Abstract
Background
Many patients report a wearing-off phenomenon with monoclonal antibody treatment for multiple sclerosis in which perceived benefits wear off before the next dose is due.
Objectives
To determine prevalence of the wearing-off effect, symptoms experienced, impact on treatment satisfaction, and associated patient characteristics.
Methods
Patients receiving natalizumab, ocrelizumab, ofatumumab, or rituximab at a tertiary multiple sclerosis center were invited to take an online survey interrogating their monoclonal antibody experience. Additional history and patient characteristic data were collected. Logistic regression was used to determine if patient characteristics predicted the wearing-off effect and linear regression to evaluate the impact of the wearing-off effect on treatment satisfaction. The models were adjusted for age, disease duration, race, sex, body mass index, education, and depression as measured by the Patient Health Questionnaire-9.
Results
We received 258 qualifying responses and 141 (54.7%) patients reported the wearing-off phenomenon. The most common symptom was fatigue (47.7%). Higher Patient Health Questionnaire-9 scores were significantly associated with the wearing-off phenomenon (OR = 1.02, p = 0.005). The wearing-off effect (β = −0.52, p = 0.04) and higher Patient Health Questionnaire-9 (β = −0.09, p < 0.01) scores were associated with significantly reduced treatment satisfaction.
Conclusion
The wearing-off phenomenon is common, associated with depression, and reduces treatment satisfaction. Research addressing mitigation strategies is needed.
Introduction
Multiple sclerosis (MS) is a demyelinating and neurodegenerative disease that leads to accumulation of irreversible physical and cognitive disability. 1 A number of highly effective monoclonal antibodies (MAbs) are approved as disease-modifying therapies (DMTs) for MS. 2 These treatments are dosed at differing intervals, including once a month for natalizumab and ofatumumab and every 6 months for rituximab, ocrelizumab, and ublituximab. Alemtuzumab is given in two cycles one year apart, and then as needed at the discretion of the treating neurologist.
Anecdotally, many MAb-treated MS patients report a wearing-off phenomenon in which symptomatic improvement is perceived shortly after dosing, but the benefit wears off before the next dose is due. This phenomenon has previously been recognized and studied in natalizumab and ocrelizumab.3,4 The reasons behind the wearing-off phenomenon are incompletely understood, particularly in agents such as rituximab and ocrelizumab, in which B-cell repopulation rarely occurs prior to the 6 month dosing interval 5 and B-cell depletion can be verified through laboratory testing. In natalizumab-treated patients, the wearing-off phenomenon has been associated with lower occupancy rates of the alpha 4-integrin receptor on circulating leukocytes. 6
We elected to further evaluate the patient-reported wearing-off phenomenon in patients receiving MAb therapy with ocrelizumab, rituximab, natalizumab, and ofatumumab. Our goal was to determine the typical wearing-off symptoms experienced with each MAb and whether there were differences in the frequency and severity of symptoms with the different DMTs. Further, we sought to determine the impact of the wearing-off phenomenon on treatment satisfaction and whether particular patient characteristics were associated with it. We hypothesized that patients receiving monthly dosing with natalizumab and ofatumumab would have a lower prevalence of the wearing-off phenomenon and less severe symptoms than those receiving rituximab and ocrelizumab, which are dosed every 6 months.
Methods
Survey instrument
We developed an online survey administered via REDCap (Vanderbilt University) to investigate the patient-reported wearing-off phenomenon in patients receiving ocrelizumab, rituximab, natalizumab, and ofatumumab. The survey included six questions that all participants answered and used branching logic to offer an additional four questions to patients who experienced the wearing-off phenomenon. The questions interrogated the MAb received, whether the wearing-off phenomenon was experienced, and if so, the typical symptoms and the timing of those symptoms. The survey also asked participants to rank their tolerance of the MAb and their treatment satisfaction on a scale of 0–10 and to indicate whether they had given serious consideration to changing treatments due to the wearing-off phenomenon. The full survey is available in Supplemental Table 1.
Data collection
All patients receiving ocrelizumab, rituximab, natalizumab, and ofatumumab at Cleveland Clinic's Mellen Center for MS, a tertiary MS referral center, were identified through the electronic medical record (EMR). A diagnosis of MS by a Mellen Center subspecialist, age ≥18 years old and at least two doses of the MAb were required. All patients receiving natalizumab and ofatumumab were invited for participation. Those receiving rituximab and ocrelizumab were randomly selected and invited due to high volumes of patients receiving these treatments. The survey invitation was sent via the MyChart feature of the Epic EMR (Madison, Wisconsin).
Additional data for survey participants was collected from the EMR. These included demographics, disease characteristics, treatment history, Patient Health Questionnaire-9 (PHQ9, a measure of depression) score, 7 and the patient's level of disability as measured by the Patient Determined Disease Steps (PDDS). 8 Individuals who had received fewer than two doses of their MAb at the time of the survey were excluded.
Statistical analysis
Descriptive statistics were used to characterize the cohort, including the prevalence of the patient-reported wearing-off phenomenon and the particular symptoms experienced. Analysis of variance was used to make unadjusted comparisons of the monoclonal antibodies with respect to frequency of the wearing-off phenomenon and treatment satisfaction.
A logistic regression model was built to determine whether specific MAbs were particularly associated with the wearing-off effect. Covariates included age, body mass index (BMI), disease duration, years of education, sex, PHQ9 score, and race. A linear regression model was used to determine if the wearing-off effect was related to treatment satisfaction, adjusting for the same covariates.
Results
We received 283 responses from 1027 invitations; a completion rate of 27.6%. Fifteen responses were excluded because the patient had not received at least two doses of their MAb, leaving 258 surveys for analysis. Details of the cohort are summarized in Table 1. There were 154 (59.7%) patients treated with ocrelizumab, 28 (10.9%) with rituximab, 39 (15.1%) with ofatumumab, and 37 (14.3%) with natalizumab. The average age was 48.1 years, average disease duration was 12.2 years, and mean BMI was 30.1 kg/m2.
Cohort characteristics stratified by monoclonal antibody.
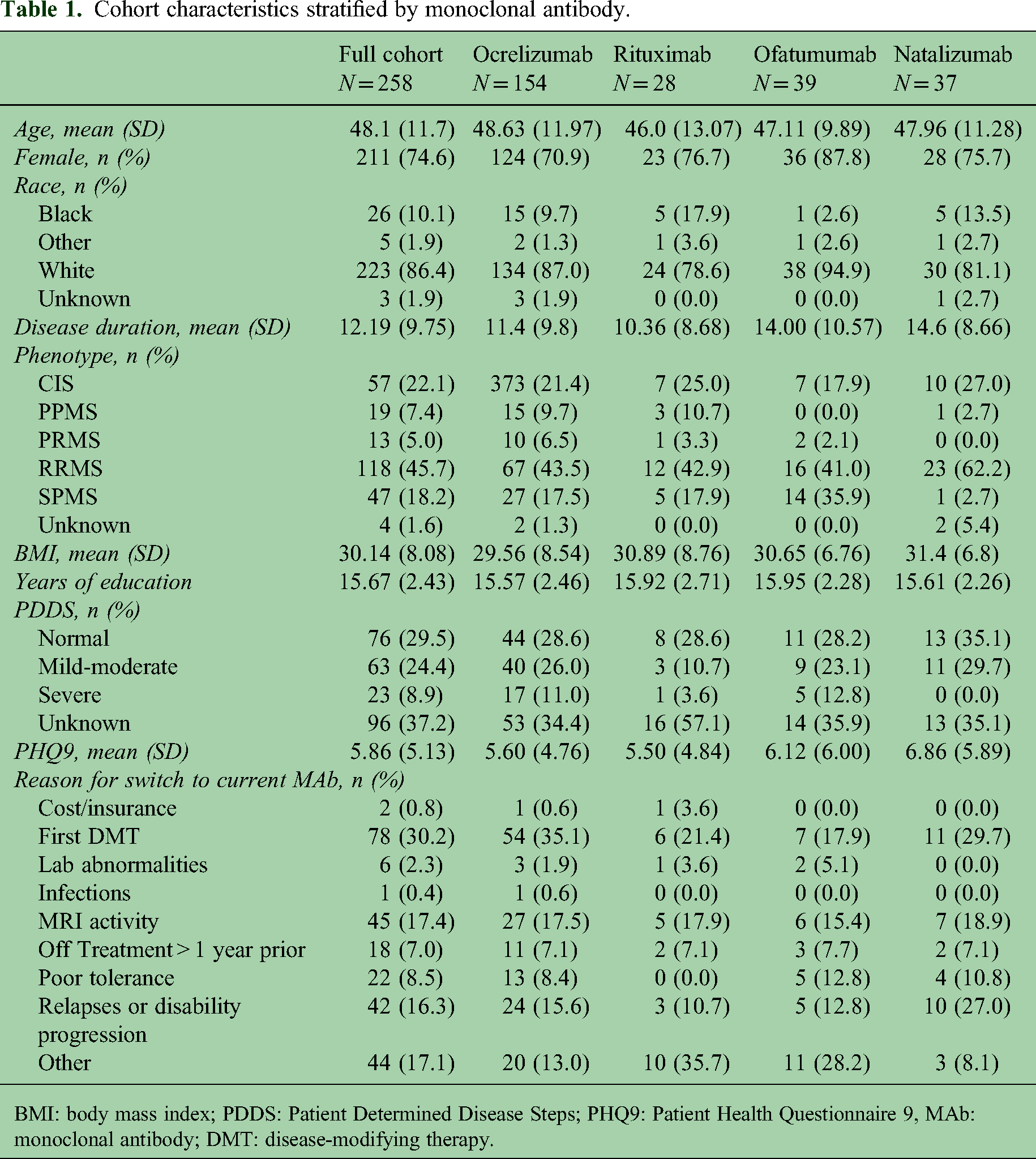
BMI: body mass index; PDDS: Patient Determined Disease Steps; PHQ9: Patient Health Questionnaire 9, MAb: monoclonal antibody; DMT: disease-modifying therapy.
The prevalence and timing of wearing-off symptoms are summarized in Table 2. Overall, 141 (54.7%) patients experienced the wearing-off phenomenon, constituting slightly more than half of the sample. The prevalence was not significantly different between MAbs (p = 0.44), with wearing-off symptoms occurring most frequently in individuals on rituximab (n = 17/28, 60.7%) and least frequently in those receiving ofatumumab (n = 18/39, 46.2%). For patients on ocrelizumab and rituximab, more than half of individuals began experiencing wearing-off symptoms within 2 months of receiving their dose. However, only 17.0% of ocrelizumab patients considered switching treatments because of the wearing-off effect, as compared to 35.3% of rituximab patients, 27.8% of ofatumumab patients and 5.6% of natalizumab patients (p = 0.098).
Presence of wearing-off symptoms stratified by monoclonal antibody.
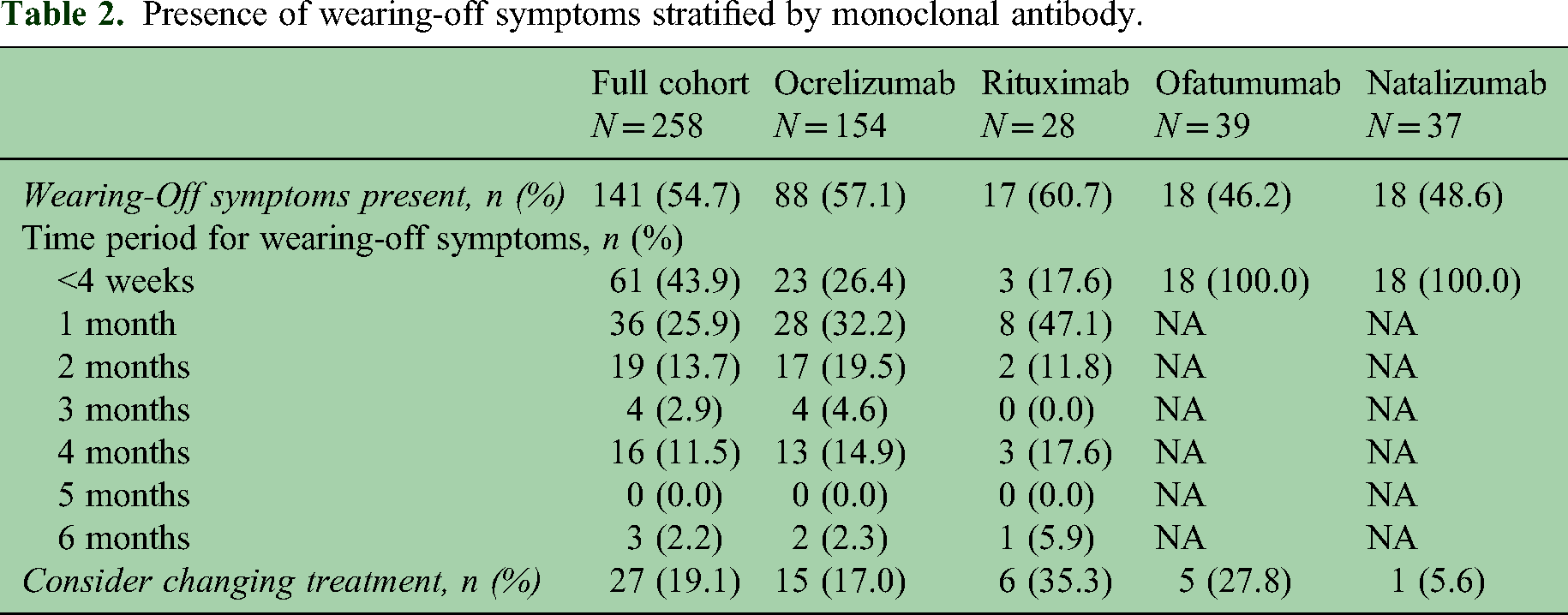
The most common symptom experienced as part of the wearing-off effect was fatigue, which ranged from 45.9% of patients receiving ofatumumab to 53.6% of patients on rituximab. The symptoms were not significantly different between monoclonal antibodies. Other common symptoms included worsened cognitive functioning (26.0%), worsening mobility (28.6%), and increased pain/paresthesias (29.9%). Table 3 summarizes wearing-off symptoms for the full cohort and by MAb.
Wearing-off symptoms stratified by monoclonal antibody.

The results of the logistic regression model predicting the presence of the wearing-off effect are provided in Table 4. Depression was a highly significant predictor of the wearing-off phenomenon. Each point a patient scored higher on the PHQ9, the odds of experiencing the wearing-off effect increased by 2% (p = 0.005). Also, relative to ocrelizumab, patients receiving ofatumumab had 18.2% lower odds of experiencing it (p = 0.046). There was no significant difference in the wearing-off effect between natalizumab and ocrelizumab or rituximab and ocrelizumab.
Multivariable logistic regression models predicting presence of the wearing-off effect.

BMI: body mass index; MAb: monoclonal antibody; PHQ9: Patient Health Questionnaire 9
Multivariable linear regression model predicting treatment satisfaction.

BMI: body mass index; MAb: monoclonal antibody; PHQ9: Patient Health Questionnaire 9
The mean reported treatment satisfaction was 8.80 (SD = 1.8) on a scale of 1–10. Unadjusted mean treatment satisfaction was not significantly different between MAbs: 9.19 (SD = 1.4) for natalizumab, 8.80 (SD = 2.34) for ofatumumab, 8.56 (SD = 1.9) for ocrelizumab, and 8.53 (SD = 1.73) for rituximab (p = 0.44). In the adjusted linear regression model predicting treatment satisfaction (Table 5), presence of the wearing-off effect reduced mean treatment satisfaction by 0.515 points (p = 0.038). Patients on natalizumab had average treatment satisfaction scores that were 0.749 points higher than those on ocrelizumab (p = 0.034). There was no significant difference in treatment satisfaction between ofatumumab and ocrelizumab or between rituximab and ocrelizumab. Interestingly, more educated patients were significantly less likely to be satisfied with their MAb, with each additional year of education reducing treatment satisfaction by 0.157 points (p = 0.002).
Discussion
This study used survey data to better characterize the patient-reported wearing-off phenomenon in MS patients receiving MAbs. We found that the wearing-off effect is common, affecting more than 50% of MAb-treated patients and that increased fatigue is the most common manifestation. Further, patients with depression appear to be at higher risk of experiencing the wearing-off effect and the wearing-off effect significantly influences treatment satisfaction, even when adjusting for depression.
With ocrelizumab as our reference group, we did not find a significant difference in the wearing-off effect when compared to once monthly natalizumab or every 6-month rituximab-treated patients. Patients taking ofatumumab, given as a once monthly subcutaneous injection, had 18.2% lower odds of the wearing-off effect as compared to ocrelizumab-treated patients. This finding is interesting in that prior data suggests that both ofatumumab and ocrelizumab provide rapid and sustained B-cell depletion.9,10 Analysis of Phase 2 ocrelizumab data suggested that the B cell nadir is maintained for 24 weeks with slow repopulation thereafter and that the median time to B cell repletion after the last infusion is 62 weeks. 5 The lower incidence of the wearing-off effect among ofatumumab-treated patients in our data could suggest additional effects of B cell depletion that in some way differ between ocrelizumab and ofatumumab versus possibly a placebo effect from more frequent dosing of the ofatumumab.
A study of the wearing-off effect in ocrelizumab-treated MS patients was previously conducted in a Dutch cohort. 4 In that study, 61% of patients reported the wearing-off phenomenon with ocrelizumab treatment, similar to the 57.1% of patients in our study. Fatigue was the most common symptomatic manifestation, affecting 62.0% of those experiencing the wearing-off effect. Additional findings of interest included no relationship between CD19 B-cell counts and the wearing-off effect, but a greater likelihood of the wearing-off effect in those with higher BMI.
Our study expanded on this approach by investigating four of the most commonly used MAbs. Unlike in the Dutch cohort, we did not find a relationship between BMI and the wearing-off effect. There is a good rationale for suspecting such a relationship because MAb dosing for MS is not weight-based, but it should be noted that the contribution of body size to pharmacokinetic variability is low for most MAbs. 11 In a Norwegian study of 40 natalizumab-treated patients, however, higher BMI was associated with lower natalizumab α4 integrin receptor occupancy. 6 It is unclear why there was no relationship between BMI and the wearing-off effect in our study, but this could be related to the inclusion of multiple MAbs with a relatively small proportion of natalizumab-treated patients. Further research is needed, including further investigation into the relationship between BMI, CD20 counts, and the wearing-off effect in patients treated with B-cell depleting agents.
To what extent the wearing-off effect represents a true physiologic phenomenon is unclear. In the previously mentioned Norwegian study, the wearing-off effect was associated with lower α4 integrin receptor occupancy. However, van Kempen et al. 12 found that the wearing-off effect was more frequently reported by patients receiving natalizumab every four weeks than in those treated with extended interval dosing. With respect to B-cell depletion, patients whose rituximab treatment is interrupted or stopped still show prolonged disease suppression, even when alternative DMT is not started. 13 A study of 103 ocrelizumab-treated MS patients sought to use several patient-reported outcomes to assess symptomatic burden across the ocrelizumab treatment cycle. 14 The patient-reported outcomes were generally found not to worsen towards the end of the treatment cycle, leading the authors to speculate that the wearing-off effect is primarily due to attribution bias rather than a biological effect. Given the lack of a physiologic correlate or even consistent patient-reported symptomatic worsening at the end of a treatment cycle, the wearing-off effect could simply be normal variations in symptomatic severity that are erroneously attributed to MAb dosing intervals.
We found that higher depression scores significantly increased the odds of the wearing-off effect. Depression is well known to occur at a higher rate in MS than in the general population and may, to some extent, be related to MS-induced structural changes in the brain. 15 Depression is also known to impact a patient's relationship with DMT. For instance, in a systematic review examining DMT adherence, Washington and Langdon 16 found five studies suggesting lower adherence in patients with depression, including one with 38.2% lower odds of adherence in patients with depression relative to those without. 17 To the best of our knowledge, the relationship between the wearing-off effect and depression has not previously been described. This association would support the notion that the wearing-off effect is psychological in nature rather than reflective of a biological phenomenon. Treatment of underlying depression may be a method to address the wearing-off effect in appropriate patients.
Patients reporting the wearing-off effect had >0.5-point lower treatment satisfaction scores on a scale of 1–10, a significant difference compared to those with no wearing-off effect. The significant impact of the wearing-off effect on MAb treatment satisfaction suggests that the phenomenon could have wide-ranging effects, such as reducing treatment adherence or leading patients to switch to less effective DMTs. Notably, however, a prior study using data from the North American Research Committee on Multiple Sclerosis registry did not find a relationship between treatment satisfaction and adherence. 18 Another study did not find any overall difference in treatment satisfaction between natalizumab, interferon beta, and glatiramer acetate, 19 suggesting that while the wearing-off effect of MAbs may be a drawback relative to other DMTs, it is offset by other advantages. Our data showed that nearly 20% of individuals with the wearing-off effect have considered switching to an alternative DMT, but further research is needed to determine how many patients follow through with treatment switching.
Increased fatigue was, by far, the most prominent symptom of the wearing-off effect. Fatigue is already a highly prevalent manifestation of MS that impacts patient quality of life and also carries a significant economic burden. 20 Strategies for management of MS-related fatigue have received significant research attention and a number of options exist including pharmacologic management, 21 physical exercise, 22 and cognitive behavioral therapy. 23 Assessing and correcting other causes of fatigue, such as vitamin B12 deficiency, thyroid dysfunction, or inadequate sleep are also important management considerations. Attempting to directly address fatigue using these strategies may be helpful for patients experiencing the wearing-off effect, but further investigation is needed. Some literature supports a response to placebo in patients with fatigue, 24 so there might also be a placebo component to the wearing-off effect in which patients anticipating symptomatic benefit feel less fatigued following MAb treatment.
Some limitations to our study deserve mention. First, the patient population was from a single tertiary-referral MS center. As such, the results may not be completely generalizable, particularly given that the center population may be slanted towards those with more severe MS. Another consideration is that patients experiencing the wearing-off effect might be more likely to respond to a survey on the topic. We attempted to mitigate this potential sampling bias by not directly mentioning the “wearing-off effect” in the survey invitation, but rather indicated that we were attempting to understand the symptoms patients experience between MAb doses. Finally, treatment satisfaction was measured with a single question asking patients to rate their satisfaction on a scale of 1–10. It would have been preferable to use a validated instrument that comprehensively assessed treatment satisfaction. However, because treatment satisfaction was not the focus of the study and we wanted to keep the survey short in order to maximize participation, we elected to use a single question. Future studies to assess the relationship more completely between the wearing-off effect and treatment satisfaction would be helpful.
Our results show that the patient-reported wearing-off effect with MAb treatment for MS is common, is most frequently characterized by increased fatigue, and that close to 20% of patients consider switching treatment because of it. The patient-reported wearing-off effect was seen least frequently with ofatumumab, and after correction for potential confounders, ofatumumab was the only treatment that had lower odds of the phenomenon relative to ocrelizumab. Despite widespread investigation in the literature, a biological mechanism for the patient-reported wearing-off phenomenon remains elusive and our finding of a relationship with depression would argue against a physiologic cause. Another consideration is the timeframe of the symptomatic worsening. Most patients in our study reported increased symptoms beginning within two months of their MAb dose, even with ocrelizumab and rituximab, which are dosed every 6 months. If the symptomatic worsening is truly due to treatment benefits degrading over time, the number of patients experiencing the effects would be expected to peak closer to the end of a cycle. As in the previously discussed study from Kister et al., 14 future research should focus on a granular evaluation of when symptomatic worsening occurs relative to MAb dosing, how the symptoms evolve over time, and their relationship to biomarkers of DMT effectiveness and disease activity.
Regardless of whether the patient-reported wearing-off effect with MAbs has physiologic plausibility or reflects misattribution of typical symptomatic variation, it has a significant impact on many MS patients. Because of its effect on treatment satisfaction, it may induce patients to switch from DMTs that are otherwise effective in controlling their disease. Thus, more research is needed, both to further characterize the patient-reported wearing-off phenomenon and to develop strategies to mitigate it, particularly as greater consideration is given to early highly effective therapy with MAbs. 25
Supplemental Material
sj-docx-1-mso-10.1177_20552173241251707 - Supplemental material for The patient-reported wearing-off phenomenon with monoclonal antibody treatments for multiple sclerosis
Supplemental material, sj-docx-1-mso-10.1177_20552173241251707 for The patient-reported wearing-off phenomenon with monoclonal antibody treatments for multiple sclerosis by Lindsey M Marian, Kathleen A Harris and Devon S Conway in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kathleen A Harris has received consulting fees from EMD Serono, TG Therapeutics, and Novartis. Devon S Conway has received research support paid to his institution by Novartis, EMD Serono, Bristol Myers Squibb, Horizon Therapeutics, Biogen, and the Department of Defense. He has received consulting fees from Bristol Myers Squibb, TG, Novartis, and Alexion Pharmaceuticals and speaking fees from Biogen. Lindsey M Marian declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
