Abstract
Background
Diffusion tensor imaging (DTI) in adults with multiple sclerosis (MS) has identified marked volume and diffusion abnormalities of the fornix, the main white matter (WM) output tract of the hippocampus.
Objective
To determine if the fornix is affected in pediatric-onset MS (POMS) using the same DTI protocols used in adult-onset MS (AOMS), which would suggest its early involvement in the disease course.
Methods
High-resolution, fluid-suppressed diffusion tractography was used to identify the fornix in 11 POMS patients (13–19 years old) and 26 controls. Fornix volume and diffusion metrics were compared between groups and with other total/regional brain volumes, and then correlated with cognitive/clinical scores.
Results
POMS showed lower fornix volumes (−26%) compared to controls, which was greater than proportional losses in total and other regional brain volumes. Notably, the hippocampus volume was not lower in POMS. DTI yielded lower fractional anisotropy (−7%) and higher mean (+12%), axial (+7%), and radial (+16%) diffusivities in POMS. There were no significant correlations between fornix volume/diffusion metrics and cognitive/clinical scores.
Conclusion
Diffusion tractography showed marked injury to the fornix in POMS that precedes injury to connected gray matter such as hippocampus, implicating the fornix as an early brain region affected in MS.
Keywords
Introduction
Pediatric-onset multiple sclerosis (POMS) accounts for up to 5% of MS cases and is characterized by disease onset and high clinical and radiographic disease activity during childhood and adolescence. 1 It was shown that within just 2 years of MS onset, approximately 30% of children and adolescents with MS become cognitively impaired. Like adult-onset MS (AOMS), POMS has shown widespread gray matter (GM) atrophy across most cortical and subcortical regions that appear to worsen over time. 2 Early and disproportionate thalamic atrophy has also been reported in POMS, particularly at the ventricular interface, presumably due to soluble MS inflammatory factors in the cerebrospinal fluid (CSF). 3 Thalamic and other GM atrophy in POMS have been shown to correlate with various measures of disability (Expanded Disability Status Scale, EDSS) and cognition (episodic memory performance).2,4 POMS also showed deviations from sex- and age-expected developmental trajectories of hippocampus volume. 2 Other POMS studies have shown hippocampal atrophy relative to controls, but there is inconsistency on whether it is associated with cognitive impairment.4,5
Beyond GM, cognitive impairment in MS may result from injury to normal-appearing WM (NAWM) from demyelination and axonal loss leading to disconnections between cortical and subcortical structures. 6 Diffusion tensor imaging (DTI) can detect changes indicative of demyelination and/or axonal injury in NAWM, as well as virtually identify WM tracts with diffusion tractography. DTI has shown reduced tract volumes and altered diffusion metrics in NAWM in adults with MS compared to controls, which are associated with physical and cognitive impairment, such as the fornix which is the main efferent tract of the hippocampus.7–13
Cognitive fornix correlations in adult MS have been shown between fornix mean (MD), axial (AD) and radial (RD) diffusivity with episodic memory scores (including the Brief Visuospatial Memory Test-Revised—BVMT-R) as well as fornix fractional anisotropy (FA) and MD with processing speed (including the Symbol Digit Modalities Test—SDMT and Paced Auditory Serial Addition Test—PASAT), fornix FA with EDSS, and fornix FA and RD with verbal memory (California Verbal Learning Test—CVLT).9–13 Fornix FA and RD also correlated with cognitive impairment in adult MS, which was determined using PASAT, SDMT, CVLT, and BVMT-R scores. 8
The fornix is a small WM tract located within the lateral ventricles making it prone to diffusion tractography errors due to partial volume effects from rapid, isotropic diffusing CSF, which contributes to the diffusion parameters per voxel; however, tractography and output diffusion metrics can be improved using high-resolution fluid-attenuated inversion recovery (FLAIR) DTI, which suppresses the confounding CSF. 14 FLAIR-DTI has shown marked volume and diffusion abnormalities of the fornix in adults with MS compared to controls, including lower tract volume and FA and higher MD, AD, and RD, which were most affected in those with cognitive impairment and greater than losses in other WM and GM volumes. 15 SDMT also correlated with fornix FA, MD, and RD.
DTI metrics are altered in POMS in most NAWM, including major tracts like the corpus callosum, posterior limb of the internal capsule, cerebral peduncle, and long association fibers (superior longitudinal, inferior fronto-occipital, and uncinate fasciculus). 16 Relative to controls, DTI metrics in POMS NAWM appear to worsen over time and fail to reach age-expected maturation. 17 Additionally, these abnormal DTI metrics are apparent even in very early-onset POMS (<12 years of age), which may suggest early NAWM injury in MS. 18 However, there is only one POMS DTI study that has mentioned the fornix region, reporting elevated MD, but it used low spatial resolution DTI protocols (voxel volume > 12 mm3) and a voxel-based analysis not ideal for measuring the fornix and which does not yield tract volume.19,20 FLAIR-DTI tractography has not been applied to study the fornix in POMS, and comparison to adults with MS previously studied using the exact same protocol 15 would examine whether it is involved early in the disease course of MS.
The purpose of this study was to: (i) determine if the fornix (volume and diffusion metrics) is affected in POMS using CSF-suppressed, high-resolution FLAIR-DTI tractography, (ii) evaluate other brain volumes (e.g., hippocampus) to determine if fornix injury precedes GM atrophy, and (iii) assess correlations between fornix volume/diffusion metrics and clinical disability/cognitive function.
Materials and methods
Participants
This study was approved by the University of Alberta Human Research Ethics Board (approval no. Pro00112979) on 17 November 2022. All 37 participants provided written informed consent (or parental consent and assent if under 16 years of age) including 11 diagnosed with relapsing- remitting POMS that were recruited from the pediatric neuroinflammatory registry and the University of Alberta Neurosciences Clinic, and 26 healthy controls chosen with a similar age/sex distribution from a previous normative study using an identical MRI protocol. The inclusion criteria for the POMS cohort were as follows: diagnosis of MS according to the 2017 McDonald criteria, disease onset prior to 18 years of age, and under 20 years of age at the time of MRI scan. The exclusion criteria included no POMS participants with medical diagnoses other than MS or with known clinical relapses at the time of the MRI scan or within 30 days of the study visit. All 11 POMS participants were on disease modifying therapies (ocrelizumab, rituximab, and fingolimod). Three POMS participants had diagnosed depression/anxiety and were on various antidepressant medications. Two POMS participants had diagnosed attention deficit hyperactivity disorder (ADHD) and were taking ADHD medications. The demographic and clinical data are summarized in Table 1.
Demographics, cognitive/clinical scores, and total lesion volume (TLV) for POMS and controls with range, mean, and standard deviation.

Abbreviations: TLV: total lesion volume; POMS: pediatric-onset multiple sclerosis.
No significant difference in age (t = 0.747, p = .460) or sex (
Cognitive assessment
Cognitive and clinical tests were administered by a trained user and MS neurologists from the University of Alberta, respectively. Tests included: EDSS for overall MS disability, Pediatric Fatigue (Neuro-QOL Item Bank v.2.1) for fatigue, Beck's Depression Inventory-II (BDI-II) for depression, SDMT to measure processing speed, and BVMT-R to test visuospatial memory (only total recall score). Pediatric fatigue, SDMT, and BVMT-R scores were converted to standardized z-scores based on the 2021 Neuro-QOL Scoring Manual (Version 3.0) and pediatric norms. 21 Cognitive/clinical test scores are summarized in Table 1.
MRI protocol
Participants were scanned on a 3 T Siemens Prisma MRI with a 64-channel head/neck radiofrequency coil. A whole-brain 3D T1-weighted magnetization-prepared rapid acquisition gradient echo (MPRAGE) was acquired using the following parameters: 0.9 mm isotropic voxels, TR 1800 ms, TE 2.37 ms, and total scan time 3.4 min. Whole-brain 3D sampling perfection with application optimized contrasts using different flip angle evolution (SPACE) FLAIR was acquired with 1.2 mm isotropic voxels, TI 1800 ms, TR 5000 ms, TE 385 ms, and total scan time 3.1 min. The FLAIR-DTI protocol was the same as used in a previous adult MS study here: 2D single-shot echo-planar imaging, 35 2 mm transverse slices centered for fornix coverage, 1.2 × 1.2 mm2 zero-filled to 0.64 × 0.64 mm2 in-plane resolution, GRAPPA R = 2, phase partial Fourier 6/8, TI 2300 ms, TR 9000 ms, TE 70 ms, 5 b = 0 and 20 b = 1000 s/mm2, and total scan time 4.1 min. 15
MRI analysis
MPRAGE brain volumes and SPACE FLAIR total MS lesion volumes were measured using volBrain and lesionBrain pipelines, respectively.22,23 VolBrain yielded segmented total and regional brain volumes including: total brain volume (TBV) (including cerebellum), WM, GM, CSF, cerebrum (total, WM, GM) excluding cerebellum, cerebellum (total, WM, GM), lateral ventricles, caudate, putamen, thalamus, globus pallidus, hippocampus, and amygdala. Left and right hemispheres were combined to limit comparisons and yielded 17 volumes per participant. LesionBrain yielded segmented total lesion volume (TLV).
DTI processing was performed in MRtrix3 (v.2.0) including denoising, Gibbs ringing, eddy current and motion, bias field correction, and tensor fitting. 24 Deterministic tractography of the fornix was performed in MRtrix3 similar to previously published lab protocols using: FA threshold 0.15, maximum turning angle 65°, step size 0.64 mm, minimum fiber length 10 mm, and maximum fiber length of 109 mm (MRtrix3 default, geometric mean of interpolated voxel size times 100). 15 Regions of interest (ROIs) were placed as follows: coronal AND ROI in the fornix body, axial NOT ROI to remove tracts superior to the fornix, coronal NOT ROI to remove tracts anterior to the fornix columns, coronal NOT ROI to remove tracts posterior to the fornix crura, and other NOT ROIs as needed to remove all remaining streamlines not attributable to the fornix. This tract yielded the whole fornix volume, as well as average FA, MD, AD, and RD across left and right sides. A binary mask was generated from the tractography, whereby each voxel contributed once to the average independent of the number of streamlines passing through each voxel.
Statistical analysis:
Fornix volume, FA, MD, AD, and RD for POMS and controls were tested for normality, and then metrics were assessed with two-sample independent t-tests to test for differences between POMS and controls. Regional brain volumes for POMS and controls were also tested for normality and then evaluated using two-sample independent t-tests to test for group differences. Pearson correlations were used to assess linear relationships between fornix volume/diffusion metrics (and other total/regional WM/GM volumes) and clinical/cognitive scores such as time since MS onset, EDSS, TLV, Pediatric Fatigue, BDI-II, SDMT, and BVMT-R. Multiple comparisons (FDR) were conducted for group comparisons and Pearson correlations; FDR corrected p-values are presented (*p < .05).
Results
CSF-suppressed, high-resolution FLAIR-DTI tractography depicted the full fornix in all 11 POMS and 26 controls. Fornix tractography showed visibly thinner fornix with bilateral regions of lower FA and elevated diffusivities (MD, AD, and particularly RD) in POMS relative to controls (Figure 1). These tractography observations were similar across all 11 POMS fornix, independent of TLV, and 11 representative age/sex matched controls, as shown on the tracts color-coded for MD (Figure 2).
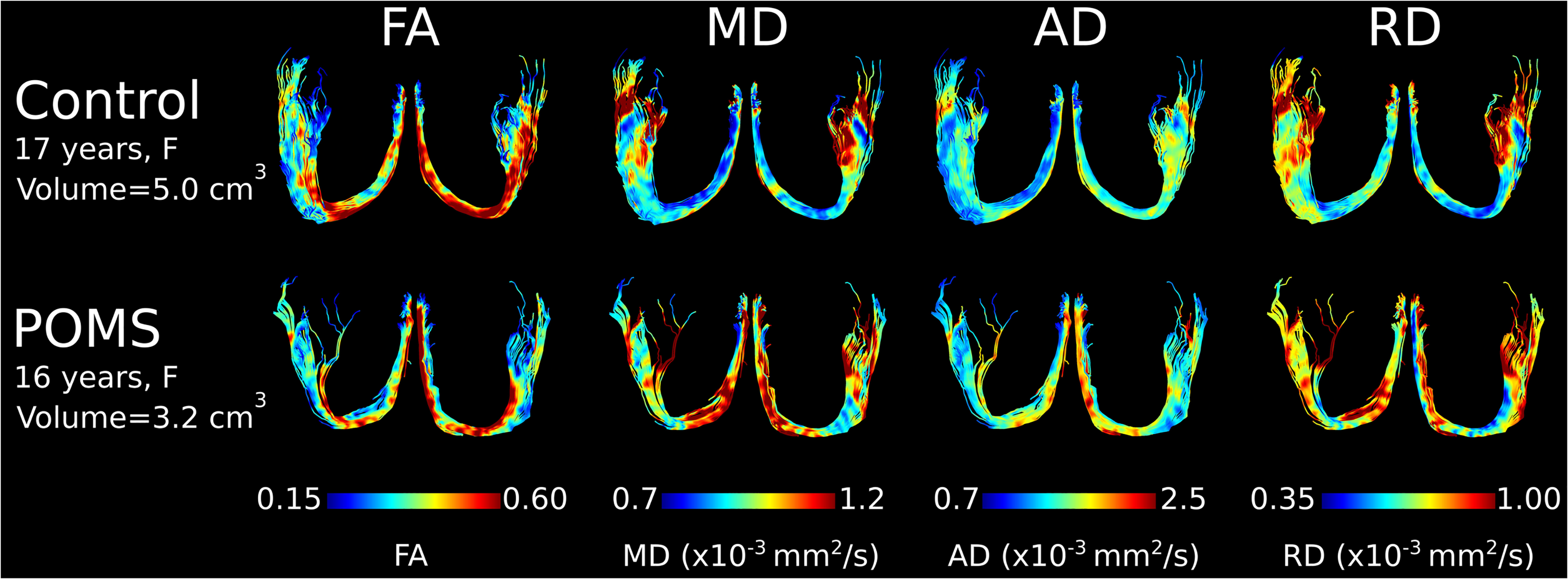
Examples of CSF-suppressed, high-resolution FLAIR-DTI fornix tractography (as seen from superior view) color-coded by FA, MD, AD, and RD in a 17-year-old female control and a 16-year-old POMS participant. In this case, the POMS fornix appears thinner with markedly smaller volume (−36%) and shows bilateral regions with lower FA (−9%), and higher MD (+14%), AD (+9%), and RD (+18%).

Examples of CSF-suppressed, high-resolution FLAIR-DTI fornix tractography (as seen from superior view) color-coded by MD in 11 representative age/sex matched controls and all 11 POMS, ordered by age. Fornix tractography depicted the full fornix in all, but the fornix in POMS appeared thinner compared to controls, and showed bilateral regions with higher MD. Representative FLAIR images of a single slice are shown for each POMS participant with a wide range of total lesion volumes (TLV).
Relative to controls, the POMS had a fornix with 26% lower volume (4.6 ± 1.1 vs 3.4 ± 0.9 cm3, p = .003), 7% lower FA (0.43 ± 0.02 vs 0.40 ± 0.03, p < .001), 12% higher MD (1.06 ± 0.05 vs 1.19 ± 0.07 × 10–3 mm2/s, p < .001), 7% higher AD (1.61 ± 0.06 vs 1.72 ± 0.08 × 10–3 mm2/s, p < .001), and 16% higher RD (0.79 ± 0.05 vs 0.92 ± 0.08 × 10–3 mm2/s, p < .001) (Figure 3). In contrast, hippocampus volume was not different between POMS and controls (p = .551).

Group comparisons of controls (n = 26) and POMS (n = 11) are shown for fornix (fx)
Relative to controls, POMS had many volume differences with the greatest proportional changes in the lateral ventricles (+56%) and CSF (+41%) volumes (Table 2). For brain subregions, the greatest volume losses in order were the thalamus (–23%), cerebellum WM (–23%), total WM (–17%), globus pallidus (–16%), cerebrum WM (–16%), putamen (–12%), total cerebellum (–9%), and then total cerebrum (–8%) and TBV (–8%). These differences were not as great as the fornix, which was proportionally smaller (–26%). Notably, there were no differences in the volumes of other GM, including total GM, cerebrum GM (cortex), cerebellum GM, caudate, or amygdala.
Total/regional left + right WM/GM volumes (cm3) derived from the 3D T1-weighted images with group comparisons for controls versus POMS with mean, standard deviation, percentage difference, and FDR corrected p-values (*p < .05; n.s. is nonsignificant). Brain regions are ordered by percent difference between POMS and controls.

Abbreviations: POMS: pediatric-onset multiple sclerosis; WM: white matter; GM: gray matter; TBV: total brain volume; CSF: cerebrospinal fluid.
For the fornix within the POMS cohort, Pearson correlations (uncorrected) were only found for SDMT versus fornix MD (R = –0.60, p = .049), SDMT versus fornix RD (R = –0.61, p = .048), and TLV versus fornix FA (R = –0.63, p = .040); however, none were significant after FDR-correction. There were also no significant correlations between fornix volume or diffusion metrics and age, time since MS onset, EDSS, pediatric fatigue, BDI-II, or BVMT-R. However, FDR-corrected significant correlations were present for other total/regional brain volumes including: TLV vs thalamus (R = –0.908, p = .005)—bigger TLVs associated with more atrophy; and SDMT versus globus pallidus (R = 0.940, p = .005), TBV (R = 0.859, p = .005), caudate (R = 0.845, p = .026), putamen (R = 0.839, p = .026), total WM (R = 0.832, p = .026), total cerebrum (R = 0.829, p = .039), and cerebrum WM (R = 0.820, p = .039)—larger brain volumes associated with better SDMT scores. Note that there were no FDR-corrected significant correlations for any brain region volume versus time since MS onset, EDSS, fatigue, depression, or BVMT-R in POMS.
Discussion
High-resolution FLAIR-DTI tractography enabled the virtual identification and quantification of the fornix in POMS (age range of 13–19 years), which showed markedly lower volume (–26%) that was affected more than TBV (–8%) or any other brain region assessed in this study (albeit with similar proportional reductions to thalamus and cerebellum WM at −23%), as well as diffusion metric differences of FA, MD, AD, and RD, compared to controls, indicative of microstructural abnormalities. A previous POMS DTI study of 12–17 year olds identified elevated MD in the fornix, among many WM regions, using a whole brain WM voxel-based skeletonized approach, but this used low spatial resolution diffusion images acquired without CSF suppression (susceptible to partial volume effects) and the actual MD values were not reported, nor was the fornix volume or other diffusion metrics such as FA, AD, and RD. 19 Minimizing the effects of CSF partial volume by smaller voxels (volume of 2.9 mm3) and CSF suppression (using inversion recovery FLAIR preparation) is important to not attribute changes of diffusion properties to merely partial volume effects in a smaller fornix. 25 Here, the largest percent change of the diffusion metrics in the fornix was a 16% larger RD that could be interpreted as demyelination; however, axonal loss (i.e., lower density) leading to a markedly smaller fornix is also possible.26,27 A potential injury mechanism is that the thin fornix is bathed in CSF, which has been shown to contain MS inflammatory immunoglobulins in POMS. 28
The large 26% volume percent reduction of the fornix in POMS is in exact agreement with the 26% volume reduction found in our cross-sectional adult MS study (age range of 32–70 years) that used the same FLAIR-DTI acquisition protocol. 15 Note that the fornix volume is reduced by 20% if TBV (which is also reduced in POMS by 8%) is corrected per participant (Supplemental Table S1). Further, POMS fornix DTI metrics were similarly affected relative to controls as in the previous adult MS study: POMS FA −7% versus AOMS −7%, MD +12% versus +9%, AD +7% versus +6%, and RD +16% versus +12%. The similarly affected fornix volume and diffusion metrics regardless of the differences in average disease durations (3 years for POMS here vs 15 years in the adult MS study) suggests that the fornix is affected early on in the disease course of MS and that fornix injury is not necessarily progressive, although this assessment would require longitudinal scans rather than the cross-sectional cohorts as in our two studies. 15 However, this hypothesis is further supported by the presence of inflammatory cytokines in CSF early on in the disease course of adult MS, which could disproportionately affect the fornix early on, as is shown for the similarly affected thalamus. 29 POMS were all relapsing-remitting MS in the current study as well as the majority (35/43 MS participants) in the adult study. 15 However, the prior adult MS FLAIR-DTI study showed far greater fornix changes in volume and all diffusion metrics in those with cognitive impairment, whereas those without cognitive impairment showed significant, albeit less changes, of lower volume and higher MD/RD with no significant changes in FA or AD. 15 Here, POMS with cognitive impairment according to the criteria used in the previous adult MS study (SDMT z-scores below −1.67 standard deviations (SD) and/or BVMT-R z-scores below −1.17 SD) and without cognitive impairment showed similar abnormal fornix volume/diffusion metric differences relative to controls. 15
Encouragingly, the uncorrected negative correlations of SDMT with fornix MD and RD, and TLV with fornix FA, agree with the prior adult MS study. 15 Other adult MS DTI studies have also shown abnormal fornix metrics with some reports of relationships to cognitive impairment, including reduced FA, and elevated MD, AD, and RD.8–13
The fornix is the primary efferent tract of the hippocampus, yet a key observation here is that the hippocampus volume was not different in POMS than controls. This suggests that the fornix abnormalities are not the result of Wallerian degeneration from neuronal death in the hippocampus, rather the fornix may be directly affected by adjacent CSF factors as mentioned earlier. This is consistent with the hypothesis that WM injury precedes injury to connecting GM in MS. 30 Some atrophy could be due to loss of axons that project from the subiculum to the anterior thalamic nuclei, which pass along the anterior portion of the fornix, 31 but its study would require a higher spatial resolution acquisition. Our previous adult MS study showed no differences in hippocampus volume (as measured using automated segmentation software on 3D T1-weighted scans) either, although FA, MD, and RD were abnormal in the hippocampus of cognitively impaired adult MS participants. 15 However, a follow-up study on the same adult MS cohort showed a 14% smaller hippocampus volume as measured by manual segmentation on 1 mm isotropic DTI scans, with no reduction of hippocampus volume in the cognitively unimpaired group although both groups had elevated MD in the hippocampus suggestive of microstructural alterations. 32 There are discrepancies in POMS with hippocampus reported as being smaller or no different than controls, which may be due to a shorter preclinical disease activity or differences in the disease process (i.e. absence of progressive MS) compared to adult MS.4,5,33 Our observations of greatly enlarged lateral ventricles (+56%) and CSF volume (+41%) as well as smaller volumes of thalamus (–23%), total WM (–17%), globus pallidus (–16%), putamen (–12%), total cerebellum (–9%), and TBV (–8%) in POMS are consistent with previous work.2,3,34,35 The thalamus volume is affected nearly as much as the fornix volume, presumably due to the thalamus being located adjacent to the CSF as well. 3 Interestingly, there was no difference in total GM volume between groups, which may suggest that brain volume losses in POMS are driven by WM atrophy. 34
There were several limitations in this study. The sample size of 11 POMS in this single site study was a result of the small local clinical population and the focus on scans under 20 years of age. A larger sample would have been beneficial to assess correlations between fornix volume/diffusion metrics and clinical disability and cognitive function. There was only one male in the 11 POMS participants limiting the analysis of sex effects. The fornix volume/diffusion metrics can depend on the tractography algorithm/parameters between studies although the same methods were used for all POMS and controls here. This study was focused on one key tract of interest, the fornix, but it would be useful to examine other WM tracts in the same cohort. The FLAIR-DTI has limited brain coverage (70 mm coverage by 35 slices 2 mm thick) as it was developed for the fornix, but future analyses of this POMS cohort will include whole brain multishell diffusion MRI analysis of other WM tracts as well as high resolution (1 mm isotropic) DTI of the hippocampus, the latter as performed in our previous adult MS study. 32
Diffusion MRI findings in POMS provide evidence that there is early involvement of the fornix in the disease course of MS, which precedes injury to connecting GM, and is likely related to its location within the ventricles leading to exposure to soluble inflammatory factors in CSF. These results suggest that the microstructural integrity of the fornix may be an early hallmark of MS and that the fornix should not be overlooked in future work examining MS pathology.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251315161 - Supplemental material for Disproportional smaller fornix with altered microstructure in pediatric multiple sclerosis shown by high-resolution fluid-suppressed diffusion tractography
Supplemental material, sj-docx-1-mso-10.1177_20552173251315161 for Disproportional smaller fornix with altered microstructure in pediatric multiple sclerosis shown by high-resolution fluid-suppressed diffusion tractography by Carly Weber, Colin Wilbur, Gregg Blevins and Christian Beaulieu in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-doc-2-mso-10.1177_20552173251315161 - Supplemental material for Disproportional smaller fornix with altered microstructure in pediatric multiple sclerosis shown by high-resolution fluid-suppressed diffusion tractography
Supplemental material, sj-doc-2-mso-10.1177_20552173251315161 for Disproportional smaller fornix with altered microstructure in pediatric multiple sclerosis shown by high-resolution fluid-suppressed diffusion tractography by Carly Weber, Colin Wilbur, Gregg Blevins and Christian Beaulieu in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-3-mso-10.1177_20552173251315161 - Supplemental material for Disproportional smaller fornix with altered microstructure in pediatric multiple sclerosis shown by high-resolution fluid-suppressed diffusion tractography
Supplemental material, sj-docx-3-mso-10.1177_20552173251315161 for Disproportional smaller fornix with altered microstructure in pediatric multiple sclerosis shown by high-resolution fluid-suppressed diffusion tractography by Carly Weber, Colin Wilbur, Gregg Blevins and Christian Beaulieu in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgments
The authors wish to thank registered MS nurse, Leah White, for her training in clinical and cognitive testing, Peter Seres for his assistance during scans, and Pablo Stack Sanchez for his teaching of MRtrix3.
Consent to participate
All 37 participants provided written informed consent (or parental consent and assent if under 16 years of age) including 11 diagnosed with relapsing-remitting POMS that were recruited from the pediatric neuroinflammatory registry and the University of Alberta Neurosciences Clinic, and 26 healthy controls chosen with a similar age/sex distribution from a previous normative study using an identical MRI protocol.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
This study was approved by the University of Alberta Human Research Ethics Board (approval no. Pro00112979) on 17 November 2022.
Funding
The authors disclosed receipt of the following financial support for research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research (CIHR), MS Canada, and a salary award by the Canada Research Chairs (coauthor CB).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
