Abstract
Background
Breastfeeding as an infant appears protective against later development of some autoimmune diseases, but research into its influence on multiple sclerosis (MS) risk has yielded inconclusive results.
Objective
We investigated the possible impact of breastfeeding on MS risk.
Methods
We used two population-based case–control studies comprising 3670 cases and 6737 matched controls. Logistic regression was used to estimate odds ratios (OR) and 95% confidence intervals (CI) for association between MS and exposure to prolonged breastfeeding (4 months or longer) versus reduced breastfeeding (less than 4 months). A meta-analysis of case–control studies that assessed the impact of breastfeeding on MS risk among women and men was conducted.
Results
Prolonged breastfeeding was associated with reduced MS risk among men (OR 0.7, 95% CI 0.5–0.9) but not among women (OR 0.9, 95% CI 0.8–1.1). Among men, a synergistic effect was observed between HLA-DRB1*15:01 carrier status and reduced breastfeeding.
Conclusions
Findings from the current study add to accumulating evidence that breastfeeding may be a modifiable protective factor for reducing the risk of MS in offspring. When possible, mothers should be supported to breastfeed their infants; however, the mechanism of a sex-specific biologic effect of breastfeeding on MS risk is unclear.
Keywords
Background
Multiple sclerosis (MS) arises from a combination of genetic and environmental factors. A large number of genetic loci have been implicated in MS susceptibility, the most prominent being the class II DRB1*15:01 allele within the human leukocyte antigen (HLA) complex. 1 Epstein-Barr virus infection,2,3 vitamin D status, 4 sun exposure habits, 5 adolescent body mass index (BMI),6,7 and smoking 8 are also linked to MS etiology.
Human breast milk is immunologically complex, containing multiple compounds that support infant growth, provide protection against infections and modulate mucosal immune responses. 9 In addition to passive transfer of humoral immunity, breast milk also contains factors that actively stimulate the infant immune system, and evidence supports the beneficial role of breastfeeding for reducing the risk of developing several immune-mediated diseases such as inflammatory bowel disease and type 1 diabetes.10–12
Prior studies assessing a potential association between breastfeeding and MS risk have yielded conflicting results. The earliest study found no association, 13 whereas later studies have observed a protective association between prolonged breastfeeding and subsequent MS risk.14–17 After stratification by sex, a strong association was found among men but not among women in both Norwegian and Italian cohorts. 17 Absence of infant breastfeeding has also been associated with increased risk of pediatric-onset MS. 18
Using two large population-based case–control studies, we investigated the influence of breastfeeding on subsequent MS risk among women and men, considering a large number of covariates and potential confounding factors including number of siblings, adolescent BMI, smoking and HLA-DRB1*15:01 status.
Methods
The report is based on two large case–control studies on environmental and genetic risk factors for MS. The Epidemiologic Investigation of Multiple Sclerosis (EIMS) is a nationwide study with a study base comprising the Swedish population aged 16–70 years. Incident cases of MS were recruited from 40 neurology clinics, including all university hospitals. All cases had been diagnosed by their treating neurologist according to the McDonald criteria.19–21 For each case, two controls were randomly selected from the national population register. Both controls were frequency matched by the case’s age in 5-year age strata, sex and residential area. If information could not be obtained from a selected control, another control was chosen using the same principles. All aspects of the study were approved by the Regional Ethical Review Board at Karolinska Institutet and all participants gave their informed consent to participate. The study has been described in more detail elsewhere. 22
Information regarding environmental exposures and lifestyle habits were collected using a standardized questionnaire. During the study period April 2005 to June 2015, completed questionnaires were provided by 2880 cases and 6122 controls. The response rate was 93% for cases and 73% for controls. Those who did not know whether they had been breastfed were excluded (203 cases and 388 controls).
The other study identified prevalent MS cases among members of the Kaiser Permanente Medical Care Plan, Northern California Region (KPNC) using electronic health records. KPNC is an integrated health services delivery system with a membership of 4.0 million that comprises about 25–30% of the population of a 22-county service area in northern California. Diagnoses were validated by chart review and radiology and pharmacy records, according to McDonald criteria.19–21 Controls were randomly selected from KPNC members and were individually matched to cases on sex, birth date, race/ethnicity, and zip code of the case residence. The study protocol was approved by the Institutional Review Boards of the KPNC Division of Research and the University of California, Berkeley, and all participants gave their informed consent to participate. The study has been described in more detail elsewhere. 23
KPNC participants completed a computer-assisted telephone interview regarding lifestyle factors and various exposures. As of the data freeze in August 2014, the study included 1163 cases and 1178 matched controls. The study participation proportions were approximately 80% for cases and 66% for controls. Those who did not know whether they had been breastfed were excluded (170 cases and 175 controls). A flow-chart presenting the number of subjects in each study is presented in Online Resource 1.
Definition of breastfeeding
Participants in both studies were asked whether they had been breastfed as infants and those who had were asked to specify the duration of breastfeeding. In EIMS, participants estimated the number of months they had been breastfed, whereas participants in the KPNC study could choose between the following categories; less than 1 month, 1–3 months, 4–6 months, and greater than 6 months. Prolonged breastfeeding was defined as having been breastfed for 4 months or longer, and reduced breastfeeding was defined as having been breastfed less than 4 months. Those who could not specify how long they had been breastfed were placed in a separate category (unknown duration).
Genotyping
In EIMS, genotyped blood samples were available for 1973 cases and 2784 controls who had answered both questions regarding breastfeeding. Genotyping was performed on the MS replication chip 24 which is based on an Illumina exome chip to which approximately 100,000 custom markers were added and HLA region alleles were imputed with HLA*IMP:02. 25 In KPNC, genotyped blood samples were available for 865 cases and 679 controls. Subjects were genotyped as previously described. 26
Statistical analysis and potential confounding variables
Differences in variables across categories of breastfeeding duration were assessed using one-way analysis of variance (ANOVA) for continuous variables and the Kruskal–Wallis test for categorical variables. Using unconditional logistic regression in EIMS and conditional logistic regression in KPNC, the occurrence of MS in subjects who had been breastfed for 4 months or longer was compared with that in subjects who had been breastfed less than 4 months, by calculating odds ratios (OR) with 95% confidence intervals (CI). 27 The analyses were performed separately for men and women. We performed a trend test for a dose–response relationship regarding duration of breastfeeding and risk of MS using a continuous variable for duration of breastfeeding (number of months) in a logistic regression model using the EIMS data. The linearity assumption was met.
Sufficient-cause interaction is present when two causal risk factors are involved in the same causal pathway toward disease. Rothman et al. have shown that sufficient-cause interaction between two causal factors results in departure from additivity of disease rates for these factors and vice versa. 28 The concept has been further developed by VanderWheele and Knol. 29 Interaction on the additive scale between carriage of HLA-DRB1*15:01 and reduced breastfeeding was assessed using the attributable proportion due to interaction (AP) together with a 95% CI. 29 The AP between two causal factors can be interpreted as the proportion of cases among those who are exposed to both factors that is due to the interaction per se. An AP larger than 0 thus indicates presence of interaction.
The potential for confounding of the association between breastfeeding and risk of MS was assessed separately in EIMS and KPNC (data not shown). A variable was identified as a confounder if the point estimate changed by more than 10% with the addition of the confounder to the crude model. The crude model for EIMS included the frequency matching factors age and residential area. Potential confounding variables for EIMS included ancestry, number of siblings, family history of MS, a history of infectious mononucleosis, smoking habits, passive smoking, alcohol consumption at inclusion in the study, BMI at age 20 years, sun exposure habits, educational level, socioeconomic index, carriage of HLA-DRB1*15:01 and absence of the protective allele HLA-A*02:01. Variable descriptions are presented in Online Resource 2. The final EIMS model included variables for age and residential area. The other variables had only minor influence on the results and were therefore not retained in the final analysis. Potential confounding variables for KPNC included number of siblings, a history of infectious mononucleosis, smoking habits, passive smoking, and BMI at age 20 years, with the same definitions as EIMS. The final KPNC model included smoking pack years. Variables such as smoking that describe adult behavior that cannot occur before being breastfed were included as potential confounders because being breastfed and adult smoking behavior may share common causes such as year of birth, parental smoking behavior, and family or cultural traditions, and thus confound our associations of interest.
Two meta-analyses were conducted as follows: (a) measures of association for prolonged breastfeeding and MS derived from EIMS and KPNC case–control studies were combined; and (b) measures of association for prolonged breastfeeding and MS derived from both EIMS and KPNC case–control studies and previously reported Norwegian and Italian MS cases-control studies 17 were combined. Specifically, ORs were combined by calculating a weighted average of the study-specific adjusted ORs, where weights were proportional to the inverse of the variance for each OR. Heterogeneity between studies was quantified using the I2 statistic. 30 Due to the small number of studies included in these meta-analyses, formal statistical tests for random versus fixed effects were not performed; however, measures of association and CIs estimated under each assumption were very similar. Summary measures of association estimated with fixed-effect models are reported here.
Analyses evaluating evidence that any observed non-null association between prolonged breastfeeding and MS was mediated by being overweight during adolescence, as well as analyses investigating potential effects of missing data and exposure misclassification are described in Online Resource 3. All analyses were conducted using Statistical Analysis System (SAS) version 9.4, STATA version 15, and R version 3.5.
Results
Our analyses of breastfeeding and MS risk included 2522 cases and 4397 controls matched by age, sex and residential area. In EIMS, there were no significant differences with regard to the matching variables or known potential confounding variables between those who could estimate the duration of breastfeeding and those who could not. In KPNC, significantly fewer female cases were ever smokers among those who reported prolonged breastfeeding compared with those who reported reduced breastfeeding or unknown duration of breastfeeding.
Characteristics of cases and controls in EIMS and KPNC, by sex and duration of breastfeeding, are shown in Table 1.
Characteristics of cases and controls in EIMS and KPNC, by sex and duration of breastfeeding.

EIMS: Epidemiologic Investigation of Multiple Sclerosis; KPNC: Kaiser Permanente Northern California; IM: infectious mononucleosis.
Reduced breastfeeding = breastfeeding less than 4 months; prolonged breastfeeding = breastfeeding 4 months or longer. Based on self-reported sun exposure habits, we constructed an index for sun exposure ranging between 3 (the lowest exposure) and 12 (the highest exposure).
Compared with subjects who had been breastfed for less than 4 months, prolonged breastfeeding was associated with a significantly reduced risk of developing MS among men (combined OR: 0.7, 95% CI: 0.5, 0.9) but not among women (combined OR: 0.9, 95% CI: 0.8, 1.0). The results based on EIMS did not significantly differ from those in the meta-analysis whereas prolonged breastfeeding showed an association with MS risk among both women and men in KPNC (Table 2).
OR with 95% CI of developing MS among subjects who were breastfed 4 months or longer, compared with subjects who were breastfed less than 4 months, by sex.

aNumber of exposed cases and controls; bAdjusted for age, sex, and residential area; cAdjusted for age and residential area; dAdjusted for smoking; eMeasure of study heterogeneity, I2 = 100×(Q-df)/Q, where Q is Cochran’s heterogeneity statistic and df is degrees of freedom. 28
OR: odds ratio; CI: confidence interval; EIMS: Epidemiologic Investigation of Multiple Sclerosis; KPNC: Kaiser Permanente Northern California.
There was no significant difference between those who had been breastfed 4–6 months versus longer than 6 months. No significant dose–response relationship was observed between duration of breastfeeding as a continuous variable and MS risk (p-value for trend was 0.1 among men and 0.3 among women). HLA-DRB1*15:01 and reduced breastfeeding showed evidence for a synergistic effect between the two risk factors among men (AP 0.4, 95% CI 0.04–0.6), but not among women (AP: 0.1, 95% CI: −0.2, 0.4) (Table 3). An AP of 0.4 indicates that the combined effect is 40% higher than the sum of the individual effects, or equivalently that 40% of the cases among the double exposed are due to the interaction per se.
OR with 95% CI of developing MS among subjects with different combinations of breastfeeding (<4 months versus ≥4 months) and HLA-DRB1*15:01 status, by sex.
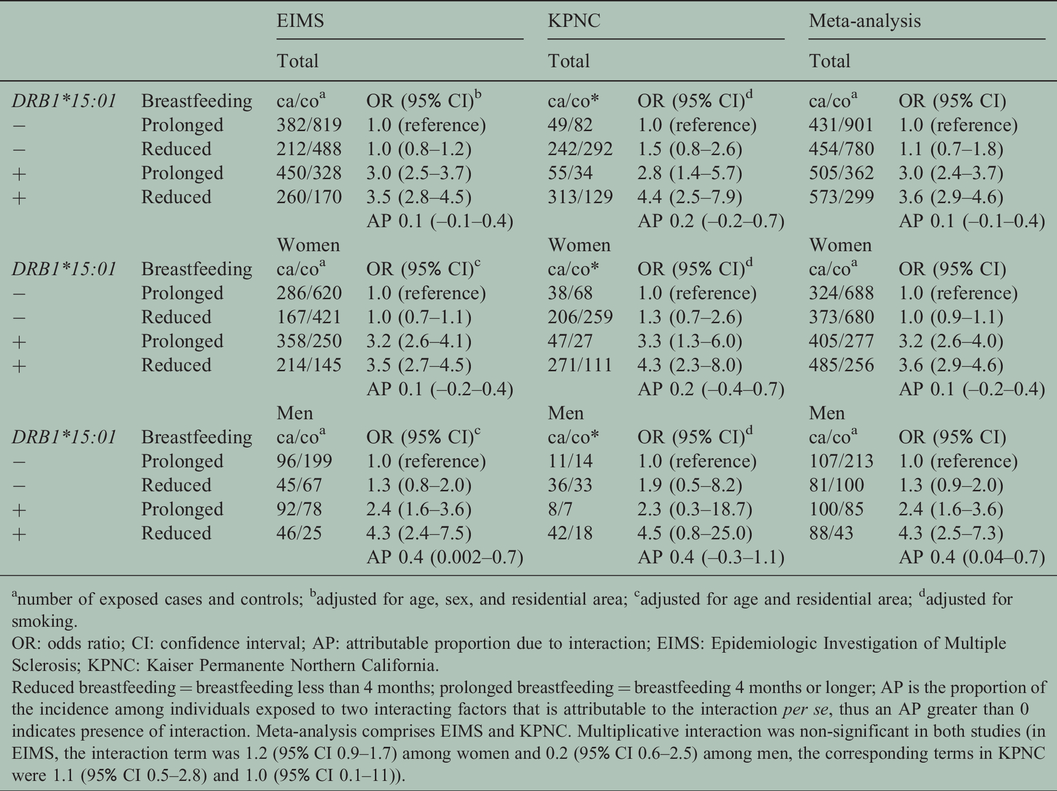
anumber of exposed cases and controls; badjusted for age, sex, and residential area; cadjusted for age and residential area; dadjusted for smoking.
OR: odds ratio; CI: confidence interval; AP: attributable proportion due to interaction; EIMS: Epidemiologic Investigation of Multiple Sclerosis; KPNC: Kaiser Permanente Northern California.
Reduced breastfeeding = breastfeeding less than 4 months; prolonged breastfeeding = breastfeeding 4 months or longer; AP is the proportion of the incidence among individuals exposed to two interacting factors that is attributable to the interaction per se, thus an AP greater than 0 indicates presence of interaction. Meta-analysis comprises EIMS and KPNC. Multiplicative interaction was non-significant in both studies (in EIMS, the interaction term was 1.2 (95% CI 0.9–1.7) among women and 0.2 (95% CI 0.6–2.5) among men, the corresponding terms in KPNC were 1.1 (95% CI 0.5–2.8) and 1.0 (95% CI 0.1–11)).
Meta-analysis of combined results from this study and from published Italian and Norwegian case–control studies demonstrated a protective effect for MS in men (OR: 0.5, 95% CI: 0.4, 0.7) (Table 4) and null effect in women (OR: 0.9, 95% CI: 0.8, 1.1). The magnitude and direction of effect was consistent across all four studies among men and women.
Individual and meta-analysis combined ORs and 95% CIs for association between breastfeeding 4 months or longer compared with breastfeeding less than 4 months and developing MS among EIMS, KPNC, Norwegian, and Italian men and women.

OR: odds ratio; CI: confidence interval; Meta: meta-analysis combined associations; EIMS: Epidemiologic Investigation of Multiple Sclerosis; KPNC: Kaiser Permanente Northern California.
aResults for Norwegian and Italian datasets are from Ragnedda et al., 2015; All models were adjusted for confounding variables.
bThe inverse of reported ORs from Ragnedda et al. were used to represent the protective associations observed between prolonged breastfeeding and risk of MS.
cCombined effect estimated using inverse-variance fixed-effect meta-analysis.
dMeasure of study heterogeneity, I2 = 100×(Q-df)/Q, where Q is Cochran’s heterogeneity statistic and df is degrees of freedom. 28
Mediation analyses did not reveal evidence for an indirect effect of prolonged breastfeeding mediated by BMI <25 among EIMS or KPNC male participants (Online Resource 4). The Baron-Kenney and causal mediation estimators yielded similar results. In addition, there was no evidence of interaction between breastfeeding and being overweight during adolescence among EIMS or KPNC males on the multiplicative or additive scale.
Results from missingness analyses generally showed no effect of missing data on observed findings in EIMS and KPNC. Among KPNC women, the observed unknown duration association was further from the null (OR: 1.5, 95% CI: 1.0–2.1) than the IPM-weighted association (OR: 0.9, 95% CI: 0.6-1.2), but otherwise, missingness results were consistent with observed associations (Online Resource 5). Results from breastfeeding exposure misclassification analyses indicate that observed protective associations among male participants were robust to moderate exposure misclassification. For both EIMS and KPNC, bias-corrected associations were very similar to observed results (Online Resource 6).
Discussion
In accordance with a recent study, 17 we observed that prolonged breastfeeding reduced the risk of MS among men, but not among women. There was evidence of a synergistic effect between HLA-DRB1*15:01 and reduced breastfeeding among men.
Human breast milk is an immunologically complex solution. In addition to passive transfer of humoral immunity, breast milk also contains factors that influence the development and differentiation of T-cell subsets and cytokine production, and has a long-term effect on the development of the immune system. 9 Human breast milk also stimulates the growth of colon bacterial species that have been found to help prevent autoimmune reactions.31,32 Prior studies have demonstrated a protective effect of breastfeeding on subsequent risk of several autoimmune diseases, such as diabetes mellitus and inflammatory bowel disease.10–12
Studies have demonstrated an elevated interferon-gamma/IL-4 ratio in formula-fed children compared with breastfed children, suggesting that formula feeding induces a Th1 cytokine shift. 33 The Th1 predominance in infants receiving formula nutrition might be explained by a higher incidence of acute infections or by the high challenge of foreign cow-milk antigens in formula feeding. The extracellular region of bovine butyrophilin (BTN) exhibits high structural similarity to human myelin oligodendrocyte glycoprotein (MOG), which is a candidate autoantigen for MS. 34 Immunological cross-reactivity has been demonstrated between these proteins. Since oral tolerance is poorly developed in infants, early exposure to bovine BTN may allow expansion of cross-reactive T-cell responses to MOG that increases subsequent MS risk, especially in the context of a susceptible genotype.
The HLA-DRB1*15:01 allele has been reported to influence the response to MOG peptides. 35 Children with Th1 autoimmune diseases such as MS and diabetes type I have increased abnormal T-cell responses against cow-milk proteins when compared with healthy controls. 36 An early disease mechanism may thus arise from antigenic mimicry between cow-milk proteins and human self-antigens, increasing the risk of Th1 autoimmune disease later in life, especially in the context of a susceptible genotype. While it is unclear why the observed breastfeeding association was restricted to males, it is possible that sex-specific differences in T-helper immune response may interact with breastfeeding. Interestingly, a protective effect of breastfeeding on respiratory infections among female but not male infants has been reported. 37 Breastfeeding may also protect children from adverse effects of environmental exposures, 38 some of which may have sex-specific effects on MS risk later in life.
In 2015, Ragnedda et al. reported evidence for an association between reduced duration of breastfeeding (<4 months) and increased risk of MS in males from Italy and Norway. 17 Associations were null among females within each population. Our meta-analysis combined ORs across EIMS, KPNC, Italian, and Norwegian studies and a consistent protective association between prolonged breastfeeding (>4 months) and risk of developing MS in males was observed. There were a several similarities across the studies that made combining them possible: (a) the cases and controls were from a national multicenter case–control study of environmental risk factors in MS,39,40 (b) the prolonged breastfeeding exposure was defined as being breastfed for >4 months in all four studies, and (c) breastfeeding exposure was determined by questionnaires similar to ones used in the current study. Our study is the first to investigate interaction between breastfeeding and HLA-DRB1*15:01.
The primary strength of this study is the use of two large population representative case–control samples that each has extensive data on breastfeeding exposure status, other established environmental MS risk factors and genetic data. Our results build upon recent case–control studies that demonstrated a protective association between prolonged breastfeeding and MS among males. In addition, the fact that our exposure of interest occurs early in life ensures that there is temporality between breastfeeding and MS onset and that reverse causality is not a concern. However, some limitations must be acknowledged. Our study was based on data from two case–control studies in which information on personal characteristics and different exposures was collected retrospectively. Since we used study designs in which controls were matched by the cases’ sex, calculating multiplicative interaction between sex and breastfeeding was not an alternative. We did not ask participants who were breastfed to provide information regarding exclusive versus non-exclusive breastfeeding, and we were therefore not able to analyze this aspect of breastfeeding.
As with all retrospective study designs, accurately determining exposure status can be difficult for exposures that occur many years before study inclusion. In order to minimize the risk of recall bias, EIMS predominantly included cases who had received their diagnosis within the past year whereas KPNC used prevalent cases. The risk of recall bias is thus greater in KPNC. The questionnaires in both studies, however, contained a wide range of questions regarding many potential environmental risk factors and no section in the questionnaires was given prime focus. Moreover, since the relationship between breastfeeding and MS risk had not been investigated until recently, the quality of the reported information would probably not differ between cases and controls.
The recruitment of cases and controls may introduce selection bias. In EIMS, the mean index year did not differ between responding and non-responding cases and controls. Among both cases and controls, the proportion of non-responders was more often men (58% among cases and 61% among controls). Selection bias was minimized by the population-based design. The Swedish health care system provides free-of-charge access to all Swedish residents and almost all cases of MS are referred to hospital-based neurological units, making them eligible to be part of EIMS. The McDonald criteria have been revised during the period in which cases have been recruited to EIMS. This should not have affected the case recruitment, i.e. those who received a diagnosis of MS according to the McDonald criteria during the study period were asked to participate in the study. However, the revised criteria have made it possible to diagnose individuals at an earlier stage of the disease.
Even though there was a relatively high proportion of non-responders among the controls, this bias is probably modest because the prevalence of lifestyle factors, such as smoking and socioeconomic status, among the controls was consistent with that of the general population in similar ages. 41 A relatively high proportion of the participants did not answer the question on breastfeeding or specify the duration of breastfeeding. However, among the responders, the reported information on breastfeeding among controls was in line with that of the general population, indicating that selection bias was unlikely to affect results. Selection bias due to the lower control response in the KPNC samples is possible; however, case–control matching should limit influence of any selection bias on results.
We comprehensively investigated the potential for bias resulting from exclusion of subjects missing any breastfeeding exposure data and missing breastfeeding duration, and found no evidence for bias in results derived from the EIMS study; only weak evidence for bias from ‘unknown duration’ on sex-stratified prolonged breastfeeding association results was observed in the KPNC study. Further, results from analysis of misclassification of breastfeeding exposure among males did not reveal evidence for bias. Therefore, findings in the current study should be generalizable to the populations from which both study samples were derived.
In conclusion, prolonged breastfeeding may reduce the risk of MS among men; however, the mechanism of a sex-specific biologic effect on MS risk is unclear. Human breast milk stimulates the growth of colon bacterial species that have been found to help prevent autoimmune reactions. Another possible explanation is that antigenic mimicry between cow-milk proteins and human self-antigens may arise, increasing the risk of Th1 autoimmune disease later in life. Our findings add to accumulating evidence that breastfeeding may be a modifiable, potentially protective factor for reducing the risk of MS in offspring, and mothers should be supported to breastfeed their infants if possible.
Supplemental Material
sj-pdf-1-mso-10.1177_2055217320928101 - Supplemental material for Breastfeeding is associated with reduced risk of multiple sclerosis in males, predominantly among HLA-DRB1*15:01 carriers
Supplemental material, sj-pdf-1-mso-10.1177_2055217320928101 for Breastfeeding is associated with reduced risk of multiple sclerosis in males, predominantly among HLA-DRB1*15:01 carriers by AK Hedström, C Adams, X Shao, C Schaefer, T Olsson, LF Barcellos and L Alfredsson in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Authorship
All authors made a substantial contribution to the concept or design of the work. AKH and CA performed the statistical analyses and drafted the manuscript. All authors commented on the manuscript and approved the final version to be published. All authors take public responsibility for appropriate portions of the content.
Conflict of Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AKH reports no disclosures. CA and XS report no disclosures. LFB and CS report no disclosures. TO served on scientific advisory boards and received speaker honoraria Novartis, Merck-Serono, Biogen Idec, TEVA and Genzyme; served as Co-editor of Current Opinion in Immunology; received from Novartis and Biogen; and receives research support from Novartis, Genzyme, Biogen Idec, the Swedish Research Council (07488), EU fp7 Neurinox, and CombiMS, the, and the Swedish Brain Foundation. LA received research support from the Swedish Medical Research Council (Dnr 2016-02349), the Swedish Council for Health, Working Life and Welfare (Dnr 2015-00195) and the Swedish Brain Foundation; has received speaker honoraria from Biogen Idec and TEVA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the Swedish Medical Research Council; from the Swedish Research Council for Health, Working Life and Welfare, the Swedish Brain Foundation, and the Swedish Society for Medical Research. National Institutes of Health grants R01NS049510 (PI: Barcellos); NIH NIEHS: R01ES017080 (PI: Barcellos); and NIH NIAID: R01AI076544 (PI: Barcellos).
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Regional Ethical Review Board at Karolinska Institutet, Sweden and the Institutional Review Boards of the KP Division of Research and the University of California, Berkeley) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
