Abstract
Background and Objectives
The genetic basis of adult-onset multiple sclerosis (MS) is well-studied, but less is known about pediatric-onset MS (pedMS), comprising approximately 5% of all MS onsets. Mendelian randomization (MR) studies have demonstrated evidence for a causal association between MS and both 25-hydroxyvitamin D [25(OH)D] serum levels and genetic variation related to vitamin D receptor (VDR) binding. The objective was to identify whether VDR binding variants (VDR-BVs) previously implicated in adult-onset MS were associated with pedMS using genetic instrumental variables (GIVs).
Methods
Using previously identified VDR-BVs to construct individual GIVs with two-sample MR, we investigated associations with pedMS in 725 cases and 592 controls of European ancestry from the US Network of Pediatric MS Centers. Associations between each VDR-BV and pedMS were estimated using logistic regression adjusting for the first three genome-wide principal components. A significant interaction between a VDR-BV and 25(OH)D GIV provided evidence for a causal association unbiased by pleiotropy.
Results
One VDR-BV, rs2531804, previously associated with adult-onset MS, was also significantly associated with pedMS after multiple testing correction.
Discussion
This study is the first to use VDR-BVs from previous MR studies to demonstrate causal differences in VDR binding at a locus contributing to pedMS susceptibility.
Keywords
Introduction
Globally, 2.9 million people live with multiple sclerosis (MS). In the United States, approximately 5% of these MS patients have disease onset before age 18 (pediatric-onset or pedMS).1,2 Because children/adolescents with MS may carry a higher genetic burden and have shorter time between exposure and symptom onset, studying this patient group offers opportunities to explore hypotheses that are challenging to test in adults. 3 PedMS shares several risk factors with adult-onset MS, including HLA-DRB1*15:01—a well-known genetic risk factor—along with exposure to tobacco smoke, Epstein-Barr virus infection, and low serum vitamin D levels.3,4
The genetic basis of MS in adults has been extensively studied—over 230 susceptibility variants have been identified through genome-wide association studies (GWAS). 5 Notably, residing in latitudes further from the equator—where ultraviolet B radiation is lower—is an established risk factor for MS. 5 As a result, the geographic distribution of MS has led to the hypothesis that high vitamin D levels may be protective. 6 Previous studies have shown that high serum 25-hydroxyvitamin D [25(OH)D] levels, greater sunlight exposure, and both vitamin supplements and diets rich in vitamin D are associated with a decreased risk of adult-onset MS.7–10 Studies have also demonstrated a strong association between both low vitamin D status and increased risk of developing pedMS and relapse rate, although supplementation trials did not prove to prevent MS relapses.3,11,12
Utilization of Mendelian randomization (MR) analysis has demonstrated a causal role for low vitamin D status in MS.13,14 MR is a method that uses one or more genetic variants associated with an exposure as a genetic instrumental variable (GIV) to estimate causal associations between that exposure and a given outcome. 15 There are three main assumptions for the application of MR: (a) the variant(s) must be associated with the exposure, (b) the genetic variant(s) only affect the outcome through the exposure, and (c) there is no unmeasured confounding of the association between the genetic variant(s) and the outcome. 15 Recent MR studies in three large, adult-onset MS case-control datasets and one pedMS study have demonstrated compelling evidence for a causal relationship between low serum 25(OH)D levels and increased MS susceptibility.11,13,15 Several of the genetic variants utilized in these MR studies play roles in the biosynthesis of vitamin D, signifying that different components of the vitamin D pathway, particularly transcription and expression regulated by the binding of the vitamin D receptor (VDR) to DNA, may contribute to the relationship between vitamin D and MS.16,17 Because single nucleotide polymorphism (SNP) genotypes are determined at birth and not likely to be influenced by potential confounding factors, MR analysis can overcome problems of both reverse causation and confounding when assessing the causal relationship between an exposure and outcome. 15
Vitamin D has immunomodulatory effects and plays a role in regulating calcium metabolism, cellular growth, proliferation and apoptosis, and other essential immune functions including the modulation of inflammatory processes. 18 25(OH)D communicates via the nuclear VDR, a ligand-regulated transcription factor that facilitates all genomic actions of 25(OH)D.13,19 Gene expression profiling studies have revealed that 25(OH)D signaling through the VDR can activate or repress the transcription of target genes. 20 The VDR, similar to other nuclear receptors, exerts its transcriptional control by binding to specific high-affinity DNA sequences known as vitamin D response elements (VDREs). These VDREs are found in the regulatory regions of genes targeted by vitamin D. Upon VDRE binding, the liganded VDR recruits a complex of coregulatory proteins, facilitating the transactivation of proximal target genes. Vitamin D exposure induces a conformational change in the DNA-bound VDR, promoting a transition in protein-protein interactions from corepressor to coactivator complexes, thereby modulating genetic transcriptional activity. 21 Studies examining gene expression profiles have demonstrated that 25(OH)D signaling through the VDR can either activate or suppress the transcription of target genes (see Figure 1). 20
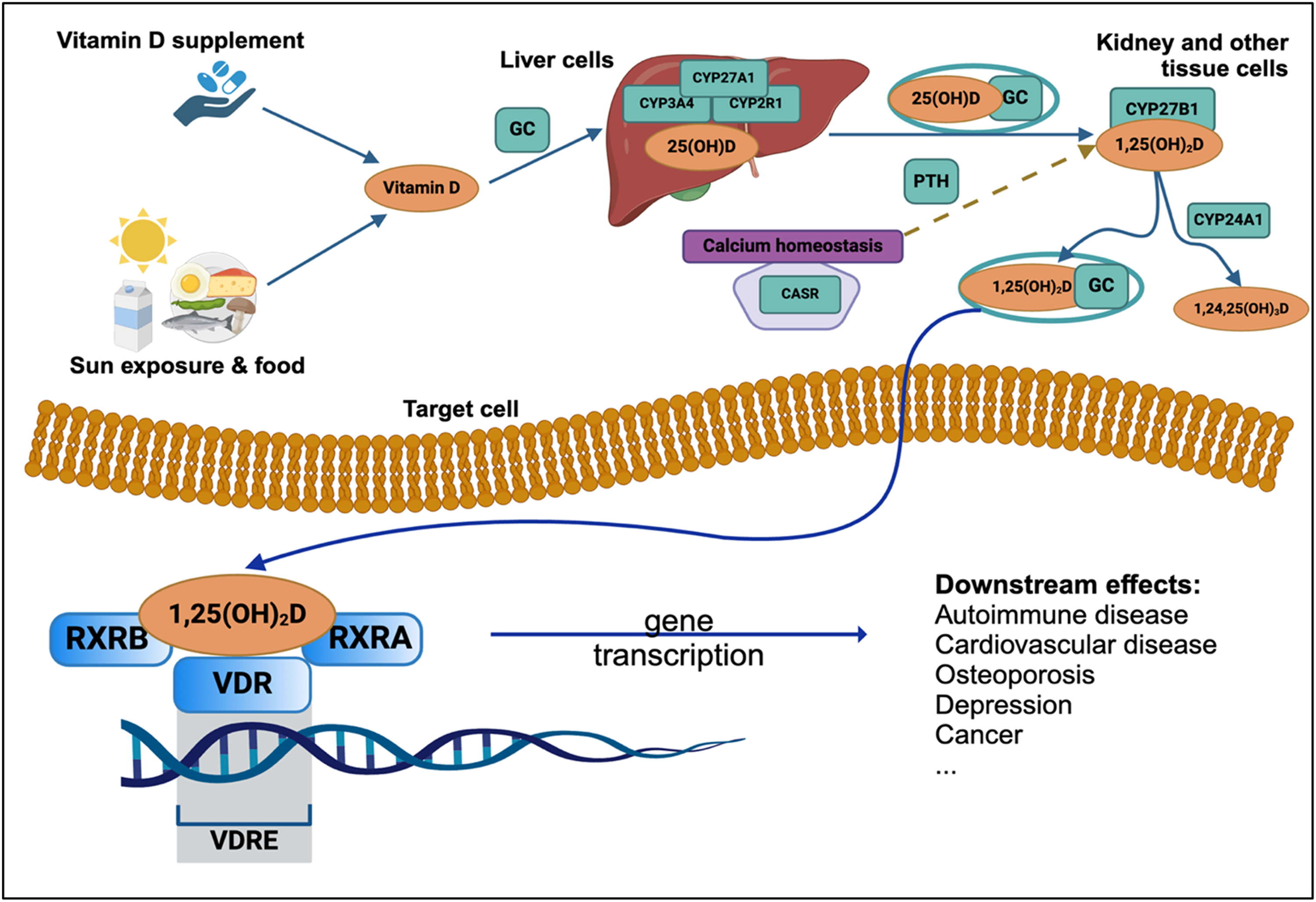
Vitamin D metabolism and receptor binding pathway in human body. Figure created using BioRender.
Prior research utilizing chromatin immunoprecipitation coupled with exonuclease digestion (ChIP-Exo) has demonstrated that VDRs can bind preferentially to one allele (VDR-binding variants, or VDR-BVs) over another at heterozygous sites in lymphoblastoid cell lines (LCLs). 22 Analysis of 1000 Genomes data has shown that VDR-BVs are enriched within genomic regions linked to autoimmune conditions, such as MS. 22 These results suggest that genetic variations influencing VDR binding may be the causal mechanisms underlying MS GWAS findings, making VDR-BVs promising candidates for exploring genetic variations pertinent to pedMS risk using MR methods.
Here, we aimed to identify whether VDR-BVs implicated in adult-onset MS are associated with pedMS cases and controls in a study of individuals recruited from the U.S. Network of Pediatric MS Centers. We utilized data on 1317 individuals from a previously-described pedMS case-control study.3,23 We hypothesized that genetic variation in VDR binding, known to disrupt the vitamin D-mediated regulation of genes and pathways, affects the risk of developing pedMS. Identifying VDR-BVs associated with pedMS susceptibility will improve our understanding of the biological pathways through which vitamin D influences pedMS.
Materials and methods
Pediatric MS cohort
As previously described, 2287 individuals were recruited as a large multiethnic cohort from the U.S. Network of Pediatric Multiple Sclerosis Centers.11,23 Of these, 725 cases and 592 controls of European ancestry were selected for the present study. PedMS cases were defined as individuals clinically diagnosed with MS onset before age 18 years. 4 Controls were participants without a diagnosis of MS or related condition (transverse myelitis, optic neuritis, or demyelination disease) recruited as part of the aforementioned pedMS case-control study. Blood samples were collected from each participant for DNA extraction. This study was approved by the local institutional review boards of participating centers. All patients and parents signed assent and consent forms for this study.
Genetic data and quality control measures
SNP genotyping was performed using the Illumina Infinium 660K OmniExpress and OmniExpressExome BeadChip arrays and imputed against reference haplotypes from Phase 3 of the 1000 Genomes Project using IMPUTE4 on the Michigan Imputation Server. Participants with missing genotypes that met quality control thresholds (INFO score > 0.8, missingness per SNP < 0.05, and minor allele frequency (MAF) > 0.05) were imputed using the mean MAF. SNPs with an INFO score < 0.1, a MAF < 1%, a genotyping rate of <90%, or deviations from Hardy–Weinberg equilibrium among controls (p < 0.000001) were removed. To reduce potential confounding by population stratification, the larger, multi-ethnic cohort (N = 2287: 1303 cases, 984 controls) was restricted to individuals with primarily European ancestry (≥80% EUR), yielding a final study sample of 1317 individuals, of which 725 were cases and 592 were controls. Genome-wide genetic ancestry was inferred using SNPWeights version 2.1.24,25
Identifying valid genetic instruments for VDR binding
Occupancy of VDR at a given genomic locus was the exposure of interest. 305 VDR-BVs were previously identified from Gallone et al. as reference candidates for the construction of the GIV for each VDR-BV (hereafter, GIVVDR) utilized in the study of adult-onset MS by Adams et al.22,26 For the present study, candidate VDR-BVs were selected from SNPs previously shown to be significantly associated with VDR binding at a locus in adult-onset MS to detect potential variations in VDR binding and related associations with pedMS susceptibility.22,26 All seven VDR-BVs significant in adult-onset MS were present in our quality-controlled dataset and available for analysis. 26 A GIVVDR was derived for each of these individual VDR-BVs by multiplying the dosage of the effect allele for VDR binding by the SNP-VDR binding effect size.
Linkage disequilibrium (LD) between VDR-BVs and MS GWAS risk variants was estimated in Plink1.9 with the --r2 function using the European LD reference panel from 1000 Genomes Phase 3. 27
Pleiotropy and bioavailability of vitamin D
MR analysis yields unbiased causal estimates in the absence of pleiotropic effects. As there was only one instrumental SNP for each GIVVDR, sensitivity analyses for multi-SNP MR instruments were not conducted in the present study. Because VDR binding is dependent upon the bioavailability of vitamin D, we used summary statistics, estimated effect sizes, and variants from two recent GWAS on serum 25(OH)D to construct the GIVs for bioavailability of vitamin D (hereafter GIV25(OH)D). The GIVs25(OH)D were constructed from variants identified by Revez et al. and Jiang et al., replicating methods previously used in adult-onset MS.16,17,26 Briefly, the MR-Base R platform was used to extract GWAS summary statistics. 28 The extract_instruments() function in the TwoSampleMR R package was used to identify independent variants with GWAS p < 1 × 10−8 within genomic windows of 10,000 kb (R2 < 0.001) using a European LD reference panel from 1000 Genomes Phase 3. 28 These measures produced 7 SNPs for the Jiang et al. GIV25(OH)D and 84 SNPs for the Revez et al. GIV25(OH)D. Each GIV25(OH)D was calculated in Plink1.9 using --score after identifying these reference SNPs and standardized (centered and scaled) using the scale() function in R.27,29 The presence of an interaction between a GIVVDR and bioavailability of 25(OH)D was evidence of an association between VDR binding and pedMS susceptibility not biased by horizontal pleiotropy.
Statistical methods
Associations between all GIVs (GIVVDR and GIV25(OH)D) and pedMS susceptibility were estimated using logistic regression for each VDR j and 25(OH)D GIV k:
In addition, to estimate associations not biased by horizontal pleiotropy, we estimated interaction between each GIVVDR and GIV25(OH)D for pedMS susceptibility:
Meta-analysis was used to combine results from the present study (pedMS) with those in adults from Adams et al. (KPNC, UKB, GSA, Human Omni) for each of the GIVs25(OH)D and the GIVVDR for rs2531804, resulting in three separate meta-analysis models. 26 All meta-analyses were executed using the metagen() function in the Meta R package. Between-study variance was estimated using the Der Simonian-Laird estimator and between-study heterogeneity was evaluated using Cochran's Q statistic and Higgins & Thompson's I’ statistic. 30 P-values calculated from associations between each GIV25(OH)D and pedMS and the interaction parameter between each GIVVDR and GIV25(OH)D were corrected for multiple tests using the Benjamini–Hochberg method (BH). 31 All models were adjusted for the first three genome-wide principal components.
All statistical analyses were conducted using R 4.0.2. 29
Results
Characteristics of pedMS cases and controls
The characteristics of pedMS cases and controls included in this study are presented in Table 1. In both the case and control groups, most subjects were female (69.4% of cases and 60.1%, respectively). Among pedMS cases, the average age of MS onset was 14.32 years, and 87 cases were diagnosed before puberty. HLA-DRB1*15:01 is a well-established genetic risk factor for MS. Its tagging SNP, the rs3135388 A allele, is significantly associated with MS susceptibility. 32 In agreement, 6.6% of pedMS cases and only 1.2% of controls had two copies of this allele (p < 0.001).
Characteristics of pedMS cases and controls.

GIV25(OH)D associated with pedMS susceptibility
The mean of the GIVs25(OH)D for both Jiang et al. and Revez et al. were marginally higher among pedMS cases compared to controls (Table 1). Logistic regression analyses adjusted for the first three genome-wide principal components and meta-analyses combining results from adult-onset MS demonstrated that decreased serum 25(OH)D was associated with increased odds of pedMS (Table 2 and Figure 2). The Jiang et al. GIV25(OH)D had a slightly larger magnitude of association with pedMS (odds ratio [OR]: 1.18, 95% confidence interval [CI]: 1.06–1.32, Table 2) compared to the Revez et al. GIV25(OH)D (OR: 1.12, 95% CI: 1.00–1.25, Table 2). Results from meta-analyses also demonstrated a greater magnitude of association for the GIV25(OH)D from Jiang et al.

(A, B) Results from meta-analyses of association between 25(OH)D genetic instrumental variables (GIV25(OH)D) and adult MS (KPNC, UKB, GSA, Human Omni from Adams et al. 26 ) and pedMS. τ2 = estimate of between study heterogeneity; I2 = proportion of variance due to heterogeneity; and p = p-value for Cochran's Q test of heterogeneity. (A) GIV25(OH)D calculated using independent GWAS variants from Jiang et al. 2019 17 and its association with adult and pedMS; (B) GIV25(OH)D calculated using independent GWAS variants from Revez et al. 2020 16 and its association with adult and pedMS. (C) Results from meta-analyses of GIVVDR for rs2531804 and its association with adult MS (KPNC, UKB, GSA, Human Omni from Adams et al. 26 ) and pedMS.
25(OH)D instrumental variables (IVs) tested for their association with pedMS at p < 0.05.

GIV 25(OH)D calculated using summary statistics from Jiang et al 2019 and Revez et al 2020 GWAS.
Odds ratios are from logistic regressions adjusted for the first three genome-wide principal components.
ORs for association between 25(OH)D GIV and pedMS demonstrate that decreased serum vitamin D is associated with increased odds of pedMS.
VDR-BVs associated with MS susceptibility
Of the seven GIVsVDR previously shown to be significantly associated with adult-onset MS (p < 0.05), rs2531804 had a significant association with pedMS (OR: 0.58, 95% CI: 0.40–0.84; Table 3). This GIVVDR also remained significant following correction for multiple testing (pBH < 0.05). Meta-analyses of the GIVVDR for rs2531804 demonstrated consistent evidence for association between VDR binding and MS susceptibility across both the adult and pediatric studies. It was associated with a decreased odds of pedMS and adult-onset MS (Meta OR: 0.84, 95% CI: 0.79–0.88, p < 0.0001, Figure 2C).
Vitamin D receptor binding variant instrumental variables previously implicated in adult MS

Abbreviations: bp, base pair; chr, chromosome; CI, confidence interval; BH, Benjamini–Hochberg correction; GIV, genetic instrumental variable; OR, odds ratio; VDR-BV, vitamin D receptor binding variant.
VDR-BV used as instrumental variable for allele-specific binding. Allele included is the allele associated with increased binding.
Odds ratios are from logistic regressions adjusted for the first three genome-wide principal components. ORs for association between VDR-BV and pedMS.
Within our pediatric population, we subsequently focused on replicating adult-onset MS findings for significant GIVVDR and GIV25(OH)D interactions. In the current study, one GIVVDR (rs2881514) showed evidence of interaction with the GIV25(OH)D for Jiang et al. in pediatric subjects and was significant at p < 0.05 but did not remain significant after correction for multiple testing (Table 4).
Vitamin D receptor binding variant instrumental variables previously implicated in adult MS

Abbreviations: bp, base pair; chr, chromosome; CI, confidence interval; BH, Benjamini–Hochberg correction; GIV, genetic instrumental variable; OR, odds ratio; VDR-BV, vitamin D receptor binding variant.
GIV 25(OH)D calculated using summary statistics from Jiang et al 2019 17 and Revez et al 2020 GWAS. 16
VDR-BV used as instrumental variable for allele-specific binding. Allele included is the allele associated with increased binding affinity.
Odds ratios are from logistic regressions adjusted for the first three genome-wide principal components. ORs are multiplicative interaction coefficient for interaction between 25(OH)D GIV and VDR-BV.
LD between VDR-BVs and established MS GWAS variants was generally low within the European reference from 1000 Genomes Project. None of the seven VDR-BVs tested in the present study had an r2 > 0.01 with a known MS GWAS variant.
Discussion
This study is the first to examine VDR binding at a locus in pedMS. Our results provide evidence for association between variation in VDR binding and pedMS. These variations may lead to modifications in the transcription of target genes dependent on vitamin D and our results suggest that differences in VDR binding play a role in pedMS susceptibility, offering additional evidence which supports the involvement of the vitamin D pathway in pedMS. Of the seven GIVsVDR previously shown to be independently associated with adult-onset MS and significant at p < 0.05, the GIVVDR rs2531804 was also shown to be independently associated with pedMS and remained statistically significant after correction for multiple testing (pBH < 0.05). 26 Three GIVsVDR, including rs2531804, indicated a protective effect of increased binding at that locus on pedMS risk (OR < 1.0), while four indicated a harmful effect of increased binding at that locus on pedMS risk (OR > 1.0).
Thirteen GIVsVDR, including rs2881514, showed evidence of interaction with one or both GIVs25(OH)D in adults, though none remained significant after correction for multiple testing. 26 Notably, of the GIVsVDR which showed significant evidence of interaction with the GIVs25(OH)D at p < 0.05 in adults, one GIVVDR (rs2881514) demonstrated significant evidence of interaction with the GIV25(OH)D for Jiang et al. and pedMS susceptibility at p < 0.05 in the current study but was not significant after multiple testing correction. The A allele of this SNP was shown to be associated with increased VDR binding in LCLs. 26 This GIVVDR also demonstrated a protective effect on the risk of pedMS in the presence of decreased bioavailability of 25(OH)D. It is important to emphasize that identifying interactions demands significantly more statistical power compared to detecting main effects and the presence of an association between a VDR-BV and pedMS in both the interaction and independent models is encouraging.
Previous studies have documented causal links between serum vitamin D levels and MS risk with the use of MR methods.13,14 The GIVs25(OH)D for Revez et al. and Jiang et al. constructed for 25(OH)D serum levels in this study were both significantly associated with pedMS at p < 0.05, with lower levels of serum 25(OH)D being associated with increased pedMS susceptibility for both GIVs25(OH)D, a finding which is also reinforced by the existing body of literature. These results also underscore the importance of vitamin D deficiency as a critical risk factor for pedMS.
Among those previously shown to be significantly associated with MS, the GIVVDR for rs2531804 was significant in the present study. 26 rs2531804 is located on chromosome 6 upstream of ZSCAN3, which enables sequence-specific double-stranded DNA binding activity and is involved in the regulation of transcription by RNA polymerase II. 33 The top VDR-BV demonstrating evidence for interaction with decreased 25(OH)D in this study was rs2881514, located on chromosome 3, 1533 bases downstream of the transcription start site of RFTN1. 26 RFTN1 encodes Raftlin, a protein that promotes binding of exogenous double-stranded RNA and is necessary for the production of lipid rafts, which are membrane microdomains that play a critical role in B cell activation through B cell receptor signaling.26,34
This study had several strengths. We utilized MR methods to investigate the relationship between VDR binding and pedMS. Because of difficulties in accounting for confounding and reverse causation in assessing the causal pathway between a given exposure and outcome, this study would have been difficult to conduct without leveraging this approach. We focused on candidate VDR-BVs previously identified using experimental methods by Gallone et al. to construct GIVs for VDR binding at a specific locus and implemented methods previously tested in adults.22,26 Utilizing candidates already shown to be significant in adult-onset MS produced increased statistical efficiency and power for the present study in pedMS. Some limitations in this study must also be acknowledged. Horizontal pleiotropy may have biased associations between GIVsVDR and pedMS and constructing each GIVVDR with only one VDR-BV precluded the use of standard sensitivity analyses to assess MR assumptions in our VDR-BV analyses. Both GIVs25(OH)D for Revez et al. and Jiang et al. were constructed from summary statistics derived from adult populations and based on the assumption that SNP information from adults could be applied directly to a pediatric population. Nonetheless, employing GIVs for 25(OH)D serum levels as an additional GIV allowed us to estimate an association between GIVVDR and pedMS unbiased by horizontal pleiotropy. The smaller study sample size had reduced statistical power to identify interactions compared to main effects. Furthermore, the study population was restricted to individuals of European ancestry. While reducing the impact of population stratification, this limits the generalizability of our findings to non-European groups. Our study design aimed to identify only causal mechanisms of pedMS susceptibility mediated by altered VDR binding attributable to DNA variation. Other causal mechanisms beyond VDR binding likely also contribute to the observed VDR-pedMS associations and future studies should explore variations in VDR binding across more loci and in more genetically diverse samples.
The findings from the present study further underscore the significance of the vitamin D pathway in the development of pedMS. Moreover, these results are pertinent to various autoimmune and inflammatory conditions linked to vitamin D. Future research on VDR binding and pedMS should prioritize larger cohorts, diverse genetic subgroups, and identifying VDR-BVs in lymphocytes beyond those found in LCLs, such as CD4+ and CD8+ T cells.
Supplemental Material
sj-xlsx-1-mso-10.1177_20552173251335625 - Supplemental material for Allele-specific vitamin D receptor binding is associated with pediatric-onset multiple sclerosis
Supplemental material, sj-xlsx-1-mso-10.1177_20552173251335625 for Allele-specific vitamin D receptor binding is associated with pediatric-onset multiple sclerosis by Defne Yilmaz, Cameron Adams, Mary K Horton, Jennifer S Graves, Carla Francisco, Alice Edwards, Hong Quach, Diana Quach, Gregory Aaen, Timothy Lotze, Soe Mar, Jayne Ness, Yolanda Wheeler, Mark P Gorman, Leslie Benson, Bianca Weinstock-Guttman, Amy Waldman, Teri Schreiner, Jan-Mendelt Tillema, Tanuja Chitnis, John Rose, T Charles Casper, Mary Rensel, Emmanuelle Waubant and Lisa F Barcellos in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
The authors acknowledge Drs. Moses Rodriguez and Lauren Krupp for their contributions to the study.
Consent to participate
This study was approved by Human Research Protection Program Institutional Review Board at the University of California, San Francisco (IRB# 10-05039) on December 18, 2023. All patients and parents signed written assent and consent forms for this study.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
This study involved human subjects and was approved as Minimal Risk and qualified for Expedited Review Approval by the Human Research Protection Program Institutional Review Board at the University of California, San Francisco (IRB# 10-05039) on December 18, 2023. This research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the following: National Institutes of Health, National Institute of Neurological Disorders and Stroke [R01NS049510 (PI Barcellos)], National Institutes of Health, National Institute of Environmental Health Sciences [R01ES017080 (PI Barcellos)], National Institutes of Health, National Institute of Allergy and Infectious Diseases [R01AI076544 (PI Barcellos)], National Multiple Sclerosis Society [RG-19707-34664 (PI Barcellos)], and National Institutes of Health, National Institute of Neurological Disorders and Stroke R01NS071463 (PI Waubant).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



