Abstract
Background
Comorbidities are common among people with multiple sclerosis (pwMS) and have been suggested to affect the usage of disease-modifying treatments (DMTs).
Objectives
We investigated the prevalence and types of comorbidities in newly diagnosed pwMS, as well as the associations between comorbidities and the choice and persistence of the initial DMT.
Methods
This retrospective register study used data from the Finnish MS register (85% of national coverage). The inclusion criteria were a diagnosis of relapsing-remitting MS between 2010 and 2022, the first DMT started between 2016 and 2022, and an age of at least 18 years. The exclusion criteria were a diagnosis of secondary progressive MS and initiation of azathioprine or mitoxantrone.
Results
The inclusion criteria were met by 1630 pwMS. At least one comorbidity was present in 50.9% of the pwMS. Respiratory comorbidities were associated with the choice of medium-efficacy DMT over high-efficacy DMT (OR = 0.58, CI = 0.36–0.91). Multicomorbidity was associated with lower persistence on all DMTs. Additionally, lower persistence on medium-efficacy DMTs was associated with autoimmune and psychiatric comorbidities and the injectable administration route.
Conclusions
Comorbidities are prevalent among newly diagnosed pwMS. Clinicians should consider comorbidities carefully, as they are associated with DMT persistence.
Introduction
Multiple sclerosis (MS) is never treated in isolation; its treatment is influenced by various confounding factors. One such factor is comorbidity, which is more common among people with MS (pwMS) than in the general population 1 and affects about 47% of pwMS. 2 The cornerstone of MS treatment is immunomodulatory medication therapy. Comorbidities have been suggested to affect the initiation 3 and persistence4–6 of these disease-modifying treatments (DMTs).
Only a few previous studies have examined the prevalence of comorbidities7–11 and their associations with early DMT persistence4,5 in newly diagnosed patients. Finnish health registries provide comprehensive data that support further investigation into the issue.
Our primary objective was to investigate the prevalence of comorbidities in newly diagnosed pwMS. Secondarily, we aimed to study whether comorbidities were associated with the choice of the initial DMT or two-year DMT persistence. The information obtained from the study helps to provide guidance for tailoring treatment decisions for individual patients.
Materials and methods
Study design
This retrospective register study used secondary data obtained from the Finnish MS register. Clinicians have collected data since 2014 (retrospective pre-2014, mostly prospective after). Diagnosis and basic patient information are auto-imported from hospital systems—along with laboratory results, medications, and imaging dates in certain units. Clinical data are entered manually. Overall, 16 out of 21 Finnish wellbeing service counties were included in the study (about 85% of national coverage of pwMS): Southwest Finland, Satakunta, Pirkanmaa, Päijät-Häme, Kymenlaakso, South Karelia, North Savo, Central Finland, South Ostrobothnia, North Ostrobothnia, Ostrobothnia, Kainuu, East Uusimaa, Central Uusimaa, West Uusimaa, and Vantaa and Kerava.
Comorbidities were identified based on ICD-9 and ICD-10 diagnoses from the Care Register for Health Care (inpatient care, day surgery operations, and outpatient care in public specialized medical care) starting from 1994 and the Register of Primary Care Visits (outpatient primary health care) starting from 2011, the earliest available data from each source.
The inclusion criteria were a diagnosis of RRMS between 2010 and 2022, the first DMT initiation between 2016 and 2022, and being 18 years or older at the time of diagnosis (Figure 1). The exclusion criteria were a diagnosis of secondary progressive MS (SPMS) at the first DMT initiation and initiation of azathioprine or mitoxantrone.

The inclusion process (DMT: disease-modifying treatment; MS: multiple sclerosis; RRMS: relapsing-remitting multiple sclerosis; SPMS: secondary progressive multiple sclerosis).
A two-year period was selected for assessing DMT persistence, as the length of the observation period (2016–2022) limited longer-term analysis. An extended observation period would have reduced the inclusion of high-efficacy DMTs (heDMTs), which became more common toward 2022. A similar approach was used in our previous study. 12
The STROBE reporting guidelines were used in this study. 13
Variables
The following variables were studied: sex, age, time and the type of MS symptom onset, the time of diagnosis, Expanded Disability Status Scale (EDSS) score at the time of DMT initiation, the history of relapses and DMT usage, reasons for DMT discontinuation (patient’s wish, pregnancy, alteration of disease course, drug antibodies, JC virus, other) and comorbidities before DMT initiation. Comorbidity was defined as a chronic disease or medical condition diagnosed prior to the initiation of the first DMT. A comorbid condition was considered to be present if the according ICD-10 code had been indexed at least twice and on different dates. The ICD-10 codes were categorized according to the relevant organ systems (Supplemental Appendix 1), except for autoimmune conditions, which were listed separately (Supplemental Appendix 2) due to their particular focus of interest since pwMS have a higher risk for autoimmune diseases prior to MS diagnosis than the general population.7,14 The list of autoimmune diseases was based on previous research evidence and agreed between the researchers. Inflammatory bowel disease (IBD) includes Crohn's disease and ulcerative colitis. Multicomorbidity was defined as ≥ 2 comorbidities.
The DMTs were classified as medium-efficacy DMTs (meDMTs) and heDMTs. meDMTs were subcategorized into injectable therapies (meINJs) and oral therapies (meORALs). meINJs included interferon betas and glatiramer acetate. meORALs encompassed dimethyl fumarate, diroximel fumarate, and teriflunomide. heDMTs consisted of alemtuzumab, cladribine, daclizumab, fingolimod, natalizumab, ocrelizumab, ofatumumab, ponesimod, and rituximab (off-label). The approval years of the DMTs are presented in Supplemental Appendix 3.
The symptoms at onset were categorized according to their clinical origin within the central nervous system (CNS): (I) brainstem (double vision, specific brain nerve and other cranial nerve symptoms); (II) cerebellum (coordination and balance disturbances); (III) spinal cord (bladder and bowel dysfunction); (IV) pyramidal tract (muscle weakness and spasticity); (V) optic nerve (optic neuritis); (VI) sensory pathways (paresthesia and dysesthesia); and (VII) unknown. Symptoms at onset were considered multifocal when ≥ 2 symptoms were present, excluding sensory and unknown symptoms, as their origin from a distinct CNS area cannot be reliably determined.
Statistical analysis
Data analysis and visualization were performed on pseudonymized data using RStudio (Version 2021.09.1). Partial dates were imputed as the middle of the month or year. Baseline EDSS score was defined as the last recorded EDSS score within one year prior to the start of initial DMT, and annualized relapse rate (ARR) was defined as the number of relapses during that year. Diagnosis date for the comorbidity group was selected based on the first observed visit with one of the group's ICD-10 codes.
Numerical variables were expressed as means with standard deviations (SDs) or medians with interquartile ranges (IQRs), and categorical variables as frequencies and proportions based on non-missing data. Group comparisons were performed using non-parametric methods. The Wilcoxon rank sum test was used for calculating p values for continuous variables and Fisher's exact test for categorical variables. Multivariable logistic regression models were fitted using a generalized linear model with logit link function to get odds ratios and confidence intervals for choosing one DMT group over another and for discontinuing a DMT within two years of initiation. Models for DMT discontinuation were restricted to meDMT patients stratifying by DMT choice and sex. Besides sex-stratified analysis, interaction models were also fitted with explanatory variables sex, age, number of prior relapses, corresponding comorbidity, and sex-comorbidity-interaction to test factors associated with meDMT discontinuation, keeping both sexes in the same model. For explanatory variables, age was the only variable to be considered as continuous, as others were used in binary form. Usage of the selected variables was confirmed using variance inflation factors, and underlying multicollinearity was assessed. Diagnostic tests were conducted to identify any potential influential outliers. The Benjamini-Hochberg procedure (1995) was used for controlling and checking the false discovery rate and for correcting p values for multiple comparisons between demographic and clinical variables.
Trend analysis for multicomorbidity and individual comorbidities was performed using the Cochran-Armitage trend test. In all analyses, two-tailed or corrected p values below 0.05 were considered statistically significant. StellarQ Ltd, Finland, was responsible for data analysis and visualization.
Standard protocol approvals, registrations, and patient consents
This study was approved (THL/4118/14.02.00/2022) by the Finnish Social and Health Data Permit Authority Findata (www.findata.fi/en). According to the Finnish legislation (552/2019), since only retrospective register data were involved in the study without contact with patients, ethics board review or patient consent was not required.
Results
Demographics
The inclusion criteria were met by 1630 patients, 72.4% of whom were female (Table 1). Patients with heDMT as the first DMT were younger than those with meDMT initiation (34.2 vs. 36.6 years at the time of DMT initiation, p < 0.001), had shorter diagnostic delay (5.8 vs. 9.9 months, p < 0.001), and higher disease activity in terms of ARR (1.1 vs. 0.8, p < 0.001) and EDSS score (2.0 vs. 1.5, p < 0.001).
Demographical data of the study patients.

ARR: annualized relapse rate; DMT: disease-modifying therapy; EDSS: Expanded Disability Status Scale; heDMT: high-efficacy disease-modifying treatment; IQR: interquartile range; m: missing; meDMT: medium-efficacy disease-modifying treatment; SD: standard deviation; MS: multiple sclerosis.
Brainstem (double vision, specific brain nerve symptoms, and other cranial nerve symptoms); cerebellum (disturbances of coordination and ataxia, vertigo, and disturbances of balance and posture); optic nerve (optic neuritis); pyramidal tract (muscle weakness and spasticity); sensory pathways (paresthesia/dysesthesia); spinal cord (bladder and bowel dysfunction).
*p < 0.05; **p < 0.01; ***p < 0.001.
Comorbidities
At least one comorbidity was present with 50.9% of the patients (Table 2) and multicomorbidity with 21.0% of the patients. The most common comorbidities were psychiatric (22.0%), neurological (12.0%), respiratory (7.9%), endocrine (7.4%), and autoimmune (7.2%) disorders. The most common autoimmune disorders were IBD (1.5%), recurring or chronic iritis (1.5%), and type 1 diabetes (T1D) (0.9%).
Comorbidities in the study population.
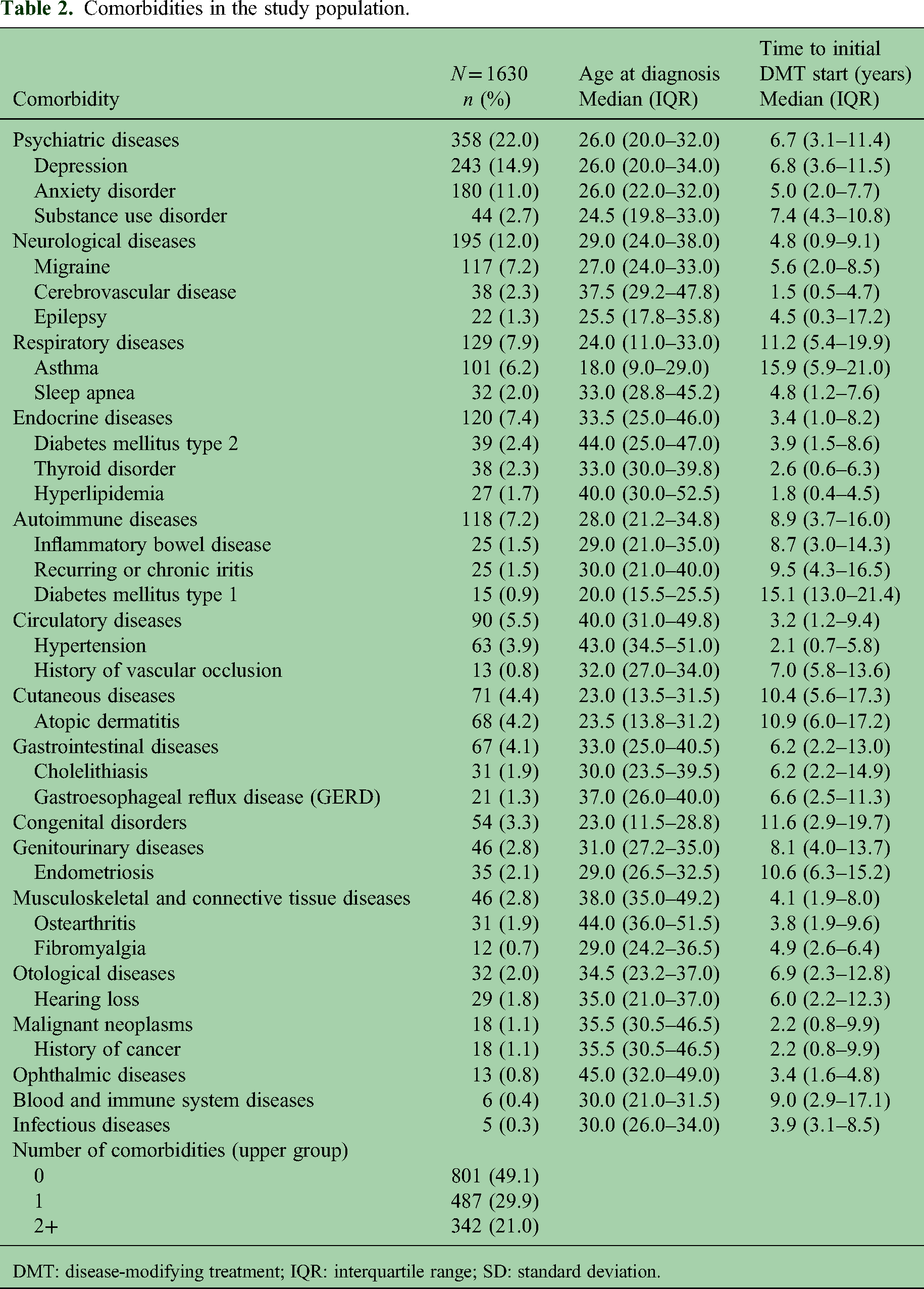
DMT: disease-modifying treatment; IQR: interquartile range; SD: standard deviation.
The proportion of newly diagnosed pwMS having multicomorbidity (p = 0.012) and psychiatric diseases (p < 0.001) increased from 2016 to 2022 (Figure 2). Interestingly, the proportion of patients with psychiatric diseases increased by 57.4% from 2020 to 2021.

Proportions of patients with a comorbidity by year of initial disease-modifying treatment (DMT) start.
DMT initiation
We conducted a multivariable model to determine the factors associated with a higher likelihood of starting heDMT over meDMT (Table 3). In Model 1, we examined multicomorbidity and demographic factors. Multicomorbidity was not associated with choosing either DMT class, yet younger age and two or more relapses in medical history were associated with a higher likelihood of starting heDMT over meDMT. Individual comorbidities were looked at in Model 2. Respiratory comorbidities were associated (OR = 0.58, CI = 0.36–0.91) with choosing meDMT over heDMT, in addition to older age (OR = 0.97, CI = 0.96–0.99) and less than two prior relapses (OR = 2.18, CI = 1.67–2.85).
Logistic regression analysis of factors influencing the choice of heDMT over meDMT and meINJ over meORAL.

CI: confidence interval; heDMT: high-efficacy disease-modifying treatment; meDMT: medium-efficacy disease-modifying treatment; meINJ: medium-efficacy injectable therapy; meORAL: medium-efficacy oral therapy; OR: odds ratio.
*p < 0.05; **p < 0.01; ***p < 0.001.
We examined meDMTs more closely to study the factors associated with starting medication with either meINJs or meORALs. In Model 1, again, multicomorbidity was not associated with either administration route. Instead, female sex and younger age were associated with a higher likelihood of starting meINJs over meORALs. In Model 2, no individual comorbidity was associated with choosing either meINJs or meORALs. Female sex (OR = 2.47, CI = 1.81–3.41) and younger age (OR = 0.98, CI = 0.97–1.00) remained the only associated factors choosing meINJs over meORALs.
DMT persistence
We investigated which factors explained the discontinuation of the initial DMT, looking only at the cases where the reason for discontinuation was either adverse effects or lack of efficacy. A multivariable model was built to investigate two-year persistence with the first DMT (Table 4). Those who started with meDMT were more likely to discontinue than those with heDMT (OR = 0.20, CI = 0.12–0.31) within the first two years. In the model of meDMT persistence, higher risk for discontinuation was associated with multicomorbidity (OR = 1.60, CI = 1.13–2.25) and injectable administration route (OR = 1.51, CI = 1.12–2.04). Multicomorbidity had a weak association with higher risk for heDMT discontinuation (OR = 2.62, CI = 0.97–6.73).
Logistic regression analysis of factors associated with DMT discontinuation within the first 2 years of use.

DMT: disease-modifying treatment; CI: confidence interval; heDMT: high-efficacy disease-modifying treatment; meDMT: medium-efficacy disease-modifying treatment; meINJ: medium-efficacy injectable therapy; meORAL: medium-efficacy oral therapy; OR: odds ratio.
*p < 0.05; **p < 0.01; ***p < 0.001.
We examined the reasons for DMT discontinuation in more detail, including individual comorbidities in the analysis (Table 5). Due to the insufficient population size for heDMTs, the analysis was limited to meDMTs. Within meORALs, lower persistence was associated with psychiatric (OR = 1.71, CI = 1.07–2.70) and autoimmune diseases (OR = 2.48, CI = 1.27–4.79). Gastrointestinal diseases were associated with lower persistence with meINJs, although weakly (OR = 3.09, CI = 1.03–9.90).
Logistic regression analysis of factors associated with meDMT discontinuation within the first 2 years of use.

CI: confidence interval; meDMT: medium-efficacy disease-modifying treatment; meINJ: medium-efficacy injectable therapy; meORAL: medium-efficacy oral therapy; OR: odds ratio.
Interaction models included sex, age, number of prior relapses, corresponding comorbidity, and sex * comorbidity interaction as explanatory variables.
p < 0.05; **p < 0.01; ***p < 0.001.
Patients with autoimmune comorbidity were slightly more likely to discontinue a DMT due to ineffectiveness, with discontinuation rates for adverse effects/lack of efficacy at 61/39 in autoimmune patients versus 68/34 in those without an autoimmune condition (a few cases having both reasons listed). However, the result was nonsignificant (p = 0.65). There was no significant difference in disease activity among autoimmune patients and others at the time of DMT initiation (mean ARR 0.90 vs. 0.83, p = 0.81), nor in the proportion of patients who had a relapse during the first 2 years (20% vs. 18%, p = 0.63).
Finally, since sex was associated with the initial DMT choice within the meDMT group, we further analyzed whether sex would associate with meDMT persistence, too. In the model for female patients (Table 5), autoimmune (OR = 2.77, CI = 1.58–4.86) and psychiatric diseases (OR = 1.58, CI = 1.06–2.34) were associated factors for lower two-year persistence. We found no associated factors for men, but the sample size was underpowered for reliable analysis.
Discussion
In this Finnish register study, we found that comorbidities are common among newly diagnosed pwMS (50.9%). We observed a growing trend of multicomorbidity and psychiatric diseases. There was a 57.4% increase in the prevalence of psychiatric comorbidities from 2020 to 2021, simultaneously with the COVID-19 pandemic. Respiratory comorbidities were associated with choosing meDMTs over heDMTs as the initial DMT. Multicomorbidity was associated with lower persistence across all DMTs. Among meDMTs, lower persistence was also associated with injectable administration and the presence of autoimmune or psychiatric comorbidities.
Multicomorbidity has been associated with worse clinical outcomes in MS. 15 In our data, 50.9% of the patients had at least one comorbidity, consistent with a prior study a previous study reporting a comorbidity present in slightly more than half of the patients at the time of MS diagnosis. 16 The most common comorbidity group in our study was psychiatric disorders (22%), aligning with a previous study reporting an 18% prevalence of psychiatric diseases at MS onset. 16 We observed neurological comorbidity in 12% of the patients, mostly migraine (7.2% of all the patients). Similarly, a recent study found that neurological comorbidity was common during the first year after MS diagnosis. 17 Prevalence of epilepsy has been reported to be higher among pwMS at diagnosis than in the general population.7,8
The prevalence of autoimmune disorders was 7.2% in our data. The most prevalent conditions were IBD (1.5%), chronic iritis (1.5%), and T1D (0.9%). PwMS have a higher risk for autoimmune diseases prior to MS diagnosis than the general population.7,14 Autoimmune diseases share common genetic 18 and environmental risk factors, such as vitamin D deficiency, 19 smoking, 20 and Epstein-Barr virus infection. 21 Polyautoimmunity is more frequently reported among pwMS. 22 Autoimmune comorbidities may be related to treatment delays and higher disability at MS diagnosis. 23 The prevalence of IBD prior to MS diagnosis has been reported as 0.6%–1.0%8,16 and 0.5% 16 with uveitis. In a meta-analysis, pwMS had a 53% higher risk of having IBD than controls, without a difference between Crohn's disease or ulcerative colitis. 24 Vice versa, patients with IBD had 91% higher prevalence of MS than controls. 25 An association between T1D and MS has been suggested, as patients with T1D have been reported to have a three-fold risk of developing MS. 26 Taken together, we confirmed that autoimmune comorbidities are common already in the early MS.
We observed a growing trend of multimorbidity and psychiatric diseases from 2016 to 2022. There was a 57.4% increase in psychiatric diagnoses in 2021 compared to the previous year, which may be explained by the COVID-19 pandemic. Similar observations were made among Finnish adolescents 27 and in older British adults 28 during the pandemic.
Respiratory comorbidities were associated with a higher likelihood of choosing meDMT over heDMT as the initial DMT, although the statistical association was weak. Moreover, the patients were characterized by older age and having less than two prior relapses. Thus, our data did not suggest an association of respiratory comorbidities and higher disease activity. A previous study found no association between comorbidities and DMT choice, although respiratory diseases were not assessed. 5 The association may be more related to age than to respiratory conditions themselves, as such conditions increase with age. 29 We found that older age and lower disease activity were associated with the choice of meDMT over heDMT. As heDMTs are considered more efficient in treating neuroinflammation, 30 patients with higher disease activity are understandably more often initiated on heDMTs. However, according to a meta-analysis, the efficacy of heDMTs exceeds meDMTs only in patients under 40 years of age. 31 Efficacy may be limited in older patients due to immunosenescence, 32 and the risk of adverse effects may increase with age. The cut-off age for considering DMT de-escalation appears to be at least 55 years, although more research is needed. 33 The clinicians are aware of the impact of age on disease activity and DMT response, which likely explains the choice of heDMTs in younger patients. The factors associated with choosing meORALs over meINJs were male sex and older age. The higher probability of choosing meINJs over meORALs among women might be explained by family planning. The updated 2024 Finnish guidelines for MS 34 suggest that injectable meDMTs can be continued during pregnancy, whereas oral meDMTs should be discontinued.
The persistence of meDMTs was lower than that of heDMTs. For meDMTs, three factors were associated with discontinuation: route of administration, comorbidities, and sex. The injectable administration route was associated with a higher risk of discontinuing the meDMT compared to peroral dosing. A meta-analysis found no difference in persistence between oral and injectable DMTs but noted that research on oral DMT persistence is limited. 35 In our study, multicomorbidity was associated with lower persistence on all DMTs. Multicomorbidity has been associated with higher disability in MS, 15 which might explain the findings. Psychiatric and autoimmune diseases were associated with lower persistence on meORALs. For meINJs, there were no associated factors, but a smaller sample size limited the reliability of the results. Psychiatric diseases have been associated with lower persistence, 6 probably due to lower tolerance against the adverse effects of DMTs. Presence of autoimmune comorbidities has been associated with higher disability in MS patients, 23 but we did not observe higher MS disease activity among autoimmune patients. However, the sample size was insufficient for reliable analysis. Sex was not associated with meDMT persistence itself, but we found that in female patients, autoimmune and psychiatric comorbidities were associated with a higher probability of stopping the meDMT. There were no persistence-associated factors in males, although the sample size was underpowered to provide reliable results.
This study has many strengths. Vast multicenter register data, with national coverage of about 85% of patients with RRMS, and well-established Finnish public healthcare, make the results of this study generalizable in western countries with similar healthcare systems. Clinical relapses, the timing of diagnosis, the onset of initial symptoms, and the reasons for DMT changes were thoroughly recorded in the register. The risk of selection bias was minimal, as all patients diagnosed with RRMS and receiving initial DMTs were included. As always, some limits need to be acknowledged. The data is missing patients who had not started DMTs during the follow-up period. The lack of a control group causes some limitations for our study, for example, comparison of the comorbidities in the general Finnish population. Assessing the quality of register data is challenging, and there is probably some overdiagnosis in the assessment of comorbidities. Finally, our approach to comorbidity classification has not been validated.
In conclusion, it is crucial to consider the comorbidities of pwMS when planning MS treatment. Especially, autoimmune and psychiatric comorbidities should be taken into account, as they were associated with lower treatment persistence.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251411076 - Supplemental material for Comorbidities in newly diagnosed multiple sclerosis patients: A population-based register study in Finland
Supplemental material, sj-docx-1-mso-10.1177_20552173251411076 for Comorbidities in newly diagnosed multiple sclerosis patients: A population-based register study in Finland by Henrik Ahvenjärvi, Johanna Krüger, Matias Viitala, Elina Jokinen, Henri Autio, Anne M Portaankorva, Merja Soilu-Hänninen and Mervi Ryytty in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-2-mso-10.1177_20552173251411076 - Supplemental material for Comorbidities in newly diagnosed multiple sclerosis patients: A population-based register study in Finland
Supplemental material, sj-docx-2-mso-10.1177_20552173251411076 for Comorbidities in newly diagnosed multiple sclerosis patients: A population-based register study in Finland by Henrik Ahvenjärvi, Johanna Krüger, Matias Viitala, Elina Jokinen, Henri Autio, Anne M Portaankorva, Merja Soilu-Hänninen and Mervi Ryytty in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-3-mso-10.1177_20552173251411076 - Supplemental material for Comorbidities in newly diagnosed multiple sclerosis patients: A population-based register study in Finland
Supplemental material, sj-docx-3-mso-10.1177_20552173251411076 for Comorbidities in newly diagnosed multiple sclerosis patients: A population-based register study in Finland by Henrik Ahvenjärvi, Johanna Krüger, Matias Viitala, Elina Jokinen, Henri Autio, Anne M Portaankorva, Merja Soilu-Hänninen and Mervi Ryytty in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
Taija Pietilä is greatly appreciated for her invaluable remarks on improving the manuscript. We would also like to recognize translator Robin Brown for valuable assistance with the language check.
Data availability statement
The research data cannot be shared due to restrictions caused by Finnish and European Union legislation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research collaboration study received funding from Novartis Finland Ltd. H Ahvenjärvi has received research grants from the Finnish Neurological Society, the Finnish Cultural Foundation, the Finnish MS Foundation and the University of Oulu Scholarship Foundation, support for conference and education session participation from Novartis and Orion, and honoraria for consultant services from Novartis. E Jokinen is a former employee of Novartis Finland Ltd. H Autio is a former employee of Novartis Ltd. M Viitala is an employee of StellarQ Ltd. M Soilu-Hänninen has served as an adviser or speaker for Biogen, Novartis, Roche, Sanofi, and Teva, and received support for congress participation from Biogen, Merck, and Novartis. J Krüger has served on the advisory board of Eisai, Lilly, Novartis, Nutricia, Roche, and Roche Diagnostics, received honoraria from lectures from Bioarctic and Lilly, and received support for congress participation from Merck and Lilly. AM Portaankorva reports no conflicts of interest. M Ryytty has received honoraria for lectures, advisory boards, congress visit, or for serving as an investigator for clinical trials from AbbVie, Biogen, Merck, Novartis, Roche, and Sanofi. The Finnish MS Foundation, Pohjois-Pohjanmaan Rahasto, Oulun Yliopiston Tukisäätiö, Finnish Neurological Society (grant numbers 60212346, 60221946, and 20230121).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
