Abstract
Background
Patients with multiple sclerosis (PwMS) could suffer from frequent and disabling motor symptoms, including balance and mobility problems, spasticity, weakness and fatigue, with an impact on patients’ quality of life. Current treatments have limited efficacy or significant side effects. The EXOPULSE Mollii Suit, a transcutaneous electrical nerve stimulation system, provides simultaneous stimulation to 40 muscle groups and may offer a therapeutic alternative.
Objectives
This study evaluated the effects of this device on balance, other motor symptoms and quality of life in PwMS.
Methods
A randomized, crossover, sham-controlled, double-blind study (phase 1) evaluated the effects of a 60-min single session of active versus sham stimulation. An open-label phase 2 evaluated the effects of stimulation over four weeks. Balance (Berg Balance Scale) was the primary outcome, with secondary measures including spasticity, mobility, pain, fatigue and quality of life.
Results
Thirty-two patients completed phase 1, and 30 completed phase 2. The intervention was well tolerated. Significant improvements in balance (p < 0.001), spasticity (p < 0.001) and fatigue (p = 0.007) were observed in phase 1. Phase 2 showed sustained improvements in balance, spasticity, mobility and quality of life (p < 0.05).
Conclusions
The EXOPULSE Molii Suit demonstrated significant benefits for motor symptoms, warranting further large-scale research into long-term effects.
This clinical trial was prospectively registered on clinicaltrials.gov as ‘EXOPULSE Mollii Suit, Motor Function & Multiple Sclerosis (EXOSEP)’ (NCT 06702137). https://clinicaltrials.gov/study/NCT06702137?term=NCT06702137&rank=1
Introduction
Patients with multiple sclerosis (MS) could suffer from frequent and disabling motor symptoms, including balance and gait problems, spasticity, motor weakness and fatigue. 1 Disorders of balance and gait affect 50–80% of patients with MS (PwMS), 1 and over half of PwMS had at least one yearly fall. 1 Imbalance could imply a reduced capacity to sustain a stable position, restricted and slowed movement toward stability limits and delayed reactions to postural shifts and external disturbances. 1 Walking difficulties, in terms of quality or speed, could be related to spasticity, motor weakness, functional mobility limitations, deficits in coordination, as well as imbalance. 2 Spasticity is present in up to 80% of PwMS, it is reported by 30–50% of PwMS as moderate to severe, and it is often associated with pain, fatigue, motor impairment and gait disturbance.3,4
These motor symptoms could arise from gray and white matter pathologies affecting the sensorimotor and cerebellar systems. They could have a drastic impact on patients’ quality of life. 5 Various conservative and pharmacological approaches have been used in this context. For instance, balance and mobility could be treated with rehabilitation interventions and fampridine in PwMS.6,7 Pharmacological treatments of spasticity include baclofen, tizanidine, benzodiazepines, dantrolene and botulinum toxin.3,8,9 In addition, several nonpharmacologic methods have been used, including exercise, muscle stretching, transcutaneous electrical nerve stimulation (TENS), cryotherapy, tonic vibration, extracorporeal shock wave therapy and acupuncture. 8 Different exercise approaches – such as exergaming, balance training, cycling and proprioceptive neuromuscular facilitation – have been explored, showing promising results in improving motor symptoms and quality of life.10,11
Despite these options, many physicians remain partially or completely dissatisfied with the available interventions. 4 This highlights an unmet need and underscores the importance of further research to explore alternative treatment options. Over the last decade, the field of rehabilitation in general – and MS care in particular – has witnessed the integration of modern methods and strategies. These novel approaches include a wide array of technologies such as robotic therapy, virtual and augmented reality interventions, functional electrical stimulation, telerehabilitation platforms, wearable sensor systems, powered exoskeletons, brain–computer interfaces, smartphone-enabled healthcare programs, gamified rehabilitation instruments and artificial intelligence-based adaptive therapy systems – all of which could enhance motor re-learning, patient engagement, real-time monitoring and personalization of care.12–16
Among these technologies, the EXOPULSE Mollii Suit, a multi-site TENS device, has emerged as a novel tool capable of stimulating 40 muscle groups simultaneously. Preliminary data is available on its application in the field of rehabilitation targeting motor symptoms in patients with cerebral palsy and post-stroke as well as pain and related symptoms in patients with fibromyalgia. 17 Addressing the distribution of spasticity in PwMS represents a potential therapeutic alternative to existing interventions.
The primary objective of this study is to evaluate the short-term effects of the EXOPULSE Mollii Suit on balance in adults with MS experiencing spasticity, with the hypothesis that its use will improve balance in this population. The secondary objectives are to evaluate the effects of this technique on mobility, spasticity, pain, fatigue and quality of life.
Methods
Clinical population
The study took place at the Henri Mondor Hospital (Créteil, France) between June 2022 and May 2024. Patients were recruited among the MS outpatients presenting to the neurology department of Henri Mondor Hospital (Centre de Ressources et de Compétences (CRC) SEP Grand Paris Est).
Adult patients aged 18–75 years with a confirmed diagnosis of MS based on 2017 revised McDonald criteria, established for at least one month, 18 and an Expanded Disability Status Scale (EDSS) score <7 (indicating the ability to walk independently or with assistance), 19 were included. Participants had to be relapse-free for at least three months, speak French, be able to understand verbal instructions, and have national health insurance. They also had to have spasticity with a minimum score of 1+ on the Modified Ashworth Scale (MAS), 20 and a Berg Balance Scale (BBS) score ≤46, indicating a risk of falling. 21 Exclusion criteria included participation in another research study, inability to attend medical monitoring due to geographical or social constraints, contraindications to the EXOPULSE Mollii Suit (cardiac stimulator, ventriculoperitoneal shunt, intrathecal baclofen pump), pregnancy, recent changes in pharmacological treatment (last three months), other somatic (arrhythmias, uncontrolled epilepsy, diseases causing osteoarticular and muscular pain) or neuropsychiatric conditions, body mass index >35 kg/m² or being under legal protection. Patients who had been introduced to another medical device during the study were also excluded.
Ethical approval and information
The protocol was approved by an independent ethics committee (Comité de Protection des Personnes Sud-Est I) on 16 May 2022. Written informed consent was obtained from all participants before enrollment. Financial compensation was limited to reimbursement of travel expenses for study visits.
Study product and intervention
The EXOPULSE Mollii Suit (EXONEURAL NETWORK AB, Danderyd, Sweden) consists of two CE-marked medical devices: (a) a set of body garments (jacket and pants, classified as Class I according to Regulation EU 2017/745) embedded with electrodes, conductive wires and connectors, and (b) a control unit (active Class IIa), a battery-powered device that transmits low-energy electrical pulses to the garments via the connectors (Figure S1). This system provides non-invasive electrostimulation to key nerves and associated muscle groups throughout the body. During active sessions, the device applied a low-intensity current (pulse width: 25–175 µs) at 20 Hz, ensuring stimulation remained below the motor threshold. During sham sessions, the same current was applied for one minute before automatically shutting off, producing cutaneous sensations that simulated active stimulation to maintain blinding.
Study design
The study was conducted in two phases (Figure 1). Phase 1 was a randomized, sham-controlled, double-blind, crossover trial (RCT) in which participants were randomly assigned to receive either a single 60-min session of active or sham stimulation, with a two-week washout interval. At the end of phase 1 (two weeks after the second session of active or sham), all participants were offered enrollment in an open-label phase 2 consisting of 14 sessions of active stimulation over four weeks (one session every other day, each session lasting 60 min). Details of each phase, study visits, randomization procedure and patients’ instructions are available in Supplementary Section 2.4.
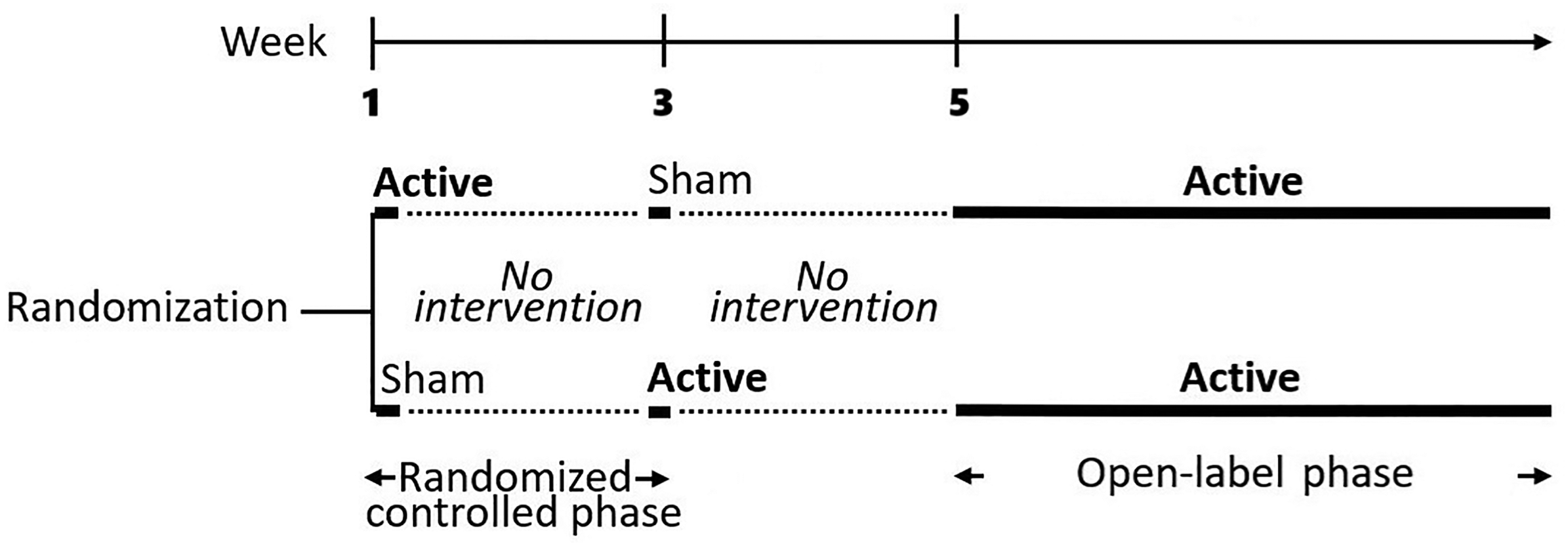
Study design illustrating the crossover randomized controlled trial (phase 1) and the open label trial (phase 2).
Study objectives and endpoints
The primary objective of this study was to evaluate the short-term effects of the EXOPULSE Mollii Suit on balance in adult PwMS using the BBS (primary endpoint) after a single active intervention (in the randomized phase 1). 22
Secondary objectives included assessing the effects of this intervention on spasticity (MAS, Visual Analogue Scale [VAS]),20,22 mobility (Timed Up and Go Test [TUG], Multiple Sclerosis Walking Scale-12 [MSWS-12], 23 and Falls Efficacy Scale-International [FES-I]),23,24 quality of life (Multiple Sclerosis International Quality of Life Questionnaire [MusiQoL]), 25 and pain and fatigue (VAS). 22 Patients reporting pain and fatigue at baseline (VAS > 4) were asked to evaluate the impact of the intervention on both symptoms. 22
In addition, patients completed the Clinical Global Impression of Change (CGI) after each intervention. 26 A questionnaire was also administered to assess the integrity of the blinding. The assessment tools’ content, scoring methods and administration timepoints are available in Supplementary Section 2.5.
Statistical analysis
Sample size calculations were performed using G*Power software (version 3.1.9.6). 27 With parameters set for a mean effect size of 0.25, a two-tailed significance level of α = 0.05 and an estimated power of 80%, a sample size of 34 was determined, accounting for potential dropouts. Statistical analysis was performed using IBM SPSS Statistics (version 30, Armonk, NY: IBM Corp). Because the quantitative data did not follow a normal distribution (Shapiro–Wilk test), comparisons for the randomized phase were made using Friedman's tests followed by Bonferroni-adjusted post-hoc Dunn's tests. Wilcoxon signed-rank tests were used for the open-label phase. Categorical endpoints (CGI and blinding integrity) were compared using the Chi2 test. The significance level was set at 0.05. Effect size estimation was based on Kendall's coefficient of concordance W for the randomized phase and r = Z/√N for the open-label phase. Quantitative variables are reported as mean ± standard deviation. Categorical variables are reported as frequencies or the number of participants.
Results
Clinical and demographic characteristics
Overall, 45 patients were screened initially; 34 patients were recruited. The participation flow chart in phase 1 is presented in Figure 2. Thirty-two patients completed phase 1 (n = 16 females, 50.00%, age: 47.81 ± 10.07 years), and 30 patients completed phase 2. The mean disease duration was 11.84 ± 9.36 years, and the mean EDSS score was 4.28 ± 1.79. The disease type, treatments and other interventions are summarized in Supplementary Section 3.1.

Participation flow chart (crossover randomized controlled trial, phase 1).
Randomized phase
Blinding integrity was preserved as the number of patients who guessed or not the type of intervention did not differ between active and sham conditions (χ2 = 1.70, p = 0.193). Additional analysis was conducted to assess whether blinding was different according to the order of intervention. The proportions of correct answers did not significantly differ between the first and the second interventions (χ² = 0.07, p = 0.794).
The CGI was significantly different between both conditions (χ2 = 10.11, p = 0.018). Post-hoc tests revealed that the active condition resulted in a significantly higher frequency of ‘much improved’ (31.25% vs. 9.38%, p < 0.05) and a lower frequency of ‘no change’ (18.75% vs. 43.75%, p < 0.05) compared to the sham condition. Any improvement (minimal, much or very much) was reported by 81.25% of patients following the active intervention and by 56.26% following the sham intervention.
Efficacy results from the randomized phase are shown in Table 1. For BBS, Friedman's test for repeated measures yielded an χ2 value of 80.77 (p < 0.001). Post-hoc analysis indicated a significant increase in scores following the active condition, with scores rising from a baseline of 38.25 ± 7.68 to 47.41 ± 7.09 post-active (Dunn's p < 0.001) (Figure 3). No significant differences were noted within the sham condition (pre-sham BBS: 37.56 ± 8.09, post-sham BBS: 37.56 ± 8.09, Dunn's p = 1.000) nor were there significant baseline differences between the active and sham conditions (Dunn's p = 1.000). Additionally, 68.75% of patients had a BBS score greater than 46 following the active condition, compared to 0.00% after the sham condition.

Berg Balance Scale before and after active and sham periods of stimulation (randomized phase 1) in patients with multiple sclerosis (phase 1). Results are displayed as mean ± SEM. ***p < 0.001.
Summary of efficacy endpoints before and after each single session of active or sham intervention (randomized phase 1) in adult patients with multiple sclerosis.
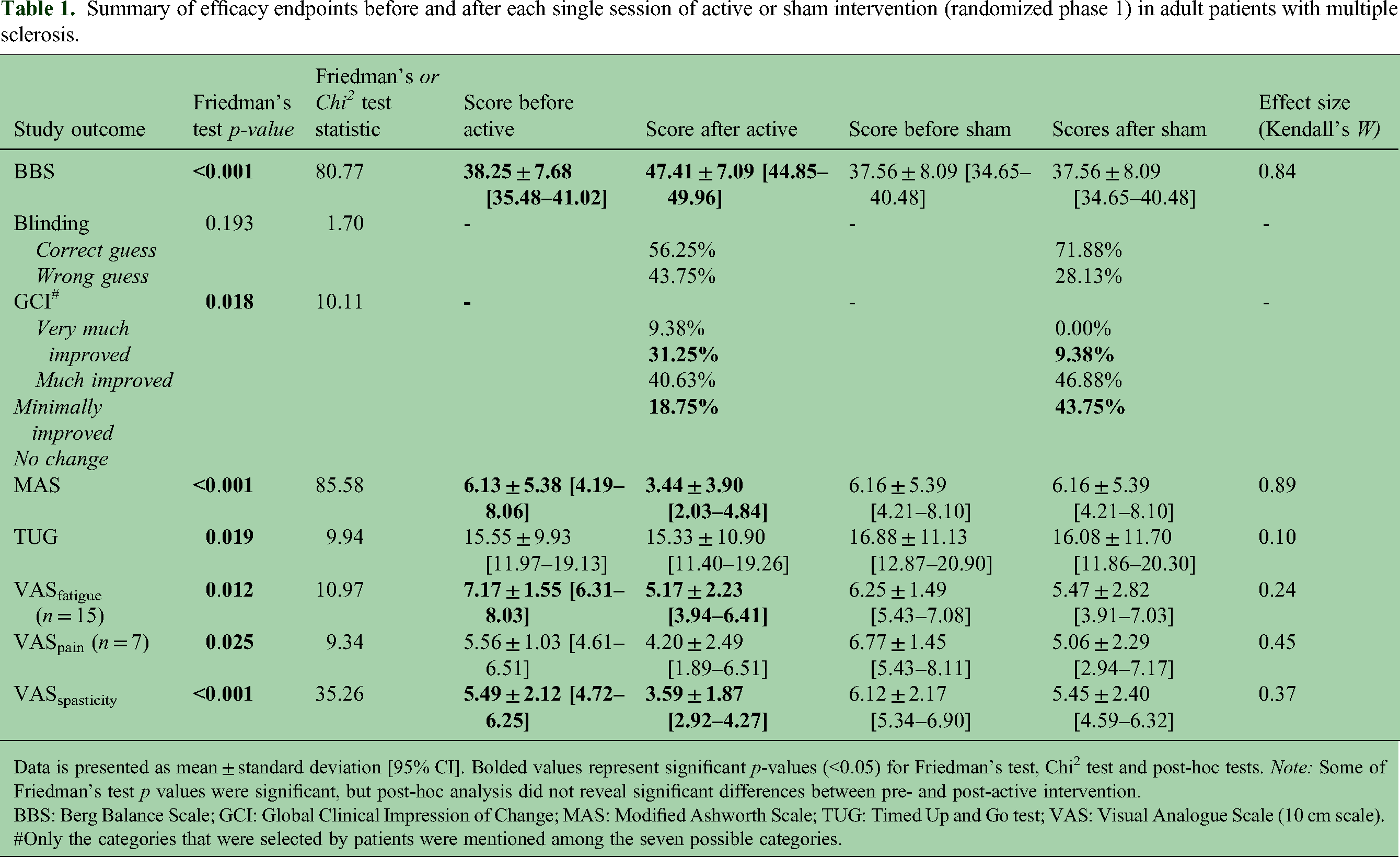
Data is presented as mean ± standard deviation [95% CI]. Bolded values represent significant p-values (<0.05) for Friedman's test, Chi2 test and post-hoc tests. Note: Some of Friedman's test p values were significant, but post-hoc analysis did not reveal significant differences between pre- and post-active intervention.
BBS: Berg Balance Scale; GCI: Global Clinical Impression of Change; MAS: Modified Ashworth Scale; TUG: Timed Up and Go test; VAS: Visual Analogue Scale (10 cm scale).
Only the categories that were selected by patients were mentioned among the seven possible categories.
Also, a significant reduction in spasticity was observed based on both VASspasticity (Friedman's χ2 = 35.26, p < 0.001) and MAS (Friedman's χ2 = 85.58, p < 0.001). Post-hoc tests indicated a notable decrease in scores after the active intervention, but not after the sham intervention, as demonstrated by VASspasticity (pre-active: 5.49 ± 2.12 vs. post-active: 3.59 ± 1.87; Dunn's p = 0.001) and MAS (pre-active: 6.13 ± 5.38 vs. post-active: 3.44 ± 3.90; Dunn's p < 0.001).
Additionally, a significant change was noted in VASfatigue (Friedman's χ2 = 10.97, p = 0.012), with post-hoc analysis showing a reduction in fatigue following the active intervention (pre-active: 7.17 ± 1.55 vs. post-active: 5.17 ± 2.23; Dunn's p = 0.007).
In contrast, neither intervention significantly affected mobility as measured by TUG nor pain according to VASpain. Detailed results are presented in Table 1.
The effect sizes for outcomes showing significant improvement in phase 1 were large for balance (0.84), medium to large for spasticity depending on the scale used (VASspasticity: 0.37 vs. MAS: 0.89) and small for fatigue (VASfatigue: 0.24).
To exclude the effect of aging, the statistical analysis was repeated on a sample of 30 patients after excluding the two patients aged above 65 years (68 and 70 years), and this exclusion did not affect the results of phase 1. Significant effects were observed on BBS (Friedman's χ2 = 76.44 and p < 0.001, Dunn's p < 0.001), MAS (Friedman's χ2 = 79.58 and p < 0.001, Dunn's p < 0.001), VASspasticity (Friedman's χ2 = 33.00 and p < 0.001, Dunn's p = 0.002), VASfatigue (Friedman's χ2 = 12.97 and p = 0.005, Dunn's p = 0.002) and CGI (χ 2 = 8.73, p = 0.033).
Open-label phase
After one month of intervention, the Wilcoxon signed-rank test showed significant effects (p < 0.05) for several study outcomes, including balance, spasticity (MAS and VASspasticity), walking (FES-I and MSWS-12, but not TUG) and quality-of-life measures (MusiQoL index score and subscores for activities of daily living, coping and rejection). After the open-label phase, 60.00% of patients achieved a BBS score greater than 46. According to CGI, 80.00% of patients reported improvement after phase 2 (mild in 63.33% and much in 16.67%). In contrast, no significant changes were observed in VASpain, VASfatigue or other quality-of-life dimensions. The results are summarized in Table 2.
Summary of efficacy endpoints before and after the four-week active intervention period (open phase 2) in adult patients with multiple sclerosis.

Data is presented as mean ± standard deviation [95% CI]. Bolded values represent significant p-values (<0.05).
BBS: Berg Balance Scale; FES-I: Falls Efficacy Scale-International; MAS: Modified Ashworth Scale; MSWS-12: 12-item Multiple Sclerosis Walking Scale; MusiQoL: Multiple Sclerosis International Quality of Life Questionnaire; TUG: Timed Up and Go test; VAS: Visual Analogue Scale (10-cm scale).
The effect sizes for outcomes showing significant improvement after phase 2 were large for balance (0.85) and spasticity (VASspasticity: 0.57 vs. MAS: 0.83), medium to large for mobility depending on the scale used (FES-I: 0.39 vs. MSWS-12: 0.50) and medium to large for quality of life (MusiQoL index: 0.43, rejection: 0.40, activities of daily living: 0.50, coping: 0.56).
After excluding both patients aged above 65 years, the previously observed significant effects were maintained: BBS (Z = 4.51, p < 0.001), MAS (Z = −4.39, p < 0.001), VASspasticity (Z = −3.02 and p = 0.003), FES-I (Z = −2.02, p = 0.043), MSWS-12 (Z = −2.91, p = 0.004) and MusiQoL (index: Z = 2.48 and p = 0.013, rejection: Z = 2.11 and p = 0.035, activities of daily living: Z = 2.69 and p = 0.007 and coping: Z = 3.07 and p = 0.002).
Tolerance
All the stimulation sessions were well tolerated, and no serious adverse events were reported at any time.
Discussion
This study consisted of two phases – an RCT and an open-label study – to evaluate the efficacy of the EXOPULSE Mollii Suit on balance, associated symptoms and quality of life in PwMS. To our knowledge, it is the first to use this technology in this clinical population. The results suggest beneficial effects on balance, as well as on spasticity, mobility, fatigue and quality of life. Spasticity reduction showed a medium to large effect after phase 1 and a large effect after phase 2 for both scales. Fatigue reduction had a small effect after phase 1 but was not significant after phase 2. Improvements in mobility and quality of life showed medium to large effect sizes after phase 2.
Balance involves the integration of inputs from the proprioceptive, vestibular and visual systems in the central nervous system (cerebellum, brainstem and cerebral cortex). The balance of a healthy person in a standing position is determined by 20% visual input, 70% proprioceptive information and 10% vestibular input.
Muscle spindles are the primary receptors involved in proprioception. 28 They offer proprioceptive feedback for the muscles in the lower legs and trunk and are crucial for maintaining postural stability. Large-diameter fibers – like A alpha fibres that innervate muscle spindles – have a lower excitability threshold compared to motor axons and muscle fibers. Here, one can assume that applying low-intensity electrical stimulation over the skin will act on the most excitable fibers, activate muscle spindles, increase proprioceptive afferent inputs and improve balance. In addition to peripheral mechanisms, literature suggests that TENS can improve postural balance via other plausible mechanisms at spinal (motoneurons excitability) and supraspinal cortical levels (neuroplasticity). 29
No previous work has applied this medical device or other TENS interventions to improve balance in PwMS. However, few works assessed the impact of TENS on mobility in PwMS and documented positive outcomes following stimulation of tibialis anterior and quadriceps muscles bilaterally.30,31
In addition, the observed clinical improvement in balance and mobility after using the EXOPULSE Mollii Suit could also be partially attributed to the reduction in spasticity observed in this study and some previous works on cerebral palsy and stroke. 17 This improvement may also indicate an increase in muscle strength and passive range of motion. 17 Very few studies have applied TENS in the context of MS spasticity, with mixed results.9,32,33 In addition, some data suggest positive effects of TENS on motor functions and mobility in this clinical population.30,31,34 The higher effect size observed with the MAS during the four-week intervention suggests that further reduction in spasticity and additional clinical improvement could be achieved by extending the stimulation protocol over a longer duration.
From a mechanistic point of view, in addition to the corticospinal pathway involvement, several other pathways seem to be involved in the pathophysiology of spasticity.35,36 For instance, spasticity could arise from structural damage impairing the integrity of the reticulospinal tract and its inhibitory effect or from a defective cortical excitation of the vestibulospinal tract resulting in reduced motoneuron firing, a low extensor tone and a flexed posture. 37 Moreover, it could arise from secondary adaptations at the spinal circuit level. 37 Following a brain injury, the loss of inhibitory control would shift the balance toward hyperexcitability of spinal reflexes, resulting in reduced reciprocal Ia and non-reciprocal Ib inhibitions as well as reduced presynaptic inhibition of Ia afferent fibers, which could contribute to the appearance of spasticity. 36 Furthermore, besides spinal hyperreflexia, modifications of the muscles and tendon mechanical properties (e.g., physiological changes in muscle fibers, altered tendon compliance and subsequently a heightened mechanical resistance of muscles) could contribute to the observed symptomatology. 36 The use of the EXOPULSE Mollii Suit primarily involves stimulating the non-spastic muscles that act as antagonists to the spastic ones, aiming to decrease muscle tone (spasticity) through the mechanism of reciprocal inhibition. The other cited mechanisms might also have contributed to the observed effects.
Interestingly, the observed clinical improvement in mobility varied depending on the assessment tool used, with significant effects noted for the FES-I and MSWS-12 but not the TUG. This discrepancy can be attributed to several factors. First, the FES-I and MSWS-12 are patient-reported outcomes, while the TUG is a performance-based measure. Second, these tools assess different outcomes: the FES-I focuses on fear of falling and perceived risk, the MSWS-12 evaluates walking limitations and the TUG examines overall mobility. Third, unlike the first two measures, which rely on patients’ perceptions, the TUG can be influenced by cognitive factors. 38
Moreover, the observed improvement in balance and mobility in this work was accompanied by a significant improvement in patients’ quality of life, as observed in a previous TENS study using MusiQoL. 34 They reported an improvement in their activities of daily living, expressed better coping skills, and a decrease in perceived social rejection.
Furthermore, in this study, fatigue significantly decreased following phase 1 but not phase 2, while pain did not change significantly regardless of the intervention type or phase. However, it is important to note that fatigue and pain were assessed as secondary outcomes in a small subset of patients, with an even smaller sample for pain assessment; thus, the results should be interpreted with caution. Positive antifatigue effects have been documented in PwMS following TENS, as measured by the Modified Fatigue Impact Scale (MFIS). 34 Fatigue is a frequent, debilitating, multifactorial and multidimensional symptom that can fluctuate in PwMS. 39 Therefore, using a multidimensional scale (e.g., MFIS) in conjunction with unidimensional tools (e.g., VAS) and obtaining multiple measures may help address the observed limitations in future studies. As for pain, reduction of neuropathic pain in PwMS was observed following TENS according to a systematic review, 40 as well as in patients with fibromyalgia following the use of EXOPULSE Mollii Suit.41,42 The mechanisms by which analgesia could be obtained primarily involve the gate control theory but might also include other central and peripheral mechanisms. 17 Evaluating the effects of this technique in a larger cohort of PwMS suffering from pain and employing a multidimensional pain assessment might help better understand its utility in this clinical context.
Finally, it is important to highlight the limitations of this current work. Although this is the first RCT assessing the utility of EXOPULSE Mollii Suit in PwMS, it could be limited by its monocentric design, number of sessions in the RCT phase, relatively short follow-up, sample size and the concomitant pharmacological and alternative interventions in the study cohort. However, this last point can also provide evidence of the utility of this medical device as an add-on intervention for PwMS with previous insufficient response to available therapeutics. Regarding the study design, the RCT phase included only a single session per condition, with evaluation conducted immediately afterward. Although sustained effects were observed during the open-label phase, placebo effects cannot be formally excluded, and the long-term effects beyond a single stimulation session remain to be elucidated. Furthermore, the study did not include an objective assessment of motor functions, such as 3D motion analysis or posturography, which would have provided complementary data on the participants’ motor performance. Therefore, larger multicenter RCTs with longer stimulation periods, extended follow-up and a multidimensional evaluation are needed to further confirm the benefits of this technique.
Conclusion
The present results suggest beneficial effects of EXOPULSE Mollii on balance, mobility, spasticity, fatigue and quality of life in PwMS, with further clinical benefits observed when stimulation sessions were repeated. The findings of this intervention appear promising in addressing the debilitating and challenging motor symptoms experienced by PwMS and the associated altered quality of life.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251348304 - Supplemental material for The effect of the EXOPULSE Mollii suit on motor functions in patients with multiple sclerosis – a randomized sham-controlled crossover trial
Supplemental material, sj-docx-1-mso-10.1177_20552173251348304 for The effect of the EXOPULSE Mollii suit on motor functions in patients with multiple sclerosis – a randomized sham-controlled crossover trial by Samar S Ayache, Joseph G Mattar, Alain Créange, Mohamed Abdellaoui, Mickael Zedet, Jean-Pascal Lefaucheur, Hana Megherbi, Hayfa Khaled, Georges Naïm Abi Lahoud and Moussa A Chalah in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgments
The authors would like to thank all the patients for their participation in this study, the editor and reviewers for the valuable comments that increased the quality of this article, and Ms Imène Lameche for study monitoring.
Author contributions
J.G.M., M.A.C., A.C., J.P.L. and S.S.A: Design and conceptualization of the study. M.A.C. and J.P.L.: Statistical analysis and visualization. J.G.M.: Data collection. G.N.A.L.: Data curation. S.S.A.: Supervision and project administration. J.G.M., M.A.C., A.C., M.Z., M.A., H.K., H.M., J.P.L., G.N.A.L. and S.S.A.: Data interpretation. J.G.M., M.A.C. and S.S.A: Writing – Original draft preparation. J.G.M., M.A.C., A.C., M.Z., M.A., H.K., H.M., J.P.L., G.N.A.L. and S.S.A.: Writing – Reviewing and Editing. All authors read, commented on and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: MC declares having received compensation from Janssen Global Services LLC, Exoneural Network AB, Sweden and Ottobock, France. AC declares having received financial support for research from Biogen, GeNeuro, MedDay, Novartis, Octapharma and Roche and having received honoraria from Biogen, GeNeuro, Novartis and Roche. SA declares having received compensation from Sanofi Aventis, France; Novartis, France; Exoneural Network AB, Sweden and Ottobock, France. The remaining authors declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study sponsor (“promoteur”) was the Association l'Institut de la Colonne Vertébrale et des Neurosciences (ICVNS, France).
Ethical considerations
The protocol was approved by an independent ethics committee (Comité de protection des personnes [CPP] Sud-Est I) on 16 May 2022. Following French legislation and regulatory requirements, the approval was sent for information to the French health authority (Agence Nationale de sécurité du médicament et des produits de santé, Saint-Denis, France) before the inclusion of the first participant. The study was conducted in accordance with the ISO 14155 standard (Clinical investigation of medical devices for human subjects – Good clinical practice) and the European Union Regulation on medical devices (2017/745).
Consent to participate
Written informed consent was obtained from all participants before enrollment into the study. Financial compensation was provided only for travel costs to study visits.
Data availability statement
Data will be made available by the corresponding authors upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
