Abstract
Background
Persistence to B cell depleting therapies (BCDT) like ocrelizumab and rituximab may be higher compared with other disease-modifying therapies (DMT) in multiple sclerosis (MS). Clinical trials directly comparing these treatments are lacking.
Objective
To compare the risk of treatment discontinuation, relapse, and confirmed disability worsening in patients starting BCDT vs other DMT within real-world settings.
Methods
In a longitudinal cohort study, patients with relapsing MS starting BCDT (ocrelizumab/rituximab, n = 269) after enrolment into the Swiss MS Cohort (SMSC) were evaluated for treatment discontinuation, occurrence of relapses, and disability worsening in comparison with platform (n = 57) and oral (n = 454) DMT, or natalizumab (n = 73) using Cox regression with double robust adjustment for baseline covariates.
Results
Patients starting BCDT were less likely to discontinue treatment than all other DMT combined (HR = 0.26, 95% CI = 0.18–0.36, p < .01), oral DMT (HR = 0.28, 95% CI = 0.20–0.39, p < .01) and natalizumab (HR = 0.35, 95% CI = 0.21–0.58, p < .01). BCDT were associated with lower risk of relapses as compared to oral DMT HR = 0.59, 95% CI = 0.39–0.88, p < .01), but not to natalizumab (HR = 0.90, 95% CI = 0.45–1.82, p = .778). Disability worsening was not significantly different between treatment groups.
Conclusion
We provide real-world evidence for patients being more persistent to BCDT than to other treatments, and better clinical outcomes may partly explain this.
Introduction
More than 20 disease-modifying therapies (DMT) have been developed for multiple sclerosis (MS) to reduce disease activity and the accrual of long-term disability. All these compounds share an anti-inflammatory mode of action, while they vary by their efficacy, safety profile, route of administration, and dosing schedule.1,2
Currently, ocrelizumab (OCR) is the only DMT approved for both relapsing forms of MS (RMS) and primary progressive MS (PPMS). Two phase 3 trials in RMS (OPERA I and OPERA II) have shown superior efficacy of OCR over interferon beta-1a (IFNβ-1a) to reduce the annualized relapse rate and confirmed disability worsening (CDW) at 3 and 6 months. 3 The ORATORIO trial in PPMS had shown decreased rates of CDW for OCR versus placebo over 120 weeks. 4 Before OCR, two clinical trials and two observational studies had shown that the chimeric monoclonal anti-CD20 antibody rituximab (RTX) is also effective in reducing radiological and clinical inflammatory disease activity in relapsing-remitting MS.5–8
The effectiveness of medications strongly depends on following the prescribed treatment regimen.9,10 Observational studies have shown greater persistence to OCR therapy relative to other DMT.11–14 For example, in an analysis of administrative claims data from 4587 MS patients in the US, higher persistence and adherence in patients treated with OCR was observed, with over 90% persistence at ≥12 months follow-up. 11 Similar results have also been found in population-based studies from Italy and in an ongoing prospective study of MS patients in Germany.13,14
However, most of these studies were retrospective, based on administrative claims, and none has attempted to provide an explanation for the increased persistence on OCR. We therefore compared persistence and clinical effectiveness measures (i.e., relapses and CDW) in patients who newly started BCDT (either OCR or RTX) vs those on other DMT over several years of follow-up in a real-world setting of the Swiss MS Cohort (SMSC). 15
Methods
Settings and patients
Data were derived from the SMSC, 15 which gathers longitudinal patient-level clinical data from MS patients undergoing regular assessments every 6–12 months from eight Swiss MS centers (University Hospitals of Basel, Berne, Geneva, Lausanne, Zurich, and the Cantonal Hospitals of Aarau, St Gallen, and Lugano). The SMSC was approved by the ethics committees of all participating centers.
Patients included in the present study had a diagnosis of RRMS according to 2010 McDonald criteria, 16 and newly started a DMT after 5 November 2013. We considered patients starting with BCDT (OCR and RTX), platform (glatiramer acetate, IFNβ-1a, and IFNβ-1b), oral (fingolimod, dimethyl fumarate, teriflunomide) DMT, or natalizumab (NTZ). RTX was largely used in Switzerland before the approval of OCR, therefore upholding the positivity assumption of non-zero probability of being treated with a DMT belonging to one of these four categories over study period.
Study design and endpoints
The first new DMT initiated after inclusion in the SMSC was considered the index treatment, and the date of initiation was the index date. The index DMT had not to be used in the same individual before SMSC entry. Patients were followed from index date to outcome or censoring event including discontinuation of the index DMT, date of last record, death, or administrative end of follow-up (6 May 2024, date of data extraction).
The primary outcome of the study was persistence on the index DMT, which was defined as the time interval between its start and discontinuation. Discontinuation was identified by proxy using a pre-defined time gap in therapy for each DMT (i.e., a minimum period during which the patient did not receive the index DMT to define discontinuation). These intervals were 18 months for OCR and RTX, 6 months for teriflunomide (3 months if eliminated by cholestyramine), and 8 weeks for fingolimod, dimethyl fumarate, glatiramer acetate, IFNβ-1a, IFNβ-1b, and NTZ. In patients who started a new treatment within these predefined intervals, the date of initiation of the new DMT was used as time of discontinuation of the index treatment.
Secondary outcomes were the time to first relapse and time to 6-month CDW since start of the index DMT. Relapses were defined as new, worsening, or recurrent neurologic symptoms, in the absence of fever, infections, or adverse reactions to prescribed medications, that lasted ≥24 h and were preceded by a stable or improving neurologic status of ≥30 days. Neurological disability was measured using the Expanded Disability Status Scale (EDSS). 17 Thresholds for CDW were defined as an EDSS increase of ≥1.5 if from a score of 0, 1 from a score of 1–5, or 0.5 from a score of ≥5.5. The date of worsening was the date of initial change in EDSS score, including if worsening occurred before censoring, but was confirmed after censoring.
Statistical analyses and neurofilament measurement
Patients were categorized into those initiating BCDT and those initiating any of the remaining DMT considered in the analysis. All analyses were adjusted using a double robust methodology, implementing inverse probability of treatment weighting (IPTW), followed by model-based multivariable adjustment. IPTW was performed using stabilized weights calculated from a propensity score model that included the following variables at index date: age, sex, disease duration, number of relapses in the previous 12 months, EDSS, and number of DMT used before index date.
Those covariates with residual imbalance after weighting (defined as standardized mean differences [SMD] > 0.1) were also carried forward into the outcome model for double robust adjustment. Values up to 6 months before index date were accepted as EDSS at baseline, and multiple imputations were performed for the remaining missing values using five imputations. IPTW and multiple imputation were combined using a “within” approach whereby a separate propensity score model was fitted, and the effect estimate was calculated, within each imputed data set. Effect estimates from the imputed dataset were combined using Rubin's rule. 18
All endpoints were evaluated using Cox Proportional Hazards regression (testing BCDT vs all other DMT grouped together as reference) and visualized using Kaplan-Meier (KM) curves. Robust 95% confidence intervals (CIs) were calculated using a sandwich variance estimator.
The comparisons of BCDT versus platform DMT, BCDT versus oral DMT and BCDT versus NTZ, respectively, were executed under the premise of appropriate balance in baseline characteristics after IPTW. In terms of relapses, events occurring during treatment with the index DMT were considered for the analysis. As to disability worsening, all events occurring up to 4 years after termination of index treatment were considered. The rationale behind this was the evidence for a longer therapeutic lag between DMT initiation and effect on disability outcomes, which is longer than for acute inflammatory relapses.19,20
As sensitivity analyses, alternative definitions of treatment discontinuation were used, first considering only time to next treatment initiation, and second reducing the time gap between BCDT infusions from 18 to 12 months to define discontinuation. In further sensitivity analyses, the same tests were performed after including serum neurofilament light (sNfL) concentrations at index date as an additional variable in IPTW and multivariable adjustment.
Blood samples within the SMSC are collected within 8 days from the clinical visit and stored at −80°C following standardized procedures. 15 sNfL concentrations were measured in duplicate with the ultrasensitive single molecule array (Simoa) technology (Quanterix). Rather than the raw sNfL concentrations, age, sex, and BMI-adjusted sNfL z scores were used for these analyses as previously shown. 21 The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Results
Baseline variables and balance between groups
The full analysis set included 853 patients: 269 (31.5%) started BCDT (OCR n = 220, RTX n = 49), 454 (53.2%) oral DMT, 57 (6.7%) platform DMT, and 73 (8.6%) NTZ (Table 1). Among the 49 patients who started RTX, 22 subsequently switched to OCR, while 27 remained on RTX up to censoring.
Number of patients included in the study categorized by index DMT.
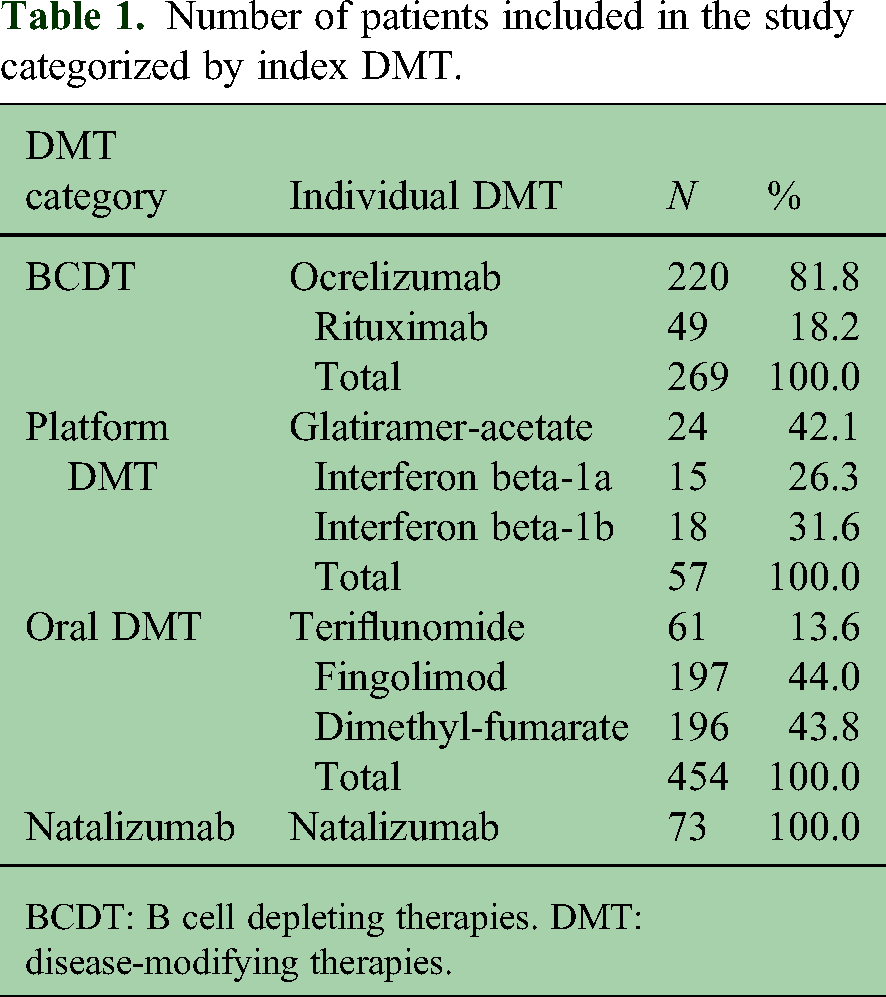
BCDT: B cell depleting therapies. DMT: disease-modifying therapies.
Baseline covariates of patients on BCDT and all other DMT are presented in Table 2, together with SMD before and after IPTW. Patients on BCDT had longer disease duration, higher EDSS scores, and were treated with more DMT before index date. After IPTW, baseline variables were well balanced, with SMD for all variables <0.1.
Demographic and clinical characteristics at baseline (count with % or mean with standard deviation) in patients initiating treatment with BCDT and other DMT, before and after inverse probability of treatment weighting (IPTW).
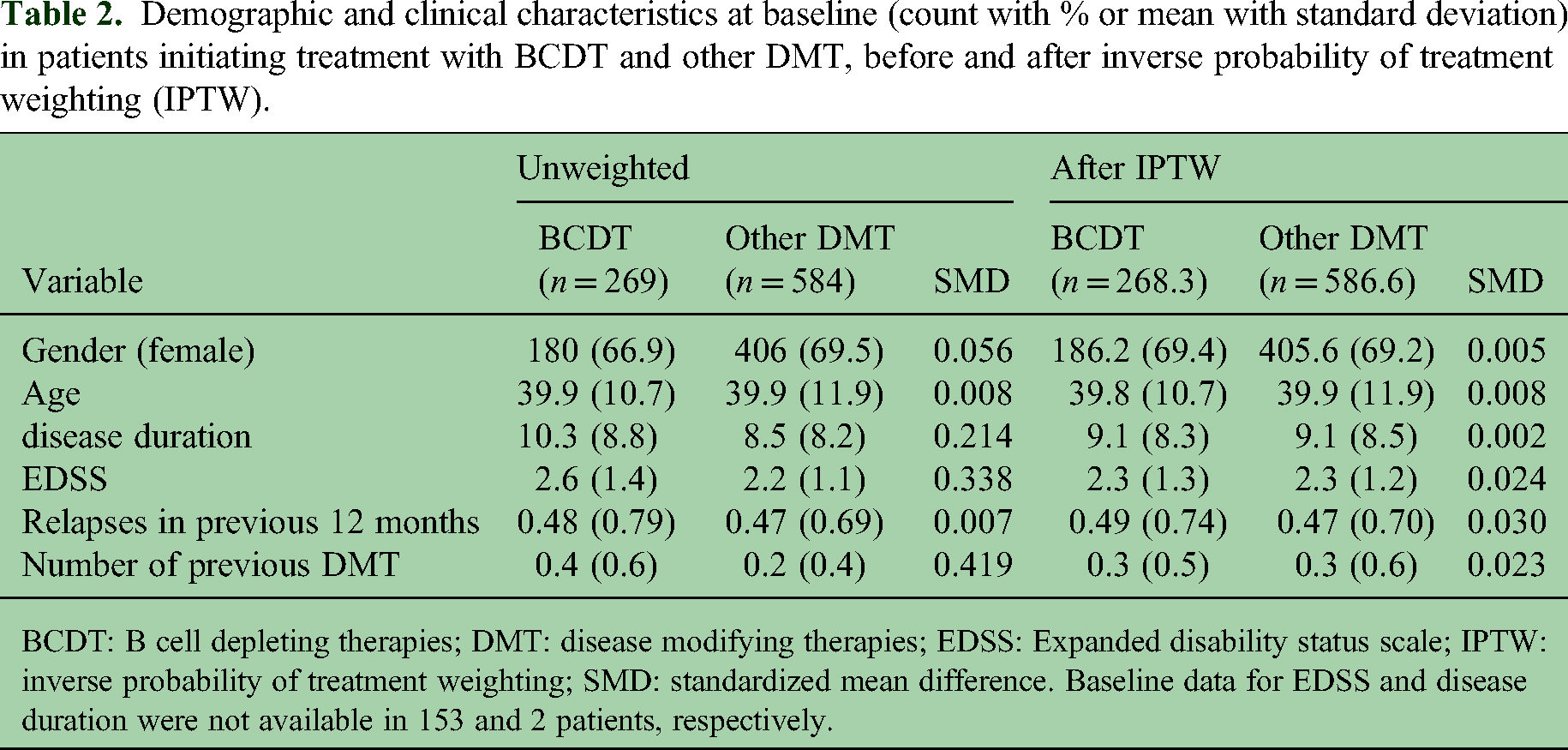
BCDT: B cell depleting therapies; DMT: disease modifying therapies; EDSS: Expanded disability status scale; IPTW: inverse probability of treatment weighting; SMD: standardized mean difference. Baseline data for EDSS and disease duration were not available in 153 and 2 patients, respectively.
Baseline characteristics were also well balanced after IPTW between patients on BCDT and those on oral DMT and NTZ, but not with those on platform DMT (Supplementary Tables 1–3). We therefore did not separately test outcomes for association with BCDT vs platform DMT. Additional baseline characteristics are presented in Supplementary Table 4.
Time to treatment discontinuation
The median (IQR) follow-up time after index date was 4.1 (2.0–5.5) in BCDT, 6.8 (4.1–8.5) in oral DMT, 6.9 (5.4–8.5) in platform DMT and 6.7 (3.1–8.3) years in NTZ treated patients.
In patients under BCDT, there were 39/269 (14.5%) discontinuation events over 944.8 patient years. Among all other DMT, there were 324/584 (55.5%) discontinuation events over 2199.0 patient years. In the model, patients who started BCDT had an approximately 75% lower risk of discontinuing treatment compared to patients under other DMT (HR = 0.26, 95% CI = 0.18–0.36, p < .01). The same was the case when BCDT was tested pairwise versus oral DMT and NTZ (BCDT vs. oral DMT: HR = 0.28, 95% CI = 0.20–0.39, p < .01; BCDT vs. NTZ: HR = 0.35, 95% CI = 0.21–0.58, p < .01, Figure 1).

Kaplan-Meier curves with numbers at risk showing the proportion of patients who remained on treatment with the index DMT over the study period (BCDT in red versus oral DMT in green on the left side, and BCDT in red versus NTZ in blue on the right side). BCDT: B cell depleting therapies; DMT: disease modifying therapies; NTZ: natalizumab.
Time to relapses and disability worsening
In terms of risk of relapse, 34/269 (12.6%) individuals on BCDT experienced a relapse over 806.2 patient-years, as compared to 134/584 (22.9%) individuals on other DMT over 1850.1 patient-years. In the model, BCDT-treated patients had a 48% lower risk of relapses (HR = 0.52, 95% CI = 0.34–0.78, p < .01). When tested pairwise, BCDT was also associated with a lower risk of relapses as compared to oral DMT (HR = 0.59, 95% CI = 0.39–0.88, p = .01), with instead no difference between BCDT and NTZ (HR = 0.90, 95%CI = 0.45–1.82, p = .778, Figure 2).

Kaplan-Meier curves with numbers at risk showing the proportion of patients who remained free of relapses during treatment with the index DMT over the study period (BCDT in red versus oral DMT in green on the left side, and BCDT in red versus NTZ in blue on the right side). BCDT: B cell depleting therapies; DMT: disease modifying treatments; NTZ: natalizumab.
Next, we investigated the relation between the occurrence of relapses and index DMT discontinuation. Among the 39 patients who later discontinued BCDT, only 3 (7.7%) had experienced a relapse between BCDT start and discontinuation (none within 1 year before discontinuation; 0%). Among the 247 patients who later discontinued oral DMT, 71 (28.7%) had experienced a relapse (48 within 1 year before discontinuation; 19.4%). Among the 34 patients who later discontinued NTZ, 5 (14.7%) had experienced a relapse (3 within 1 year before discontinuation; 8.8%).
As to disability, 43/252 (17.1%) individuals on BCDT experienced 6-month CDW over 907.7 patient-years, as compared to 127/516 (24.6%) individuals on other DMT over 2507.1 patient-years, which was not statistically significant (HR = 0.92, 95% CI = 0.64–1.34, p = .683). The risk of disability worsening was marginally lower in patients on BCDT than on oral DMT, with KM curves starting to separate approximately 4 years after index DMT initiation (Figure 3) (HR = 0.85, 95% CI = 0.59–1.23, p = .386). The risk of disability worsening was very similar between BCDT and NTZ (HR = 1.21, 95% CI = 0.63–2.32, p = .577, Figure 3).

Kaplan-Meier curves with numbers at risk showing the proportion of patients who remained free of 6-month CDW stratified by index DMT (BCDT in red versus oral DMT in green on the left side, and BCDT in red versus NTZ in blue on the right side). All EDSS worsening events occurred up to 4 years after termination of index DMT were considered. BCDT: B cell depleting therapies; CDW: confirmed disability worsening; DMT: disease-modifying therapies; NTZ: natalizumab.
Sensitivity analyses
The first sensitivity analysis using an alternative definition of treatment discontinuation (i.e., treatment switch), confirmed the main analysis. Here, 35/269 (13.0%) of patients on BCDT and 297/584 (50.9%) of patients on any other DMT discontinued and switched (HR = 0.24, 95% CI = 0.17–0.35, p < .01). In the second sensitivity analysis, reducing the time gap between infusions from 18 to 12 months to define BCDT discontinuation did not change the results, and patients on BCDT were still less likely to discontinue treatment (HR = 0.41, 95% CI = 0.30–0.56, p < .01).
In further sensitivity analyses, we implemented NfL Z scores as an additional variable included in the IPTW. As compared to all other DMT, patients who started BCDT remained at lower risk of treatment discontinuation (HR = 0.26, 95% CI = 0.18–0.36, p < .01) and relapses (HR = 0.51, 95% CI = 0.34–0.78, p < .01). Time to 6 months CDW was not significantly different between the two groups (HR = 0.93, 95% CI = 0.64–1.36, p = .724).
Reasons for discontinuation
We attempted to further investigate the reasons behind discontinuation of the index DMT. Within the SMSC, it is possible for the neurologist to specify whether DMT discontinuation was related to lack of efficacy, tolerance/side effects, JCV status, and pregnancy. Considering patients with BCDT, oral DMT and natalizumab as index DMT, specific discontinuation reasons were available for 228 out of 320 events.
Discontinuations related to lack of efficacy were 0/39 (0%) for BCDT, 75/247 (30.3%) for oral DMT and 1/34 (2.9%) for NTZ. Discontinuations related to lack of tolerance/side effects were 1/39 (2.6%) for BCDT, 64/247 (25.9%) for oral DMT, and 1/34 (2.9%) for NTZ. Discontinuations related to JCV status were 0/39 (0%) for BCDT, 36/247 (14.6%) for oral DMT (all on either fingolimod or dimethyl fumarate), and 11/34 (32.3%) for NTZ. Discontinuations related to pregnancy were 8/39 (20.5%) for BCDT, 21/247 (8.6%) for oral DMT, and 10/34 (29.4%) for NTZ.
Discussion
This real-world study provides non-randomized evidence that patients starting BCDT are less likely to discontinue therapy at any time over several years of follow-up when compared with platform and oral DMT, as well as NTZ. These results are in line with those provided by other observational studies,11,12 and were also confirmed when we considered the switch to a different DMT and a shorter (12 months) time gap between infusions, as alternative more restrictive definitions of treatment discontinuation.
Novel in present results is that we found suggestive evidence for a link between the superior effectiveness of BCDT in terms of relapse, especially over oral DMT, as an explanation of their more frequent discontinuation. Interestingly, the risk of relapses was similar between BCDT and NTZ suggesting that most probably safety considerations were causing NTZ discontinuation, for example, risk of progressive multifocal leukoencephalopathy. Accordingly, the available data regarding reasons for discontinuations within the SMSC database, showed that lack of efficacy and side effects were the most frequent reason to discontinue oral DMT, whereas JCV status was the most frequently reported reason for NTZ discontinuation.
Compared with the approval trials of OCR in RMS, 3 our cohort was slightly older (Mage = 39.9 vs. 37.0), had longer disease duration (10.3 vs. 6.7 years), and lower mean number of relapses in the 12 months prior to BCDT (0.48 vs. 1.3). Clinically important is that, despite these differences, our results showed a very similar clinical benefit of BCDT in reducing relapse frequency over the entire observation period, hereby confirming previous non-randomized studies.22–24 Similar findings on relapse reduction have also been demonstrated for RTX in randomized and observational studies.5–7,25,26
On the other hand, analyses of time to CDW showed no significant difference between BCDT and comparator therapies. Reduced disability progression in OCR has been demonstrated in clinical trials with controlled settings vs IFNβ-1a, 3 but there is accumulating evidence from the follow-up of these studies, as well as from non-randomized real-world evidence, that disease progression continues. In a recent review of observational studies of OCR with variable follow-up, the percentage of RRMS patients treated with OCR with confirmed disability worsening ranged between 0% and 20%, in line with our findings. 23 Nevertheless, some evidence has shown the risk of disability worsening also depends on how early efficacious treatments are initiated, time gaps between previous treatments and OCR, 27 and likely suboptimal drug exposure by fixed-dosed OCR in function of patients’ various distribution volume. Two randomized trials with double (<75 kg body weights) and triple (≥75 kg body weight) doses of OCR for both for RMS and PPMS patients are under way. 28 While the KM curves related to disability worsening were closely overlapping between BCDT and NTZ, patients starting oral DMT appeared to have more disability worsening events after 4 years of treatment. Despite being not statistically significant, this finding suggests that more time may be needed to observe the full effect of DMT on disability accrual, congruent with evidence of a longer expected therapeutic lag for disability than for inflammatory relapses.19,20
The main strength of the study is the availability as part of the SMSC of a large prospective collection of clinical data over several years of follow-up, which makes it possible to adjust for imbalances in relevant variables between treatment groups outside the context of randomized studies. Among the limitations, we acknowledge first the small sample size of groups on platform DMTs, which is the natural consequence of the availability of more efficacious drugs. Second, pooling of several DMTs in the same group (e.g., the oral DMT group) is also problematic because of their potential different efficacy, safety, and tolerability profiles. Despite being a large cohort, it was not feasible to compare BCDT against each separate DMT and preserve a good balance in baseline characteristics between groups. Third, despite the statistical efforts to correct for imbalances for some potential known confounders, the lack of randomization limits the certainty of evidence as residual bias cannot be excluded and calls for a cautious interpretation of these findings. Finally, it will be interesting to implement in future follow-up studies potential surrogate markers of disability progression and smouldering MS including neurofilament light chain, glial fibrillary acidic protein, and MRI measures.21,29–31
To conclude, MS patients were more persistent to BCDT than to other DMT. The longer persistence on BCDT as compared to oral DMT might be explained by a higher effectiveness in reducing inflammatory disease activity, whereas for NTZ-treated patients we speculate that the higher rate of discontinuation is more likely related to safety reasons (e.g., anti-JCV seroconversion). We highlight the need of pragmatic randomized trials comparing different DMT including BCDT in terms of both efficacy and safety outcomes in the long term.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251315457 - Supplemental material for Treatment persistence and clinical outcomes in patients starting B cell depleting therapies within the Swiss MS Cohort
Supplemental material, sj-docx-1-mso-10.1177_20552173251315457 for Treatment persistence and clinical outcomes in patients starting B cell depleting therapies within the Swiss MS Cohort by Giulio Disanto, Sabine Schaedelin, Johanna Oechtering, Johannes Lorscheider, Riccardo Galbusera, Sebastian Finkener, Lutz Achtnichts, Patrice Lalive, Stefanie Müller, Caroline Pot, Robert Hoepner, Anke Salmen, Chiara Zecca, Lars G Hemkens, Marcus D’Souza, Bettina Fischer-Barnicol, Renaud Du Pasquier, Patrick Roth, Özgür Yaldizli, Maximilian Einsiedler, Tobias Derfuss, Ludwig Kappos, Claudio Gobbi, Cristina Granziera, Marjolaine Uginet, Aleksandra Maleska Maceski, Keltie McDonald, David Leppert, Pascal Benkert, Jens Kuhle and in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgments
The authors would like to thank all participants, study nurses, technicians, and physicians for their engagement in the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was supported by Roche. The SMSC receives grant funding from the Swiss Multiple Sclerosis Society and Biogen, Bristol Myers Squibb, Merck, Novartis, Roche.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
