Abstract
Background
Neuromyelitis optica spectrum disorder (NMOSD) is an inflammatory autoimmune disorder that damages optic nerves, brainstem, and spinal cord. In vivo corneal confocal microscopy (IVCM) is a noninvasive technique that provides corneal images with dendritic cells (DCs) and corneal subbasal nerve plexus (SBP), which arises from the trigeminal nerve.
Objective
We investigated corneal SBP changes in NMOSD and proposed IVCM as a potential new disease severity biomarker for NMOSD.
Methods
Seventeen age-sex matched NMOSD patients and 19 healthy participants underwent complete neurologic and ophthalmologic examinations. The duration of disease, first symptom, presence of optic neuritis attack, antibody status, Expanded Disability Status Scale(EDSS) score and disease severity score(DSS) were recorded. Retinal nerve fibre layer (RNFL) thickness was measured with optical coherence tomography, and corneal SBP images were taken with IVCM.
Results
NMOSD patients had significantly reduced corneal nerve fibre lenght-density and corneal nerve branch lenght-density compared with controls, while DC density was increased. NMOSD patients also showed significantly reduced RNFL thickness compared with controls. EDSS,DSS levels were inversely correlated with IVCM parameters.
Conclusion
We observed significant corneal nerve fibre loss in NMOSD patients in relation to disease severity. IVCM can be a candidate noninvasive imaging method for axonal damage assessment in NMOSD that warrants further investigation.
Keywords
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is an inflammatory autoimmune disorder of the central nervous system (CNS) that mainly damages the optic nerves, brainstem, and spinal cord. Astrogliopathy, inflammation, and demyelination in the later stage have the main role in the disease course associated with axonal loss and perivascular lymphocytic, eosinophilic and neutrophilic infiltration. Hallmarks of NMOSD include acute attacks of severe visual loss, limb weakness, sensory loss or bladder dysfunction with a typically relapsing course.1,2 Approximately half of NMOSD patients present with isolated optic neuritis (ON), which causes a severe loss of visual acuity (<0.1) during an acute attack in approximately 80% of patients and leads to reduced visual quality.3,4 Before aquaporin-4 (AQP4) antibody detection for diagnosis, the NMOSD course was described as a highly disabling disorder.3,5 Patients with suspected NMOSD are evaluated with brain and spinal cord magnetic resonance imaging (MRI), serum AQP4 and myelin oligodendrocyte glycoprotein (MOG) antibody status, and additional cerebrospinal fluid analysis. 6
Axonal degeneration during the disease course plays the main role in disability accumulation in NMOSD patients. Early detection of degeneration prevents further deterioration through treatment and management planning. The major challenge is to monitor progressive axonal degeneration in NMOSD. Currently, the mainstay of investigations evaluating disease progression is MRI and the Expanded Disability Status Scale (EDSS). Recent studies have focused on new imaging techniques for the assessment of axonal damage in neurological diseases such as multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), diabetic neuropathy, Fabry disease, and Parkinson’s disease.7–9 According to those studies, in vivo corneal confocal microscopy (IVCM) could be used as an indirect method to show axonal degeneration in the periphery.7,8
IVCM is a noninvasive imaging technique that provides high-resolution corneal images at the cellular level by visualizing corneal nerve fibres and dendritic cells (DCs). 10 Corneal nerve fibre density has been proposed as a new parameter to assess axonal damage by detecting the corneal subbasal nerve plexus (SBP), which is derived from the axons of the trigeminal nerve. According to recent data, the analysis of DCs by IVCM could be a marker to show local corneal inflammation. 11 Thus, IVCM is a unique imaging technique for analysing both the peripheral nervous system and the immune system. There is no published study evaluating axonal and DC changes in NMOSD using IVCM.
The primary aim of our study was to assess morphological changes in corneal SBP and differences in corneal DC density as well as changes in the retinal nerve fibre layer (RNFL) in patients with NMOSD using IVCM and optical coherence tomography (OCT). 10 An additional objective was to investigate the association between those changes and the clinical severity of the disease.
Methods
Patients and controls
In this cross-sectional study, seventeen patients diagnosed with NMOSD according to criteria defined by the 2015 International Panel of Neuromyelitis Optica and nineteen age- and sex-matched healthy controls were included. 6 Both the eyes of patients and healthy controls were treated as independent groups and were included separately. All procedures in the study adhered to the tenets of the Declaration of Helsinki. Regular clinical and radiological follow-ups of eleven NMOSD patients were performed at Koç University Hospital, Neurology Department, Turkey. 6 The remaining six patients were included in this study through collaboration with Charité–Universitätsmedizin Berlin, Berlin, Germany. The study was approved by the local ethics committees of Koç University and Charité–Universitätsmedizin Berlin. Written informed consent was obtained from all subjects beforehand. A detailed history of disease was obtained, including duration of the disease symptoms, duration of the diagnosis, first symptom type, existence of any optic neuritis attack, AQP4 and MOG antibody status, total number of attacks, date of last attack, Expanded Disability Status Scale (EDSS) level in the 2nd and 5th years after diagnosis, current EDSS level, and DSS, which relates disability level (EDSS) to disease duration (year). 12 NMOSD patients were subgrouped according to their EDSS levels according to the 30-year longitudinal study of Chung et al 13 (Subgroup 1: EDSS level 3.5 and below; Subgroup 2: EDSS level above 3.5).
The exclusion criteria were a history of any other ocular surgery, glaucoma, cornea or ocular surface disorders, contact lens usage, neurologic disorders, diabetes mellitus and other autoimmune diseases. Healthy controls were recruited from among volunteers. Complete ophthalmological evaluations included visual acuity testing, slit-lamp biomicroscopic examination, tonometry (Canon TX-20P, Japan), autorefractometry (Topcon KR-800, Japan), OCT and IVCM.
Optical coherence tomography (OCT)
Fundoscopic examination was performed, and patients were examined under spectral-domain OCT (Spectralis, Heidelberg Engineering GmbH, Heidelberg, Germany) by a single masked trained examiner at each institution. Peripapillary RNFL thickness (pRNFL) was measured with a circular scan around the optic nerve head using the standard protocol with a scanning angle of 12°, resulting in a 3.4-mm retinal diameter, optimal centring, and a minimum signal strength of 20 per default settings. All examinations were checked for sufficient quality using the OSCAR-IB criteria. 14
In vivo corneal confocal microscopy (IVCM)
All subjects underwent IVCM (Heidelberg Retinal Tomography 3 Rostock Cornea Module, Heidelberg Engineering GmbH, Heidelberg, Germany) using section mode to scan the full thickness of the central cornea. IVCM was performed to quantify corneal nerve fibre length (CNFL), density (CNFD), corneal nerve branch length (CNBL), density (CNBD), and DC density. Before the examination, two drops of local anaesthetic (proparacaine hydrochloride 0.5% or oxybuprocaine hydrochloride 4 mg/ml) and a lubricant gel (carbomer-containing gel 2 mg/g) were applied to both eyes. Both centres followed a similar protocol for confocal imaging of the corneal apex with a single experienced examiner in each facility who was masked to each subject’s disability status. The duration of the examination was approximately 5 minutes for each eye. The IVCM images that were collected in Charité–Universitätsmedizin Berlin were sent to Koç University Hospital in .tif format. An experienced masked ophthalmologist (AS) from Koç University Hospital selected the three best-quality nonoverlapping images per eye and analysed 216 images in total using NeuronJ, a semiautomated ImageJ software plug-in (http://imagej.nih.gov/ij/). Due to the presence of relationship between inflammation and DCs maturation, only mature DCs were evaluated, and their number was counted using ImageJ’s Cell Counter plug-in. Then, DC density was calculated as cells/mm2. All visible nerve fibres were traced, while their length was measured as mm/mm2, and their density was calculated as fibres/mm2. Four measures were quantified: (1) CNFL, the total length of major nerves in millimetres per square millimetre; (2) CNFD, the total number of major nerves per square millimetre; (3) CNBL, the length of branches emanating from major nerve trunks; and (4) CNBD, the number of branches per square millimetre.
Statistical analysis
Statistical analyses were performed using SPSS Statistics software (Version 20.0, IBM, Chicago, USA). To assess the normality of the distribution of the data, the Shapiro-Wilk test was used. Normally distributed quantitative data were analysed using Student’s t test and other data were analysed with the Mann-Whitney U test. Demographic features of the NMOSD and control groups were analysed using a nonparametric Mann-Whitney U test for age and Fisher’s exact test for sex. An unpaired t test was used for comparison between the NMOSD and healthy groups. For the correlation analysis between clinical parameters, IVCM and RNFL values, Spearman's correlation coefficient correlation (r) was used. Additionally, receiver operating characteristic (ROC) curves were calculated to compare the diagnostic abilities of RNFL and IVCM measurements. A two-sided p-value lower than 0.05 was considered statistically significant. A Mann-Whitney U test was performed to compare the medians of two subgroups according to recent EDSS levels. In the NMOSD group, eyes with and without ON were investigated and compared with the control group regarding RNFL and IVCM values using the Mann-Whitney U test.
Results
Demographic and clinical status
The mean ages were 47.00 ± 13.93 years for the 17 NMOSD patients and 48.47 ± 18.65 years for the 19 healthy participants. The mean Snellen visual acuities (VAs) were 0.68 ± 0.37 for the NMOSD patients and 1.0 for all healthy subjects. Patients with NMOSD had a mean disease duration of 12.29 ± 7.51 years since their first symptom. The mean duration since diagnosis was 6.45 ± 4.80 years. The patients had a median total number of attacks of 4.00 (2.5–6.5), while the mean duration (± standard error [SE]) since their last attack was 5.60 ± 1.06 years. Forty-seven percent of the patients presented with transverse myelitis (TM) as their onset symptom, 41% presented with ON, and 12% presented with both ON and TM. Seventy-one percent of the patients had at least one previous episode of ON during the disease course. Eighty-two percent of the patients had positive AQP4 serum antibody, and 13% had positive serum MOG antibody. The median EDSS scores at the 2nd year, 5th year and last visit were 1.0 (0.0–3.5), 3.0 (1.12–4.75) and 4.75 (2.0–5.12), respectively. Three NMOSD patients were followed up without any medication, and the rest were receiving immunosuppressive treatment (eight patients were taking rituximab, while five were taking azathioprine, and one was taking mycophenolate mofetil) (Table 1).
Demographic and clinical features of control subjects and NMOSD patients.

NMOSD: neuromyelitis optica spectrum disorders; AQP4: aquaporin 4; MOG: myelin oligodendrocyte glycoprotein; EDSS: Expanded Disability Status Scale; SD: standard deviation; IQR: interquartile range.
Optical coherence tomography (OCT)
In the NMOSD patient group, there were significant reductions in superior, inferior, temporal, nasal and global RNFL thickness compared with the controls (Table 2). Furthermore, in the NMOSD patient group, the eyes that experienced ON attack/s presented with significantly lower values of superior, temporal, nasal and global thickness compared with the eyes without ON attack/s (Table 3). There was no difference in the pRNFL between NMOSD eyes without ON attacks and controls.
Optical coherence tomography results for patients with NMOSD and control subjects.

NMOSD: neuromyelitis optica spectrum disorders; RNFL: retinal nerve fiber layer; SD: standard deviation.
Optical coherence tomography results for NMOSD patient eyes with and without ON attacks.

NMOSD: neuromyelitis optica spectrum disorders; ON: optic neuritis; RNFL: retinal nerve fiber layer; IQR: interquartile range.
In vivo corneal confocal microscopy (IVCM)
NMOSD patients had significantly reduced CNFL, CNFD, CNBL and CNBD compared with controls. The DC density was increased significantly in NMOSD patients compared with healthy controls (Table 4 and Figure 1). When the diagnostic ability of CNBL was compared with CNFL and global RNFL values via ROC curve analysis, CNBL showed significantly higher sensitivity and specificity (AUC = 0.8940, P < 0.001) (Figure 2). The eyes of NMOSD patients who had not experienced ON attacks also had significantly reduced CNFL, CNFD, CNBL and CNBD compared with healthy controls. The DC density was increased significantly in NMOSD eyes without ON attacks compared with control eyes (Table 5). Furthermore, the OCT and IVCM values of NMOSD patients were analysed to identify the correlations between optic and trigeminal nerve changes. Analysis identified significant negative correlations between DC density and superior (r = −0.408, P = 0.028), nasal (r = −0.533, P = 0.003), temporal (r = −0.405, P = 0.029) and global (r = −0.580, P = 0.001) RNFL values. Other than DC density, the IVCM parameters were not correlated with RNFL values for NMOSD patients. Furthermore, in the NMOSD group, the IVCM values of the eyes that experienced ON attack/s were not significantly different compared with the eyes without ON attack/s. Last, there were no differences in either IVCM or pRNFL parameters between the eyes of NMOSD patients without ON attacks and the fellow eyes of patients with unilateral ON attacks (Supplementary Table S1).
In vivo corneal confocal microscopy results in NMOSD patients and control subjects.

CNFL: corneal nerve fiber length; CNBL: corneal nerve branch length; CNFD: corneal nerve fiber density; CNBD: corneal nerve branch density; DC: dendritic cell; SD: standard deviation.
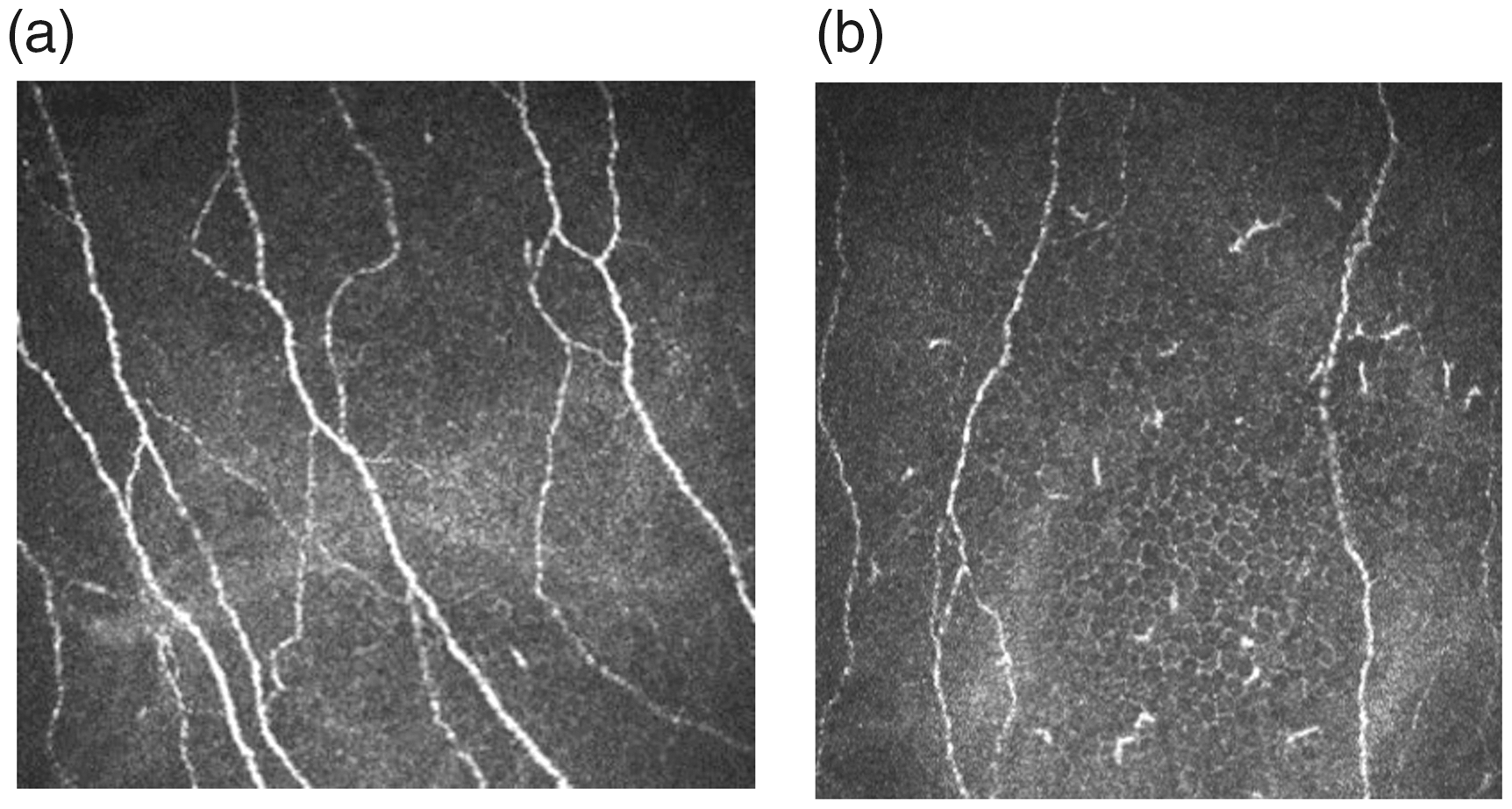
Representative in vivo corneal confocal microscopic images of a healthy control (a); and a patient with NMOSD (b).
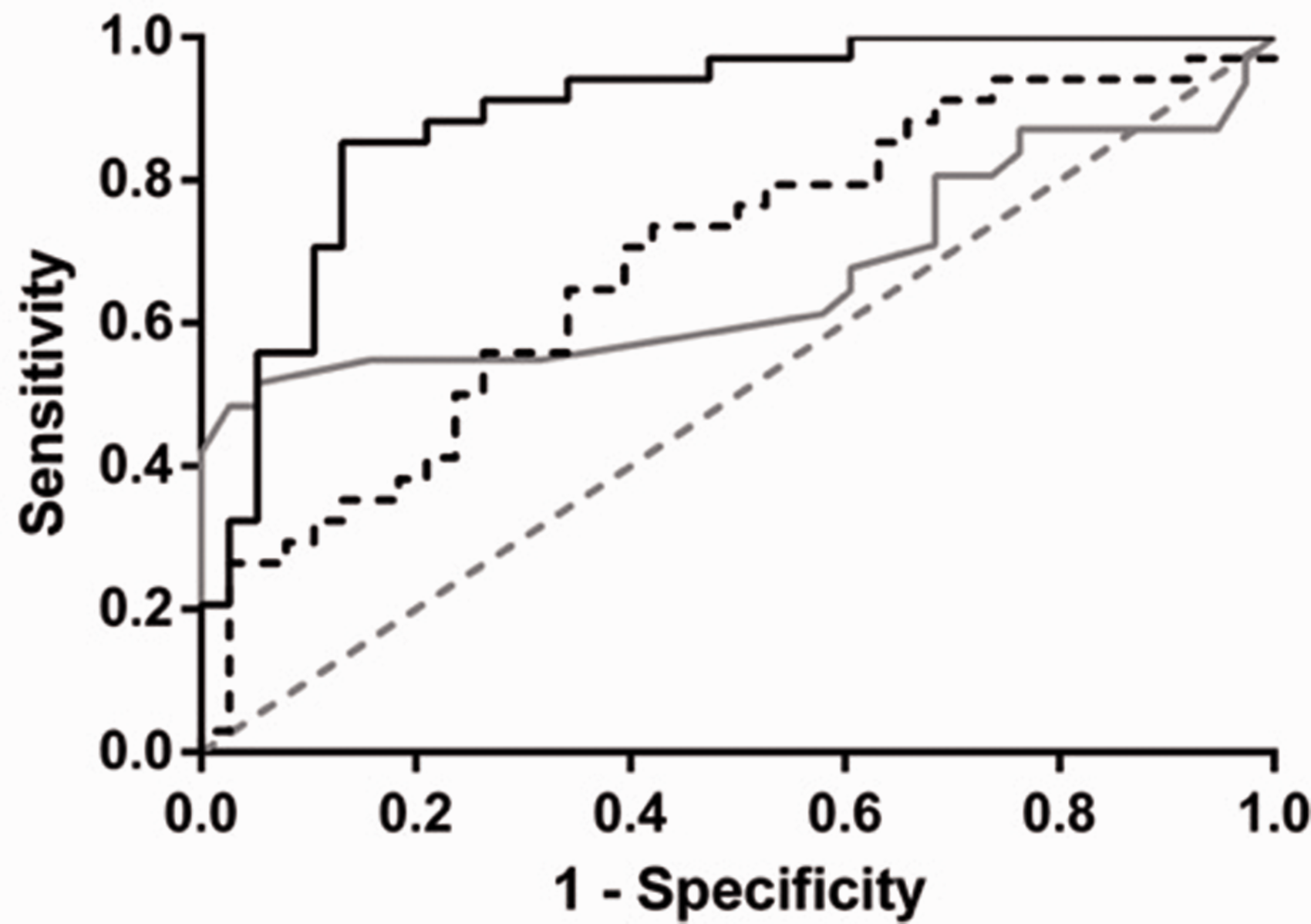
ROC curves for CNBL (solid black line), CNFL (dashed black line) and global RNFL (solid grey line). The line of identity is shown as dashed grey line.
In vivo corneal confocal microscopy results for eyes of NMOSD patients without on attacks and control subjects.

CNFL: corneal nerve fiber length; CNBL: corneal nerve branch length; CNFD: corneal nerve fiber density; CNBD: corneal nerve branch density; DC: dendritic cell; SD: standard deviation.
Clinical disability and disease severity score relationships with axonal loss
We divided NMOSD patients into two subgroups according to recent EDSS levels: subgroup 1 (n = 9) consisted of patients with a recent EDSS level of 3.5 or below, and subgroup 2 (n = 8) consisted of patients with a recent EDSS level above 3.5. 13 An EDSS level of 3.5 was selected as the cut-off because it is the limiting value between moderate and significant disability levels. The patients included in this study were free of relapse for at least six months before the investigation. Subgroup 1 had significantly higher CNFL (30.15 (32.97–26.01) vs. 26.77 (30.39–15.14) mm/mm2, P = 0.045) and CNFD (188.45 (206.11–162.56) vs. 167.32 (189.98–94.68) fibres/mm2, P = 0.045) values than subgroup 2 (Figure 3). Furthermore, the evaluation revealed that recent EDSS levels significantly inversely correlated with the CNFL and CNFD of NMOSD patients (r = −0.391, P = 0.022). The disease severity scores of NMOSD patients were significantly inversely correlated with CNBL and CNBD (r = −0.385, P = 0.025) and CNFL and CNFD (r = −0.341, P = 0.049). Moreover, the correlation between disease duration and IVCM parameters was not statistically significant.

NMOSD patients with higher EDSS than 3.5 have significantly lower average corneal nerve fiber length compared to NMOSD patients with lower or equal EDSS to 3.5 (*p < 0.05).
Discussion
In our study, we demonstrated significantly reduced CNFL and CNFD, CNBL and CNBD in NMOSD patients compared with controls. DC density was increased significantly in NMOSD patients compared with controls. These results are consistent with the findings of previous studies evaluating axonal changes in MS using corneal IVCM.7,8,10,15
Until recently, NMOSD was believed to be confined to the optic nerves, brainstem, and spinal cord without involvement of the peripheral nervous system (PNS). Recent studies have reported PNS damage in autoimmune diseases of the central nervous system, such as MS and NMOSD. Although central and peripheral demyelination is a widely investigated feature of MS, the characteristics of PNS involvement in NMOSD patients are still unknown. 16 There have been few reports investigating peripheral neuropathy in NMOSD. Kim et al. reported that a 31-year-old woman with NMOSD also showed additional findings corresponding to PNS damage. 17 Mizuno et al. showed that a woman diagnosed with NMOSD with AQP4 antibodies 2 years ago presented with numbness and tingling sensations in the distal parts of all limbs due to multifocal peripheral nerve involvement. 18 Some other NMOSD cases presented with various symptoms involving the peripheral nervous system, such as leg weakness or gait disturbance as an initial symptom.16,17,19 There are a few published reports highlighting the relationship between peripheral neuropathies and NMOSD and MOG antibody-associated disorders. All the reported cases had AQP4 antibody positivity except for one report by Feyissa et al., and in all cases in which electrodiagnostic testing was conducted, axonal loss and demyelination-type radiculopathy were revealed.16,17,20
Chronic inflammatory demyelinating polyradiculopathy (CIDP) can occur in patients with MS and results in combined central and peripheral demyelination (CCPD). 21 Ogata et al. found that patients with CCPD met the McDonald criteria for MS and the EFNS/PNS definite criteria for CIDP with percentages of 67.5 and 87.5, respectively. 22 Another study by Misawa et al. reported abnormal nerve conduction in 5% of MS patients. 23 Antibodies against myelin antigens may also lead to CNS and PNS involvement. Anti-neurofascin antibodies are frequently present in these patients. 24 Another view is that MS causes neurodegenerative changes in the grey matter of the spinal cord, resulting in radiculopathy. 25 Spinal cord grey matter involvement was demonstrated by postmortem examinations or in vivo evidence of atrophy using phase-sensitive inversion recovery MRI or electrodiagnostic testing.25,26 To explain the mechanism, it is suggested that axo-glial antigens are a target of the immune system in MS. 27
There are two different viewpoints on PNS damage in NMOSD. The first approach suggests that AQP4 in the transitional zone between the CNS and PNS at the spinal root may be disrupted by immune attack, which leads to radiculitis in NMOSD. 28 The other approach suggests that an unknown antibody against an axoglial antigen may cause PNS damage, causing axonal and grey matter damage, as in MS. 19
The cornea is innervated exclusively by small-fibre nociceptive axons similar to those in the skin. While skin biopsy is an invasive method to diagnose small-fibre peripheral neuropathy, IVCM images of nerves can easily be acquired from the cornea. Therefore, IVCM can be used to evaluate PNS involvement in neurodegenerative diseases instead of skin biopsy. 29 In our study, we found that NMOSD patients had significantly reduced CNFL and CNFD and CNBL and CNBD compared with control subjects. These findings support that NMOSD may cause damage in small nerve fibres in the PNS even before PNS symptoms emerge. Two MOG antibody-positive patients were included in this study. One patient without a history of ON experienced longitudinally extensive transverse myelitis (LETM) attack once and fully recovered following steroid treatment without any sequelae. Another patient had paediatric-onset NMOSD and experienced recurrent myelitis and ON attacks with double positivity for antibodies (mild MOG+, strong AQP4+). Additionally, while 14 patients had AQP4 antibodies, two patients did not have AQP4 or MOG antibodies, although their corneal nerve fibre density was reduced, similar to patients with positive anti-AQP4 antibodies. This supports the idea that an unknown antibody, other than AQP4 and MOG antibodies, against axo-glial antigens may lead to PNS damage.
In further analysis, we demonstrated that the recent EDSS level was significantly and inversely correlated with the CNFL and CNFD levels of NMOSD patients. We also showed that the disease severity scores of NMOSD patients were significantly inversely correlated with the CNBL and CNBD measures. Our findings may argue that IVCM can be used as a surrogate marker for the extent of axonal damage in patients with NMOSD; however, further studies are needed to contribute to our results.
We also showed a significant reduction in RNFL thickness in NMOSD patients compared with controls. Our findings were compatible with former studies. OCT has become a useful tool for analysing retinal damage in both MS and NMOSD. 30 Previous OCT studies reported significantly reduced RNFL thickness in patients with NMOSD, reflecting severe axonal injury.31–33 Bennett et al. showed RNFL thickness reduction, especially in the superior and inferior quadrants, while our findings demonstrated greater reduction in the inferior and temporal quadrants. Two studies showed that optic neuritis in NMOSD results in more severe RNFL thinning than in MS.31–33 Naismith et al. found that after ON attack, the adjusted mean RNFL for 22 NMOSD patients was 56.7 ± 3.1 micrometres, whereas it was 66.6 ± 2.4 micrometres for 47 MS patients. 32 Ratchford et al. suggested that the identification of substantial RNFL thickness loss after ON in a patient without MS should be considered in terms of NMOSD. 33 An analysis of the effect of the number of ON episodes on RNFL thickness found that the first episode of ON caused greater thinning on the RNFL than subsequent episodes in both NMOSD and MS. 33 Comparing patients without ON attacks, Bennett et al. showed that RNFL thinning is rare in patients with NMOSD, while it presents in MS even without a history of ON. 31 Conversely, Ratchford et al. found that the RNFL was mildly thinner in both MS and NMOSD patients than in controls. 33 In our study, we demonstrated that NMOSD patients have significantly lower RNFL thickness than controls and that the RNFL thicknesses of the eyes with ON attack/s were significantly reduced compared with eyes without ON attack/s.
These findings suggest that ON is a more destructive process with more widespread axonal injury in the affected eyes and a higher visual disability risk in NMOSD than MS. In MS, ON attacks tend to be less severe with a better prognosis. The severe thinning of the RNFL in NMOSD is presumably due to optic nerve damage, which could cause retrograde degeneration of retinal ganglion cell axons. We suggest that earlier diagnosis of NMOSD may decrease future disability through earlier initiation of systemic immunomodulatory treatment. There is a need for further longitudinal research on RNFL thickness to assess its prognostic role.
Our data showed that DC density increased significantly in NMOSD eyes without ON. There are studies showing the presence of neuroimmune cross-talk between different structures through the interaction of immune system molecules. 34 Recently, Lee et al. reported that asymptomatic brain lesions could be observed on MRI in clinically stable NMOSD patients, which may indicate ongoing inflammation. 35 The possible explanation of the increased DC density in our study could be that ongoing immune activation occurs in NMOSD.
We would like to acknowledge some limitations of our study. The sample size of the NMOSD patient group was limited in number. Considering the low prevalence of the disease, this number can be acceptable to obtain significant results. Moreover, it would be better to repeat the IVCM and OCT tests after some time for each patient to assess their prognostic role in the disease course. However, we performed the tests only once, which allowed us to assess their clinical situation at that time point but not in a longitudinal setting. Recent data have shown that the most promising OCT marker is macular layer segmentation to identify neurodegeneration, especially segmentation of the ganglion cell-inner plexiform layer. 36 We acknowledge that in our study, we only measured the pRNFL of the participants, which creates a limitation. In addition, IVCM findings in demyelinating diseases are still controversial and deserve further clarification. The underlying pathophysiological mechanisms that cause a reduced number of corneal nerve branches and increased DCs should be further studied.
Our findings in this study suggest that evaluation of SBP using IVCM may be a novel biomarker for the detection of neurodegeneration and inflammation in NMOSD during the disease course.
Conflict of Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data for this study are available from the corresponding author upon reasonable request.
Supplemental Material
sj-pdf-1-mso-10.1177_2055217321998060 - Supplemental material for A novel investigation method for axonal damage in neuromyelitis optica spectrum disorder: In vivo corneal confocal microscopy
Supplemental material, sj-pdf-1-mso-10.1177_2055217321998060 for A novel investigation method for axonal damage in neuromyelitis optica spectrum disorder: In vivo corneal confocal microscopy by Ayşe Altıntaş, Ayse Yildiz-Tas, Sezen Yilmaz, Betul N Bayraktutar, Melis Cansu Comert, Hanna Zimmermann, Alexander U Brandt, Friedemann Paul and Afsun Sahin in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the local ethics committee of each participating centre.
Informed consent
Informed consent was obtained from all participants.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
