Abstract
Background
SPRINT-MS was a placebo-controlled phase 2 trial of ibudilast in secondary and primary progressive multiple sclerosis. The trial included multimodal imaging to assess brain tissue integrity. This contribution focuses on improved analysis methods of diffusion tensor imaging to refine its application in clinical trials.
Objective
Reassess diffusion tensor imaging from the SPRINT-MS trial.
Methods
Postprocessing incorporated corrections for bulk motion, eddy current distortion, outlier replacement, and intra-volume movement. The ICBM-DTI-81 white matter parcellation map was coregistered into native space. Six unilateral and 21 bilateral regions of interest were identified. Median radial diffusivity was the primary outcome measure for this analysis. A linear mixed-effects model was used to assess the interaction between time and treatment for the outcome measure with Holm correction for multiple comparisons.
Results
Radial diffusivity in the cingulum and cerebellar peduncles showed a significant difference in rate of change between treatment and placebo groups (2.7–7.4 × 10−3 mm2/s per 24-week time period, p < 0.04). Radial diffusivity was unchanged (declined) in the treatment (placebo) groups, consistent with preservation (deterioration) of tissue integrity.
Conclusion
Our results suggest that the diffusion tensor imaging of the cingulum and cerebellar peduncles may be useful target outcome metrics in neuroprotective trials in progressive multiple sclerosis.
Keywords
Introduction
SPRINT-MS was a multicenter, double-blind, placebo-controlled phase 2 trial of ibudilast in secondary and primary progressive multiple sclerosis (MS). 1 The trial met its primary outcome, finding a 48% slowing in the progression of whole brain atrophy in the ibudilast-treated subjects. Ibudilast also showed a benefit on key secondary outcomes, cortical atrophy and magnetization transfer, in normal-appearing brain tissue. 1 Subsequent analyses by different groups found a benefit of ibudilast on gray matter atrophy, 2 slowly expanding lesions, 3 and ganglion cell + inner plexiform layer (GCIPL) as measured by optical coherence tomography. 4
Despite a benefit of ibudilast on these imaging measures of tissue injury, no benefit was seen on diffusion tensor imaging (DTI). 1 DTI provides quantitative measures of tissue integrity on a voxel-wise basis, 5 thereby providing more granular information than summary measures such as whole-brain atrophy. The preplanned DTI analysis for the SPRINT-MS trial focused on radial diffusivity and axial diffusivity in the corticospinal tracts based on their large size and clinical relevance, but not based on clinical trial performance. After motion correction and eddy current distortion correction, 6 the corticospinal tracts were identified by probabilistic tractography, 7 connecting hand-drawn regions of interest (ROIs) on the hand knob of motor cortex and on the midbrain.
Given the benefit of ibudilast on many imaging measures but not DTI, we examined alternative image analysis methods to see if a benefit of ibudilast is seen using DTI, with the ultimate goal of applying the refined method to future MS therapeutic clinical trials. Initial postprocessing used software8,9 that, in addition to motion correction and eddy current distortion correction, could also account for outliers 10 and within-volume motion. 11 To enable investigation of regions other than corticospinal tracts, we applied an atlas-based approach. 12
Methods
Study population
Patients with secondary or primary progressive multiple sclerosis were enrolled and followed at 27 sites across the US. Imaging was scheduled to occur at five times: baseline and 24, 48, 72, and 96 weeks. Baseline images were acquired prior to initiation of treatment. Of the 255 who were randomized to treatment or control arms of the study, 244 completed more than one imaging session. More comprehensive details about the study population can be found by Fox et al. 13
Imaging
All magnetic resonance imaging (MRI) scanners were 3 T and manufactured by Siemens (Trio, Skyra) or GE (Signa HDxt, Signa EXCITE, DISCOVERY MR750, DISCOVERY MR750 W). Detailed information about scanner models, software versions, and scan parameters is provided by Zhou et al. 14 Included in each scan session was a DTI acquisition (axial, FOV = 255 mm × 255 mm × 150 mm. 2.5 mm isotropic voxels acquired, 64 b = 700 s/mm2 volumes, eight b = 0 s/mm2 volumes. Approximately eight b = 0 s/mm2 volumes are recommended per diffusion-weighted volume for tensor estimation. 15 On GE systems, images were interpolated by the scanner to 1 mm × 1 mm in-plane resolution with no through-plane interpolation. TE = 69/80/93 ms on Siemens Skyra/Siemens Trio/all GE. TR = 7000/7400/8500 ms on Siemens Skyra/Siemens Trio/all GE. TA = 8.4/8.9/10.2 min on Siemens Skyra/Siemens Trio/all GE). Other imaging, not used in this study, included axial T1-weighted, proton-density weighted, T2-weighted, FLAIR, and matched spoiled gradient echo with and without magnetization transfer weighting for calculating magnetization transfer ratio (MTR).
Image post processing
Images were inspected visually for artifacts as part of the quality control process. For this study, we incorporated a number of advances in image analysis that were not available at the time of the original trial. Motion and eddy current distortion correction were performed with FSL/eddy,8,9 including outlier correction 10 and correction for within-volume motion. 11 As no fieldmap or reverse phase encoding scan was acquired, susceptibility distortion correction was not performed. FSL/dtifit was then used to calculate the diffusion tensor-based measures of tissue integrity in each voxel—radial diffusivity (RD), axial diffusivity (AD), fractional anisotropy (FA), and mean diffusivity (MD). Each FA map was directly registered to the JHU-ICBM FA template 16 using the antsRegistrationSyN.sh script, with default values off all parameters, provided by ANTs (transform type: rigid, affine and nonlinear deformable SyN. Similarity metric is mutual information for the rigid and affine stages, cross-correlation for the deformable SyN stage), 17 and the inverse transform was applied to the ICBM-DTI-81 white matter parcellation map (WMPM) to place six unilateral and 21 bilateral white matter ROIs in native space. Alignment of the WMPM to the FA maps in native space was inspected visually. The median, across voxels in each ROI, of RD, AD, FA, and MD was calculated. The JHU-ICBM FA template and WMPM are provided with FSL. 8 The use of ANTs for such voxel-based analysis has been found to perform better than tract-based spatial statistics (TBSS).12,18 Images were organized into the brain imaging data structure (BIDS) 19 to facilitate future analyses and data sharing.
Statistical analysis
The null hypothesis was that DTI measures do not differ over time for subjects in the treatment vs. control arms of the study. The primary outcome is the difference between treatment and placebo groups in the rate of change of RD in each ROI. AD, FA, and MD were also examined as secondary outcomes. To test this hypothesis, the relationship between time and DTI measures was assessed with a linear mixed effects model with interaction between time and treatment. Each DTI measure (RD, AD, FA, and MD) of each ROI was modeled separately. The summary statistic for each DTI measure was the median across voxels in each ROI. Time was treated as a continuous variable, and it was assumed that data were missing at random. Participants with missing data were included in the analysis as long as they completed at least one imaging session. All analyses were conducted using R version 4.3.1. 20
Among the ROIs, some are bilateral, for example, the right and left corticospinal tracts. For bilateral ROIs, a likelihood ratio test was performed to determine if the random effects structure should include only a random intercept for each individual or separate random intercepts for subject and subject-side combinations. This test indicated that including both random intercepts significantly enhanced the model's predictive ability. Consequently, for bilateral ROIs, two random intercepts were included. For unilateral ROIs (e.g. body of corpus callosum), models included only a random intercept for each subject. There are 21 bilateral ROIs and six unilateral ROIs.
RD was chosen as the primary outcome for this study because it has been shown to correlate with demyelination in animal 21 and human 22 imaging-pathology validation studies. RD is therefore relevant to MS, a demyelinating disease, and an increase in RD is thought to reflect, in part, myelin integrity, and changes over time can be interpreted as being correlated with changes in myelin integrity. RD was therefore chosen as an outcome for the SPRINT-MS trial and continues to remain relevant. 1 However, the correspondence between myelin integrity and RD should be approached with caution due to limitations of the tensor model 23 and contributions of other cellular structures as well as physiological factors such as edema, inflammation, and complex fiber geometries. 24 Although reduction in AD can indicate axonal injury, such reductions seem to only reflect acute, not chronic injury. 22 Although commonly used in DTI studies, FA and MD are mathematically derived from the same three eigenvalues that are used to derive RD and AD, introducing inherent correlations that affect the statistical power of a study.
To address multiple comparisons due to the simultaneous testing of 27 primary outcome variables (RD of 27 regions), a Holm correction was performed. 25 The difference in the rate of change (slope) of RD between the treatment and placebo groups was derived from the fixed-effect coefficient for the interaction term of time and group in 27 separate models, one for each of the six unilateral and 21 bilateral ROIs. The correction was implemented using the p.adjust function in R. 20 No corrections for multiple comparisons were performed for AD, MD, and FA because they were not primary outcomes.
Results
Of the 244 who completed more than one scan session, 236 had DTI that passed quality control at the second time point (24 weeks). At subsequent time points, fewer subjects had DTI due to a combination of either dropping out of the study, failure to complete the imaging study, or failure of quality control. Imaging acquired upon early withdrawal from the study was not included in the analysis. 220, 209, and 208 had DTIs that passed quality control at the third (48 weeks), fourth (72 weeks), and fifth (96 weeks) time points, respectively. A total of 39 subjects switched scanner type during the trial, leading to detectable changes in RD, but no detectable impact on trial outcomes. 26
Table 1 summarizes the primary results—the rate of change per 24-week time period of RD for placebo and treatment groups for each ROI, along with 95% confidence intervals (CIs). Also reported is the difference (placebo–treatment) in the slopes. After correction for multiple comparisons, the difference in rate of change was significant in the part of the cingulum that is adjacent to the cingulate gyrus (7.4 × 10−3 mm2/s per 24-week time period, adjusted p < 0.02) and in the cerebellar peduncles (2.7 × 10−3 mm2/s per 24-week time period, adjusted p < 0.04). Figure 1 illustrates the location of these ROIs. For the placebo group, the 95% CI covers a positive range of values, consistent with an increase over time. In other words, the change in RD over time is consistent with a decline in tissue integrity over time. For the treatment group, the 95% CI straddles zero, consistent with relatively stable tissue integrity. Figure 2 illustrates the change over time of RD for the treatment and placebo groups. The WMPM also specifies a parahippocampal segment of the cingulum, for which no statistically significant results were found. The dividing line between the two segments of the cingulum is at the axial level of the splenium of the corpus callosum. 16
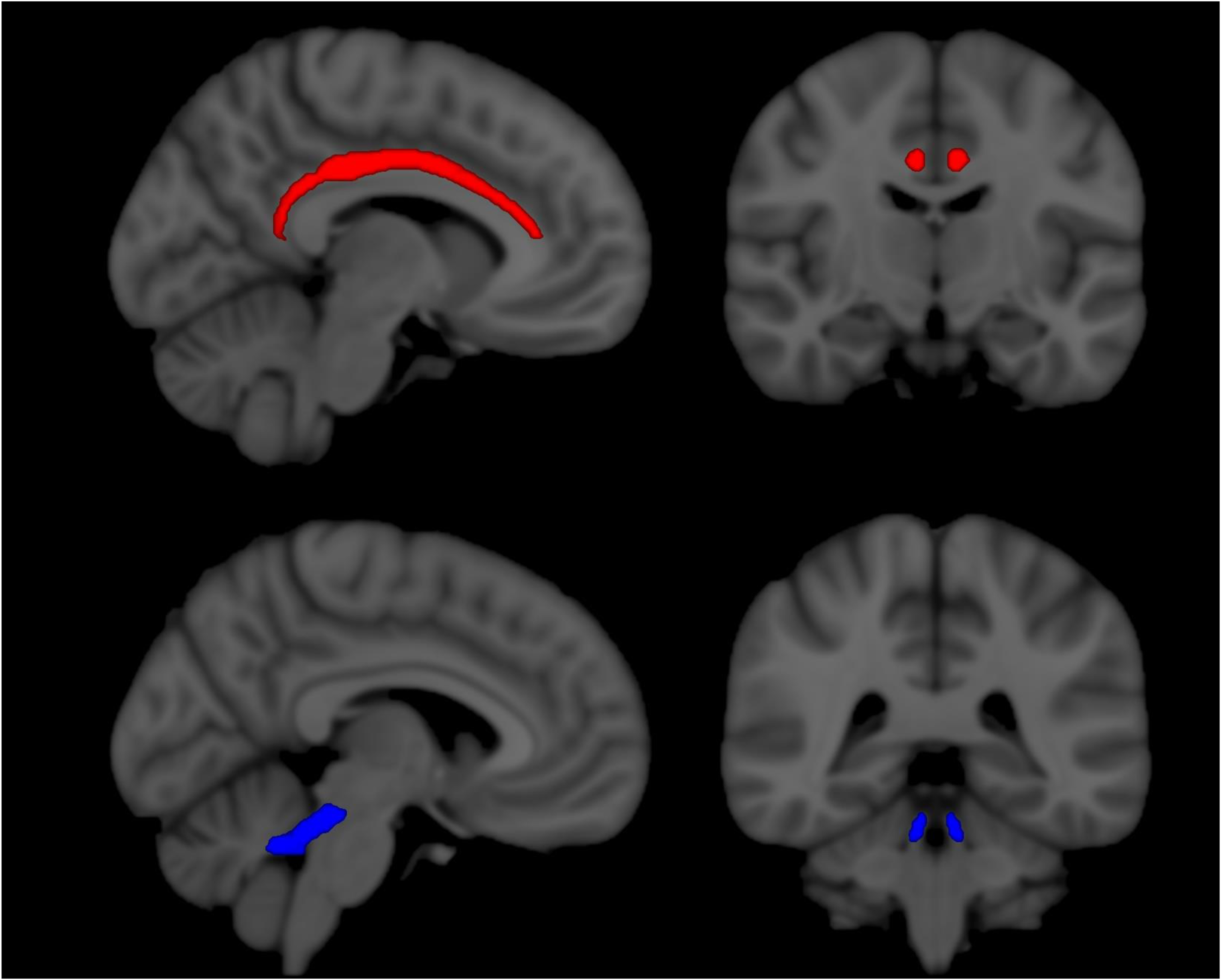
ROIs as defined by the ICBM-DTI-81 WMPM in MNI space. Cingulum (red) and superior cerebellar peduncles (blue) are shown.

Change over time by treatment group of the median, across voxels, of radial diffusivity (RD) in (a) cingulum and (b) superior cerebellar peduncle. Units along the vertical axis are × 10−3 mm2/s.
Rate of change over time and 95% CI of RD in each ROI for placebo and treatment groups, and the difference (placebo–treatment) between rates of change.

Units are × 10−3 mm2/s per 24-week time period. All values have been scaled by 1000 for readability. Both unadjusted and adjusted p-values are shown. Adjusted p-values are corrected for multiple comparisons with a Holm's correction. Superior cerebellar peduncle and cingulum (
CIs: confidence intervals; ROIs: regions of interest; RD: radial diffusivity.
Part of the middle cerebellar peduncle.
Includes optic radiations.
Includes inferior longitudinal fasciculus and inferior fronto-occipital fasciculus.
Includes stria terminalis
Results for other tissue integrity parameters are provided in the supplementary tables. As these were secondary outcomes, p-values were not corrected for multiple comparisons. For FA, differences between treatment and placebo groups were found in the corticospinal tract, anterior corona radiata, sagittal stratum, and unicinate fasciculus. In each of these regions, the 95% CI covers a negative range of values for the placebo group, indicating a decline in FA over time and consistent with a decline in tissue integrity. For the treatment group, the 95% CI straddles zero, consistent with stabilization. For MD, differences between treatment and placebo groups were found in different regions: posterior corona radiata, cingulum, and superior longitudinal fasciculus. In each of these regions, the 95% CI covers a positive range of values for the placebo group, indicating an increase in MD over time and consistent with worsening tissue integrity. For the treatment group, the 95% CI straddles zero, consistent with stabilization. AD shows differences between treatment and placebo groups in the sagittal stratum. For both groups, the 95% CI straddled zero.
Discussion
In this hypothesis-generating study, we leverage advances in image postprocessing to determine if alternative approaches to analysis of MRI might help make DTI useful in neuroprotective clinical trials in progressive MS. In particular, we explored the use of a parcellation of white matter regions without imposing a priori expectations about which white matter region or regions might show a treatment effect. In this study, we found that ibudilast treatment was associated with decreased RD in the cingulum. The cingulum is a large white matter pathway that is associated with a wide array of cognitive functions, including executive control, emotion, pain, and episodic memory. 27 In MS, RD in the cingulum bundle has been found to correlate with deficits in episodic memory. 28 A more recent analysis of diffusion MRI found strong associations between microstructural damage in the cingulum and cognitive impairment. 29 MS has long been recognized as being associated with cognitive impairment, with important consequences for quality of life, but can be difficult to detect reliably with standard diagnostic tools. 30 DTI may hold potential for examining interventions that are believed to have an impact on cognition. In a cross-sectional study, RD was found to be higher among clinically impaired subjects with MS than with unimpaired subjects with MS in both middle and superior cerebellar peduncles, and significant differences were also found for MD and FA. 31 Thus, it is possible that RD in the cerebellar peduncle is a clinically relevant area to measure changes in a clinical trial. Although other work has demonstrated an association between RD and myelin integrity,21,22,32 the interpretation of increased RD as reflecting myelin status and demyelination should be treated with caution due to limitations of the tensor model 23 and contributions from other cellular structures as well as physiological factors such as edema, inflammation, and complex fiber geometries. 24 However, in the SPRINT-MS trial, MTR, an alternative proxy for demyelination, did demonstrate trends consistent with demyelination and preservation of tissue integrity in the placebo and treatment groups, respectively. 1
More broadly, the approach examined here lies between two extremes. The first, taken by the original SPRINT-MS, declared a priori that a single white matter pathway, the descending pyramidal tracts, should be the focus of investigation. Other examples include work by Harrison et al., 33 which proposed the body of the corpus callosum as the most appropriate region for use in longitudinal, placebo-controlled trials, as well as work by Bodini et al. 34 showing associations between DTI measures in this region and disability progression. Neither of these regions turned out to show significant treatment effects in this analysis. Note that the pyramidal tracts used in SPRINT-MS were generated using probabilistic tractography and roughly correspond to ROIs in the WMPM that are named cerebral peduncles, posterior limb of the internal capsule, and superior corona radiata. The second extreme, which examines normal-appearing white matter or lesional tissue, ignores differences among white matter pathways. Although sensitive to disease progression, 35 this approach has not, to our knowledge, found effective use in a clinical trial. Focusing on white matter pathways, but not a specific pathway a priori, may be appropriate for use in clinical trials.
Limitations of this study largely derive from being a retrospective analysis of a completed clinical trial. The outcomes, therefore, cannot be interpreted as being a true treatment effect and can only be used for planning future trials. This study was also intentionally limited in scope to focus on one aspect of the analysis– the choice of white matter pathway. The absence or presence of lesions was not accounted for, so it is possible that lesions could have influenced our measures and observations. The study population consisted of patients with secondary and progressive MS, in whom lesion burden was expected to be stable over time. As this study examined changes over time of DTI measures, it was assumed that the lesions could be ignored. However, recent work on the SPRINT-MS dataset found that 6% of T2 lesions were slowly expanding lesions (SEL). 3 The change over time of MTR differed between treatment and placebo groups within SEL, but not within non-SEL. Accounting for the impact of lesions on longitudinal studies of imaging biomarkers is an important avenue for future research, but may require care in accounting for different lesion types.
There are a number of other approaches that could be applied to this dataset. Different microstructure metrics besides the diffusion tensor 29 could be explored. Previous work demonstrated high concordance in DTI measures across scanner platforms when scan parameters were matched, 36 and analysis within-subject changes of scanner type showed limited impact on study outcomes. 26 However, scanner-dependent factors were not explicitly accounted for in this study. Data harmonization methods that are appropriate to longitudinal studies have been developed, 37 and could affect trial outcomes.
The absence of susceptibility distortion correction may have affected alignment to the JHU-ICBM template. Postprocessing software such as TORTOISE (https://github.com/QMICodeBase/TORTOISEV4) can correct susceptibility distortion by nonlinear coregistration to a T2-weighted image, obviating the need for a field map or reverse phase-encoding scan, and may reveal trends not found in this study.
Alignment of FA maps to an FA template can be affected by the presence of lesions and atrophy, which can severely alter FA values. An alternative approach is to use multi-contrast registration. In one subject with severe lesions and atrophy, FA and T1-weighted (T1w) images were simultaneously coregistered to FA and T1w templates in MNI space using the default parameters of antsRegistrationSyN.sh. The inverse transform was used to visualize the JHU-ICBM atlas on the T1w images, and the results are shown in Supplemental Figure 1. The ROIs used in this study are also shown on the T1w images for comparison. These latter ROIs were generated by coregistering the FA map to the T1w image with antsRegistrationSyN.sh with default parameters, concatenating this transform to the one used in this study to transform ROIs from the JHU-ICBM atlas to DTI space, and applying the concatenated transform to the ROIs. The comparison of methods suggests that multi-contrast registration can improve alignment of white matter ROIs with underlying anatomy. The impact of alignment, particularly on small ROIs, is a matter that deserves more detailed analysis in future studies. Work comparing different spatial normalization methods in the presence of lesions and atrophy found that nonlinear warping, which was used in this study, performed better than linear registration, with some additional benefit conferred by lesion filling. 38 Improved methods that account for lesions and atrophy could improve sensitivity to treatment effects.
Partial volume averaging between tissue and cerebrospinal fluid (CSF) may have affected results because of the large disparity between tissue and CSF in terms of diffusion parameters. The median, rather than the mean, across voxels in each ROI was chosen to limit the influence of partial volume averaging. An alternative approach, erosion of the edge voxels of the ROIs, was not explored. By structuring the data in the BIDS format, we hope to facilitate additional analyses in the future 19
Conclusions
In this study, we performed a retrospective ROI-based analysis of DTI from a 24-month, placebo-controlled drug trial to inform the conduct of future clinical trials. Significant differences between treatment and placebo groups were found in the RD of the cingulum and cerebellar peduncles. The cingulum and cerebellar peduncles may therefore be attractive areas for focus in the evaluation of putative therapies in progressive MS.
Supplemental Material
sj-doc-1-mso-10.1177_20552173251361225 - Supplemental material for Diffusion tensor imaging in the SPRINT-MS clinical trial: Advancing trial methodology
Supplemental material, sj-doc-1-mso-10.1177_20552173251361225 for Diffusion tensor imaging in the SPRINT-MS clinical trial: Advancing trial methodology by Ken Sakaie, Mengke Du, Nancy Obuchowski, Mark J Lowe, Jian Lin and Robert J Fox in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
This work was supported by the National Multiple Sclerosis Society (SI-2301-40767). The original SPRINT-MS trial was supported by grants from the National Institute of Neurological Disorders and Stroke (NINDS) (U01NS082329), the National Multiple Sclerosis Society (RG 4778-A-6), and by MediciNova through a contract with the National Institutes of Health (NIH). The trial was performed with the NeuroNEXT Network with support from the NINDS (Central Coordinating Center, U01NS077179; Data Coordinating Center, U01NS077352).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: KS, MD, NO, MJL, and JL have no relevant disclosures. RJF has received personal consulting fees from Astoria Biologica, Biogen, Bristol Myers Squibb, Cognito, EMD Serono, Galvani, Immunic, INmune Bio, Kiniksa, Novartis, Sanofi, Siemens, TG Therapeutics, and Viracta. RJF has served on advisory committees for AB Science, Biogen, Immunic, Novartis, and Sanofi, and received clinical trial contract and research grant funding from Biogen, Novartis, and Sanofi.
Data availability statement
Underlying materials will be made available upon request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
