Abstract
Background
Inflammatory and neurodegenerative processes are hallmarks of multiple sclerosis (MS). The synthesis of the major stress-inducible heat shock protein 70 (Hsp70) is induced by inflammation.
Objective
The purpose of this study is to determine whether Hsp70 in serum can serve as a potential biomarker to distinguish inflammatory and neurodegenerative processes in MS.
Methods
Serum was obtained from 94 patients: 26 clinically isolated syndrome (CIS), 40 relapsing–remitting MS (RRMS), 19 secondary progressive MS (SPMS), and nine primary progressive MS (PPMS). As controls, serum samples were collected from patients with non-inflammatory neurological diseases (NINDs, n = 41), other inflammatory neurological diseases (OINDs, n = 28) and healthy donors (HDs, n = 114). Serum levels of Hsp70 were quantified using the enzyme-linked immunosorbent assay detecting free and liposomal Hsp70 (lipHsp70 ELISA).
Results
Patients with MS displayed significantly higher Hsp70 serum levels than HDs (p < 0.001) and significantly lower levels than OINDs (p = 0.001). A subgroup analysis revealed that Hsp70 serum levels of CIS/RRMS patients are significantly higher than those of patients with progressive MS (SPMS/PPMS) (p < 0.05).
Conclusion
Inflammation causes the release of Hsp70 into the blood. As CIS/RRMS are associated with higher Hsp70 serum levels than progressive MS, serum Hsp70 levels might provide a marker for inflammatory processes.
Introduction
MS is considered as a chronic inflammatory disease of the central nervous system (CNS) that is characterised by inflammation, demyelination, gliosis and neurodegeneration. 1 It is widely believed that the underlying pathomechanism of relapsing–remitting MS (RRMS), including clinically isolated syndrome (CIS) and progressive forms of MS (secondary progressive MS (SPMS)/primary progressive MS (PPMS)), differ. In RRMS, the disease seems to be driven by peripheral immune responses targeting the CNS. In progressive MS compartmentalised immune responses within the CNS and secondary neurodegeneration appear to play a key role in disease progression 2 whereas within the group of progressive subtypes, PPMS seems to go along with less inflammation than SPMS. 3
Heat shock proteins (HSPs) are molecular chaperones which are highly conserved among different species from bacteria to humans. 4 Depending on their molecular weights, HSPs can be grouped into the following main families: HSP40, HSP60, HSP70, HSP90, HSP100 5 and the small HSPs with a molecular weight of approximately 20 kDa. 6 The human HSP70 family comprises a variety of different isoforms, with molecular weights ranging between 66 kDa and 78 kDa. 7 The main representatives of the HSP70 family are the constitutively expressed Hsc70 (73 kDa) and the major stress-inducible Hsp70 (72 kDa). 8
Hsp70 is predominantly localised in the cytosol 7 but is also found in the extracellular space.9,10 In malignantly transformed tumour cells and virally infected cells,11,12 Hsp70 is present on the plasma membrane. The expression of Hsp70 is highly upregulated upon a large variety of stress stimuli including thermal stress 13 , anoxia, ethanol, heavy metals, 4 inflammation, 14 infection and tissue injury. 15 Depending on its intra- or extracellular location, Hsp70 fulfils different functions. Intracellular Hsp70 protects cells against lethal damage induced by stress, supports the synthesis and transport of other proteins, and prevents misfolding and aggregation.7,8 Extracellular and cell surface-bound Hsp70 plays an essential role in eliciting immune responses. It can support specific immune reactions by assisting antigen presentation 16 and it has been shown to activate natural killer cells 17 in the presence of interleukin 2. 18 Furthermore, it can modulate the immune defence by triggering the transcription and release of pro-inflammatory cytokines. 19
Hsp70 also has been assumed to play a role in the development of MS.20,21 It is known that Hsp70 knock-out mice are better protected against the development of experimental autoimmune encephalomyelitis (EAE), which represents an animal model of MS. 22 In contrast to these findings, elevated intracellular Hsp70 levels can also exert beneficial effects for MS by protecting cells within the CNS against apoptotic cell death. 20
To date only Hsp70 gene polymorphisms and intracellular Hsp70 levels in peripheral blood mononuclear cells (PBMCs) have been studied in MS patients. 23 Different publications show a great diversity of intracellular Hsp70 levels in PBMCs of MS patients ranging from low 24 to normal 25 to elevated levels. 26 In the extracellular milieu, only Hsp70-specific antibodies 20 but not Hsp70 proteins have been determined in the serum of MS patients.
The aim of this study was to measure Hsp70 protein levels in the serum of patients with different MS subtypes and inflammatory diseases. It was further assessed whether extracellular Hsp70 can serve as a potential biomarker to distinguish inflammatory and neurodegenerative processes in MS. For this purpose, serum of patients with MS, non-inflammatory neurological diseases (NINDs), other inflammatory neurological diseases (OINDs), and healthy control donors (HDs) was collected.
Materials and methods
Sample collection and enzyme-linked immunosorbent assay (ELISA) experiments
All blood samples of patients (n = 163) were collected between 2008 and 2016 by the Department of Neurology, Klinikum rechts der Isar, TU München (TUM). After centrifugation, serum samples were aliquoted and immediately stored at –80°C. The Hsp70 levels of an age- and gender-matched control group (n = 114) was provided by the Department of Radiation Oncology, Klinikum rechts der Isar, TU München (TUM). The data of this control group have been published before, 27 but several samples of HDs were re-analysed with identical results in parallel to the patient samples. Informed written consent was provided by all patients and healthy donors. The diagnoses of the patients were obtained from medical records. Approval of the study protocol was granted by the ethical committee of the Klinikum rechts der Isar, TUM.
Serum samples of a discovery cohort consisting of MS, NIND and OIND patients (n = 87) and an independent validation cohort (n = 76) were measured blinded. Since both cohorts showed similar results as illustrated in supplementary Figures 1 and 2, mean values of both cohorts were summarised and used for further analyses. Lipid-bound and free Hsp70 in the serum was quantified by using the lipHsp70 ELISA following the protocol described previously in detail. 27 The Hsp70 results represent the means of up to three independent experiments, each measured in duplicates ± standard deviation (SD). The mean intra- and inter-plate coefficients of variation were 5.1% and 16.3%, respectively.
Patient characteristics, therapy and magnetic resonance imaging (MRI)
The NIND group comprises patients with epilepsy (n = 1), head pressure (n = 1), headache (n = 25), lumbosciatica (n = 2) and pseudotumour cerebri (n = 12). The OIND group is composed of patients with encephalitis (n = 2), herpes zoster neuritis (n = 2), meningitis (n = 15) and meningoencephalitis (n = 9).
The group of MS patients (n = 94) can be subdivided into CIS (n = 26), RRMS (n = 40), SPMS (n = 19), and PPMS (n = 9). For diagnosis of MS the revised McDonald criteria from 2005 28 and 2010 29 were applied. Three patients with CIS/RRMS received high-dose methylprednisolone (MPS) therapy over a period of five to six days. Five additional patients received glucocorticoids due to a relapse (n = 4) or an uncertain relapse (n = 1) within the last 30 days before blood sampling. Twenty-two patients diagnosed with CIS/RRMS received disease-modifying therapy (DMT) (interferon substances, glatiramer acetate or natalizumab).
MRI was performed at the time point of blood sampling in 29 patients. A new lesion is defined as a gadolinium enhancement in either spinal cord or brain, as detected by MRI scan. A patient with no new lesions is characterised by the absence of gadolinium enhancement in the spinal cord and brain, as detected by MRI scan.
Statistical analyses
Statistical analyses were performed using MATLAB R2015a (MathWorks Inc, Natick, MA, USA). A comparison of two subgroups was performed by using the Student’s t test; a comparison of more than two subgroups was performed with the analysis of variance test using the Bonferroni method as post hoc analysis. In order to identify potential correlations between Hsp70 levels, age and gender multiple linear regression analyses with the diagnoses as an additional independent variable were performed. A p value of <0.05 was defined as statistically significant.
Results
Clinical characteristics of patients and HDs
In this study, Hsp70 protein levels were quantified in the serum of a total of 163 patients (MS, n = 94; NINDs, n = 41; OINDs, n = 28). Patients with MS were further subclassified into CIS (n = 26), RRMS (n = 40), SPMS (n = 19) and PPMS (n = 9). Table 1 summarises the clinical characteristics of patients in the discovery and validation cohort and HDs.
Patient characteristics.
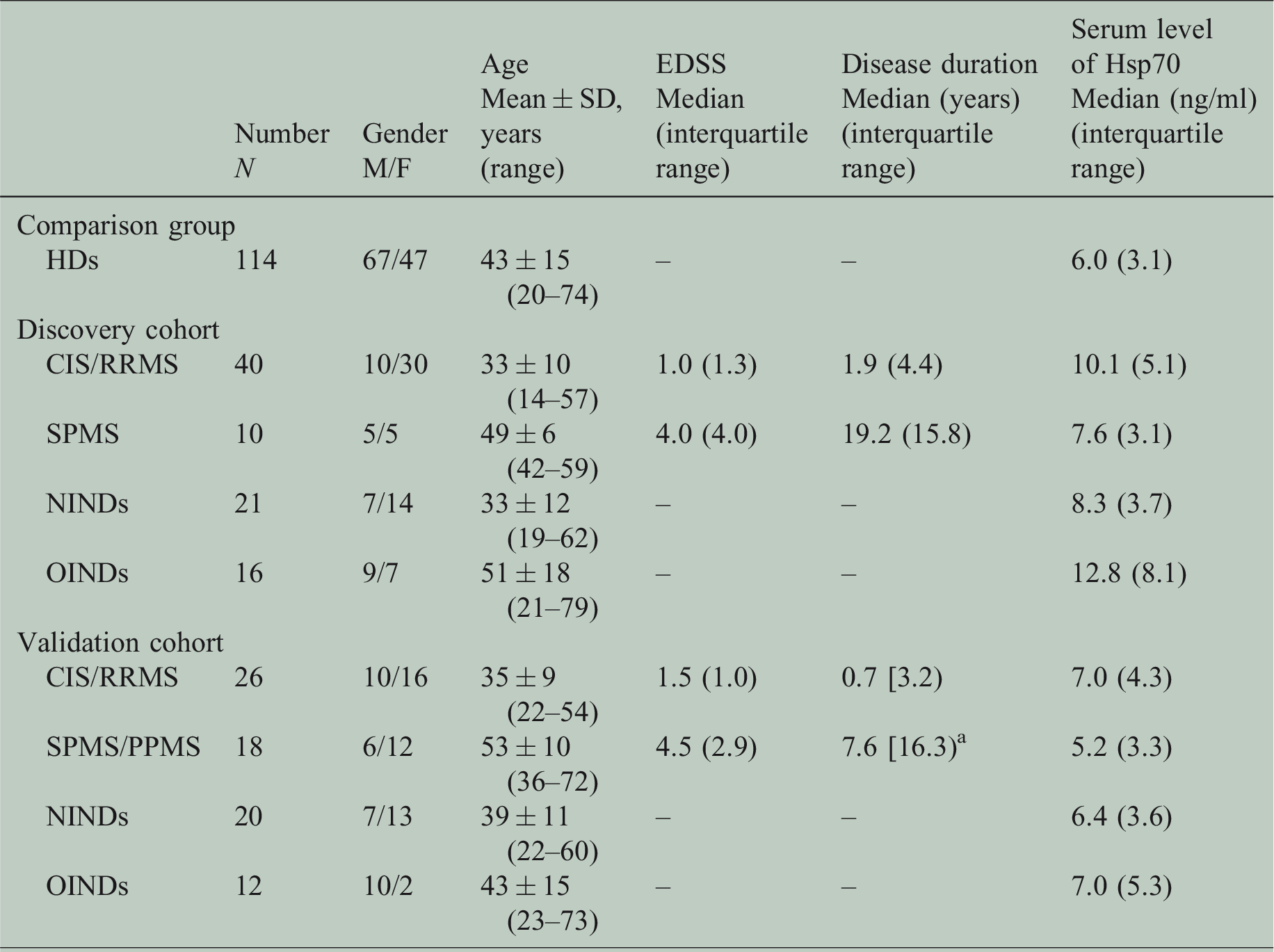
The disease duration is the time between initial manifestation and blood sampling.
aThe information about initial manifestation was missing from four patients.
CIS: clinically isolated syndrome; EDSS: Expanded Disability Status Scale; F: female; HDs: healthy donors; Hsp70: heat shock protein 70; M: male; NINDs: non-inflammatory neurological diseases; OINDs: other inflammatory neurological diseases; PPMS: primary progressive multiple sclerosis; RRMS: relapsing–remitting multiple sclerosis; SD: standard deviation; SPMS: secondary progressive multiple sclerosis.
Regarding age distribution, the four main groups (HDs, MS, NINDs and OINDs) were in a similar age range. Nevertheless, within the subgroups of MS, patients with SPMS and PPMS were, as expected, older than those with CIS and RRMS (Table 1).
Hsp70 serum levels are not influenced by age and gender
The role of age and gender as potential parameters that could potentially influence Hsp70 serum levels was evaluated. Regression analyses displayed no age- (p = 0.435) or gender-related (p = 0.422) differences in Hsp70 serum levels.
Inflammation causes increased Hsp70 serum levels
In order to evaluate the general influence of inflammatory processes on Hsp70 serum levels, Hsp70 was measured in patients with NINDs and OINDs (Figure 1). Patients with OINDs displayed significantly higher Hsp70 serum levels compared to HDs (p < 0.001) and NINDs (p < 0.001), whereas no significant differences were determined in Hsp70 serum levels of HDs and NIND patients (p = 0.383).
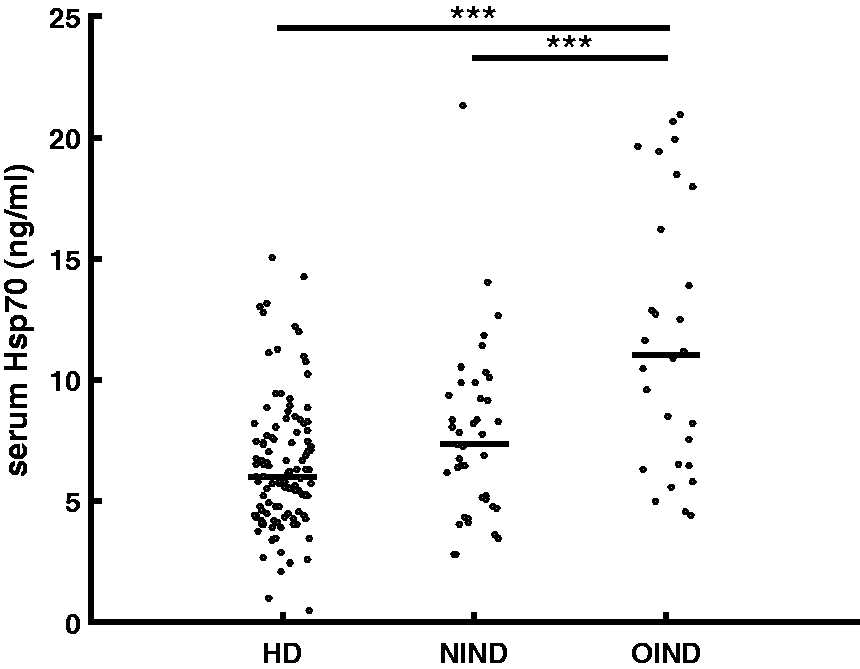
Serum Hsp70 levels of HDs compared to patients with NINDs and OINDs. Patients with OINDs (n = 28) display significantly higher Hsp70 serum levels than HDs (n = 114, p < 0.001) and patients with NINDs (n = 41, p < 0.001). HDs: healthy donors; Hsp70: heat shock protein 70; NINDs: non-inflammatory neurological diseases; OINDs: other inflammatory neurological diseases. Each data point represents one patient. The lines show the median value.
Furthermore, we evaluated whether MS patients suffered from additional diseases (data not shown). As documented in the medical records, 91 out of 94 MS patients showed no additional inflammatory disease or cancer at the time point of blood sampling. Three MS patients with pre-existing inflammatory conditions several years ago had low serum Hsp70 levels. Therefore, we speculate that elevated Hsp70 serum levels are mainly caused by MS-induced inflammatory processes.
Hsp70 expression varies in different MS subtypes
Comparison of Hsp70 serum levels in HDs and MS patients revealed significantly elevated Hsp70 levels in the serum of MS patients (p < 0.001; Table 2). Furthermore, the Hsp70 serum levels of MS patients were significantly lower compared to patients with OINDs (p = 0.001). Regarding the different subtypes, CIS/RRMS displayed significantly higher levels than the progressive forms (SPMS/PPMS) (p < 0.05; Figure 2(a)). With respect to the four different subtypes of MS, CIS and RRMS showed highest Hsp70 serum levels followed by SPMS and PPMS patients (Figure 2(b)). Hsp70 serum levels of CIS (p = 0.001) and RRMS (p < 0.001) patients were significantly higher than those of HDs, whereas serum levels of SPMS, who displayed lower Hsp70 levels, did not show significant differences compared to HDs (p = 0.831). Regarding PPMS, the Hsp70 serum levels were similar to those of HDs (p=1.000) (Figure 2(b)).
Serum Hsp70 levels related to the different diagnoses.

The disease duration is the time between initial manifestation and blood sampling.
aThe information about initial manifestation was missing from two patients.
P values refer to comparisons to HD and were calculated with the analysis of variance test using the Bonferroni method as post hoc analysis. A p value <0.05 is defined as statistically significant.
CIS: clinically isolated syndrome; F: female; HDs: healthy donors; Hsp70: heat shock protein 70; M: male; MS: multiple sclerosis; PPMS: primary progressive multiple sclerosis; RRMS: relapsing–remitting multiple sclerosis; SD: standard deviation; SPMS: secondary progressive multiple sclerosis.
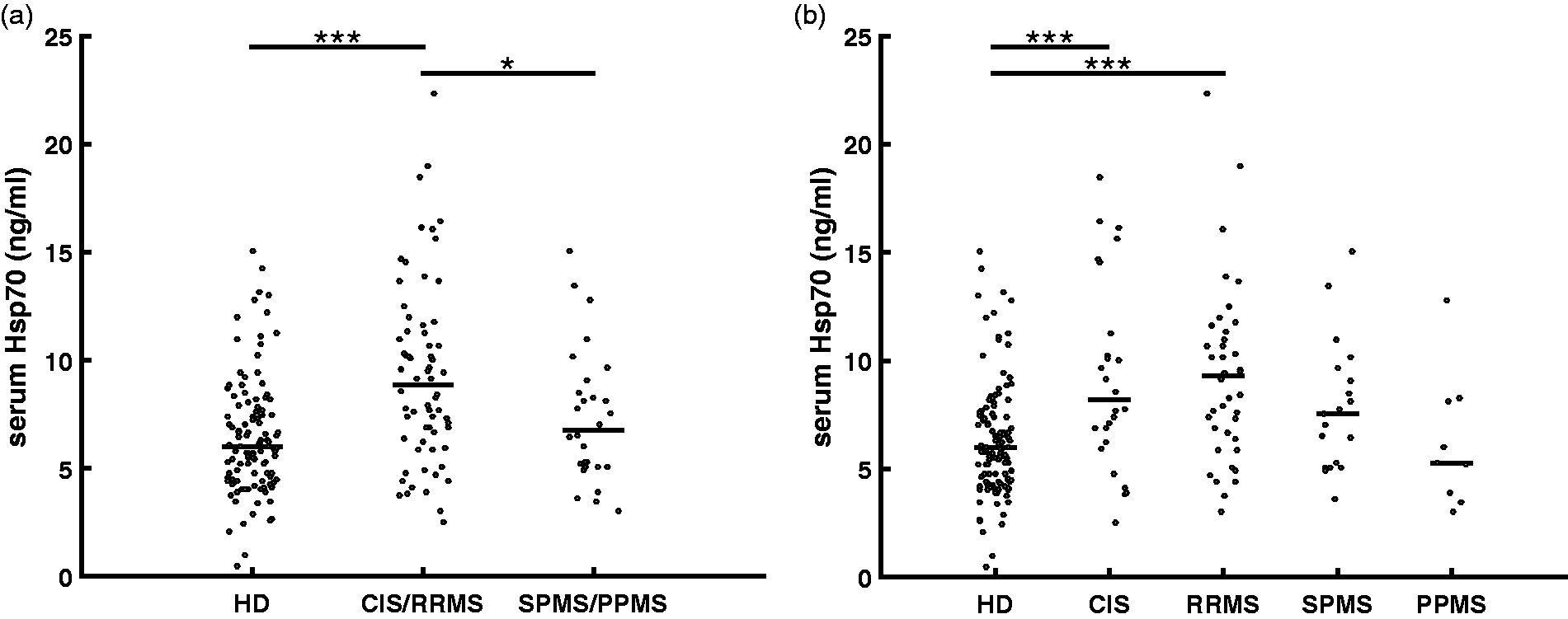
Serum Hsp70 levels of HDs compared to subgroups of MS patients. (a) Samples of patients with CIS/RRMS (n = 66) display significantly higher levels than HDs (n = 114, p < 0.001) and SPMS/PPMS (n = 28, p < 0.05). (b) Samples of patients with CIS (n = 26, p = 0.001) and RRMS (n = 40, p < 0.001) have significantly higher Hsp70 serum levels than HD (n = 114). SPMS (n = 19) and PPMS patients (n = 9) display a reduced trend toward lower levels compared to CIS and RRMS. Both showed no significant differences in the Hsp70 serum levels compared to HDs. CIS: clinically isolated syndrome; HDs: healthy donors; Hsp70: heat shock protein 70; MS: multiple sclerosis; PPMS: primary progressive multiple sclerosis; RRMS: relapsing–remitting multiple sclerosis; SPMS: secondary proressive multiple sclerosis. Each data point represents one patient. The lines show the median value.
Further analysis of disease activity parameters of the CIS and RRMS groups revealed that 36 patients were in remission and 27 patients had experienced a relapse within the last 30 days before blood sampling. A comparison of both groups showed no significant difference with respect to their Hsp70 serum levels (p = 0.249; Table 3). We also detected no significant differences in Hsp70 serum levels in patients with newly detected gadolinium-enhanced lesions (n = 21) compared to patients with no detectable new lesions in MRI scans of the brain and spinal cord (n = 8) (p = 0.628; Table 3).
Serum Hsp70 level related to disease activity parameters and disease-modifying therapy.

P values refer to comparisons between each pair and were calculated with the Student’s t test. A p value <0.05 is defined as statistically significant.
Hsp70: heat shock protein 70.
Stable Hsp70 levels in the serum were demonstrated in five out of six patients who were diagnosed with RRMS over a period of time ranging from 22 to 1216 days (Figure 3).

Serum heat shock protein 70 (Hsp70) levels over longer time periods. Follow-up samples over a period of time ranging from 22 to 1216 days were measured. Levels remained stable over time in five out of six patients diagnosed with relapsing–remitting multiple sclerosis. The graph represents mean values ± standard deviation (SD).
Responders to high-dose cortisone therapy show decreasing Hsp70 serum levels
Three patients with CIS/RRMS and an acute relapse were recruited to determine the influence of high-dose MPS therapy on Hsp70 serum levels. The first blood sample was taken immediately before start of therapy (day 1); the second blood sample was taken on the next day (day 2) after the first and second intravenous injection of MPS (1g). The following blood samples were taken every next day after the daily drug administration. As shown in Figure 4(a), the Hsp70 serum values of two patients who responded to MPS therapy by a regression of symptoms significantly decreased (41% and 52%, respectively) after the whole treatment cycle. However, the mean (8.1 ± 1.2 ng/ml, 10.8 ± 0.7 ng/ml) and median (8.2 and 10.7 ng/ml) Hsp70 values still remained elevated after therapy compared to those of HDs (mean 6.4 ± 2.7 ng/ml; median 6.0 ng/ml). In contrast, the Hsp70 serum level of a third patient showing only minor responses to steroid treatment increased (Figure 4(b)).
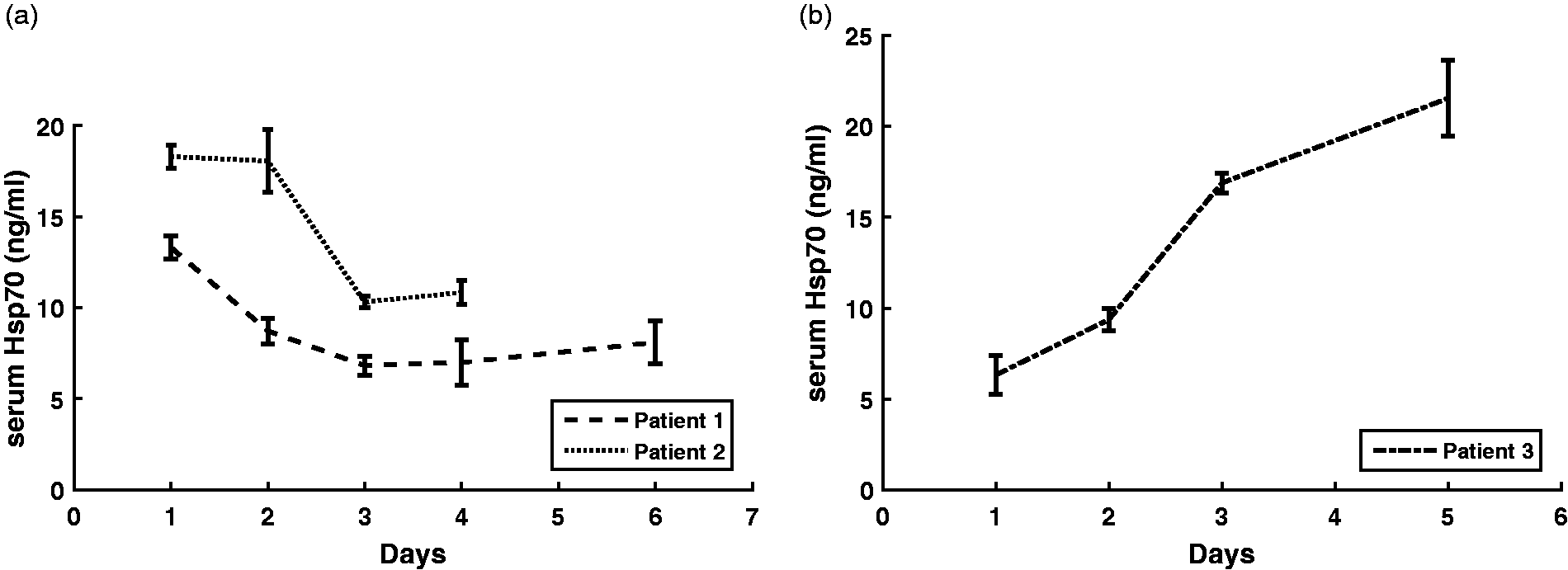
Serum heat shock protein 70 (Hsp70) levels of relapsed multiple sclerosis patients before and during high-dose cortisone therapy. Day 1 reflects the serum Hsp70 value of patients with acute relapse before start of the treatment. Directly thereafter the patient received a dose of 1 g methylprednisolone intravenously, which was repeated every day thereafter. The blood samples were collected after the daily drug administration. (a) Kinetics of serum Hsp70 levels of two patients showing good clinical responses to the steroid treatment. (b) Kinetics of serum Hsp70 levels of a patient showing only a minor clinical response to the steroid treatment. Data show the mean value of two independent experiments measured in duplicates ± standard deviation (SD).
A cohort of 22 patients with CIS/RRMS receiving DMT displayed no significant differences in serum Hsp70 levels (p = 0.171) compared to CIS/RRMS patients with no DMT (n = 44) (Table 3). Nevertheless, both subgroups showed significantly higher Hsp70 serum levels than HDs (p < 0.001, p < 0.001).
Discussion
In this study, the question was addressed whether serum Hsp70 levels, as determined by the lipHsp70 ELISA, can serve as a biomarker to distinguish inflammatory and neurodegenerative processes in MS patients. Patients with OINDs and MS displayed significantly higher Hsp70 serum levels than HDs. In patients with MS Hsp70 serum levels were found to be lower than in OIND patients. This finding can be explained by the fact that inflammation in MS is generally less pronounced compared to other inflammatory diseases of the CNS. In line with these findings, we were able to show that the anti-inflammatory effect of high-dose MPS therapy in a small cohort of MS patients with acute relapse was accompanied by a decrease in Hsp70 serum levels, although the mean and median Hsp70 values remained elevated compared to HDs. Nevertheless, the decreased Hsp70 serum levels under MPS therapy were determined in only two patients and, therefore, further studies with larger patient cohorts are needed.
Since it appears that Hsp70 serum levels remain stably elevated over a longer period of time, we hypothesise that increased Hsp70 serum levels are not directly caused by an acute exacerbation, but may be the result of a chronically stressed peripheral immune system. This notion is supported by our results that acute clinical relapses and the detection of gadolinium-enhanced lesions by MRI in the brain and spinal cord were not associated with a further increase in serum Hsp70 levels.
Hsp70 serum levels did not change significantly with age and gender. Therefore, the lower Hsp70 levels in patients with SPMS and PPMS compared to CIS and RRMS cannot be explained by an unequal distribution of age in the different patient groups, but rather are triggered by different pathomechanisms. High Hsp70 serum levels in CIS and RRMS and low Hsp70 levels in SPMS and PPMS most likely reflect the degree of systemic inflammation. However, future studies with larger patient cohorts are necessary to confirm this observation.
The origin of Hsp70 in the serum, however, could not be specified as it is released by both living and dead cells. 30 Nevertheless, since Hsp70 serum levels decreased in patients who responded to high-dose cortisone therapy, we speculate that Hsp70 might originate from activated immune cells such as T or B cells. As MS is believed to be predominantly mediated by T 31 and B cells, 32 we hypothesise that these cells might be considered as a possible source for elevated Hsp70 serum levels. The fact that liposomal Hsp70, which is believed to be actively released by living cells, 19 is increased during inflammation, underpins this assumption. However, additional studies are needed to identify the origin of extracellular Hsp70 in the serum of MS patients.
Concerning the role of Hsp70 in the pathogenesis of MS, presently there is no general consensus as to whether Hsp70 mediates beneficial or harmful effects. High cytosolic Hsp70 levels, which are known to exert cytoprotective functions, might reduce cell death in brain cells and thus elevated cytosolic Hsp70 levels might be considered beneficial for the treatment of MS. 20 In the extracellular milieu, however, it is assumed that Hsp70 can activate and enhance inflammatory processes. Therefore, medications that can inhibit the production and release of Hsp70 might also provide a potential treatment strategy for MS. 22 In an effort to personalise this therapeutic approach, it is necessary to know the actual Hsp70 serum levels of an individual patient during the course of disease. Patients with high basal Hsp70 serum levels might respond better to Hsp70-reducing drugs than patients with low levels. As a result, serum Hsp70 levels could provide a useful biomarker in the future for monitoring inflammatory processes in MS and for an adaptation of therapy. In order to confirm serum Hsp70 levels as a diagnostic biomarker, further prospective studies are warranted.
Supplemental Material
Supplemental material for Serum heat shock protein 70 levels as a biomarker for inflammatory processes in multiple sclerosis
Supplemental material for Serum heat shock protein 70 levels as a biomarker for inflammatory processes in multiple sclerosis by Patricia Lechner, Dorothea Buck, Lisa Sick, Bernhard Hemmer and Gabriele Multhoff in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
The authors would like to thank Verena Grummel for selecting the blood samples and for providing patient data.
Conflicts of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Bernhard Hemmer has served on scientific advisory boards for F. Hoffmann-La Roche Ltd, Novartis, Bayer Schering, AllergyCare and Genentech; has received speaker honoraria from Biogen Idec, Teva Neuroscience, Merck Serono, Medimmune, and F. Hoffmann-La Roche Ltd; has received research support from Chugai Pharmaceuticals; and holds part of two patents: one for the detection of antibodies and T cells against KIR4.1 in a subpopulation of MS patients and one for genetic determinants of neutralising antibodies to interferon-beta. Dorothea Buck has received compensation for activities with Bayer HealthCare, BiogenIdec, Merck Serono, and Novartis. She is supported by the Abirisk Consortium. The other authors have nothing to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Research Foundation (DFG) and the Technische Universität München within the funding programme Open Access Publishing. Gabriele Multhoff’s laboratory is funded by grants from the DFG (SFB824/3, STA1520/1-1), German Federal Ministry of Research and Education (BMBF; 01GU0823, 02NUK031B, 02NUK038A), BMWi (AiF project GmbH), by the Cluster of Excellence: Munich Centre for Advanced Photonics (MAP), Alexander von Humboldt stipend, and by the German Cancer Consortium Radiation Oncology Group Munich (DKTK-ROG). Bernhard Hemmer’s laboratory is supported by grants from the DFG (Transregio 128), the BMBF (Competence Network Multiple Sclerosis) and the European Union project MultipleMS.
Supplementary material
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
