depression, insomnia, and fatigue between PwMS and healthy controls
Table 2 provides the descriptive and inferential statistical overview of scores for CDS, depression, insomnia, and fatigue between PwMS and healthy controls.
Descriptive and inferential statistical indices of CDS, symptoms of depression, insomnia, and fatigue between persons with multiple sclerosis and healthy controls.

T: trivial effect size; S: small effect size; M: medium effect size.
Compared with healthy controls, PwMS reported higher scores for CDS (t(418) = 4.29, p < 0.001, d = 0.42 (small effect size), higher scores for depression (t(418) = 2.21, p = 0.014, d = 0.22 (small effect size), higher scores for insomnia (t(418) = 1.66, p = 0.049, d = 0.17 (trivial effect size), and higher scores for fatigue (t(418) = 5.25, p < 0.001, d = 0.51 (medium effect size).
Correlations between cognitive disengagement syndrome and MS symptoms in the MS sample
Table 3 provides the overview of Pearson's correlation coefficients between CDS and MS-related symptoms. Coefficients are only reported in Table 3, but not in the text. Higher scores for CDS were statistically significantly associated with higher scores for depression, insomnia, fatigue, and paresthesia (small to medium to large effect sizes) in the MS sample.
Descriptive and correlational statistical overview of cognitive disengagement syndrome, insomnia, symptoms of depression and insomnia, fatigue, and paresthesia among individuals with multiple sclerosis.
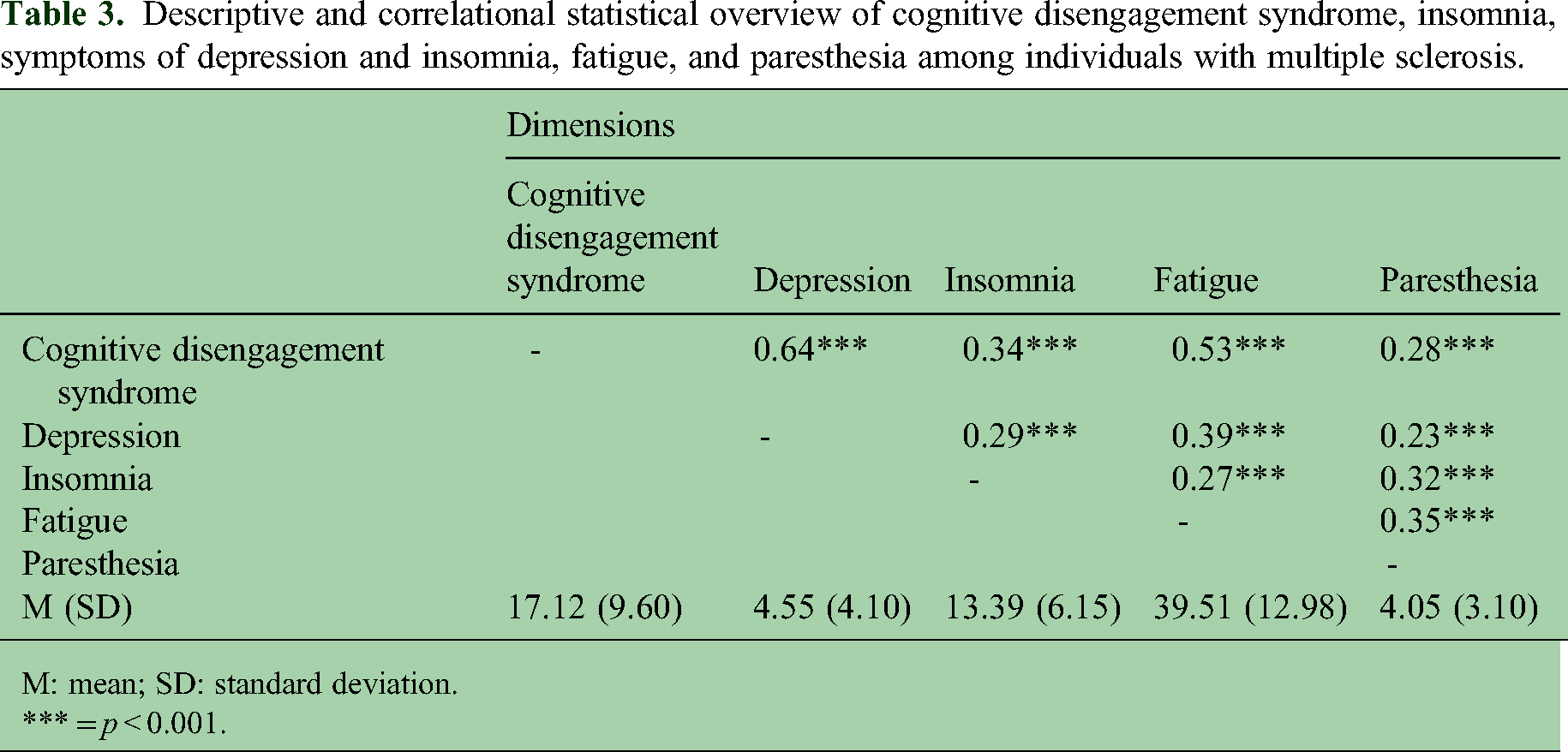
M: mean; SD: standard deviation.
*** = p < 0.001.
MS-related symptoms in relation to cognitive disengagement syndrome in the MS sample
Last, with a multiple regression analysis, we explored whether symptoms of depression, insomnia, fatigue, and paresthesia were independently associated with CDS scores among the MS sample.
Table 4 provides the statistical overview of the multiple regression model. Higher scores for depression, insomnia, and fatigue were independently associated with higher scores for CDS in the MS sample, while paresthesia did not reach statistical significance.
Multiple regression model to associate CDS as a function of depression, insomnia, fatigue, and paresthesia among individuals with multiple sclerosis.

CDS: cognitive disengagement syndrome.
Discussion
The present study examined whether scores for CDS of PwMS differed from those of healthy controls. We further examined correlations between CDS and MS-related symptoms of insomnia, depression, fatigue, and paresthesia among the MS sample, and we ran a multiple regression model to explore whether scores for depression, insomnia, fatigue, and paresthesia were more strongly and independently associated with CDS scores. PwMS had higher scores for CDS, when compared with healthy controls. In the MS sample, higher scores for CDS were associated with higher scores for all MS-related symptoms (depression, insomnia, fatigue, paresthesia). Higher scores for depression, insomnia, and fatigue, but not for paresthesia, were independently associated with higher scores for CDS.
Cognitive disengagement syndrome in PwMS
Scores for CDS of PwMS were higher than those of healthy controls. This study shows, for the first time, that adults with MS are more likely than the control group to experience CDS symptoms. Individuals with MS often experience a wide range of cognitive and attentional difficulties,11,40 and the current study findings suggest that these difficulties may also include CDS symptoms. It is possible that MS contributes to or exacerbates CDS symptoms, and additional research is needed to investigate both of these possibilities.
Associations between cognitive disengagement syndrome and MS-related symptoms
Higher scores for CDS were associated with higher MS-related symptoms of depression, insomnia, fatigue, and paresthesia.
For depression, as shown in Table 3, higher CDS scores and higher depression scores were associated. Accordingly, we have confirmed what has been already observed among non-clinical samples of children, adolescents, and adults.18,23,32,41–46 Similarly, we confirmed that CDS was more closely associated with internalizing than externalizing symptoms.12,13,24,25 The novelty of the present pattern of results is that such a pattern of association was observed among a sample of adults with neurological disorders, in general, and among PwMS, more specifically.
The observed CDS-insomnia link is consistent with findings from non-clinical samples of children and adolescents, where higher scores for CDS were associated with higher scores for insomnia.18,19 However, the novelty is that such a CDS-insomnia link was observed among a sample with a neurodegenerative condition (i.e., PwMS).
Fatigue and paresthesia are two typical MS-related health issues, which could be understood as a proxy of the cognitive-emotional processing of pain. Results from studies in the context of MS showed that poor sleep, higher scores for fatigue, and pain were associated. 47 Further, symptoms of depression and fatigue often co-occur among PwMS,7,48–51 possibly due to common immune and inflammatory pathways. 52 In the present study, higher scores for fatigue, but not paresthesia, were independently associated with CDS. While the quality of the data does not allow a deeper understanding of the underlying psychological and psychophysiological mechanisms, it is conceivable that fatigue, that is, the generalized and complex state of mental and physical impairment of an individual to function at their normal capacity, 53 could be the cause, the effect, or the co-occurrence of CDS. We have no conclusive answer, and future studies among clinical and non-clinical samples should further investigate this fatigue-CDS-link. To conclude, given that adults with MS report a broad variety of cognitive dysfunctions,11,40,54,55 in general, and impairments in attention and concentration,56,57 more specifically, it is conceivable that CDS may be an important factor, which might accelerate the onset or maintain cognitive impairments.
Strengths, limitations, and future directions
We recognize the following strengths of the study: (1) We assessed CDS scores among a sample of PwMS, and to our understanding, this is the first study to investigate CDS among an adult clinical sample. Among a pediatric sample (n = 122) with children and adolescents with spina bifida, nine (7.4%) showed clearly elevated CDS scores, 58 which longitudinally were associated with a lower cognitive performance. 59 (2) We compared CDS scores between PwMS and healthy controls. (3) We associated CDS scores with common and burdensome MS-related symptoms; more specifically, we showed that higher scores for depression, insomnia, and fatigue, but not paresthesia, were more strongly associated with higher scores for CDS.
Despite the novelty of the results, the following limitations should be considered. First, MS-specific medications were assessed (namely interferon beta, glatiramer acetate, natalizumab, fingolimod, and dimethyl fumarate), but we did not further investigate if the patterns of results systematically changed as a function of the medication. While, thus, theoretically the type of medication might have systematically modified mood and sleep, we assume that such an impact occurred randomly; further, one inclusion criterion was the EDSS score < 6, which is considered a robust indicator to assess a quite homogeneous sample of PwMS. However, future studies should provide detailed information about the medical treatment; this would enable a better understanding of the associations between the type of medication and the stability and changes of outcomes. Second, we focused on CDS. However, it would have been interesting to know whether and to what extent CDS and MS-illness-related dimensions were associated with scores for ADHD, in general, and with the inattentive type (ADHD-IN), more specifically. 13 Third, in future work, it would be interesting to assess whether CSD scores were associated with lower scores for cognitive60–62 and workplace-related performances. Fourth, we did not assess the level of exercise/sports. In future work, such assessments would be valuable because there is extensive evidence that regular exercise and sports interventions favorably impact both physical and psychological issues among PwMS.63,64 As such, it is conceivable that individuals with higher levels of exercise and physical fitness might have reported lower symptoms of insomnia, depression, fatigue, and paresthesia, along with lower scores for CDS. Fifth, there is some sparse evidence that higher scores for CDS were associated with social withdrawal 65 and suicidal ideation. 66 As such, future studies should consider these social and behavioral risk factors. Sixth, a longitudinal study design would have allowed to understand the causal associations between CDS and MS-related symptoms over time. Seventh, CDS is not a nosological category within the current classification systems, such as the DSM-5 TR 67 or the ICD-11. 68 In this line, future studies might further investigate whether CDS, as a standalone syndrome, may affect PwMS independently of symptoms of depression and fatigue. On the flip side, CDS may be considered a transdiagnostic concept,12,13 which, however, is clearly distinct from ADHD-IN, autism spectrum disorder, or dimensions of alexithymia.
Footnotes
Data availability
Data are made potentially available under specific conditions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent
All participants signed the written informed consent. The Ethical Committee of the Kharazmi University (Tehran, Iran; code IR. KHU.REC.1401.014) approved the study, which was performed in accordance with the current edition of the Declaration of Helsinki.
Funding
D S-B disclosed receipt of the following financial support for the research of this article: This research was partially supported by a Postdoctoral Fellowship Program of the Swiss National Science Foundation (P400PS_191009).
