Abstract
Background
People with multiple sclerosis (MS) have an increased risk of ischemic heart disease as compared to people without MS after accounting for traditional vascular risk factors.
Objective
We assessed whether subclinical atherosclerosis, an inflammatory disease of arteries, occurs in persons with MS who do not have traditional vascular risk factors, and whether the Framingham Score (FRS) predicted carotid intima media thickness (CIMT) similarly in people with and without MS.
Methods
We recruited participants with and without MS who did not have vascular disease. Participants completed questionnaires, physical assessments, underwent an ultrasound (CIMT), and provided samples for HbA1c and lipid measurements. We defined subclinical atherosclerosis as an average CIMT ≥75th percentile, and tested the association between MS/not-MS, FRS, and atherosclerosis using logistic regression.
Results
We recruited 106 participants with MS 101 without MS. The average (SD) CIMT did not differ between the MS (0.60 [0.11]) and non-MS (0.61 [0.12]) cohorts (p = 0.69), nor did the proportion with atherosclerosis (MS: 11.3% vs. non-MS 13.4%, p = 0.58). On regression analysis a 1-point increase in the FRS was associated with 11% increased odds of having atherosclerosis (95%CI: 1.04, 1.19) but MS was not.
Conclusion
MS was not associated with subclinical atherosclerosis.
Keywords
Introduction
People with multiple sclerosis (MS) experience an increased risk of ischemic heart disease (IHD) as compared to people without MS. 1 Most IHD events occur in individuals with traditional risk factors 2 ; however, the increased risk of IHD in people with MS is not accounted for by traditional vascular risk factors including diabetes, dyslipidemia, hypertension, and smoking. 1 Insufficient physical activity, obesity, alcohol use disorder, smoking history, and lower socioeconomic status (SES) also contribute to vascular risk.
People with chronic inflammatory diseases such as rheumatoid arthritis (RA) also have an increased risk of cardiovascular disease, and have premature subclinical atherosclerosis (progressive inflammatory disease of the arteries) even without traditional vascular risk factors 3 ; guidelines recommend heightened screening in these conditions. 4 If MS similarly is associated with premature subclinical atherosclerosis, then screening and management of vascular risk factors should be modified.
Coronary angiography remains the gold standard for diagnosis and stratification of atherosclerotic disease but its' costly and invasive nature limits its utility for screening. Carotid intima media thickness (CIMT) has been extensively studied and can be reliably, non-invasively measured using B-mode ultrasound. The American Heart Association Prevention Conference V and National Cholesterol Education Program (NCEP) Adult Treatment Panel III concluded that CIMT can be used for IHD risk assessment. 5 The prevalence of abnormal CIMT rises with the number of vascular risk factors. 6 CIMT is strongly associated with the future risk for cardiovascular and cerebrovascular events, 7 independent of strong vascular risk factors. 8 A 0.10 mm increase in CIMT is associated with a 15% increased risk of myocardial infarction and 18% increased risk of stroke. 8 Carotid plaque reflects a different aspect of atherosclerosis which may have different determinants than CIMT, 9 but approaches to measurement and grading of plaque have been inconsistent. Little is known about subclinical atherosclerosis and CIMT measures of MS; findings have been mixed.10,11
We aimed to determine: (i) whether subclinical atherosclerosis is present in persons with MS who do not have traditional vascular risk factors; and (ii) whether a commonly used cardiovascular risk prediction tool, the Framingham risk score (FRS), predicted CIMT similarly in people with and without MS, to guide future work regarding the prevention and management of vascular disease in MS. We hypothesized that persons with MS without traditional vascular risk factors would have a higher frequency of subclinical atherosclerosis than persons without MS, and that the FRS would perform differently in people with and without MS.
Methods
Design
From April/2022 to July/2023, we conducted a cross-sectional comparative study in Manitoba, which has a population of approximately 1.4 million people.
Study populations
We recruited participants with and without MS. Participants with MS met the prevailing diagnostic criteria at the time of diagnosis. Participants without MS did not have immune-mediated disease. All participants were aged ≥18 years; provided informed consent; had sufficient knowledge of English to complete questionnaires; and could stand without assistance (required for the carotid ultrasound). No participant had been diagnosed with clinical atherosclerosis (IHD, stroke/transient ischemic attack or peripheral vascular disease). For each of the MS and non-MS groups, we sought two subgroups who had: (i) none of these traditional vascular risk factors: hypertension, diabetes, dyslipidemia, current smoking (so we could measure subclinical atherosclerosis in the absence of strong risk factors); (ii) ≥1 traditional risk factor (see E-methods for sample size calculation). We did not individually match the groups, but frequency matched by age.
Participants with MS were recruited from the Winnipeg MS Clinic, the sole clinic providing specialized MS care in Manitoba (see E-methods). Ethics approval was obtained from the the University of Manitoba Health Research Ethics Board.
Data collection
Each participant completed questionnaires either on paper or online, physical assessments, underwent a carotid ultrasound, and was provided a blood sample (see E-methods).
We captured gender, birthdate, ethnicity, and measures of SES and social status (highest education level attained, annual household income, and marital status). 12 Gender was reported as female, male, other (specify) and decline to answer. Ethnicity was captured using Statistics Canada's categories. Highest education level attained was collapsed into ≤ high school, and post-secondary education; <5 participants had less than a high school education. Annual household income was collapsed into <$50,000 (low income), ≥$50,000, and do not wish to answer. Marital status was summarized as single (single/never married/divorced/widowed/separated) and partnered (married/common-law).
Using a validated questionnaire, 13 participants reported whether a doctor had ever diagnosed them with comorbidities and current treatment status (yes/no). We summarized physical comorbidities as a count, excluding vascular comorbidities captured in the FRS. Participants reported a history of IHD or stroke among first-degree relatives. Depression and anxiety are associated with increased CIMT and atherosclerosis. 14 We used the Hospital Anxiety and Depression Scale (HADS), a questionnaire validated in MS, 15 to measure symptoms of depression and anxiety. The HADS uses 7 items each to assess current symptoms of anxiety (HADS-A) and depression (HADS-D); scores ≥ 11 indicate clinically meaningful symptom elevations.
Individuals who reported ever smoking ≥ 100 cigarettes were defined as ‘ever smokers’; current smoking status (yes/no) was also captured in keeping with definitions and questions from the Behavioral Risk Factor Surveillance System, 16 and US National Health Interview Survey. Even past low intensity smoking is associated with increased cardiovascular risk. 17 Alcohol intake was captured using the 3-item AUDIT-C, a valid measure for detecting past-year alcohol abuse or dependence, 18 the type of excessive alcohol intake that increases IHD risk. 19 Active alcohol abuse or dependence are indicated by scores ≥ 4 in men and ≥3 in women. Participants reported physical activity (PA) using the Godin Leisure Time Activity questionnaire, which is valid and reliable in the MS population. 20 Total scores <14 indicate insufficient PA, which increases IHD risk. 21
A measured blood pressure ≥ 135 systolic or ≥80 diastolic indicated hypertension. 22 BMI was derived from measured height and weight, where ≥25 and <30 were classified as overweight, and BMI ≥ 30 as obese; obesity is the threshold where IHD risk increases. Because BMI may underestimate adiposity in people with MS, 23 we measured waist and hip circumference. Waist circumference ≥101.6 cm in men and ≥88.9 cm in women indicates abdominal obesity.
To characterize physical and cognitive function, trained research assistants administered the Timed 25-foot walk test, Nine-Hole Peg test, and the Symbol Digit Modalities Test (SDMT) (see E-methods for interpretation). 24 The SDMT scores were converted to age, sex and education-adjusted z-scores using regression-based methods. 25 Participants with z-scores < −1.5 were classified as impaired. For participants with MS, we recorded age at symptom onset, clinical course, and disease-modifying therapy (DMT) use.
The day of the physical assessment we collected a non-fasting serum sample to measure lipid levels and hemoglobin A1c (HbA1c); this captured undiagnosed diabetes or dyslipidemia. A HbA1c ≥ 6.5% is diagnostic of diabetes. A low-density lipoprotein cholesterol (LDL-C) ≥ 5 mmol/L is an indication for treatment across all cardiovascular risk categories. We measured CRP as a measure of inflammation associated with increased vascular risk.
Carotid intima media thickness (CIMT), the thickness of the carotid artery wall between the innermost layer of the artery [intima] and the middle layer of the artery [media], was measured at the data collection site for the Canadian Longitudinal Study on Aging (CLSA), 26 according to their standard protocol (E-methods). CIMT measurements include average (based on six measurements), minimum and maximum measurements; and standard deviations (SD, in millimeters) for each carotid (right & left).
Analysis
We summarized participant characteristics using means (SD), median (interquartile range [IQR]) and frequencies (percent). We compared characteristics between groups using student's t-tests, Mann Whitney tests, chi-square tests and Fisher's exact tests. We compared average CIMT values between groups after age and sex adjustment using linear regression.
We calculated the FRS, which estimates the 10-year cardiovascular risk of an individual based on age, total cholesterol, high density lipoprotein cholesterol (HDL-C), current smoking history (smoking of any intensity), systolic blood pressure and hypertension treatment status (yes/no). 27 The scores differ for men and women.
We report the average CIMT for the right and left carotids, and the maximum CIMT because it may be a better index of atherosclerosis, since it will be elevated in the presence of atherosclerotic plaque. We determined the proportion of participants with a CIMT measurement ≥ 75th percentile based on published Canadian age and gender-stratified norms for average CIMT and maximum CIMT based on participants in the CLSA. 28 These norms were restricted to individuals aged 45–85 years; for participants were <45 years we assumed the norms for those aged 45–49 years. Although norms are available at younger ages for non-Canadian populations, these may not be generalizable. 29 Measurements ≥ 75th percentile without clinical evidence of atherosclerosis indicate subclinical atherosclerosis. We report the average CIMT, maximum CMIT and frequency of subclinical atherosclerosis stratified according to FRS-assigned risk: low (<10%), intermediate (10–19%) or high (>20%).
We used logistic regression analysis with Firth's procedure to test the association between MS status (yes/no), FRS and atherosclerosis (average CIMT ≥ 75th percentile vs not). Given the low number of participants with atherosclerosis, the covariates in the logistic regression model were limited to one at a time. Age and sex were not included as they were incorporated into the FRS and CIMT percentiles.
We used quantile regression analysis at the 50th quantile to test the association between FRS and average CIMT, reporting the pseudo-adjusted R2 for each regression model (see E-methods). To test if the association between MS status (yes/no) and FRS on CIMT outcomes differed, we included an interaction term between MS and FRS. Covariates included potential risk factors not captured in the FRS including abdominal obesity (no as reference), excess alcohol intake (no as reference), insufficient physical activity (no as reference), high school education (no as reference), household income (>$100,000 as reference), number of comorbidities (0 as reference), symptoms of depression and anxiety (continuous). Complementary analyses used the 20th and 80th quantiles.
In exploratory analyses, we repeated the regression analyses in MS cases after adjusting for MS-specific DMT (yes vs. no) and age of MS onset (continuous).
Statistical analyses were conducted using SAS V9.4 (SAS Institute Inc., Cary, NC).
Data availability
Data may be available to investigators with the appropriate ethical approvals and data use agreements.
Results
Participants
We enrolled 106 participants with MS and 101 participants without MS. Two additional individuals with MS were excluded after being consented because they could not stand independently long enough for the ultrasound. Overall, most participants were women, self-identified as white, and had a post-secondary education (Table 1). Compared to participants without MS, those with MS were more likely to be women, insufficiently physically active, with higher mean HADS-D scores HADS-D. Participants with MS had lower performance on the SDMT than participants without MS, and 18.9% of participants with MS had z-scores ≤1.5 indicating cognitive impairment. Ten participants with MS (9.4%) had an average timed 25-foot walk of 8 seconds or longer.
Participant characteristics.

CIS = clinically isolated syndrome, RRMS = relapsing remitting MS, SPMS = secondary progressive MS, PPMS = primary progressive MS, FRS = Framingham Risk Score, HADS = Hospital Anxiety and Depression Scale (D-Depression, A-Anxiety); MS = multiple sclerosis.
Missing: income 1 in controls, LDL 1 with MS, CRP 2 in controls.
Comparing entire MS vs. non-MS cohorts.
*Income $<50,000 vs. ≥$50,000.
**Refused to report income vs. ≥$50,000
Twenty-five participants in the MS (23.6%) and 31 in the non-MS (30.7%) groups had ≥1 traditional vascular risk factor (p = 0.25, Table 1). Among the 151 participants with no traditional risk factors, 79 (52.3%) had ≥1 non-traditional vascular risk factors, including obesity, abdominal obesity, insufficient physical activity, and excessive alcohol use.
CIMT
Neither the average nor maximum CIMT differed between the MS and non-MS groups, even when we limited the analysis to the participants with no traditional vascular risk factors, or with no vascular risk factors at all (Table 2), or after adjustment for age and sex (average CIMT MS: 0.61 (0.008), non-MS: 0.61 (0.008). The proportion of participants in each group with an average CIMT ≥ 75th percentile, indicating subclinical atherosclerosis, did not differ between the MS (11.3%) and non-MS (13.4%, p = 0.58) groups. Twenty-percent of participants had a maximum CIMT ≥ 75th percentile (MS: 25.5%, non-MS: 15.8%, p = 0.09).
Carotid intima media thickness (CIMT) stratified by cohort and presence or absence of any traditional vascular risk factors.

MS vs. non-MS cohorts:
All.
No traditional vascular risk factors.
No vascular risk factors.
FRS and CIMT
The FRS correlated strongly with the average (r = 0.74; 95%CI: 0.67, 0.80, Figure e1) and maximum CIMT (r = 0.66; 95%CI: 0.57, 0.73, Figure e2). When we stratified the sample by low, intermediate or high cardiovascular risk based on the FRS, one-quarter of those in the intermediate-risk group and one-third of those in the high-risk group had an average CIMT indicating atherosclerosis (≥75th percentile); this did not differ between those with and without MS (Table 3).
Carotid intima media thickness (CIMT) according to Framingham Risk Score predicted level of risk.

CMH = Cochran-Mantel-Haenszel test.
Association of disease and proportion with average CIMT ≥75th controlling for Framingham Risk Score.
P-value comparing MS vs. non-MS groups.
On logistic regression analysis, MS was not associated with atherosclerosis based on the average CIMT in any adjusted model. However, a 1-point higher FRS was associated with 1.11 (95%CI: 1.04, 1.19) increased odds of atherosclerosis, regardless of which covariate was included (Table 4); no covariates were associated with atherosclerosis. Based on the maximum CIMT, MS was associated with increased odds of atherosclerosis ranging from 2.17 to 2.60 but this should be interpreted cautiously given small numbers (Table 5).
Odds ratios (95% confidence intervals) for the association of multiple sclerosis, Framingham risk score with atherosclerosis (average carotid carotid intima media thickness ≥75th percentile).

Odds ratios (95% confidence intervals) for the association of multiple sclerosis, Framingham risk score with atherosclerosis (maximum carotid intima media thickness ≥75th percentile).
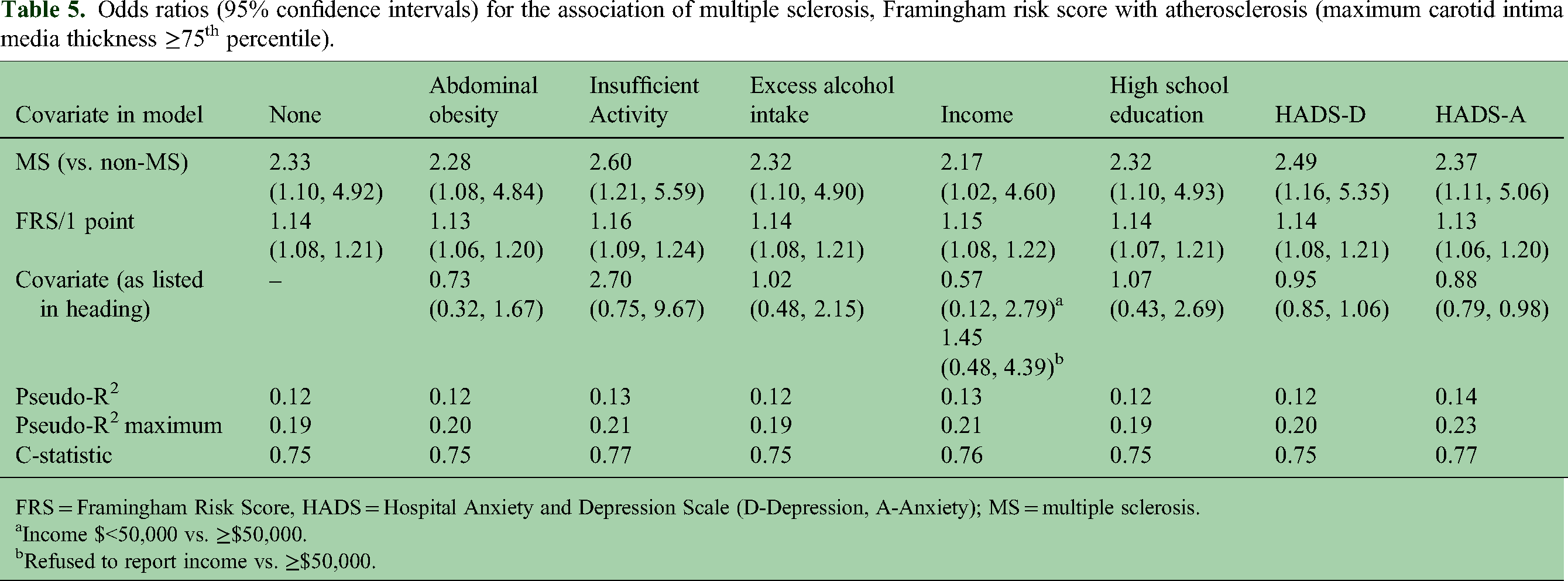
FRS = Framingham Risk Score, HADS = Hospital Anxiety and Depression Scale (D-Depression, A-Anxiety); MS = multiple sclerosis.
Income $<50,000 vs. ≥$50,000.
Refused to report income vs. ≥$50,000.
In unadjusted and adjusted quantile regression models, MS was not associated with average (Table e1) or maximum CIMT (Table e2). A one-point higher FRS was associated with a higher CIMT at the 20th, 50th and 80th quantiles of average CIMT; the association was non-linear at the 80th quantile. Covariates were not associated with CIMT. We did not observe any interaction between the disease group and FRS score on CIMT. When we repeated the analyses restricted to the MS cohort, the findings were similar (Table e3). Among those at the 20th quantile of FRS scores, older age of onset was associated with higher CIMT. DMT use was not associated with CIMT.
Discussion
In this cross-sectional study, we found that average CIMT did not differ between groups, nor did the proportion with subclinical atherosclerosis as defined by an average CIMT ≥ 75th percentile. Participants with MS were more likely to have atherosclerosis based on exceeding the 75th percentile of maximum CIMT, but this should be interpreted cautiously. Among participants with intermediate risk based on the FRS, one in four had elevated CIMT indicating a higher risk of atherosclerosis, raising the possibility that the FRS underestimates cardiovascular risk, although caution is warranted given the small sample. When we limited the sample to participants with no traditional or non-traditional vascular risk factors, approximately one in eight had subclinical atherosclerosis.
An increased prevalence of subclinical atherosclerosis, as measured using CIMT,3,30 has been reported in persons with other IMID, such as RA and psoriatic arthritis as compared to controls, even in the absence of vascular risk factors. 3 For example, among 53 participants with psoriatic arthritis, 30% had atherosclerosis based on the presence of carotid plaque, or CIMT >0.90 mm; the latter is a less stringent threshold than the one we employed and does not account for the strong association of age with CIMT. In contrast, 9.4% of controls had atherosclerosis. 30 Our findings differed. However, it is important to note limitations identified in a systematic review of the RA literature with respect to CIMT; sample sizes were generally small (n < 100), in many studies, participants with RA had a higher prevalence of vascular risk factors than control participants, and the studies with the largest sample sizes were less likely to report differences in CIMT. 31 We also did not measure carotid plaque. Moreover, inflammatory disease may increase the risk of vulnerable plaque at risk for rupture, and carotid ultrasound cannot discriminate between stable and unstable plaque. 32
Limited prior work has examined CIMT and atherosclerosis in people with MS, and the findings have been conflicting.10,11 In a small study comparing 35 people with MS and 34 controls, CIMT was 0.06 mm higher in those with MS; the effect was driven solely by differences in the right carotid. 11 Although participants with hypertension, diabetes, peripheral artery disease and other chronic diseases were excluded, the investigators did not account for other traditional and non-traditional risk factors. We carefully sought to identify a subgroup of participants who had no traditional or non-traditional risk factors which may account for the differences between our findings and that prior study. In a similarly-sized study comparing 33 people with MS and 33 controls matched on age, sex and BMI, the CIMT did not differ between groups. 10 Although this measure of carotid arterial structure did not differ between groups, functional arterial measures such as forearm blood flow and carotid arterial compliance were worse among those with MS. 10 Future studies should seek to further examine functional arterial measures.
The FRS may have underestimated the extent of subclinical atherosclerosis based on the CIMT. Although the FRS performs adequately at the population-level, it may mis-estimate cardiovascular risk at the individual level, overestimating risk in those who are low risk and underestimating risk in those who are high risk. 33 A recent study using CLSA data suggested that biologic age, operationalized as frailty, may predict cardiovascular risk better than chronologic age. 28 They found a positive curvilinear relationship between average CIMT and increased frailty. This may be valuable to explore in the MS population which is more likely to be frail than the non-MS population. 34
This study had strengths, including a larger sample size than prior studies, and evaluation of a spectrum of traditional and non-traditional vascular risk factors. We adopted the CIMT measurement protocol and team used by a large Canadian cohort study, providing highly standardized measurements, and Canadian normative values for those aged ≥ 45 years. Limitations also warrant consideration. The study was cross-sectional. We included participants as young as age 18 years because elevated CIMT has been observed in youth with inflammatory bowel disease compared to controls, 35 although rates of atherosclerosis are lower at younger ages in general and we lacked normative values for individuals aged <45 years which may have underestimated the degree of subclinical atherosclerosis in our cohorts. Because participants had to be able to stand independently to complete the carotid ultrasound, and the proportion of participants with progressive MS and severely impaired mobility was low, our findings may not generalize to those groups. The proportion of males was modest thus larger studies of males are needed. While this study was not designed or powered to evaluate the influence of disease-modifying therapy on subclinical atherosclerosis this warrants future attention.
In summary, the prevalence of subclinical atherosclerosis did not differ in people with and without MS regardless of vascular risk factors. Future larger, longitudinal studies are needed to clarify the performance of the FRS and to elucidate the factors that account for the increased risk of cardiovascular disease observed in the MS population.
Supplemental Material
sj-docx-1-mso-10.1177_20552173241238627 - Supplemental material for Subclinical atherosclerosis in multiple sclerosis
Supplemental material, sj-docx-1-mso-10.1177_20552173241238627 for Subclinical atherosclerosis in multiple sclerosis by Ruth Ann Marrie, Ronak Patel and Stephen Allan Schaffer in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
This study was supported by MS Canada (EGID #3476). Dr Marrie is supported by the Waugh Family Chair in Multiple Sclerosis. The authors acknowledge the use of Shared Health Facilities, and the use of the data collection site for the Canadian Longitudinal Study on Aging in Winnipeg.
Data availability
The datasets presented in this article are not readily available because some participants did not agree to data sharing. Components of the datasets may be made accessible to qualified investigators with the appropriate ethical approvals and data use agreements upon request. Requests to access the datasets should be directed to RAM, rmarrie@hsc.mb.ca
Declaration of conflicting interests
Ruth Ann Marrie receives research funding from CIHR, MS Canada, Crohn's and Colitis Canada, National Multiple Sclerosis Society, CMSC, the Arthritis Society and the US Department of Defense, and is a co-investigator on studies receiving funding from Biogen Idec and Roche Canada. She holds the Waugh Family Chair in Multiple Sclerosis.
R Patel receives research funding from MS Canada.
SA Schaffer receives research funding from MS Canada, Merck, Pfizer and Novo Nordisk.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by MS Canada (grant number EGID 3476).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
