Abstract
Introduction
Fatigue is a highly prevalent symptom in people with multiple sclerosis. It demands careful assessment and prompt intervention to improve their quality of life and overall burden of disease. This scoping review aims to provide a comprehensive synthesis and update of the existing evidence on the effectiveness of different pharmacological and nonpharmacological interventions for multiple sclerosis (MS)-related fatigue.
Methods
To ensure the transparency and quality of the articles chosen for this scoping review, the Preferred Reporting Items for Systematic Reviews and Meta-analysis Protocols extension for Scoping Reviews was used. Exclusively randomized controlled trials published between 2016 and 2023 were included.
Results
Twenty-eight articles were analyzed. We found that pharmacological interventions are few and have included the use of Amantadine, Ondansetron, Methylphenidate, and Modafinil, with little effects on fatigue. Nonpharmacological interventions are diverse and include cognitive behavioral therapy, guided imagery, phototherapy, exercise, brain stimulation, and lavender administration with evidence of a statistically significant decrease in fatigue.
Conclusions and Discussion
Current evidence on the effectiveness of pharmacological and nonpharmacological interventions is inconclusive. Lack of knowledge of the pathophysiology of fatigue limits its prevention, control, and management recommendations. A comprehensive and interdisciplinary approach is required to manage this symptom in patients with MS.
Keywords
Introduction
Multiple sclerosis (MS) is an autoimmune-mediated, chronic, neurodegenerative disease of the central nervous system.1,2 It affects 2.8 million people worldwide, mainly young adults between the ages of 20 and 40 years. It manifests with focal neurological signs and symptoms that compromise locomotion, vision, sensory perception, cognition, gait, coordination, among others. 3 Fatigue, defined as “a subjective lack of physical or mental energy that is perceived by the individual or caregiver to interfere with usual and desired activities,” 4 is one of the most prevalent symptoms in patients with MS. 5 It has been reported to have a prevalence in this population of 36.5%–97%. 6 It generates a negative impact on the quality of life of patients 7 compromising their physical, psychosocial, and cognitive domains, leading to high costs for the health system. 8
The physiopathology associated with fatigue in MS can be divided into primary and secondary. Regarding primary fatigue, different theories have been proposed. There are immune mechanisms mediated by cytokines (especially IL-6 and TNF-alpha), monocytes, and microglia. These alterations in cytokines impact neuronal signaling, monoaminergic pathways, and neuronal metabolism. Furthermore, cytokines can alter the neuroendocrine system in the hypothalamic–pituitary–adrenal axis, stimulating cortisol production. 9 Additionally, typical white matter lesions in MS can lead to impaired nerve conduction, reduced axonal transmission, and disruption of communication between centers related to planning, execution, and motivation. Also, gray matter lesions that affect connections and neural circuits result in a loss of the ability to perform precise movements, thus requiring greater energy expenditure and consequently increasing fatigue. Moreover, lesions in the brainstem nuclei can lead to a reduction in neurotransmitters, particularly dopaminergic, serotonergic, and noradrenergic, which contributes to decreased motivation and mood, further exacerbating fatigue. 9 In contrast, the secondary mechanism is associated with concomitant circumstances or diseases, such as anemia, thyroid disturbances, sleep disorders, and poor physical fitness. 10 The poorly understood and heterogeneous pathophysiology has been a barrier to the development of effective treatments for managing fatigue. Currently, pharmacological and non-pharmacological strategies have been developed that aim to impact the mechanisms associated with primary fatigue. The results of these studies are controversial, making the management of fatigue one of the most complex and challenging issues. Pharmacological strategies, such as amantadine, modafinil, and methylphenidate, have been used to manage fatigue, based on their effects on the dopaminergic and noradrenergic systems 9 ; however, some studies have documented that these molecules are not superior to placebo in improving fatigue. 11 These medications only address one cause of fatigue, and additional alterations associated with fatigue remain unaddressed. Other nonpharmacological interventions, such as transcranial direct current stimulation (tDCS), have been investigated and show a recommendation level between high and moderate for managing fatigue in MS, impacting cortical excitability. 12
Miller and Soundy conducted a systematic review up to 2016 to provide evidence-based recommendations for the treatment of fatigue in people with MS. They found that nonpharmacological interventions, such as energy conservation techniques and exercise, could improve fatigue in the context of MS. Moreover, psychobehavioral interventions demonstrated potential effectiveness, although the available information remains limited. Conflicting evidence was found regarding pharmacological treatments, particularly with amantadine and Prokarin; however, modafinil showed potential benefits. 13 This article highlights the potential benefits of both pharmacological and nonpharmacological interventions, though stronger evidence is still needed to make more robust clinical recommendations. Therefore, the purpose of this review is to update the findings from 2016 to 2023, aiming to assess whether other pharmacological and nonpharmacological strategies and additional evidence have emerged that could lead to new recommendations and changes in clinical practice.
Methods
To synthesize the published evidence on the pharmacological and nonpharmacological interventions for MS-related fatigue, an initial Systematic Review was proposed for the development of this study. However, due to the heterogeneity of the interventions, methods to study fatigue, and results, the research team decided to conduct a scoping review. This type of review leads to identifying knowledge gaps and types of available data on a specific topic 14 which were aligned with the intended purpose of this study. This scoping review followed the Preferred Reporting Items for Systematic Reviews and Meta-analysis Protocols extension for Scoping Reviews (PRISMA-ScR) methodology 15 in order to facilitate the transparent reporting of the available data.
The protocol of the study was registered in the Open Science Framework registries for Scoping Reviews under the registration DOI: https://doi.org/10.17605/OSF.IO/EUCJA.
Eligibility criteria
The researchers conducted independent searches covering two time periods as follows: from the deadline of the Miller and Soundy review (May 2016) until December 2023. Search strategies (Table 1) were established by an experienced researcher and discussed with the team for additional refinement. Search was performed on three databases containing medical and psychological information: PubMed, Academic Search Complete/EBSCO, and ScienceDirect. Articles based on clinical trials (CT), randomized controlled trials (RCTs), and experiments written in English and Spanish were included. The selection of the articles was based on the title and abstract. Pilot studies were excluded as well as gray literature. Studies that involved adult patients diagnosed with MS, with the presence of fatigue, independent of gender, type of sclerosis, stage of disease, and degree of disability were included. For the methodological quality, three researchers simultaneously evaluated the full text using the Joanna Briggs Institute guidelines. The PRISMA diagram illustrated in Figure 1 exhibits the process used to initially identify 5389 records to finally include 26 RCT studies in this review. Table 2 details the inclusion and exclusion criteria for the selection of articles.
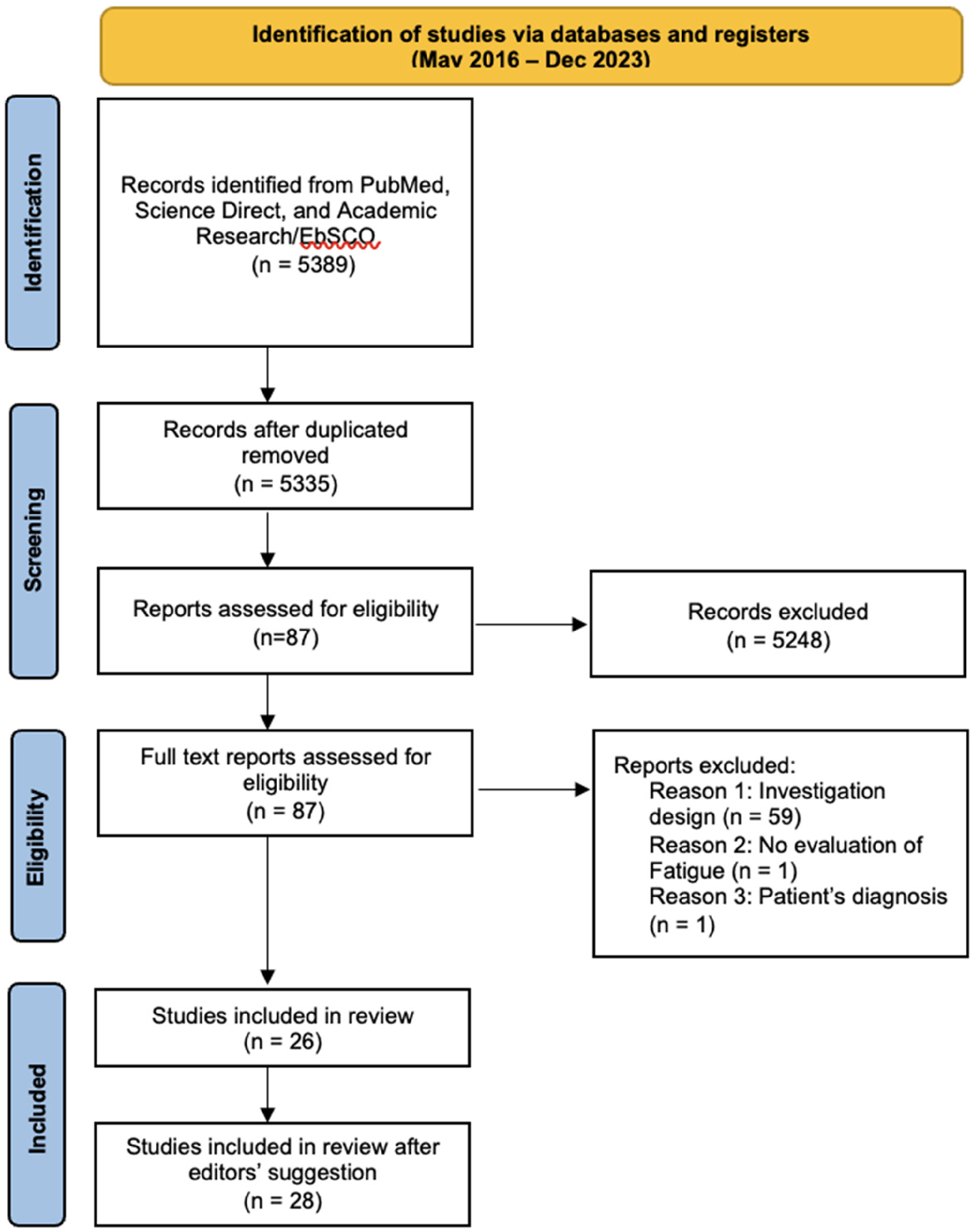
PRISMA flowchart.
Search strategies.

Inclusion and exclusion criteria considered for the selection of articles.

The database search yielded 5389 records. Of these, 5248 were excluded due to duplication or failure to meet the inclusion criteria based on the title and abstract. Out of the 87 eligible articles, 59 had research designs that did not meet the inclusion criteria, including pilot studies, observational studies, studies without a control group, without sample randomization, or with low-quality randomization processes. In one study, fatigue was not assessed, and in another, the patients had tuberous sclerosis.
Data extraction
The careful reading of each of the articles included in this scoping review allowed us to extract relevant data that provided insights into both pharmacological and nonpharmacological interventions for managing fatigue associated with MS. As a result, the researchers created an analysis matrix (Table 2) where the following information was recorded: authors, year of publication, journal, study objective, study design, population and sample, MS type, country of the sample's origin or where the study was conducted, instruments used to measure fatigue, fatigue treatment or interventions utilized, and intervention outcomes on fatigue: fatigue behavior (increase, decrease, or no change), previous and posterior score, and p value. Each of the articles was read, and data were extracted and charted by two researchers to ensure the reliability of the recorded information.
Results
Demographic characteristics
A total of 1819 participants were included in the studies reviewed, with the majority of the reported being female (73.99%; n = 1346). Most of the studies’ mean age ranged between 40 and 50 years. Fifteen of the studies included patients with Relapsing-Remitting MS (RRMS), seven studies included both phenotypes (RRMS and progressive forms). All the studies were RCTs.
Fatigue measures
The most predominantly used measure for Fatigue in the studies was the Fatigue Severity Scale (FSS) (17 studies), followed by the Modified Fatigue Impact Scale (MFIS) (12 studies). Other measurements used were the Visual Analogue Fatigue Scale (VAFS), followed by the Fatigue Scale for Motor and Cognitive Function (FSMC), Patient-Reported Outcomes Measurement Information System (PROMIS) fatigue short form, Fatigue Impact Scale (FIS), and the Fatigue Assessment Scale and the Checklist individual strength (CIS20r) subscale for fatigue severity.
Checklist Individual Strength subscale fatigue severity is a self-report instrument that measures four dimensions of fatigue (severity, decrease in motivation, concentration, and activity) to report the perception of fatigue intensity. Fatigue Impact Scale is a three-dimensional (cognitive, physical, social) self-report instrument designed by Fisk et al. 16 to measure the extent to which fatigue has caused problems for people with MS, through 40 items. Modified FIS is a 21-item version of the FIS developed to be used when responding to the full version may be exhausting for patients.17,18 Patient-Reported Outcomes Measurement Information System fatigue short form is a multidimensional (fatigue, physical functioning, pain, emotional distress, social role participation) self-report instrument that enhances communication between patients and healthcare professionals, designed for researching the impact of variables such as fatigue on the quality of life of people with chronic illnesses, so that the measurement of reports and quantification of changes in this variable can be optimized. 19 Specifically, it differentiates two dimensions of fatigue: experience measured through the topography of the response (intensity, frequency, and duration) and its impact on social, physical, and mental activities. 20 Fatigue Severity Scale is an unidimensional brief self-report instrument, created by Krupp et al. to measure fatigue in people diagnosed with MS or systemic lupus erythematosus and to measure the impact of fatigue by identifying the degree of its severity. 21
Intervention categories
After careful review, interventions were grouped into two broad categories: (1) Pharmacological (n = 2) and (2) Nonpharmacological (n = 26). Pharmacological interventions (Table 2) included the use of amantadine, modafinil, methylphenidate, and ondansetron. Due to the heterogeneity of the studies in the Nonpharmacological category, three subgroups were established to classify the interventions as follows (a) psychological, (b) exercise and other interventions, and (c) brain stimulation.
Psychological interventions (n = 5) (Table 3) included those based on psychological intervention approaches, exercise, and other interventions (n = 17) (Table 4) used diverse therapeutic methods ranging from exercise to the use of lavender. The last category was the use of brain stimulation interventions (n = 4) (Table 5), including pulsed electromagnetic field, and transcranial direct current stimulation.
Psychological interventions.
Exercise and other interventions.

Brain stimulation interventions.

Pharmacological interventions
Characteristics
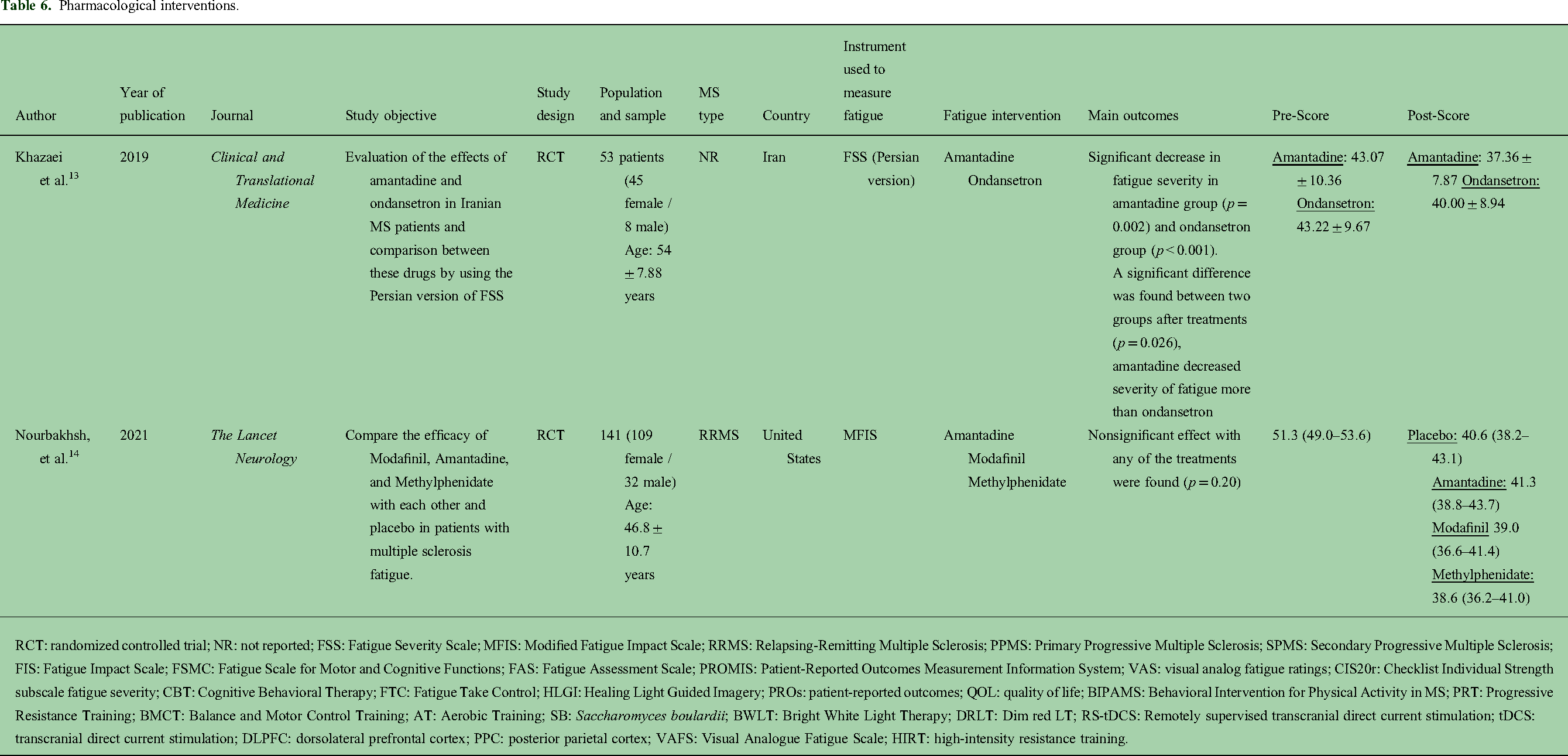
Two published articles were found to evaluate the use of pharmacological interventions to treat fatigue in MS patients (Table 6). A total of 194 individuals were included in these studies, with one involving patients with RRMS. These randomized crossover CTs assessed treatment sequences of 4–6 weeks with a washout period of two weeks, alternating the use of the following interventions: amantadine, modafinil, methylphenidate, ondansetron, and placebo.
Pharmacological interventions.
RCT: randomized controlled trial; NR: not reported; FSS: Fatigue Severity Scale; MFIS: Modified Fatigue Impact Scale; RRMS: Relapsing-Remitting Multiple Sclerosis; PPMS: Primary Progressive Multiple Sclerosis; SPMS: Secondary Progressive Multiple Sclerosis; FIS: Fatigue Impact Scale; FSMC: Fatigue Scale for Motor and Cognitive Functions; FAS: Fatigue Assessment Scale; PROMIS: Patient-Reported Outcomes Measurement Information System; VAS: visual analog fatigue ratings; CIS20r: Checklist Individual Strength subscale fatigue severity; CBT: Cognitive Behavioral Therapy; FTC: Fatigue Take Control; HLGI: Healing Light Guided Imagery; PROs: patient-reported outcomes; QOL: quality of life; BIPAMS: Behavioral Intervention for Physical Activity in MS; PRT: Progressive Resistance Training; BMCT: Balance and Motor Control Training; AT: Aerobic Training; SB: Saccharomyces boulardii; BWLT: Bright White Light Therapy; DRLT: Dim red LT; RS-tDCS: Remotely supervised transcranial direct current stimulation; tDCS: transcranial direct current stimulation; DLPFC: dorsolateral prefrontal cortex; PPC: posterior parietal cortex; VAFS: Visual Analogue Fatigue Scale; HIRT: high-intensity resistance training.
Measurements and findings
One of the studies 22 assessed fatigue through the FSS evaluating the score before and four weeks after each treatment. Significant results were found for the use of amantadine and ondansetron to decrease fatigue; however, Amantadine decreased severity more than ondansetron (43.07 (±10.36) before to 37.36 (±7.87) after treatment vs. 43.22 (±9.67) before and 40.00 (±8.94) after, respectively).
The study performed by Nourbakhsh, 11 evaluated fatigue through the MFIS at week 5 of each study period including placebo, amantadine, modafinil, and methylphenidate. No statistically or clinically significant results were obtained for the use of these treatments. However, there was a significant effect of the treatment on the psychosocial subscale of the MFIS.
Nonpharmacological interventions
Psychological interventions
Characteristics
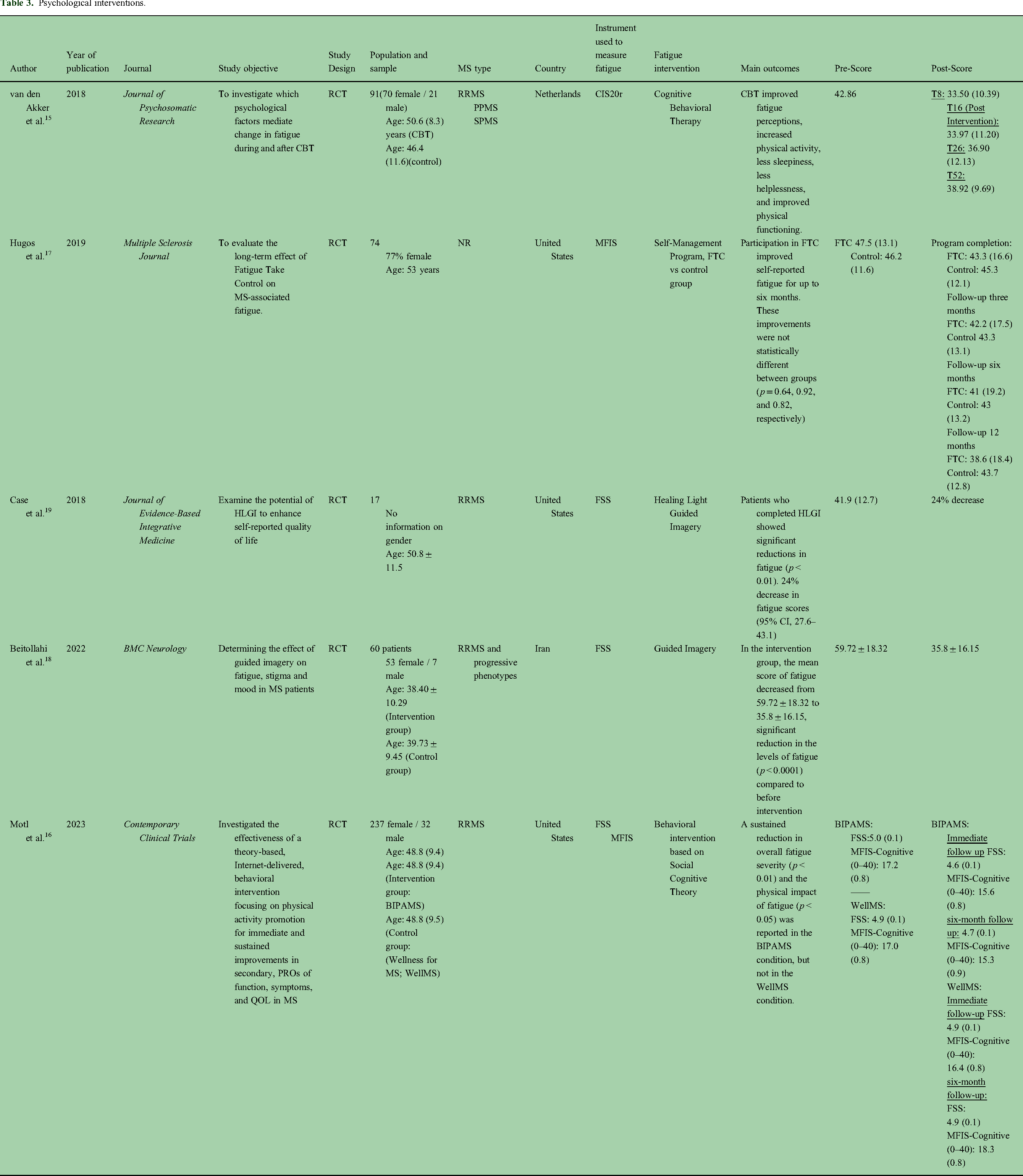
The five psychological interventions included in this review focused on diverse therapeutic methods, including Cognitive Behavioral Therapy (CBT) (n = 1), behavioral intervention based on Social Cognitive Theory (n = 1), Fatigue Take Control (FTC) (n = 1), and Guided Imagery (n = 2) (Table 3) 479 patients were included. Two studies included patients with RRMS and two studies included both phenotypes (RRMS and progressives phenotypes). The mean time of intervention for the studies was 12.0 weeks,4–24 with the majority (n = 4) implementing individual interventions vs. small groups (n = 1).
Measurements and findings
Four studies assessed fatigue through the FSS, while the others through the MFIS and the CIS20r. Overall, patients reported improvement in fatigue through most of the interventions, being statistically significant; however, one of them did not report p value. Cognitive behavioral therapy reported a considerable decrease in fatigue, 23 evaluated through the CIS20r, during the first eight weeks of intervention (42.86 (8.46) at baseline vs. 33.50 (10.39) at T8), however, when compared to the control group at week 52, there was no significant difference between groups (38.92 (9.69) in CBT vs. 39.54 (9.01) in the control group). The other study 24 that evaluated a behavioral intervention for physical activity focusing on measuring Patient Reported Outcomes showed significant results with a change of the FSS1–7 from 5.0 (0.1) to 4.6 (0.1) and 4.7 (0.1) in the immediate and six-month follow up, respectively. Another study 25 reported fatigue scores improving significantly between baseline and follow-ups with the greatest mean change in the intervention group (FTC) at 12 months, being significantly greater than the control group, with a mean improvement of 8.9 points on the MFIS. The guided imagery intervention showed improvement with a decrease of up to 23.91 points in the FSS in the guided imagery intervention with audio files, 26 being statistically significant.
Exercise and other interventions
Characteristics
The majority of studies (n = 10) in this category used exercise as a therapeutic approach, including: aerobic exercises, tailored activity pacing, interval exercise training, high-intensity resistance training (HIRT), pilates, progressive resistance training (PRT), aquatic exercise training, and hippotherapy (Table 4) Other interventions included in this category involved acupressure, diet, vestibular rehabilitation, probiotic supplementation, natural products, and the use of lavender and Bright White Light Therapy (BWLT). In total, 988 participants were included in these studies, with 10 of these involving patients with RRMS and 3 with both phenotypes (progressives and RRMS). Most of the interventions were supervised (n = 11), while the rest (n = 6) were self-administered.
Measurements and findings
Several methods were used to evaluate fatigue, with FSS (n = 10) and MFIS (n = 6) being the most used, followed by FSMC, FIS, FAS, and VAFS. Most of the exercise intervention studies showed statistically significant results when compared to control groups, except for the Tailored Activity Pacing Intervention 27 and yoga intervention, 28 which showed no significant results. Exercise intervention studies that evaluated fatigue through the MFIS demonstrated a decrease in fatigue with the greatest mean decrease in the Home-based pilates intervention 29 43.6 ± 9.8 at baseline vs. 31.0 ± 13.5 at week 8 of follow-up, followed by the PRT intervention 30 with a decrease of −12.8 [−17.7; −7.8] and the Hippotherapy intervention 31 44.2 ± 19.0 to 32.3 ± 18.5. Aquatic exercise training 32 showed a decrease from 43.1 ± 14.6 to 32.8 ± 5.9, while the AT + Vitamin D intervention 33 35.57 ± 4.20 to 29.69 ± 5.46. Those in which exercise interventions were evaluated through the FSS included the HIRT 34 and exercise training 35 which showed a decrease in fatigue mean scores. One article on aerobic exercise 36 was evaluated through the FIS with a change from 41.00 ± 33.71 to 27.75 ± 27.72. The strategies, involving those not-related to exercise, that applied the FSS included the use of Mediterranean Diet 37 with a decrease of 9.8 ± 11.5 at six-month follow-up, the use of Saccharomyces boulardii Probiotic 38 from 28.50 ± 14.29 to 22.95 ± 10.86, the use of a Herbal derived product MS14 39 (46.00 ± 11.69 to 40.44 ± 13.21) 40 Vestibular rehabilitation and Self-Accupressure. 41 The use of Lavender 42 showed a statistically and clinically significant decrease in the MFIS from 40.56 (7.63) to 7.04 (4.91). Lastly, the BWLT group reported a reduction in the FSS after four weeks of therapy, but no statistically significant differences were reported between the BWTL and control group study arms (p = 0.8).
Brain stimulation interventions
Characteristics
There were four RCTs identified in this category, including 126 patients, based on the use of tDCS (n = 3) and the use of pulsed electromagnetic field therapy (n = 1) (Table 5). Two studies included RRMS and progressive phenotypes and 2 studies included patients with RRMS. The mean time of intervention was 2.35 weeks.4,5
Measurements and findings
Methods used to evaluate fatigue include FSS, PROMIS, mFIS, and VAFS. Fatigue scores evaluated through the FSS showed statistically significant results for intervention with tDCS 43 with a decrease from 26.6 ± 9.2 to 21.0 ± 6.4 between baseline and posttreatment analysis. Moreover, five-day tDCS in the bilateral whole-body somatosensory cortex showed improvement of fatigue symptoms compared to placebo group measured by the mFIS (score preintervention: 52.3, score post intervention: 27.6) 65 and active tDCS over the left DLPFC resulted in a significant improvement in FSS. 68 However, in the pulsed electromagnetic field therapy, 44 there were no differences in changes between the intervention and the placebo group.
Discussion and conclusions
The findings show that nonpharmacological interventions for MS-related fatigue could be effective in reducing or alleviating fatigue symptoms. Given the heterogeneity of nonpharmacological options and the diversity of possible mechanisms involved in fatigue, psychological aspects must be considered in this experience. Expectations are considered as mediating variables involved in the experience of feeling relieve 45 and, consequently, to the concepts of placebo and placebo effect. Literature refers that about 40% of prescriptions act as placebo. 46 People with MS experience suffering, 47 frustration with therapeutic failure, 48 the unknown etiology of their disease, 49 progression, and general discomfort inherent to the course of the disease or treatment. 50 With these conditions, expectations about a treatment that can alleviate a disabling symptom are desirable and quickly established. 45
Placebo has nonspecific mechanisms of action, with explanations being broad, including behavior similar to that of conventional drugs (e.g., latency time, maximum effect, dose–response relation, tolerance, addiction, adverse effects, and reinforcing or reducing the effects of other drugs). 51 Mechanisms of brain empathy 52 resulting from the activation of nuclei of the prefrontal cortex, the cerebral amygdala, the limbic system, as well as the increased release of dopamine 53 and oxytocin 54 explain the learning of expectations of healing and pleasurable experiences. In fact, placebo will depend on psychological (individual differences) and brain responses in a therapeutic context, which affects the construction of meanings and the perception of well-being. Oxytocin would have a significant effect on decreasing anxiety and increasing confidence. 55 For example, the study by Motaghi et al. highlights the effect of lavender in decreasing MS-related fatigue and, at the same time, this substance has been described to have an anxiolytic effect, 56 which could be mediating the experience of fatigue relief.
Psychological interventions such as psychoeducation and CBT contribute to acquiring knowledge and developing or optimizing coping skills that modulate stress and fear and increase self-efficacy, which in turn contributes to modifying the interpretation of the experience associated with fatigue. 57 Psychoeducation allows addressing emotional concerns caused by the distress of being overwhelmed or confused 58 due to fatigue, and improves the perception of emotional well-being. 59 Mindfulness is a technique that contributes to reducing anxiety and achieving greater states of relaxation, results that also increase the perception of emotional well-being and quality of life, and can contribute to the transformation of the emotional experience 60 associated with fatigue.
The effects of all nonpharmacological interventions are indirect. 61 That is, they increase the perception of well-being and quality of life through various mechanisms such as the reduction of symptoms and tension, increased perception of control, heightened positive expectations, and even the production of endorphins, as seen with exercise and physical activity, which can also improve endurance and energy, and whose practice may be associated with better mental health and greater psychological well-being. 62 It is common for people to believe that interventions of this type do not produce side effects, leading to greater acceptance and trust, in contrast to what occurs with pharmacological treatments, which have a clear aversive association with these effects.
Previous studies largely align with our findings. Miller et al. 13 reported that nonpharmacological interventions led to improvements in fatigue, including resistance training, aerobic training, combined exercise training, CBT, energy conservation programs, and acupuncture. However, only yoga showed a significant reduction in this symptom. In fact, Harrison et al. 63 published a meta-analysis based on publications from nine databases up to August 2018, examining the most promising exercise-based and behavioral interventions for treating fatigue in MS. They found that all types of exercise have moderate to high effects in reducing this symptom, with balance exercises being superior. CBT also outperforms energy conservation programs, with strong supporting evidence.
One of the novel nonpharmacological strategies is tDCS
The results regarding tDCS are heterogeneous, which can be explained by methodological differences among the studies. Firstly, different cortical areas have been targeted, such as bilateral motor or sensory areas, 65 which appear to be effective, whereas stimulation of the right posterior parietal region seems ineffective, and the response to left dorsolateral prefrontal stimulation has been inconsistent. Furthermore, protocols vary in terms of current intensity and session duration; greater positive effects are observed when sessions are repeated. 64 Conducting studies with larger samples and standardized protocols will help determine the optimal approach for tDCS. This field holds significant potential for future research.
Regarding pharmacological interventions, there are few studies published to date, and the results have not documented a clinically significant impact. In addition to pharmacological therapies currently being investigated to treat fatigue, disease-modifying therapy (DMT) could impact fatigue by reducing inflammation, disability, and the number of brain lesions. However, very few RCTs have included fatigue as an outcome of DMT. In fact, fatigue as an outcome of DMT RCTs is underrepresented, there are few studies evaluating it, and the data are suboptimal. Another point to strengthen in research is for RCTs to include fatigue as an outcome. 66
The evidence so far has shown that the efficacy of pharmacological therapy is heterogeneous. Miller and Soundy 13 found that amantadine, pemoline, and carnitine did not lead to significant improvement in fatigue, although pemoline combined with acetylsalicylic acid showed positive results in one study. Modafinil demonstrated improvement in some studies. In our study, we observed a significant decrease in fatigue severity in both the amantadine-treated group (p = 0.002) and the ondansetron-treated group (p < 0.001). 22 However, in another RCT, no significant effect was found for modafinil, amantadine, and methylphenidate (p = 0.20). 11
The studies included in this scoping review involve a heterogeneous population in terms of phenotype, disease duration, and DMT use; additionally, various severity scales were employed. Although these are specific instruments for measuring fatigue, they are heterogeneous in their construction, purpose, and outcome, accounting for both the impact and the topography of the response (frequency, duration, or intensity). All the instruments used in the various studies included in this review were brief, self-report measures, and have adequate psychometric properties. With the exception of FSS and VAFS all are multidimensional. Therefore, this may explain the heterogeneity of results.
It is not possible to make a strong recommendation regarding the optimal type of intervention to manage fatigue in patients with MS. The studies published to date have heterogeneous evaluation methods, intervention times, results, and populations.
Conclusion
Fatigue is a secondary symptom that is difficult to manage and the evidence of its impact on the quality of life of patients is sufficient and growing,6,67 so that ignoring or undervaluing the symptom results in a decrease in quality of life. Greater attention and commitment from the treating team toward this aspect is recommended. This review highlights the need for a multimodal, interdisciplinary, and comprehensive therapeutic approach to the fatigue experienced by patients with MS. This represents several challenges, such as the conformation of multidisciplinary teams, which maintain a continuous interprofessional dialogue that allows adjustments based on the evolution of this symptom; the design of personalized, contextual, and flexible palliative interventions that address the biological, psychological, and physical dimensions. Moreover, consider the design and implementation of economically viable and sustainable proposals which improve the quality of life and decrease disease burden, ultimately aiming to reduce the costs of healthcare interventions in the long term.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
