Abstract
Background
Eye movements can reflect brain alterations and inform on the presence of motor disabilities and cognitive impairments in people with multiple sclerosis (pwMS).
Objective
The aim of the study was to determine the correlation between motor and cognitive measurements and eye movement parameters when performing the n-back task (NBKT).
Methods
This was a cross-sectional study carried out at Ramos Mejía Hospital, a center specialized in demyelinating diseases in Buenos Aires, Argentina. The study population consisted of 66 patients with relapsing-remitting multiple sclerosis (RRMS) and 5 patients with secondary progressive multiple sclerosis (SPMS). pwMS performed the n-back test while using a device head mounted display (HMD) with eyetracking capabilities in order to capture eye movement. Clinical motor and cognitive measures were assessed with Expanded Disability Status Scale (EDSS), Nine Hole Peg Test (NHPT), Timed 25-Foot Walk (T25FW), and Symbol Digit Modalities Test (SDMT).
Results
pwMS showed strong and statistically significant correlations between gaze duration; number of fixations, saccade amplitude and motor disabilities and cognitive impairments as measured by EDSS, NHPT, T25FW, and SDMT.
Conclusion
This study found significant correlations between eye movement behavior and motor and cognitive disability in pwMS. These findings suggest that eye movements have the potential to be used as a surrogate biomarker in MS progression.
Introduction
Multiple sclerosis (MS) is a cause of neurological disability in young adults and is associated with long-term disability, at considerable cost to families and the community. 1 The pathophysiological process of MS is complex and dynamic, and involves the interaction of damage and repair mechanisms, the results of which determine the evolution of the disease. Consequently, the course of the disease is unpredictable and the symptoms are heterogeneous. 2 The ocular motor system can be affected when MS lesions involve the brainstem and cerebellar structures. 3 In people with MS (pwMS), ocular motor impairments are associated with more advanced disability. 4 Previous research have shown that eye movements can reflect brain alteration and inform on the presence of neurodegeneration and cognitive impairment.2,5 Eye movements are controlled by diverse networks and circuits of the brainstem and cortical structures6,7 that are susceptible to a variety of degenerative processes. 8 Furthermore, the analysis of gaze patterns and memory-guided saccades can provide insights into the integrity of various motor and cognitive processes. 9 For instance, the networks and structures involved in the control of saccades are well defined and provide a sensitive measure of working memory and attention processes, which are disrupted early in the course of MS. 10 Working memory task require the synchronization of connections between the prefrontal, cingulate, and parietal regions, integrating complex information to generate appropriate responses. These connections are also altered due to the diffuse damage to white and gray matter observed in pwMS. 11 MS literature has documented diverse oculomotor anomalies, such as fixation instability, slowed saccades, and increased antisaccade error rates.12,13 Further, it was shown that pwMS also performed shorter saccades and longer gaze durations.8,14,15 In addition, several studies have linked these anomalies to brain health via correlations between eye movement parameters and disease or cognitive status, as measured by tools such as the Expanded Disability Status Scale (EDSS) and the Symbol Digit Modalities Test (SDMT).3,16
The aim of this study was to determine the correlation between validated motor and cognitive measurements and eye movement parameters when performing the n-back task (NBKT). To assess motor and cognitive status, we used four of the most employed MS assessment tools, that is, EDSS, Nine Hole Peg Test (NHPT), Timed 25-Foot Walk (T25WF), and SDMT, were used forming our clinical outcome measures of interest.
Methodology
Study design and population
A cross-sectional study was carried out at the Ramos Mejía Hospital, a center specialized in demyelinating diseases in Buenos Aires, Argentina. The study population consisted of 66 patients with relapsing-remitting multiple sclerosis (RRMS) and 5 patients with secondary progressive multiple sclerosis (SPMS). A nonrandom convenience sampling was carried out; patients who were evaluated at the center between July 2022 and March 2023 were included consecutively and prospectively (see Demographic description in Table 1). To be included, patients had to be over 18 years of age, not have visual acuity alterations that prevented the completion of the tests and be able to understand the evaluator's instructions. Patients who experienced a fall less than 30 days before the evaluation and those who were unable to complete the calibration of the equipment used were excluded.
Demographic, clinical, and oculomotor variables for patients with MS who were screened for disability using the EDSS, NHPT, T25FW, and SDMT.

EDSS: Expanded Disability Status Scale; MS: multiple sclerosis; NHPT: Nine Hole Peg Test; RRMS: relapsing-remitting multiple sclerosis; SDMT: Symbol Digit Modalities Test; SPMS: secondary progressive multiple sclerosis; TNF: total number of fixations.
Study variables
Demographic and clinical data were collected, including age, gender, clinical phenotype, and disease duration. Clinical measures included EDSS, 17 NHPT, 18 T25FW, 19 and the SDMT. 20
The behavior of eye movements was recorded using a specific head mounted display (HMD) with embedded eye tracking (see below for a technical description). The NBKT was carried out by a single evaluator (AM).
n-Back task
The task stimuli were presented on a black background and consisted of red circles (30 mm diameter). There was a central fixation cross (15 mm × 15 mm) surrounded by six squares: two located 10 degrees to the left or right of the central fixation cross, and four located at 45-degree angles from the central fixation cross (top right, bottom right, top left, and bottom left). The boxes measured 58 mm by 55 mm and were differentiated from the foreground by a white border. Participants were instructed to fixate their gaze on the central fixation cross while three red circles appeared sequentially, in any one of the six peripheral boxes for a duration of 250 ms each. Participants were instructed to remember both the location (box) and the order of appearance of the stimuli. Then, the central fixation cross disappeared, and participants were instructed to quickly look at the boxes in the reverse order in which the red circles had appeared: first the box where the last red circle appeared, second the box where the second red circle appeared, and finally the box where the first circle appeared. Working memory conditions (WMem) and WMem loads (0-back, 1-back, and 2-back) were reflected in the instructions. An example of the eye movements recorded during NBKT, showing eye fixations of pwMS with an EDSS score of 1.0 (left) and with an EDSS score of 3.0 (right), is shown in Figure 1.

Viewmind's n-back task. Each red circle represents an ocular fixation. In this example, a participant with MS with an EDSS score of 1.0 (left) made fewer fixations and performed longer saccades than one with EDSS score of 3.0 (right). EDSS: Expanded Disability Status Scale; MS: multiple sclerosis.
Equipment
Participants sat on a chair and wore a HMD (HP Reverb Omniset, Hewlett-Packard, Palo Alto, CA, USA) with Eye Tracking. VIEWMIND software ran on a computer suitable for the mentioned equipment (https://vr.tobii.com/integrations/hp-reverb-g2-omnicept-edition/). All stimuli were generated using VIEWMIND's registered algorithms (https://www.viewmind.com). Stimuli were presented on the center line of the HMD. Eye movements were recorded with an eye tracker included in the HMD, with a sampling rate of 120 Hz. All recordings and calibration were binocular.
Statistical analysis
Statistical analyses were performed in R version 3.1.1 (RDevelopment Core Team). Measures of central tendency and measures of dispersion were calculated for continuous variables, while frequencies and percentages were presented for categorical variables. A correlation matrix was constructed to explore associations between EDSS, NHPT, T25FW, and SDMT with gaze duration (defined as the sum of fixations on a target), total number of fixations (TNF) (defined as the number of fixations a person performs to make a task) and saccade amplitude (defined as the distance between one fixation and the next one). The Spearman correlation coefficient was calculated, depending on the nature of the variables, and the statistical significance of the correlations was evaluated.
Data availability statement
Any requests for data by qualified scientific and medical researchers for legitimate research purposes will be subject to the Data Sharing Policy of the health care business of ViewMind Inc, USA. All requests should be submitted in writing to ViewMind portal https://www.viewmind.com/contact/.
Results
Seventy-one patients with MS were included in this study. The mean age was 40.03 ± 12.12 years. Mean EDSS was 3.07 (standard deviation ± 1.84). The average NHPT time was 24.70 ± 8.34 s, while the average time for the T25FW was 6.08 ± 1.90 s. Table 1 presents MS oculomotor features and clinical outcome measures. Eye movement metrics were analyzed alongside demographic variables. Elderly patients exhibited longer gaze duration, a higher TNF, and shorter saccade amplitude when analyzing aging (p < 0.003, p < 0.001, p < 0.001, respectively). We found no differences between gender. On the other hand, patients with longer disease duration of MS exhibited longer gaze duration and shorter saccade amplitude when analyzing aging (p < 0.003, p < 0.001, respectively). The TNF seems to be not affected by disease duration.
There were strong Spearman's correlations between eye movement parameters (i.e. gaze duration, number of fixations, and saccade amplitude) and clinical outcome measures (i.e. EDSS, NHPT, T25WD, and SDMT). Patients with MS showed longer gaze duration with increased disability level as measured by EDSS ρ = 0.48(rs = 0.48, p < 0.001), NHPT (rs = 0.50, p < 0.001), and T25FW (rs = 0.48, p < 0.001), and shorter gaze duration with a higher score in the SDMT (rs = −0.76, p < 0.001), meaning that longer gaze durations are correlated with altered motor and cognitive capabilities.
A similar pattern was observed when considering TNF, where the TNF increased when more disability was found in EDSS (rs = 0.40, p < 0.008), NHPT (rs = 0.53, p < 0.001), and T25FW (rs = 0.38, p < 0.003), and decreased with a better performance in the SDMT (rs = −0.55, p < 0.001) meaning that a bigger number of ocular fixations is correlated with an increase in motor disabilities. At the same time, a lower number of fixations is correlated with less cognitive complaints.
Finally, the saccade amplitude pattern also followed a similar trend when correlating with clinical outcome measures, where pwMS produced shorter saccades with more disability in EDSS (rs = −0.35, p < 0.003), NHPT (rs = −0.55, p < 0.001), and T25FW (rs = −0.40, p < 0.004), and longer ones with a higher score in the SDMT (rs = 0.63, p < 0.001), reflecting that shorter saccades are correlated with increased motor disabilities; and longer saccades with less cognitive complaints (see Figure 2 for a Spearman's rs linear relationship between the oculomotor and clinical measures and Figure 3 for a Matrix Correlation).

Linear Regression Model. Four main effects for gaze duration, total number of fixations and saccade amplitude as dependent variables. Predictors are EDSS, NHPT, T25FW, and SDMT. EDSS: Expanded Disability Status Scale; NHPT: Nine Hole Peg Test; SDMT: Symbol Digit Modalities Test; T25FW: Timed 25-Foot Walk.
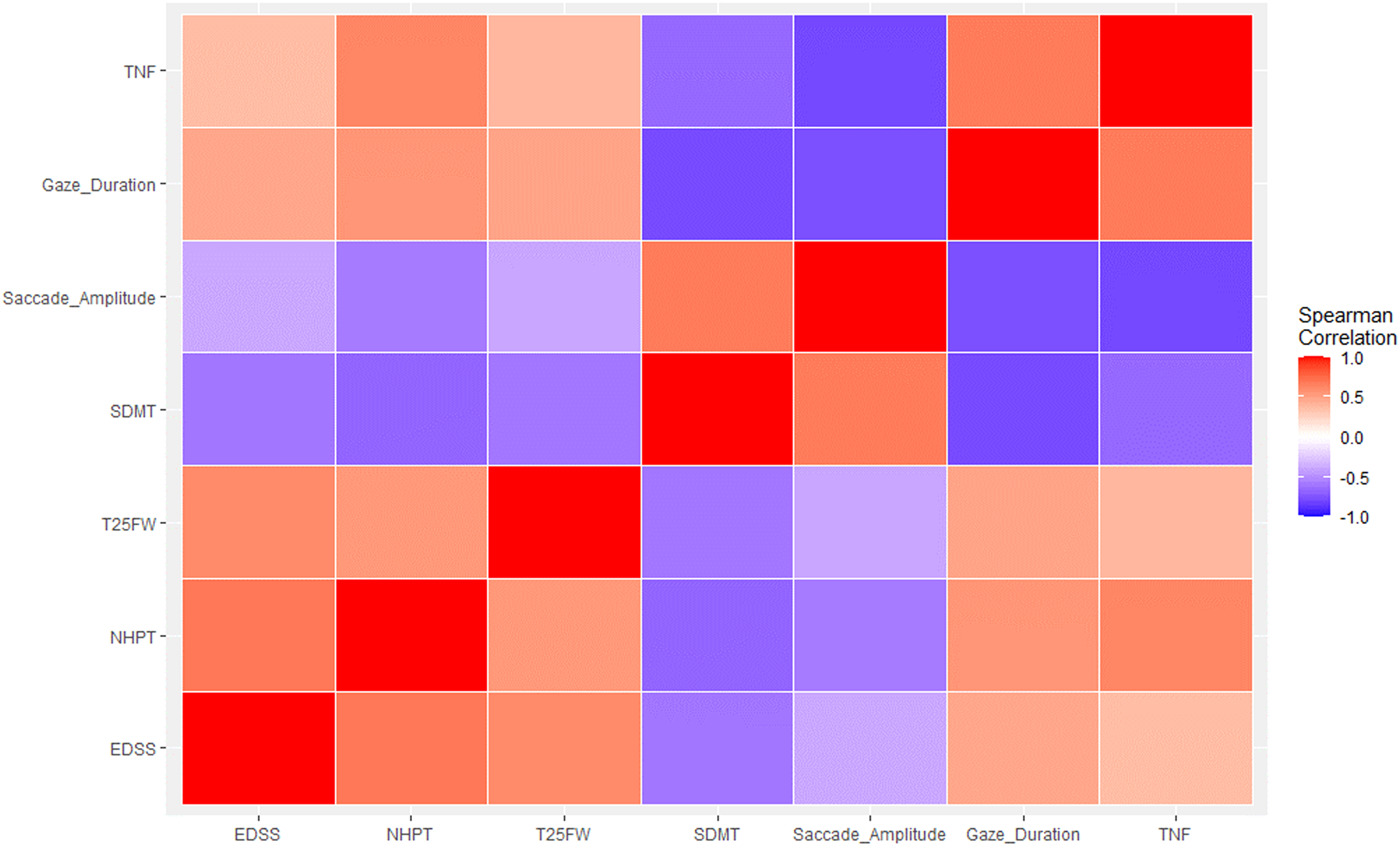
Correlation matrix. EDSS, NHPT, T25FW, SDMT, and oculomotor measures. Blue color tones represent a negative correlation. This means that as the values of one variable increase, the values of the other variable tend to decrease. The darker the shade of blue, the greater the magnitude of the negative correlation. Red color tones represent a positive correlation. This indicates that as the values of one variable increase, the values of the other variable also tend to increase. The darker the shade of red, the greater the magnitude of the positive correlation. EDSS: Expanded Disability Status Scale; NHPT: Nine Hole Peg Test; SDMT: Symbol Digit Modalities Test; T25FW: Timed 25-Foot Walk.
Discussion
In this study we evaluated the relationship between eye movement parameters as surrogate measurements of widely used clinical measures of motor disabilities and cognitive dysfunctions in MS. Our findings unveiled noteworthy correlations between eye movement parameters and both physical disability and cognitive dysfunction, as assessed by EDSS, NHPT, T25FW, and SDMT.
The basic processes involved in eye movements during complex tasks have been thoroughly described in the literature. 21 There is growing consensus that eye movement behavior can be used to evaluate cognitive and motor processing given that several cognitive processes, including visual scanning, working memory and executive processes, have been shown to influence gazing and saccade parameters. 22
In the NBKT, it is crucial for the participant to actively keep the order and location of three visual stimuli, placing high demand on active memory maintenance processes (i.e. encoding stage). The participant must also select the correct answer, which relies on executive control processes (i.e. recognition stage). An error occurs when there is a disruption of the activity underpinning either process. Given that disruption on both processes increased proportionally with MS disabilities, 5 we expected that both TNF and prolonged gaze durations would show incremental alterations when MS manifestations were more pronounced. As it was shown, pwMS made more fixations and had longer gaze durations as the disability level increased, showing significant correlations with motor disability and cognitive dysfunction (EDSS, NHPT andT25FW, and SDMT respectively). Recently, Villers-Sidani et al. 16 have shown that pwMS exhibited an increased number of target's displaced ocular fixations which correlated with a lower performance on the Paced Auditory Serial Addition Test, SDMT and the California Verbal Learning Test (CVLT), all of which assess memory, visuospatial ability, and/or inhibitory control. In our study, this increased number of fixations and the extended gaze durations could reflect a major dysfunction within executive processes, meaning that pwMS needed more time to process information—reflected on longer gaze durations—to make a decision about where to move the eyes next. Previous studies23,24 proposed that the control of the gaze was essential to retaining mentally represented information and using it to form and sustain appropriate stimulus–response relationships. Retention of goal-relevant information is recognized as being mediated by the pre-frontal cortex (PFC), with task goals reflected as sustained activity within topographically organized recurrent thalamo-cortico-striatal circuits, which serve to connect discrete regions of the PFC with distinct regions of the basal ganglia and thalamus.25,26 As in other pathologies, in MS, extended gaze durations may also represent the output of inefficient cortical integration mechanisms responsible for forming or holding the master map of target locations. 27
In our task, as cognitive demands on saccade generation increase, more-extensive recruitment of neural regions occur, particularly in prefrontal regions that subserve executive control and task learning.28,29 The PFC is critical in regulating goal-directed behavior, connecting directly and indirectly with the association cortex, limbic cortex, and subcortical structures via the orbital and medial prefrontal cortices. The PFC is in a unique position to collect and integrate diverse sets of information, especially those associated with attention, motivation, and memory. 30 Once integrated, this information is transmitted to premotor ocular motor areas. 31 The dorsolateral PFC, in particular, is thought to control memory-guide saccade. 32 In this study, the striking modifications observed in saccade amplitude pwMS may reveal a considerable deterioration of one or more brain structures involved in controlling these behaviors, in particular in the dorsolateral PFC which is responsible for sending saccades to a particular location. 6 As we reported, saccades in pwMS were correlated negatively with EDSS, NHPT, and T25FW, meaning that when longer saccades were required to reach each target during the NBKT, only pwMS with fewer disabilities were able to produce the expected appropriate landing saccades. Additionally, saccades in pwMS correlated positively with the SDMT, meaning that only those patients with lower cognitive dysfunction were able to timely generate the ongoing saccade when required. These findings add support to the validity and utility of ocular motility measures as a surrogate measurement of motor and cognitive disabilities in pwMS.
It is important to highlight some limitations of our study. First, the study used a cross-sectional design, with data collection at a single time point. This design does not allow establishing causal relationships and nor does it allow evaluating longitudinal changes in eye movement behavior and motor disability over time. In addition, our study focused only on pwMS and did not include a control group of healthy individuals for comparison. Although the research question did not include a comparison with the non-MS population, the inclusion of a control group would have allowed us to better evaluate the differences between people with MS and the general population.
In conclusion, this study found significant correlations between eye movement behavior and motor and cognitive disability in people with MS. These findings suggest that eye movements could be used as a surrogate biomarker of cognitive dysfunction in MS. Consequently, these ocular motor measures could have utility in clinical drug trials aimed at ascertaining whether agents are capable of reducing and mitigating disease progression.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Fernandez G is the Chief Scientific Officer of ViewMind Inc. No other disclosures were reported.
Funding
The authors declare that they have not received funds to carry out the study, nor to analyze the data or to write the paper.
