Abstract
Background
Cladribine shows efficacy in multiple sclerosis (MS), but Latin American (LATAM) real-world data is limited, despite potential sociodemographic variations.
Objective
Investigate baseline characteristics and clinical response in highly active MS patients in Mexico, identifying predictors of early treatment response.
Method
A multicenter cohort study analyzed retrospective data from individuals with “highly active” MS in the Cladribine Patient Support Program across 11 Mexican clinics. Criteria included one-year prior treatment with another disease-modifying treatment and recent relapse with specific MRI findings. Primary outcomes focused on achieving NEDA-3 status after 12 months.
Results
In the follow-up, 67.5% maintained NEDA-3 status. Baseline EDSS scores decreased significantly from 1.50 to 1.00 (p = 0.011), with no confirmed disability worsening. No significant differences were observed between NEDA-3 achievers and non-achievers in demographic and clinical variables. No severe adverse events were reported.
Conclusion
Cladribine showed early and effective control of active MS in Mexican patients, demonstrating a secure profile with minimal adverse events. This study provides valuable real-world evidence in the LATAM context.
Introduction
In the last two decades, there has been a growing availability of disease-modifying treatments (DMT) for people with multiple sclerosis (pwMS) in Latin America (LATAM), particularly in Mexico. This trend presents a unique opportunity for the optimal treatment profiling of pwMS but is severely limited by the accessibility of DMT in our country.1,2 Cladribine, a purine analog that induces transitory lymphocytic depletion through apoptosis, has demonstrated a high efficacy profile with safety in pivotal trials and cumulative real-world evidence. 3 In LATAM, cladribine was approved as a DMT for pwMS during 2018 and 2019. 4 Nevertheless, regional evidence of the experience is scarce and is mainly extrapolated from Argentina,5,6 where the low incidence of infections and lymphoma, along with good adherence from the patients, are reported. However, genetic, and environmental differences are expected within regions and countries of LATAM with a dissimilar interethnic admixture7–9 and associated clinical differences.10,11
The pivotal CLARITY trial demonstrated a noteworthy reduction in the annualized relapse rate (ARR) and a diminished risk of 3- and 6-month confirmed disability worsening (CDW) among patients treated with cladribine 3.5 mg/kg, compared to the placebo group, observed over 96 weeks. Furthermore, the treated group exhibited a No Evidence of Disease Activity (NEDA-3) status in 44% of cases at the 2-year follow-up.12,13 The swift onset of cladribine's effects is of particular interest, evidenced by a significant difference in MRI parameters within the initial month of treatment initiation. 14 Consequently, efforts have been directed toward identifying characteristics that may aid in selecting cladribine “responders.” However, despite these endeavors, there is no clear identification of predictive factors associated with cladribine response, except for the number of previous DMTs.15–17 In response to the demand for real-world data from Mexico, this study seeks to elucidate the baseline characteristics of individuals and their clinical response in the subset of highly active MS within our nation. Additionally, the study aims to identify predictors associated with early treatment response in these individuals.
Methods
This study constitutes a multicenter, observational, post-marketing cohort. Anonymized data were retrospectively gathered from pwMS diagnosed with “highly active” disease upon entry into the Cladribine Patient Support Program attending one of eleven tertiary MS outpatient clinics dispersed across Mexico. All enrolled patients satisfied the McDonald 2017 diagnostic criteria. The categorization as “highly active” was determined if at least one of the following conditions was met: pwMS with a minimum of one year of treatment using another DMT and the occurrence of a relapse in the preceding year, accompanied by either ≥1 gadolinium-enhancing lesion and/or ≥9 lesions in T2-FLAIR; or ≥2 relapses in the previous year with or without prior DMT use. The primary outcomes focused on NEDA-3 status at the 12 months of treatment. NEDA-3 was defined by the absence of relapse, CDW (specified as a 1.5-point increase in EDSS if the baseline EDSS score was 0, a 1.0-point increase if the baseline EDSS score was ≤5.5, or a 0.5-point increase if the baseline EDSS score was >5.5), and the absence of new lesions observed in control MRI T2-FLAIR, all assessed during the 12-month follow-up period. Use of previous DMT was cataloged as moderate efficacy (MeDMT) or if the patient was previously treated with interferon, glatiramer acetate, dimethyl fumarate, or as high efficacy DMT (HeDMT) if the treatment was fingolimod, natalizumab or ocrelizumab. Lymphopenia was characterized by an absolute lymphocyte count below 1.2 cells/μL × 103. In comparison, severe lymphopenia was defined by an absolute lymphocyte count below 0.5 cells/μL × 103, corresponding to grade III–IV in other classifications. The baseline clinical, radiological, and demographic characteristics are presented in frequencies and percentages for categorical variables. In contrast, continuous variables are described using mean ± standard deviation (SD) for those following a parametric distribution and median with interquartile range (IQR) for those with a non-parametric distribution. Per prescription information, a completed blood count was obtained before treatment onset and the second cycle. Baseline comparisons of characteristics between patients who achieved NEDA-3 and those who did not were assessed using either the T-Student or Wilcoxon test, depending on the normality of the distribution. The non-parametric Dunn–Bonferroni test was employed to investigate changes in lymphocyte, monocyte, and neutrophil counts over the study period. Additionally, univariate logistic regression was conducted to elucidate further the impact of baseline characteristics in predicting NEDA-3 status. The analyses were performed using R version 4.3.1 and packages tidyr, ggplot, ggstatdplot, ggrepel, ggsignif, tableone, and lme4. A significance level of ≤0.05 was applied. The study received approval from the local Ethics committee of the Civil Hospital of Guadalajara (CEI 130/23).
Results
Baseline characteristics
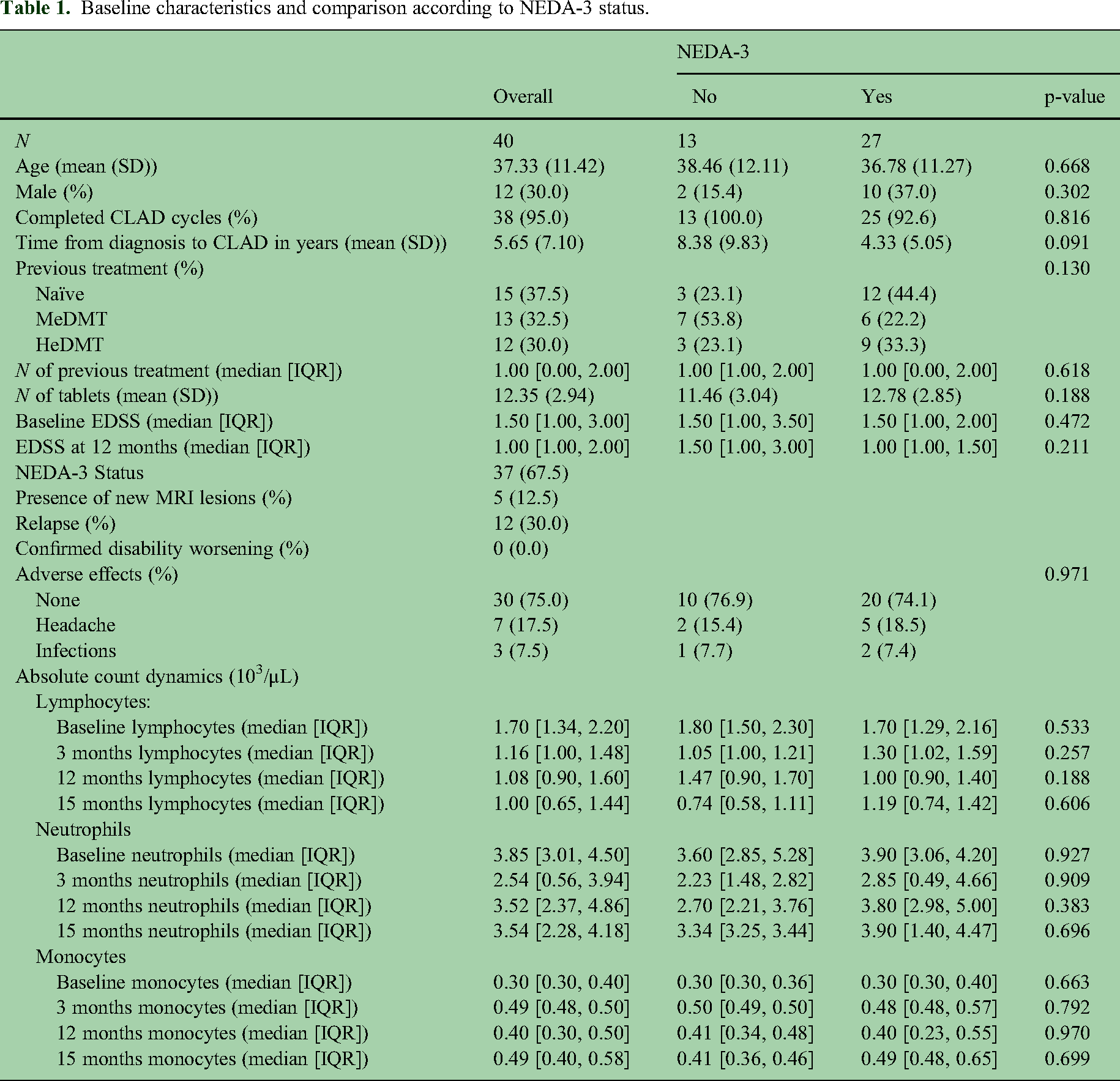
A total of 40 (19.6%) pwMS from a total of 204 fulfilled the definition of “highly active MS” at the start of cladribine treatment and were included in the study, of whom 12 (30.0%) were male. The mean age was 37.33 ± 11.42 years, presenting with a disease duration of 5.65 ± 7.10 years. At entry, 15 (37.5%) patients were treatment-naïve, while 13 (32.5%) were undergoing a previous MeDMT, and 12 (30.0%) were on HeDMT. The median number of previous DMT in non-naïve patients was 1.00 (IQR 1.00, 2.00)—Table 1. The median EDSS score at baseline was 1.50 (IQR 1.00, 3.00), which decreased to 1.00 (IQR 1.00, 2.00) at the one-year follow-up (Wilcoxon signed-rank test, V = 93, p-value = 0.011) Figure 1. Patients were revaluated at a mean of 1.07 ± 0.22 years.

Evolution of expanded disability status scale (EDSS) from baseline to 12 months following cladribine treatment onset. We found a significant reduction in EDSS scores one year post-initiation of cladribine treatment.
Baseline characteristics and comparison according to NEDA-3 status.
Cladribine effect on NEDA-3
During the follow-up period, 27 patients (67.5%) sustained a NEDA-3 status. However, relapse occurrence in 12 patients (30.0%) and radiological breakthrough activity in 5 patients (12.5%) prevented the remaining individuals from achieving this status. Notably, none of the patients experienced CDW during the follow-up. No differences were found between the patients who reached NEDA-3 and those who did not in age, sex, treatment-naïve status, initial EDSS, or CBC dynamics at baseline and follow-up assessment (Table 1). Univariate logistic regression analysis substantiated the absence of statistically significant results in these observations (Table 2).
Logistic univariate regression model.

None of the baseline variables exhibited a statistically significant effect on the probability of achieving NEDA-3 status in the first year.
Abbreviation: CLAD, cladribine. MeDMT, moderate efficacy disease-modifying treatment; HeDMT, high efficacy disease modifying-treatment; DMT, disease-modifying treatment; OR, odds ratio; CI, confidence interval at 95%.
Safety
No severe adverse reactions or infections were reported throughout the follow-up period among any of the patients. No events of special interest, including documented cases of cancers potentially associated with Cladribine treatment, severe infections, opportunistic infections, or cases of death were detected. Eight patients (25.0%) experienced minor side effects, with headache being the most common (17.5%) and minor infections observed in three patients (7.5%). Notably, only 16 patients attended their control labs at the 3-month mark. At 3 months after treatment initiation, lymphopenia was identified in eight patients (25.0%), with only one case classified as severe. Comprehensive laboratory profiling was conducted for all patients at 12 months. Before the initiation of the second cycle of cladribine, asymptomatic lymphopenia was observed in 22 patients (55.0%), with only one case deemed severe enough to warrant a 2-month treatment delay. The Dunn–Bonferroni test demonstrated a significant decrease in absolute lymphocyte count sustained from 12 to 15 months. However, no overall alteration was observed in the counts of neutrophils or monocytes within our cohort (Figure 2).

Temporal dynamics of white blood cell subpopulations following cladribine treatment initiation. (A) Fluctuations in lymphocyte counts, (B) monocyte counts, and (C) neutrophil counts are presented at various evaluation time points after treatment onset. Only a significant lymphocyte decrease is observed at 12 and 15 months.
Discussion
Our nationwide real-world longitudinal study showed that cladribine treatment is effective in the short term in patients with highly active MS. In a 1-year follow-up, 67.5% of the patients reached a NEDA-3 status regardless of being naïve or previously treated with MeDMT or HeDMT. However, we did not find clinical factors associated with achieving this status. Along the same line, our study suggests a safety profile in the first year of use of cladribine with a low incidence of non-serious side effects.
Our study revealed that the use of cladribine in the group of highly active MS maintained an appropriate degree of NEDA-3 within the first year but also showed a median decrease in the EDSS of 0.5 (1.50 [IQR 1.00, 3.00] to 1.00 [IQR 1.00, 2.00], p = 0.011). Although reduction in the EDSS is not a standard measure of efficacy when evaluating treatment response, this finding seems to be in line with a preliminary meta-analysis of a pool of 7244 MS patients that calculated a reduction of −0.54 (95%CI: −1.46 to 0.39; p = 0.030, I2 = 72%). 18 However, we theorize this observation is based on the effect of the inflammatory component in pwMS with the highly active phenotype in our cohort. In both clinical and radiological assessments, increased inflammatory activity has been associated with a less favorable prognosis in pwMS, predicting the progression of the disease and the accumulation of disabilities. 19 Consequently, scores assessing treatment response are specifically crafted to identify such outcomes within the initial year post-treatment initiation.20–22 While highly active MS and HeDMT definitions may vary, our findings demonstrate that two-thirds of patients achieved NEDA-3 status within the first year of treatment onset. Our cohort comprised both treatment-naïve and previously treated pwMS, transitioning from both MeDMT and HeDMTs, all exhibiting a satisfactory response with a decline in the EDSS within the first year. Similarly, a sub-analysis utilizing a comparable definition of highly active MS revealed a substantial response, with an 82% reduction in CDW observed in the initial 6 months, 23 independent of the individuals’ prior treatment status. 24 Regionally, our results align with a previous work published in Argentina, where 73.9% of pwMS treated with cladribine reached NEDA-3 at year two. 25
In contrast to findings indicating lymphopenia linked to cladribine reported elsewhere, 26 our results exhibited a noteworthy and sustained reduction persisting up to one year after the administration of the initial cycle and preceding the second cycle. Despite the limitation of not acquiring specific counts for lymphocyte subsets, these alterations did not demonstrate an association with the likelihood of achieving NEDA-3 status or correlate with adverse events. Furthermore, contrary to other studies, grade III-IV lymphopenia was infrequently observed in our population.27,28 In addition, it is crucial to highlight that the threshold applied in our study is based on the normal values provided by the reference laboratory, which may differ from the criteria used to define lymphopenia in other centers. 29
While the brief duration of the follow-up constrains our research, our sample did not manifest severe adverse events linked to the treatment. Minor repercussions were documented, such as headaches and uncomplicated upper respiratory tract infections. Notably, this safety profile aligns with the information from the pivotal trials conducted thus far. 30 Similarly, our results showed no significant effect on the innate immune system, preserving the absolute count of both neutrophils and monocytes, providing backing for the idea that cladribine tablets selectively and temporarily diminish cells of the adaptive immune system only.26,31 Finally, our study provides real-world evidence of the efficacy and safety in a subset of pwMS with well-delineated criteria of highly active MS in a subgroup of the underrepresented Hispanic/Latin population. 32 Real-world evidence is a reliable source of post-approval evaluation of DMT in pwMS. It may help evaluate efficacy and treatment patterns, 33 particularly in our country.
Various limitations were identified in our study, encompassing the absence of crucial baseline data, including the ARR and the sub-scores of EDSS, alongside the constraint of a brief follow-up period. Moreover, a subset of patients exhibited suboptimal adherence to the follow-up protocol, explicitly concerning the absence from the control laboratories at the 3-month mark following the onset of the treatment cycles. Methodologically, the restricted number of patients curtailed the magnitude of influence that could be attributed to our findings. Similarly, we only evaluate the effect of the treatment during an early period, while other agents with different mechanisms of action have proven that the NEDA-3 status may be reached over longer periods of exposition in different stages of the disease. 34
Despite these limitations, our study underscores the secure profile and minimal occurrence of adverse events associated with cladribine therapy in Mexican pwMS. Notably, two-thirds of the patients achieved NEDA-3 status within the initial year, irrespective of prior exposure to MeDMT or HeDMT, with a continual reduction in absolute lymphocyte count observed in the first year, albeit with unclear correlation to treatment efficacy.
Footnotes
Data availability
Anonymized data is available at reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: No company influenced the decision to write this article or its content, but we acknowledge Dr Merari Duarte for helping with the meeting and organization of the authors involved. Within the past two years, SSS, SQ, BBC, FRL, and EGF have received speaking and consulting honoraria from Merck KGaA, Novartis, Synthon, and Biogen. These disclosures are provided in the interest of transparency and do not influence the article's integrity.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
