Abstract
We evaluated the occurrence of infusion-associated reactions, severe adverse events and no evidence of disease activity 3 status of a therapeutic course consisting of natalizumab followed by alemtuzumab in paediatric-onset multiple sclerosis. Five paediatric-onset multiple sclerosis (age range 16–17 years) were followed for a median of 3.9 years (interquartile range 3.1–5.0). At a natalizumab break (mean infusions 25.6 ± 1.3) patients were switched to alemtuzumab and completed the two therapy courses. Few mild/moderate infusion-associated reactions were observed during alemtuzumab infusion. No severe adverse events were detected. Natalizumab followed by alemtuzumab proved to be a well-tolerated therapeutic course in paediatric-onset multiple sclerosis. Moreover, paediatric-onset multiple sclerosis maintained the no evidence of disease activity 3 status throughout the follow-up.
Introduction
Natalizumab has proved to be very effective in suppressing paediatric-onset multiple sclerosis (POMS) often characterised by an aggressive course. 1 However, a natalizumab break in POMS at high risk of progressive multifocal leukoencephalopathy (PML) 2 exposes these patients to the risk of disease reactivation or rebound. 3 Thus, we investigated the safety (occurrence of infusion-associated reactions, IARs, and severe adverse events, SAEs) and the achievement of no evidence of disease activity (NEDA)-3 status in natalizumab followed by alemtuzumab in highly active naive POMS.
Methods
POMS patients, diagnosed according to the modified McDonald diagnostic criteria (2011), were enrolled in a prospective, observational, open label study.
After having obtained the parent's informed consent, natalizumab was administered every 28 days according to the manufacturer’s guidelines. Treatment was started in agreement with the Agenzia Italiana del Farmaco (AIFA)-defined criteria, i.e. clear evidence of rapidly evolving relapsing–remitting multiple sclerosis (RRMS) (at least two relapses and the appearance of one or more new gadolinium-enhancing lesions at magnetic resonance imaging (MRI) in the past year). Expanded Disability Status Scale (EDSS) examination was performed every 3 months. The presence of anti-John Cunningham virus index (JCV) antibodies in patient serum was assessed by means of the test Stratify JCVTM (Unilabs, Copenhagen, Denmark) every 6 months. After 2 years of natalizumab therapy, POMS patients reaching the age of 18 years and having a high risk of PML (JCV index >0.9) were switched to alemtuzumab without a wash-out period (i.e. one month after the last natalizumab infusion). Before switching, all patients underwent a detailed MRI examination, including three-dimensional (3D) fluid-attenuated inversion recovery (FLAIR), 3D-T1, 3D double inversion recovery (DIR), phase-sensitive inversion recovery (PSIR) and post-contrast T1-weighted sequences, performed with a 3.0 Tesla scanner, in order to excluded PML.
Alemtuzumab was administered by intravenous infusion at the dosage of 12 mg/day on five consecutive days (60 mg total dose) in the first year and on three consecutive days in the second year. During alemtuzumab treatment, clinical and MRI examinations were performed every 6 months, while blood analyses (including complete blood count with differential, serum creatinine and thyroid-stimulating hormone levels, urinalysis with microscopy) were performed monthly.
Patients were carefully assessed for any IARs and SAEs during drug infusion and then monthly according to CTCAEv5.
The NEDA-3 status, defined as no clinical relapses, no increase in disability and no MRI activity 4 was scored annually. Disability improvement was defined as a decrease of 1 point EDSS confirmed at 6 months (in patients with baseline score ≥2.0) and further confirmed at the end of the follow-up period.
Results
Between June 2014 and May 2016, 12 patients with POMS were diagnosed. Nine patients, six JCV negative and three JCV positive, met the clinical and MRI criteria of aggressive multiple sclerosis (MS) and, after informed consent, were treated with natalizumab. The therapeutic course described above (see Methods), consisting of alemtuzumab following natalizumab, was offered and accepted by all nine patients and their parents. Among these, four patients that remained JCV negative continued natalizumab treatment, while five (female/male 3/2) were enrolled in this study. The demographic and clinical features of these five patients are reported in Table 1. The interval between clinical onset and diagnosis was 5.8±5.3 (2.4–14.4) months. At clinical onset the mean EDSS score was 2.8±0.6 (2.0–3.5). At natalizumab initiation the mean EDSS score was 1.5±0.4 (1.0–2.0), the mean age was 16.6±0.6 (16–17) years and three patients were JCV positive (JCV index ranging from 0.76 to 3.29). The mean number of natalizumab infusions was 25.6±1.3 (range 24–27). At alemtuzumab initiation the mean age was 18.7±0.6 (18–20) years and all the patients had JCV index greater than 0.9 (ranging from 1.71 to 3.40) (Figure 1). The median overall follow-up was 3.9 years (interquartile range 3.1–5.0).
Demographic and clinical features of the five POMS patients.
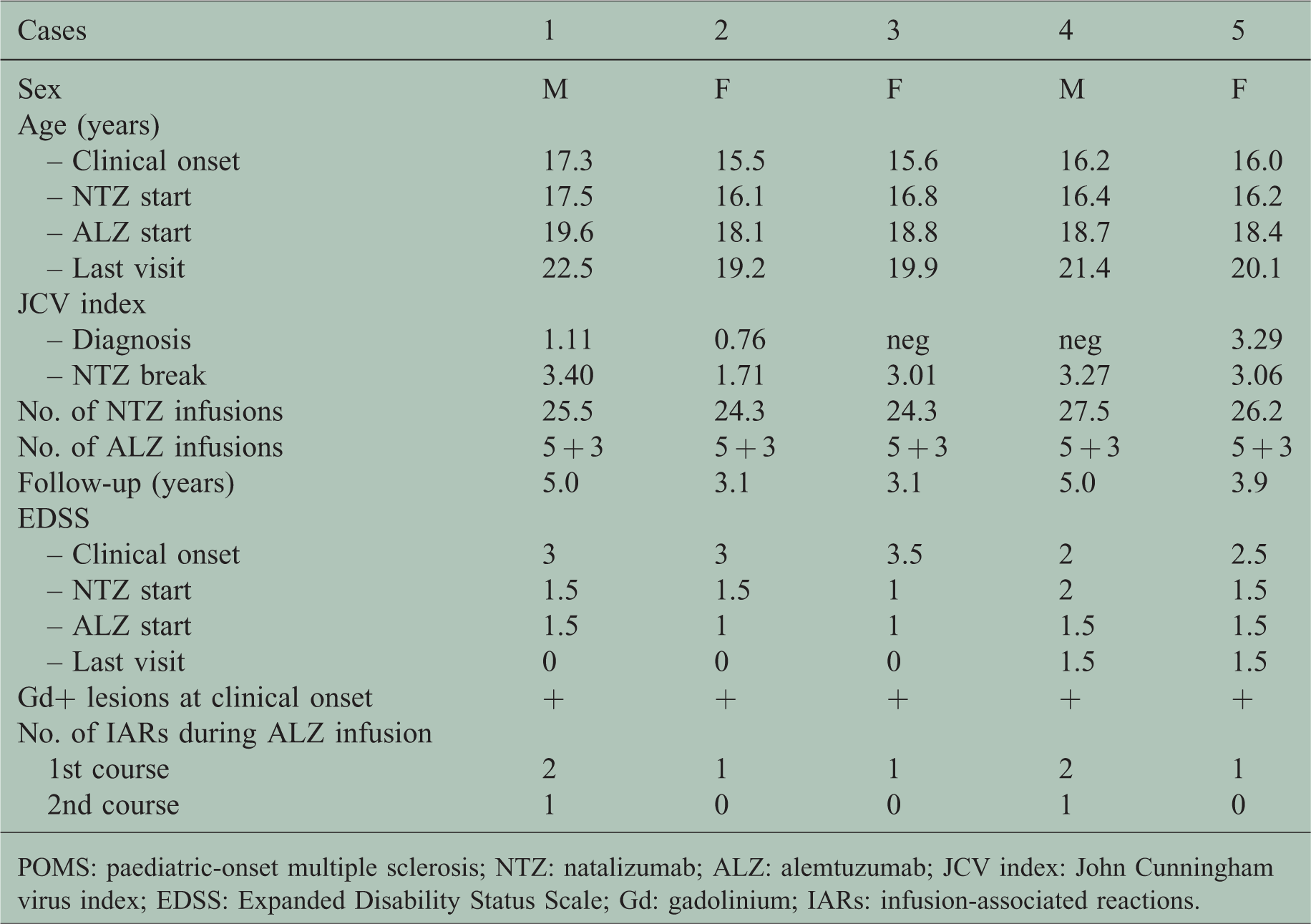
POMS: paediatric-onset multiple sclerosis; NTZ: natalizumab; ALZ: alemtuzumab; JCV index: John Cunningham virus index; EDSS: Expanded Disability Status Scale; Gd: gadolinium; IARs: infusion-associated reactions.

Time course of paediatric-onset multiple sclerosis patients treated with alemtuzumab following natalizumab. See text for explanation.
Safety
No SAEs were observed during natalizumab treatment.
During the first alemtuzumab course, all the patients experienced at least one IAR. Namely, a mild, transient and localised skin rash was observed in two patients at days 2 and 5, hyperpyrexia, always less than 38°C, in four patients at days 1, 4 and 5, bradycardia (41 bpm) in one patient at day 4, for a total number of seven adverse events. All these side effects were graded mild to moderate (grade I or II CTCAEv5), with quick resolution after either slowing the alemtuzumab infusion rate or supplemental doses of paracetamol, steroids or antihistamines.
During the second alemtuzumab course, only two patients experienced one infusion-related reaction, namely hyperpyrexia (<38°C) at days 1 and 3. Up to now, no SAEs, especially infections and autoimmune disorders (i.e. immune thrombocytopenia, glomerular nephropathies, thyroid endocrine disorders, autoimmune cytopenia, autoimmune hepatitis) have been observed.
NEDA-3 status
NEDA-3 status was achieved and maintained during natalizumab therapy in all five patients. At the natalizumab break the mean EDSS score was 1.3±0.3 (1.0–1.5). After a median period of 1.9 years (interquartile range 1.1–3) from the first alemtuzumab course, NEDA-3 status was maintained in all patients. Moreover, three patients had a clinical improvement and at the time of the last visit the mean EDSS score was 0.6±0.8 (0.0–1.5).
Discussion
MS patients treated for 2 years with natalizumab and having a high JCV index are at risk of PML. However, severe disease reactivation and dramatic cases of brain inflammation can occur at therapy break. 2 This risk can be even higher in POMS patients, whose disease is particularly active from clinical onset. 1 Recently, a high number of self-reactive, auto-proliferating T helper type 1 CD4+ clones have been demonstrated in the blood of natalizumab-treated MS patients. 5 These CD4+ clones may recognise central nervous system (CNS) antigens and, once allowed to cross the blood–brain barrier, power up a severe brain inflammation.
Thus, in order to maintain the excellent disease control obtained by natalizumab and avoid serious disease rebound at its break, the switch to a lymphocyte-depleting drug with a similar high efficacy profile has to be strongly considered. Indeed, drugs with a lower efficacy, e.g. dimethyl fumarate and fingolimod, have been evaluated for this purpose, but results were contradictory. 2 Recently, alemtuzumab was demonstrated to be superior to fingolimod in suppressing MS reactivation following natalizumab break in an adult MS population: alemtuzumab-treated patients had a lower risk of relapse (55.4% vs. 76.7%, P = 0.024) and MRI activity (49.5% vs. 74.4%, P = 0.029) compared to the fingolimod cohort.
We planned a therapeutic course in naive POMS patients that consisted of starting with natalizumab and, when patients reached the age of 18 years and became JCV positive at high index, switching to alemtuzumab. This strategy proved to have an acceptable safety profile and to maintain the NEDA-3 status throughout the follow-up. Natalizumab followed by alemtuzumab might be a promising therapeutic strategy in POMS, often characterised by a quite severe widespread brain inflammation that needs to be suppressed early and with long-lasting effect. Up to date, no similar data are available in the literature. Despite the recent warning of the European Medicines Agency on alemtuzumab safety, this drug proved to be well tolerated in POMS, further confirming that young patients, with a short disease duration and lacking comorbidities are the ideal subjects for immunosuppressive treatments.
Footnotes
Author contributions
Monica Margoni: study concept and design, critical revision of the manuscript, statistical analysis of the data. Francesca Rinaldi: critical revision of the manuscript. Silvia Miante: critical revision of the manuscript. Silvia Franciotta: critical revision of the manuscript. Paola Perini: critical revision of the manuscript. Paolo Gallo: study concept and design, critical revision of the manuscript, study supervision.
Conflicts of Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Monica Margoni reports grants from Teva, grants from Genzyme Sanofi, grants from Merck Serono, grants from Biogen Idec, grants and personal fees from Novartis, during the conduct of the study. Francesca Rinaldi reports grants from Almirall, grants and personal fees from Teva, grants and personal fees from Genzyme Sanofi, grants and personal fees from Merck Serono, grants and personal fees from Biogen Idec, grants from Novartis, during the conduct of the study. Silvia Miante reports grants from Teva, grants from Genzyme Sanofi, grants from Merck Serono, grants from Biogen Idec, grants from Novartis, during the conduct of the study. Silvia Franciotta has nothing to disclose. Paola Perini reports grants and personal fees from Merck Serono, grants and personal fees from Biogen Idec, grants and personal fees from Genzyme Sanofi, grants and personal fees from Bayer Schering Pharma, grants and personal fees from Novartis, grants and personal fees from Teva, during the conduct of the study. Paolo Gallo reports grants and personal fees from Merck Serono, grants and personal fees from Biogen Idec, grants and personal fees from Genzyme Sanofi, grants and personal fees from Bayer Schering Pharma, grants and personal fees from Novartis, grants and personal fees from Teva, grants from University of Padua, Department of Neurosciences DNS, grants from Veneto Region of Italy, grants from the Italian Association for Multiple Sclerosis (AISM), grants from Italian Ministry of Public Health, during the conduct of the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
