Abstract
Background
Multiple sclerosis remains under-recognized in Zambia and much of sub-Saharan Africa, often leading to misdiagnosis and delays. Understanding diagnostic pathways and barriers can inform strategies to improve multiple sclerosis recognition and care.
Objectives
This study explores the diagnostic journeys of adults with multiple sclerosis in Zambia, emphasizing the challenges encountered at each stage.
Methods
We conducted a qualitative study using semi-structured interviews with adults diagnosed with multiple sclerosis in Zambia. Thematic analysis identified patterns in diagnostic experiences, enabling the mapping of common pathways and barriers, along with patient perspectives on the impact of diagnostic delays.
Results
Thirteen individuals with relapsing-remitting multiple sclerosis (mean age 34 ± 11 years; 70% female) were enrolled. Diagnostic pathways involved multiple healthcare visits, frequent misdiagnoses, and substantial delays. Key barriers included limited magnetic resonance imaging access, low multiple sclerosis awareness, and financial constraints. Thematic findings highlighted misinterpretation of symptoms, lack of specialists, and systemic health system limitations. Delayed diagnosis had a significantly negative impact on patients’ lives.
Conclusion
Multiple sclerosis diagnosis in Zambia is markedly delayed, adversely affecting patients. Greater awareness and policy reforms targeting diagnostic barriers are needed to promote earlier diagnosis and improve outcomes in Zambia and similar resource-limited settings.
Introduction
Multiple sclerosis (MS) is an important neurological cause of disability worldwide. 1 The global prevalence of MS has increased in the last decade, justifying continued investigation into the factors that drive the occurrence of the disease. 2 Understanding unique factors affecting the occurrence of MS in different populations is important in understanding the epidemiology of MS as a whole. However, despite recent work that has highlighted that MS does occur among people in Africa, 3 sub-Saharan Africa (SSA) remains relatively underrepresented in data on MS occurrence. 4
Zambia is a land-locked country in south-central Africa with a gross domestic product (GDP) per capita of USD 1456 5 and a low doctor-to-patient ratio of 1–12,000. 6 Most doctors in the country practice at secondary or tertiary level hospitals, with primary level healthcare largely served by physician assistants (known as clinical officers) and nurses. Public health facilities run by the country's Ministry of Health dominate health care delivery, accounting for 93% of all health facilities nationally. Private facilities represent only 7% of all facilities, and these are heavily concentrated in urban areas. 7 Many rural health facilities have no specialist doctors, and most surgical and complex medical cases have to be referred to secondary or tertiary hospitals in urban cities. This is challenging given that most rural health facilities are in hard-to-reach areas with a poor road network and rely on public funds to transport patients; funds which are not always sufficient for national distribution and coverage. The country only has 13 MRI machines serving a population of ∼22 million. Diagnostic tests are significantly limited and often paid for out-of-pocket by the patient8,9 and disruptions in stocks of medicines are frequent. 10 The national health insurance scheme, established in 2017, is available to all Zambians, but only 30% of the population (mostly those in formal employment) is registered, and medicines and diagnostics covered by the scheme are often limited. 11 The main priorities of the Ministry of Health include HIV, malaria, tuberculosis, and maternal and child health. 12
Before 2018, there were no Zambian neurologists practicing in Zambia, and the entirety of specialized neurologic care was a single half-day outpatient clinic once weekly at the University Teaching Hospital (UTH) in the capital city of Lusaka, staffed by several expatriate neurologists. Zambia's first-ever neurology training program was launched in 2018, alongside the establishment of a dedicated neurology inpatient service at UTH in Lusaka. 13 Through this neurology service, MS has consequently been diagnosed among Zambians for the first time.
Timely and accurate diagnosis of MS is essential for initiating appropriate treatment and management, which can significantly improve patient outcomes and prevent accrual of disability. 14 However, diagnosing MS in resource-limited settings like Zambia poses unique challenges. Therefore, this study was conducted to describe the diagnostic pathway of people with MS in Zambia in order to provide insights into challenges and barriers to the diagnosis of MS in Zambia and inform future strategies to improve the diagnostic process for MS in Zambia.
Materials and methods
Study design
This was a qualitative sub-study of the Zambia Multiple Sclerosis Study (ZAMSS). The ZAMSS study was a prospective cohort study of all adults diagnosed with MS at the Neurology Clinic of the University Teaching Hospital (UTH) in Lusaka, Zambia. ZAMSS examined the epidemiology, clinical characteristics, and outcomes of adults diagnosed with MS in Zambia.
Study population
The study population comprised of adults evaluated at the UTH Neurology Clinic with a confirmed diagnosis of MS between October 2018 and December 2022. MS diagnoses were made using the 2017 McDonald criteria, and all diagnoses were confirmed with MRI studies. Inclusion criteria for this qualitative sub-study were that participants must have had their MS diagnosis made or confirmed by the neurology unit at UTH in Lusaka, be Zambian residents, and consent to a recorded semi-structured interview.
Sampling and recruitment
All eligible participants in the ZAMSS cohort were invited to participate in in-depth interviews. This approach ensured the inclusion of a diverse range of experiences and perspectives related to the diagnosis of MS. Recruitment was carried out from the ZAMSS cohort database, and potential participants were contacted via telephone or in-person meetings to explain this sub-study's objectives and obtain informed consent.
Data collection
Semi-structured interviews were conducted between December 2021 and December 2022. Consecutive participants were recruited from ZAMSS from December 2021 to December 2022. An interview guide (Supplemental Material) was developed based on the authors’ clinical expertise in diagnosing MS in Zambia. The guide included open-ended questions designed to elicit detailed narratives about participants’ experiences from the onset of symptoms to the eventual diagnosis of MS. These questions were focused on participants’ initial MS symptoms and their disease progression, clinical encounters with healthcare providers, diagnostic procedures obtained during evaluations, diagnoses obtained during evaluations, types of providers seen, and duration between MS symptom onset and MS diagnosis. We also asked about the impact of delayed diagnoses on participants.
Interviews were conducted by a neurologist (MC) who was not the participants’ primary treating neurologist. Interviews were conducted in the participants’ preferred language of English, Nyanja, or Bemba, lasted ∼45–60 minutes, and were audio-recorded with participants’ permission. Participants were compensated for their time and data costs (∼$4 USD) incurred during the interviews.
Data analysis
Audio recordings were transcribed verbatim, and transcripts were used for data management and analysis. A reflexive thematic analysis approach was used to identify and interpret patterns and themes within the data. This analysis followed a standard qualitative research approach, which included reading transcripts, coding, and identification of themes. 15 Based on the themes emerging from the interviews, barriers to MS diagnosis in Zambia were then identified.
To systematically describe the pathways to diagnosis, we adapted methodologies from previously published studies on diagnostic pathways in neurological and other chronic diseases based on the model of pathways to treatment (MPT) by Scott et al.16–18 The diagnostic journey was categorized into the following stages:
Symptom onset—The initial manifestation of symptoms, including early patient experiences and symptom recognition. Seeking medical attention—The process of deciding to seek care, including initial healthcare encounters (traditional healers, general practitioners, or hospitals). Referral and specialist evaluation—Movement through the healthcare system, including referrals to higher-level facilities and specialist consultations. Diagnostic testing—Access to and completion of necessary investigations (e.g. MRI, lumbar puncture, and laboratory tests). Diagnosis communication and treatment initiation—The final confirmation of MS diagnosis, patient acceptance, and initiation of disease management.
For this study, traditional healers were defined as any individual who offered the respondents herbal or spiritual therapies that were based on Zambian cultural practices outside the established Zambian Western-style healthcare systems.
Transcribed interviews were coded first by applying Scott's model of pathways to treatment and then using an inductive thematic approach to understand the impact of diagnostic delay on participants. We identified recurring themes related to delays, misdiagnoses, and healthcare barriers at each step. Data were analyzed independently by two researchers (MC and DS) to ensure consistency, with discrepancies resolved through discussion.
In addition to qualitative thematic analysis, we performed a descriptive quantitative analysis to quantify key diagnostic indicators, including time from symptom onset to first healthcare visit (median and range), time from first healthcare visit to confirmed MS diagnosis, and proportion of patients experiencing delays at different pathway stages. These findings were summarized using frequency distributions, medians, and interquartile ranges.
Ethical considerations
The study received ethical approval from the ERES Converge Institutional Review Board, the Zambia National Health Research Authority (NHRA), and the Johns Hopkins Institutional Review Board. Confidentiality was maintained by anonymizing transcripts and securely storing all data. Participants were assured that their decision to participate or not would not affect their access to care. All participants gave written informed consent for study participation.
Results
A total of 13 (81%) of 16 potentially eligible participants consented to be interviewed for this study. Three potential participants could not be reached by phone for consent at the time of this study, despite multiple efforts made throughout the study period. The demographic and clinical characteristics of the 13 participants are summarized in Table 1. Nine participants (70%) were women, and the mean age of all participants was 34 ± 11 years. All participants had a Relapsing Remitting type of MS. All but three participants were receiving rituximab for their treatment, and five were also receiving physiotherapy at the time of the interviews.
Demographic and clinical encounter characteristics of participants.

MS: multiple sclerosis; MRI: magnetic resonance imaging; IQR: interquartile range; UTH: University Teaching Hospital.
The median time between onset of MS symptoms and diagnosis was 14 months (interquartile range [IQR] 6–36 months), with the shortest being 3 months and the longest being 14 years. Multiple clinical encounters prior to MS diagnosis were reported, and these were at first-level public/government health facilities (47%), private non-specialized facilities (41%), and/or specialized hospitals outside Zambia (38%). Four participants also had at least one clinical encounter at UTH, which did not result in an MS diagnosis. The mean number of physicians/clinicians seen before MS diagnosis was 4 ± 3, with a range of 2–8. Three participants (23%) reported seeking a traditional healer and/or receiving traditional herbal medication at least once before their MS diagnosis. Eight participants (65%) completed at least one MRI scan that did not lead to a diagnosis of MS, most commonly because only an MRI of the lumbar spine was obtained.
Figure 1 shows typical examples of an individual diagnostic pathway for two participants, and Figure 2 shows the composite pathway to diagnosis for all participants interviewed. All participants had multiple clinical encounters, which led to multiple misdiagnoses prior to receiving their MS diagnosis. Types of physicians seen included general practitioners, orthopedic surgeons, neurosurgeons, internists, ophthalmologists, and neurologists. During these visits, the misdiagnoses included stroke, type 2 diabetes, orthopedic diseases, anemia, brain tumor, and drug abuse. Eight of the 13 participants had at least one MRI of the brain and/or spine that were also misdiagnosed as brain tumors or stroke.
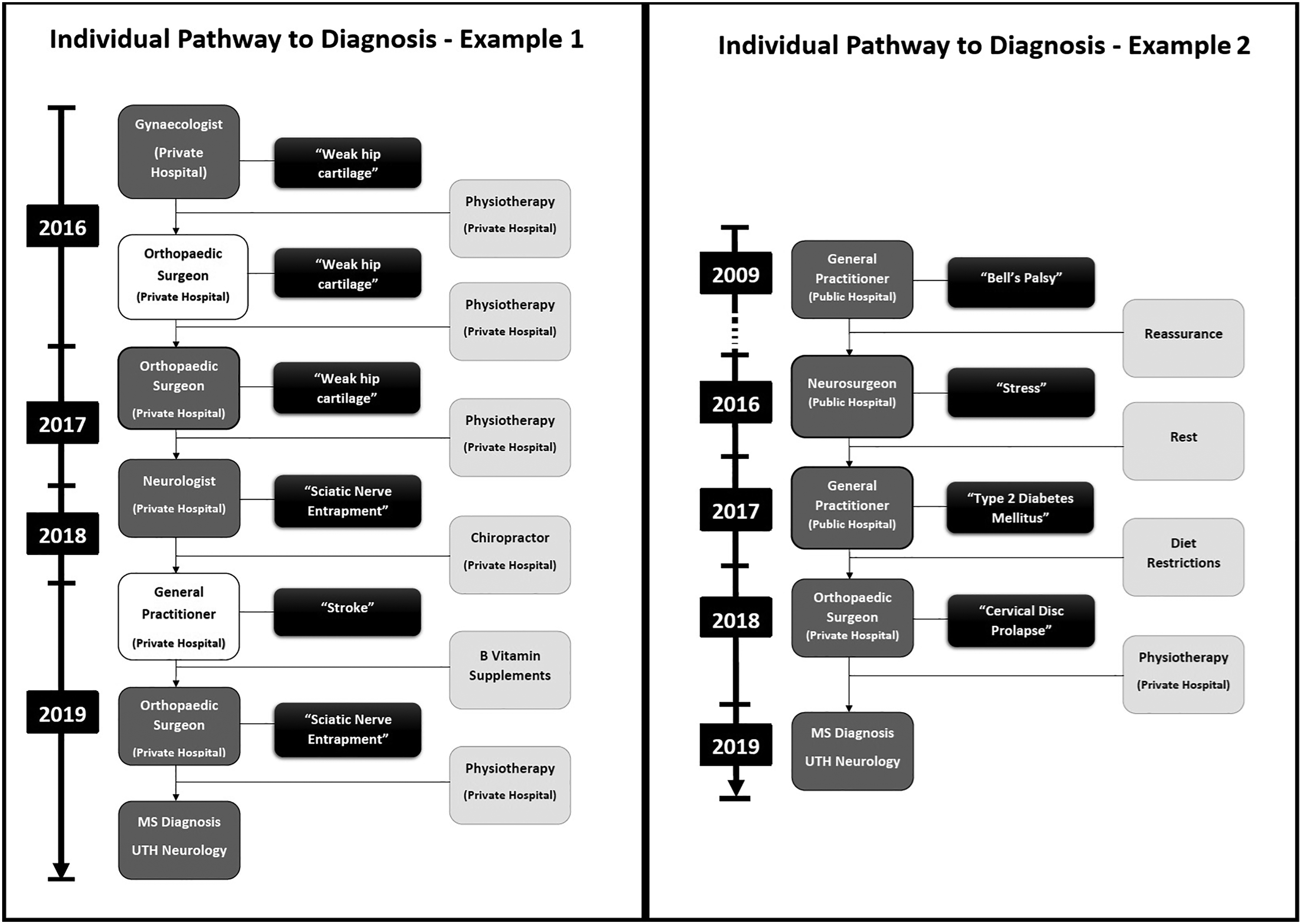
Examples of individual pathways to multiple sclerosis (MS) diagnosis in Zambia.

Composite pathway to multiple sclerosis (MS) diagnosis in Zambia.
Following the misdiagnoses, all participants reported having to self-refer to other hospitals outside the country or to UTH. Five participants self-referred to hospitals outside Zambia, and three (23%) received their MS diagnosis at those hospitals (two in India and one in the USA). Of note, these referrals all occurred prior to the opening of Zambia's first neurology unit in 2018. Regardless, two (15%) participants who sought evaluation outside of the country were not diagnosed with MS until they returned to Zambia and were eventually evaluated at the UTH neurology clinic. Therefore, ten (77%) participants, including the two participants who were misdiagnosed outside Zambia, self-referred to the UTH neurology clinic, where they were diagnosed with MS.
Existing barriers to timely and accurate MS diagnosis were identified based on participants’ diagnostic journeys and impacted each step along the diagnostic pathway (Figure 3). Barriers included the need for multiple clinical consultations that did not lead to an MS diagnosis and MRI scans that did not lead to an MS diagnosis, either due to incorrect interpretation of the scans or inappropriate diagnostic tests being ordered (e.g., lumbar spine MRI scans were the most common type of MRI ordered that did not lead to a diagnosis). Lack of trained radiologists and an absence of neuroradiologists also contributed to improper interpretations of scans. Another barrier to diagnosis was the cost of clinical encounters, with all patients paying for fees and MRI scans out of pocket. These health system and financial barriers led to some patients seeking traditional medicine, which further delayed their MS diagnosis.

Barriers to diagnosis of multiple sclerosis mapped onto a theoretical pathway to diagnosis.
The delayed diagnosis of MS impacted patients in many ways. These included significant disability at time of diagnosis; depression and frustration from living with undiagnosed symptoms; loss of employment or income resulting from inability to work due to significant disability; incurring of large costs due to multiple clinical encounters both locally and internationally, often requiring medical tests that were paid for out-of-pocket; loss of confidence in services provided by medical health workers in Zambia, even after MS diagnosis; lack of clarity in understanding MS disease severity and the role of neurologists post-diagnosis, often leading to poor compliance to treatment; and significant strain on patients’ families and loved ones (Figure 4).

Impact of delayed diagnosis on people with multiple sclerosis (MS) in Zambia.
Discussion
This study explored diagnostic pathways, common barriers to diagnosis, and the impact of delayed diagnosis on Zambian adults living with MS. The findings highlight significant delays in diagnosis, with a median time of 14 months from symptom onset to diagnosis and one patient waiting more than a decade for a diagnosis. Participants often consulted multiple physicians and visited a variety of health facilities before receiving a correct diagnosis. The limited access to and high cost of MRI, and the frequent use of traditional healers, contributed to these delays. Furthermore, these delays ultimately led to long-term consequences for people with MS even outside of the accrual of potentially preventable disability, including loss of confidence in healthcare providers, financial harm, depression, isolation, and family stress.
The results of this study bring attention to the under-recognition of MS in SSA, where MS has been thought to be largely non-existent for many decades.19–21 However, SSA has one of the lowest number of neurologists per capita at 0.03 neurologists per 100,000,22–24 and access to MRI is limited,25,26 resulting in diagnostic challenges. Therefore, rather than an absence of MS, there is a paucity of data on MS from SSA and, likely, significant under-diagnosis in this region. Studies such as this one show high rates of misdiagnosis and a need for improved recognition at many levels of the healthcare system.
Significant delays in MS diagnosis, as shown in this study, have also been shown in other cohort studies from SSA. In Kenya, a cohort of people living with MS showed a mean delay of 4 years between the onset of symptoms and diagnosis. 27 Although the study did not specifically investigate factors leading to this delay, it was observed that access to MRI was delayed by up to 5 years in some participants. 27 The poor distribution of MRI has been shown to be a significant factor affecting recognition of MS in low-resource regions. 28 Coupled with the high cost for those with access, this becomes a major barrier to diagnosis of MS in SSA, which makes our finding that lack of skilled interpretation of MRIs and appropriate investigations (i.e. MRI lumbar spine) resulted in missed opportunities for diagnosis even in those who were able to access MRI studies significant.
Lack of awareness among healthcare workers about MS and its presentations is also common in SSA. Neurology specialist training is available in only a few countries in SSA, 29 and there is limited exposure to clinical or academic neurology for non-neurologist doctors. 30 Therefore, recognition of MS symptomatology is very low, and underdiagnosis of MS is widespread in SSA, as evidenced by our findings. Lack of awareness of MS symptoms among healthcare professionals and lack of neurologists have both been shown to significantly hinder MS diagnosis. 31
Lumbar puncture completion rates are low in Zambia owing to multiple factors, including patient/family preferences, provider factors, and health systems factors. 32 Therefore, patients who require the procedure don’t often receive it on time. 33 Moreover, cerebrospinal fluid and serum oligoclonal band (OCB) testing and evoked potentials are currently not available in Zambia. Although it was not clear if any of our respondents were offered a lumbar puncture prior to diagnosis, these existing challenges for CSF collection and lack of OCB testing are likely another barrier to timely MS diagnosis in Zambia.
Given that clinical outcomes among people living with MS improve with early recognition and treatment, 34 high levels of preventable disability are likely common among people with MS in Zambia and SSA. Diagnostic delays negatively impact MS outcomes around the world and have been shown to also occur in high-income countries. Therefore, investigating barriers to MS diagnosis in all settings is essential in order to improve outcomes globally. The recommended time between onset of symptoms and diagnosis is three months, with some studies recommending an MRI within a week of symptom onset.35,36 In the DIMES study conducted in the United Kingdom and Ireland, the mean time from onset of symptoms to diagnosis was 10 months, 37 and Uher et al. 36 reported a median time to diagnosis of 4 months in their study in the Czech Republic. However, the findings of our study showed a much longer diagnostic delay of 14 months. Furthermore, the factors contributing to the diagnostic delays in these European studies are due to misdiagnoses of MS, reportedly due to MS mimics rather than a lack of awareness of MS among clinicians, as was the case in our study. Therefore, there is a need to raise awareness among health workers and policy makers about people with MS in regions like SSA and to innovate ways to improve diagnosis in these regions. This is especially important because our study also demonstrated that delayed diagnosis of MS led to significantly reduced quality of life, mistrust of the healthcare system, and skepticism about their diagnosis among people with MS in Zambia.
Despite the importance of the findings, this study had limitations. The small sample size of 13 people may make generalizability of our findings challenging for populations within the region and globally. However, at the time of the interviews, the 13 individuals represented more than 80% of all confirmed MS cases in Zambia and, therefore, were representative of typical experiences among people with MS in Zambia. It is also worth noting that the 13 individuals interviewed likely represented only a small fraction of the true number of people with MS in Zambia, as it is likely there are many more living with undiagnosed MS. Our participants also had higher education levels and incomes than the average Zambian, suggesting that they likely had higher levels of health literacy and financial resources in order to pursue a definitive diagnosis. Given that all participants interviewed also resided in urban Zambia, there are likely even greater diagnostic challenges among Zambians living with undiagnosed MS who have less education, fewer financial resources, and reside in rural Zambia. The second limitation was the possible recall bias resulting from the interviews that relied on self-reported data; electronic medical records are not available in Zambia, so we could not verify the accuracy of these self-reported data. However, this study is one of the first of its kind in SSA to systematically assess diagnostic pathways, barriers to diagnosis, and the impact of delayed diagnosis among people with MS in this region.
Future prospective studies with larger sample sizes made up of participants from all socioeconomic settings in Zambia and across SSA are recommended. Tracking patients from symptom onset through to the diagnostic process could provide more comprehensive data on delays and barriers. Additionally, interventions designed to address the identified barriers should be developed, implemented, and evaluated for their effectiveness in improving MS diagnosis and care in Zambia.
Supplemental Material
sj-docx-1-mso-10.1177_20552173261417446 - Supplemental material for Diagnostic pathways and barriers in multiple sclerosis in Zambia: A qualitative study
Supplemental material, sj-docx-1-mso-10.1177_20552173261417446 for Diagnostic pathways and barriers in multiple sclerosis in Zambia: A qualitative study by Mashina Chomba, Dominique Mortel, Sarah Braun, Malya Sahu and Deanna Saylor in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
We extend our sincere gratitude to the individuals living with MS in Zambia who generously shared their experiences and insights for this study. We also acknowledge the dedicated staff of the UTH Neurology Unit and the ZAMMS team for their invaluable support. Special thanks to the translators, transcribers, and data collectors who contributed to the qualitative work. This research would not have been possible without their commitment and collaboration.
Data availability statement
The data supporting the findings of this study consist of anonymized qualitative interview transcripts and are not publicly available due to the sensitive nature of the content and the need to protect participant confidentiality. However, de-identified excerpts relevant to the study's findings may be made available from the corresponding author upon reasonable request and with approval from the ethical review board.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Multiple Sclerosis International Foundation Du Pre Award; National Multiple Sclerosis Society Pilot Grant, National Institutes of Health Fogarty International Center (K01 TW011771-01), National Institutes of Health Fogarty International Center (D43 TW009340).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
