Abstract
Background and Objectives
Cardiovascular diseases (CVD) and their risk factors supposedly occur frequently in patients with multiple sclerosis (pwMS). We investigated prevalence of comorbidity particularly CVD among pwMS.
Methods
Two cohorts from Tehran and Isfahan were investigated retrospectively with longitudinal follow up and were invited to participate prospectively with measurement of biomedical parameters including determination of metabolic syndrome (MetS), and insulin resistance (IR). The 10-year office-based Framingham risk score (FRS) was calculated.
Results
Out of 856 pwMS 329 (38.4%) had at least one comorbidity and 97 (11.3%) had > 2 diseases, i.e., multiple comorbidity. PwMS and comorbidity were older (p < 0.0001) and had higher age at MS onset (p < 0.0001) compared to the non-comorbidity group. The prevalence of comorbidity increased from 24.0% at age 15–29 years to 37.3% at 30–49 and to 52.6% at 50–76 years (p < 0.0001) and was associated with odds of EDSS ≥ 4. FRS was for men 7.1 (4.2, 10.5) and for women 2.0 (1.3, 3.4). Of 255 with prospective blood testing, 35 (13.7%) had MetS, and 106 (41.6%) had IR.
Conclusion
A high prevalence of comorbidity, associated with disability and high FRS was observed in pwMS. Our data suggest that MetS and IR occur frequently in this population.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory demyelinating disorder, affecting more than two million people worldwide. 1 Comorbidities, defined as the coexistence of additional chronic diseases, are common among patients with MS (pwMS). Comorbidities significantly associated with MS include hypertension (HTN), hyperlipidemia (HLP), cardiovascular diseases (CVD), lung diseases, and psychiatric disorders. 2 These comorbidities are associated with more severe physical disability, longer diagnostic delays, and increased mortality rates. 3
Cardiovascular diseases are recognized as a leading cause of global mortality 4 and are linked to various risk factors such as age, gender-specific predispositions, HTN, HLP, type 2 diabetes (T2DM), and smoking. The spectrum of cardiovascular comorbidities in MS ranges from subclinical issues to severe complications such as ischemic heart diseases (IHD), valvular diseases, heart failure, and stroke. 5 Geographical variations in comorbidities, particularly CVD, underscore the importance of gathering information from diverse regions. Iran, located in the Middle East 6 has one of the highest prevalences of MS in the region. 7 We designed a study to investigate comorbidities, with specific attention to CVD among MS patients.
Methods
Participants
Two cohorts, from the MS clinic of Kashani Hospital in Isfahan and from the MS Clinic of Sina Hospital in Tehran, Iran, were investigated retrospectively with longitudinal follow up and prospectively with measurement of biomedical parameters (Figure 1). The MS clinic at Sina Hospital and Isfahan are tertiary care centers.7,8 All enrolled patients had received a definitive diagnosis of MS.9–11 We obtained from medical records demographic and clinical data including age, sex, educational level, employment status, age at MS onset, MS duration, disease course including relapsing-remitting MS (RRMS), secondary-progressive MS (SPMS), or primary-progressive MS (PPMS), and disease-modifying therapy (DMT) exposure. Severity of MS was measured based on expanded disability status scale (EDSS) score 12 by a trained neuroogist. Employment status was classified as employed or unemployed.

Diagram of the study.
This study was approved by the Ethics Committee of Isfahan University of Medical Sciences, (ethical code: IR.ARI.MUI.REC.1401.180). Written informed consent was obtained from all participants before inclusion. The research adhered to the principles of the Helsinki Declaration II.
Comorbidity assessment
We collected data on comorbidities from medical records and chart reviews. Two investigators, conducted patient interviews at each center using a standardized procedure as previously described. 13 Comorbidities other than CVD and CVD-related risk factors were classified as non-CVD comorbidity.
Framingham risk score
The Framingham risk score (FRS) was calculated for all enrolled patients aged between 30 to 74 years. 14 The sex-specific office-based FRS was computed using two mathematical equations that both incorporate age, sex, systolic BP (SBP) and diastolic (DBP), use of anti-hypertensive agents, smoking status, presence of T2DM, and BMI. For FRS a normal individual was defined as nonsmoking and non-diabetic, with a SBP of 120 mm Hg and a BMI of 22.5 kg/m². Patients with an FRS of less than 10% are classified as low risk, 10% to 20% as intermediate risk, and greater than 20% as high risk.
Paramedical profile and metabolic syndrome
We invited all patients to participate in the second part of the study at the time they were interviewed for the Phase 1 study (Figure 1). We assessed fasting blood sugar (FBS), the lipid profile, insulin levels, and the presence of MetS. FBS and lipid profile were determined by routine methodologies. Human Insulin levels were measured by ELISA (MONOCENT kit, Iran). To assess insulin resistance, we calculated the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) using the formula: FBS (mg/dl) × insulin (µU/ml)/405. Insulin resistance was defined as HOMA-IR ≥1.85 in women and HOMA-IR ≥2.17 in men, as reported in the Iranian population. 15
MetS was defined as presence of three of the following five risk factors: (a) elevated BP (SBP ≥130, DBP ≥85 mm Hg, or antihypertensive use in a patient with a history of HTN,), (b) elevated triglycerides (TG) (≥150 mg/dL or using related medication), (c) reduced HDL (<40 mg/dL in males; < 50 mg/dL in females or using related medication), (d) elevated FBS (≥100 or using related medication), and (e) elevated waist circumference (≥102 and ≥88 centimeters for men and women). 16 In the study, a BMI ≥30 kg/m² was considered equivalent to an elevated waist circumference. 17
Statistical analysis
Continuous variables, with and without a normal distribution, were presented as mean ± standard deviation (SD) and median (interquartile range [IQR]), categorical variables were reported as frequency (%). Variables between two groups were compared by independent samples t-test or Mann-Whitney test for normally and non-normally distributed continuous variables, and Chi-square or Fisher's Exact Test for categorical variables. One-way ANOVA with Tukey Post Hoc and chi-square tests were used to compare continuous and categorical variables among more than two groups. The age-stratified prevalence of comorbidity was analyzed by dividing the cohort into predefined age groups including 15–29, 30–49, and 50–76 years. Differences in comorbidity prevalence across these age groups were assessed using the Chi-square test. Furthermore, binary logistic regression analysis was performed to examine the association of age with comorbidities. This model was adjusted for sex, education level, employment status, smoking status, BMI, course of MS, and disease duration. To evaluate the association between the presence of comorbidity, FRS, and paramedical parameters, with the probability of EDSS ≥ 4, we conducted binary logistic regression analyses. To identify risk factors for MetS and insulin resistance, we conducted another binary logistic regression analysis. The assumptions for binomial logistic regression analysis were satisfied, including independent observations with mutually exclusive and exhaustive outcome categories, as well as a linear relationship between continuous independent variables and the logit transformation of the dependent variable. The results of logistic regression were reported as odds ratios (ORs) and 95% confidence intervals (95% CIs). A p-value < 0.05 was considered significant (two-tailed). All statistical analyses were performed using IBM SPSS Statistics version 22 (IBM Corporation, Armonk, NY, USA).
Results
Study participants
A total of 856 pwMS, 447 from Isfahan and 409 from Tehran were included. The mean age of the patients was 40.1 ± 9.4 (15–76) years and 714 (83.4%) were female. The age distribution was: 104 (12.1%) patients between 15–29 years, 600 (70.1%) between 30–49 years, and 152 (17.8%) between 50–76 years. The mean age at MS onset was 29.7 ± 9.0, with a mean disease duration of 10.49 ± 5.87 and a median EDSS score of 1.5 (0.0–3.0). Regarding clinical course, 723 (84.5%) patients had RRMS, 111 (13.0%) had SPMS, and 22 (2.6%) had PPMS. The most common DMT was rituximab (28.7%), followed by ocrelizumab (25.9%), interferon (15.8%), and teriflunomide (7.8%). Table S1 presents the characteristics of patients from the two hospitals.
Prevalence of comorbidities
The crude prevalence of comorbidity is summarized in Table 1. Overall, 329 (38.4%, 95% CI: 35.2%–41.7%) patients had at least one comorbidity, 97 (29.5%, 95%CI: 24.6%-34.7%) of them experienced multiple comorbidity. Non-cardiovascular disease was found in 253 patients, with a crude prevalence of 29.6% (95%CI: 26.5%–32.7%) among all participants. A total of 34 patients had CVD comorbidity, with a crude prevalence of 4.0% (95%CI: 2.7%–5.3%). Crude prevalence of documented HTN, HLP and T2DM among all enrolled participants were 52 (6.1%, 95%CI: 4.6%–7.9%), 44 (5.1%, 95%CI: 3.8%–6.8%) and 27 (3.2%, 95%CI: 2.0%–4.6%), respectively. A history of smoking was present in 248 (29.0%, 95%CI: 25.9%–32.1%) patients and 88 (10.3%, 95%CI: 8.3%–12.5%) patients were current smokers. Obesity was also observed in 91 (10.6%, 95%CI: 8.6%–12.9%) patients of all participants. A greater percentage of female patients had at least one comorbidity (p = 0.046) and non-CVD comorbidity (p = 0.005). The distribution of comorbidities across participating hospitals is shown in Table S2.
Crude prevalence of comorbidity among all MS patients.

Prevalence of comorbidity based on age
Figure 2 displays the prevalence of comorbidities based on the age groups. The presence of any comorbidity increased from 24.0% (95%CI: 15.7%–32.3%) during the period of 15–39 years to 37.3% (95%CI: 33.4%–41.2%) at 30–49, and to 52.6% (95%CI: 44.7%–60.5%) at 50–76 years (p < 0.0001). The prevalence of HTN and T2DM increased with age (p < 0.0001). The age-standardized prevalence of comorbidities is presented in Table S3. In an adjusted model, older age was significantly associated with a higher prevalence of any comorbidity (OR = 1.051, 95% CI: 1.032, 1.070), non-CVD comorbidity (OR = 1.022, 95% CI: 1.003, 1.041), HTN (OR = 1.130, 95% CI: 1.089, 1.173), HLP (OR = 1.057, 95% CI: 1.020, 1.096), and T2DM (OR = 1.056, 95% CI: 1009, 1.106) (Table 2).

Age-specific prevalence of comorbidities. Autoimmune comorbidity included systemic lupus erythematosus, immune thrombocytopenic purpura, rheumatoid arthritis, Hashimoto thyroiditis, lichen planus, vitiligo, and psoriasis were considered as autoimmune comorbidity. Psychiatric comorbidity included major depression disorder, obsessive-compulsive disorder, and other reported psychiatric disorders.
Association of age with comorbidities.

Model 1: Unadjusted
Model 2: Adjusted for sex (females vs. males), education level (diploma or lower vs. above diploma), employment status (employed vs. unemployed), smoking status (current smoker vs. non-smoker), BMI (continuous variable), course of MS (RRMS vs. progressive MS), and disease duration (continues variable)
Note. CVD: cardiovascular diseases, HTN; hypertension, HLP: hyperlipidemia, T2DM: type II diabetes mellitus
Prevalence of comorbidity and date of MS onset
Data on the age of comorbidity and MS onset, except for CVD, were available only from one center (Figure 3). The highest change during the 5 years before and after MS onset was observed in psychiatric disorders, increasing from 0.2% to 2.9%. There was no change in the prevalence of CVD and autoimmune disorders, and a decrease in prevalence of cancer (−0.3%).

Prevalence of comorbidities based on the date of MS onset.
Comparison between patients with and without comorbidity
Clinical characteristics of patients is summarized in Table 3. Compared to pwMS without any comorbidity, those with at least one comorbidity had a higher age (p < 0.0001), a higher age at MS onset (p < 0.0001), were more often female (p = 0.046), and were more often employed (p = 0.046). There is also a significant difference in DMT exposure (p = 0.030).
Characteristics of all enrolled MS patients based on the presence of comorbidity.

★ Comparison between patients with and without any comorbidity
‡ Comparison between patients with CVD and those with non-CVD comorbidity.
Note. EDSS: expanded disability status scale; RRMS: relapsing-remitting MS SPMS: secondary-progressive MS; PPMS: primary-progressive MS; IFN: interferon; GA: glatiramer acetate; FNG: fingolimod; TRF: teriflunomide; DMF: dimethyl fumarate; NTZ: nataliumab; RT: rituximab; OCR: ocrelizumab; IQR: interquartile range
Association of comorbidity with MS severity
In adjusted model, the association of CVD with EDSS score returned to non-significance (OR = 1.536, 95%CI: 0.404, 5.844). There was a significant association between any comorbidity (OR = 1.895, 95% CI: 1.017, 3.530) and the odds of EDSS ≥ 4, after adjustment.
Framingham risk score
After excluding 120 patients (due to missing data in 15, age higher than 74 years in one, younger than 30 years in 104 patients), a total of 736 patients (females = 623 and males = 113) were included to measure FRS. The median office-based FRS scores in men and women were 7.1 (4.2, 10.5) and 2.0 (1.3, 3.4), respectively. Among women, 596 (95.7%) were classified as low risk, 26 (4.2%) as intermediate risk, and 1 (0.2%) as high risk for CHD over 10 years. Among men, 84 (59.2%) were low risk, 25 (17.6%) were intermediate risk, and 4 (2.8%) were high risk. Figure 4 displays FRS scores in MS patients and compared them to the normal range based on age. In male MS patients, the FRS score exceeded the normal range across all age groups (Figure 4(A)). In female MS patients, the FRS was lower than the normal range until ages 40–44 years and increased for the 45–49 years age group and upwards (Figure 4(B)).
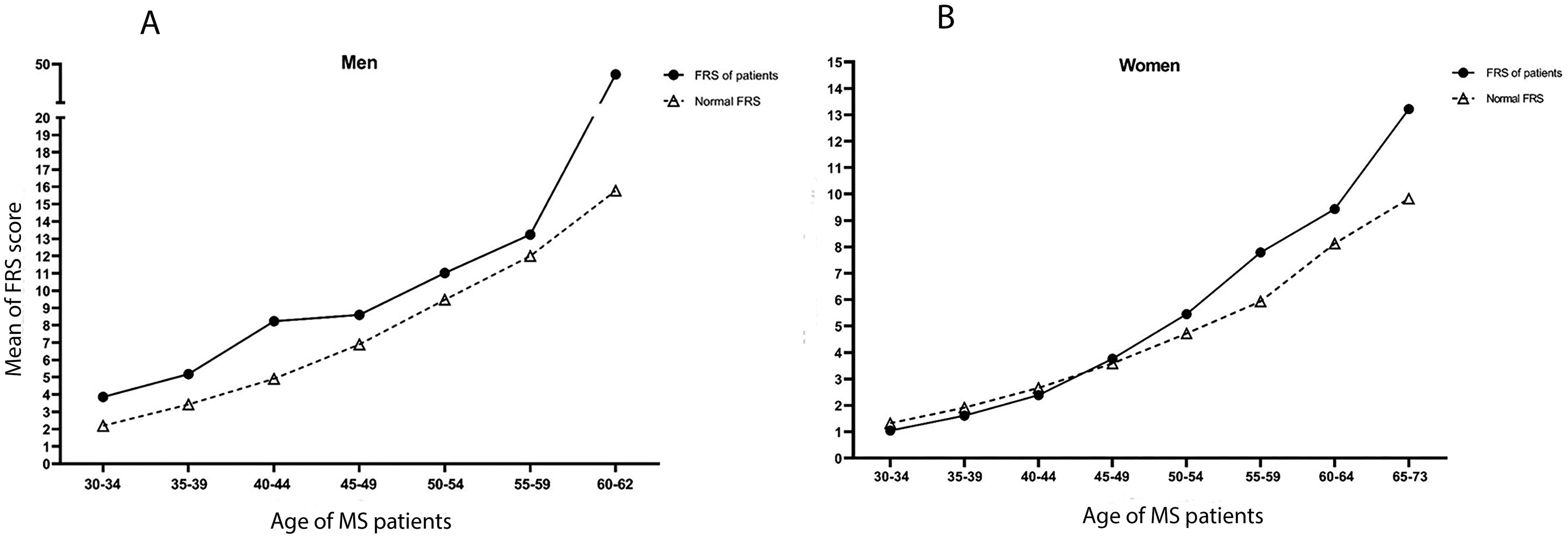
Framingham risk score based on the sex and age.
In an unadjusted model, we found an association between increased FRS and probability of occurrence of EDSS ≥ 4.0 (OR = 1.085, 95%CI: 1.039, 1.113). This did not remain statistically significant after adjustment (OR = 0.979, 95%CI: 0.895, 1.070) (Table 4).
Association of comorbidities and Framingham risk score with EDSS score.
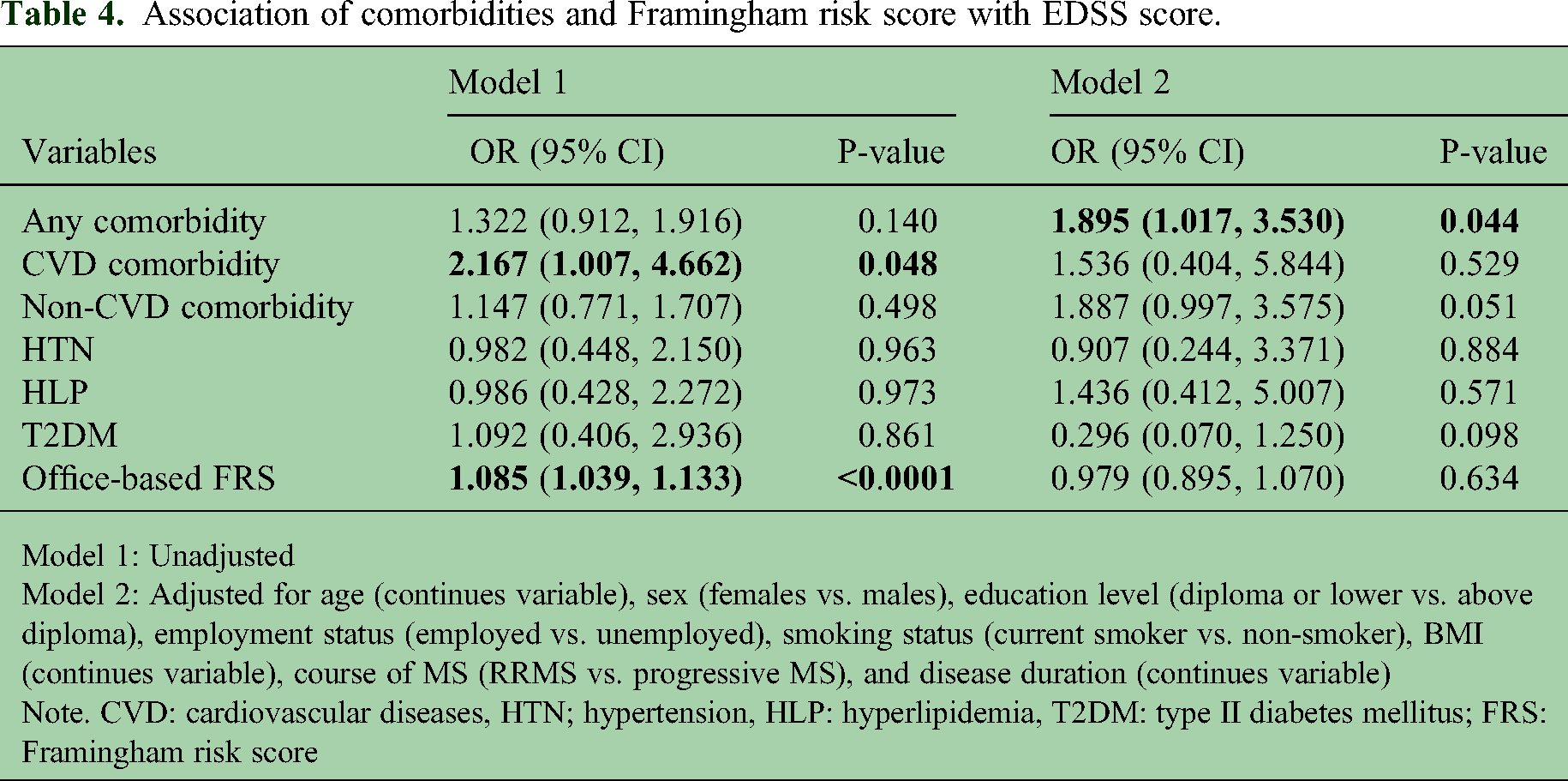
Model 1: Unadjusted
Model 2: Adjusted for age (continues variable), sex (females vs. males), education level (diploma or lower vs. above diploma), employment status (employed vs. unemployed), smoking status (current smoker vs. non-smoker), BMI (continues variable), course of MS (RRMS vs. progressive MS), and disease duration (continues variable)
Note. CVD: cardiovascular diseases, HTN; hypertension, HLP: hyperlipidemia, T2DM: type II diabetes mellitus; FRS: Framingham risk score
Biomedical profile and metabolic syndrome
A total of 255 patients accepted to undergo evaluation of their biomedical profile. The patients tested had a longer disease duration (p = 0.019), were more frequently employed (p = 0.041), and had a higher proportion of SPMS (p = 0.001) with differences in DMT exposure (p < 0.0001). Clinical features of tested patients based on the comorbidity are shown in Table 5. A total of 35 (13.7%) patients were diagnosed with metabolic syndrome, and insulin resistance was observed in 106 (41.6%) patients.
Characteristics of MS patients who were tested.
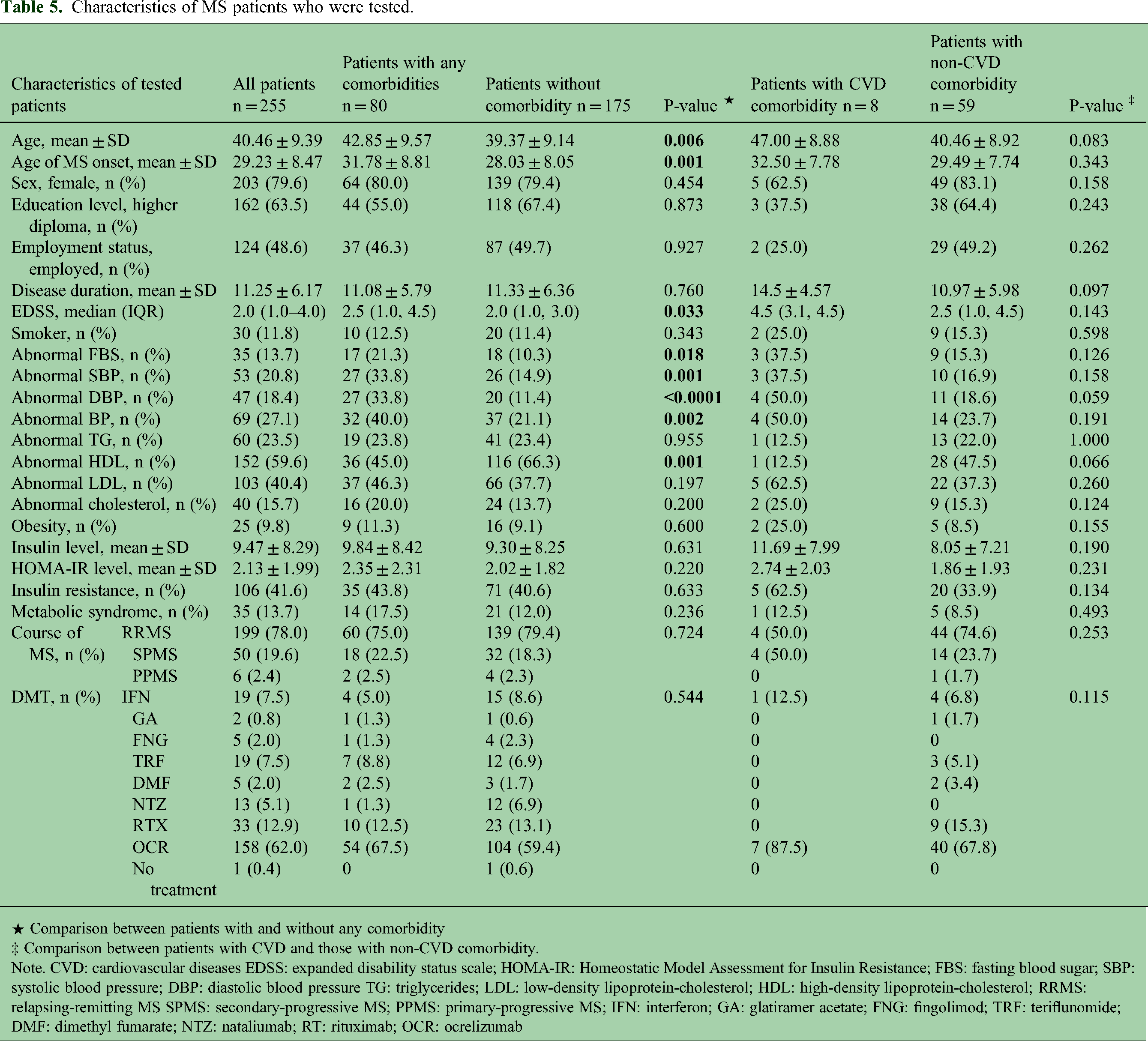
★ Comparison between patients with and without any comorbidity
‡ Comparison between patients with CVD and those with non-CVD comorbidity.
Note. CVD: cardiovascular diseases EDSS: expanded disability status scale; HOMA-IR: Homeostatic Model Assessment for Insulin Resistance; FBS: fasting blood sugar; SBP: systolic blood pressure; DBP: diastolic blood pressure TG: triglycerides; LDL: low-density lipoprotein-cholesterol; HDL: high-density lipoprotein-cholesterol; RRMS: relapsing-remitting MS SPMS: secondary-progressive MS; PPMS: primary-progressive MS; IFN: interferon; GA: glatiramer acetate; FNG: fingolimod; TRF: teriflunomide; DMF: dimethyl fumarate; NTZ: nataliumab; RT: rituximab; OCR: ocrelizumab
Sex and age-related differences in biomedical profile
We observed a significant increase in the level of all measures except HDL (Figure 5). In post-hoc analysis, a significant increase in all measures, except HDL was observed in the 50–76 age group compared to younger ages (Table S4). No significant differences were found among females (Table S5). In males, there were no age-related increases in biomedical parameter levels, except for significant differences in SBP (p = 0.018) and DBP (p = 0.026) between patients aged 15–29 and 30–49 years (Table S6).

Age-related measure of biomedical profile based on sex.
Risk factors for insulin resistance and MetS
In the multivariable regression analysis, increasing age (OR = 1.053, 95% CI: 1.017, 1.090) and male gender (OR = 2.159, 95% CI: 1.070, 4.354) were independent risk factors for insulin resistance (Figure 6(A)). There was decreased odds of insulin resistance in patients with SPMS (OR = 0.291, 95% CI: 0.102, 0.825). Only age was as an independent risk factor for the presence of MetS (OR = 1.104, 95% CI: 1.051, 1.160) (Figure 6(B)).

Risk factors of metabolic syndrome and insulin resistance.
Association of biomedical profile and MetS with MS severity
HDL abnormality was inversely associated with probability of occurrence of EDSS ≥ 4.0, both before (OR = 0.374, 95%CI: 0.207, 0.678) and after adjustment (OR = 0.183, 95%CI: 0.046, 0.733) (Table 6). In both unadjusted and adjusted models, no significant association was observed between HOMA-IR, insulin resistance, MetsS, BMI or other lipid parameters with EDSS score (Table 6).
Association of paramedical parameters and metabolic syndrome with EDSS score.

Model 1: Unadjusted
Model 2: Adjusted for age (continues variable), sex (females vs. males), education level (diploma or lower vs. above diploma), employment status (employed vs. unemployed), smoking status (current smoker vs. non-smoker), BMI (continues variable), course of MS (RRMS vs. progressive MS), and disease duration (continues variable)
Note. HOMA-IR: Homeostatic Model Assessment for Insulin Resistance; FBS: fasting blood sugar; BP: blood pressure; TG: triglycerides; LDL: low-density lipoprotein-cholesterol; HDL: high-density lipoprotein-cholesterol
Discussion
In this study the prevalence of comorbidities in pwMS was estimated, and the association between comorbidity and disability was explored. Additionally, we measured the biomedical profile and assessed CVD related risk factors such as metabolic syndrome, insulin resistance, and lipid profile. As a main finding 38.4% patients had at least one comorbidity and 29.5% hereof had multiple comorbidity. A prevalence of 29.6% was estimated for non-cardiovascular disease and 4.0% for CVD. PwMS with comorbidity were older and had later onset of MS compared to the non-comorbidity group. The prevalence of comorbidity increased with age i.e., to 52.6% at 50–76 years, and was associated with increased disability. The 10-year risk of CVD was higher for men than women. Insulin resistance was observed in more than 40% of pwMS who were tested, and HDL abnormality was inversely associated with disability. Such estimates and calculations may be implemented in clinical practice.
The most common CVD in our sample (2.0%), fell within the range reported previously. 5 . 18 Similarly, the prevalence of IHD in 1.3% of our patients was consistent with previous findings. 5 The relatively young age of our sample may also contribute to the lower prevalence of CVD. Patients with comorbidities had a later age of MS onset. This finding aligns with a study from the US population, which observed a dose-response relationship between the number of comorbidities and an older age at MS onset. 19
The association of CVD with EDSS was non-significant after adjustment, comparable with a study from Poland that found no difference in the frequency in IHD and PDA with EDSS. 20 However, studies from Canada, 21 USA, 22 and Serbia 23 found independent associations of vascular comorbidities with ambulatory disability. The Canadian study found an association between IHD and epilepsy with higher EDSS scores. 21 The study from USA found association between MetS, but not heart disease, with ambulatory disability. 22 A recent study by Salter et al. involving patients from phase 3 trials showed that the presence of two or more cardiometabolic comorbidities was associated with an increased risk of MS activity. 24
The prevalence of MetS in our MS sample, at 14%, consistent with previous studies on MS patients 25 but lower than reported rates in other MS studies26,27 and the general Iranian population. 28 This difference could be attributed to the lower rate of abdominal obesity in our sample compared to other MS studies.26,27 Insulin resistance was observed in approximately 42% of our tested patients, consistent with previous MS studies.25,29 For both MetS and IR age was a risk factor. We found a greater probability of IR in male MS patients. Men have a greater amount of adipose tissue and lack of estrogen which predispose them to IR. 30 The inverse association between SPMS and IR supports previous findings showing a protective effect of IR/ T2DM in progressive MS. 31
Consistent with previous studies, we failed to find an association between MetS and physical disability in MS patients. The association between IR and physical disability is complex, with some studies reporting a significant association,29,32 others no association. 25 We found no association between MetS components and EDSS scores, except for an increased probability of a greater EDSS score in patients with elevated HDL levels. This is an unexpected finding, as HDL is known for its anti-inflammatory functions. Previous studies have shown an association between high HDL levels and lower inflammatory activity and disability in MS.33,34 However, inflammation and oxidative stress can alter the size of HDL, leading to dysfunction and a proinflammatory shift in its activity. 35 In line with this, studies by Palavra et al. 36 and Jorissen et al. 37 reported higher HDL levels in MS patients compared to controls, suggesting an impairment in the anti-inflammatory function of HDL in patients with MS. The impact of HDL level on inflammatory activity in pwMS is mutual and dependent on the size of HDL subfractions, 38 suggesting a possible explanation for our results.
In this study we observed an increased FRS in women aged 45–49 years. We found a higher FRS score in patients with comorbidity, particularly those with CVD, likely due to the greater prevalence of T2DM in these patients. It should bear in mind that about 80% of our sample had low-risk FRS.
A notable strength of our study design is provision of the data from two different locations, and measurement of biomedical parameters. We evaluated all comorbidities and CVD risk factors based on age and sex, providing a clearer understanding of the impact of comorbidities in pwMS. Additionally, this study represents a large study of pwMS with measurement of the FRS. However, our study has several limitations including the retrospective nature of the study and the absence of a control group . . Our sample was generally young, with only three cases older than 70 years, which may have affected the prevalence of CVD and related risk factors observed in our study. A high proportion of patients enrolled in phase 1 declined to participate in phase 2, which limited the second part of our study. We used BMI instead of waist circumference to measure MetS. Lastly, we did not assess physical activity levels and diet status in this study.
In conclusion, our study determines the prevalence of comorbidity, associated with heightened disability, and related risk factors for CVD in pwMS. We recommend that clinicians actively screen for comorbidity in pwMS. Larger longitudinal studies are necessary to determine specific CVD risk factors in the MS population.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251352735 - Supplemental material for Comorbidity and cardiovascular risk factors in multiple sclerosis
Supplemental material, sj-docx-1-mso-10.1177_20552173251352735 for Comorbidity and cardiovascular risk factors in multiple sclerosis by Mahdi Barzegar, Sara Samadzadeh, Kosar Kohandel, Aysa Shaygannejad, Naghmeh Abbasi Kasbi, Saeed Vaheb, Sajjad Ghane Ezabadi, Omid Mirmosayyab, Abdorreza Naser Moghadasi, Alireza Afshari-Safavi, Nasim Rezaeimanesh, Majid Ghasemi, Vahid Shaygannejad, Mohammad Ali Sahraian and Nasrin Asgari in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Isfahan University of Medical Sciences, (grant number IR.ARI.MUI.REC.1401.180).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
