Abstract
Background
Gait disturbance is a major contributor to clinical disability in multiple sclerosis (MS). A sensor was developed to assess walking speed at home for people with MS using infrared technology in real-time without the use of wearables.
Objective
To develop continuous in-home outcome measures to assess gait in adults with MS.
Methods
Movement measurements were collected continuously for 8 months from six people with MS. Average walking speed and peak walking speed were calculated from movement data, then analyzed for variability over time, by room (location), and over the course of the day. In-home continuous gait outcomes and variability were correlated with standard in-clinic gait outcomes.
Results
Measured in-home average walking speed of participants ranged from 0.33 m/s to 0.96 m/s and peak walking speed ranged from 0.89 m/s to 1.51 m/s. Mean total within-participant coefficient of variation for daily average walking speed and peak walking speed were 10.75% and 10.93%, respectively. Average walking speed demonstrated a moderately strong correlation with baseline Timed 25-Foot Walk (rs = 0.714,
Conclusion
New non-wearable technology provides reliable and continuous in-home assessment of walking speed.
Introduction
Gait abnormality is a major contributor to clinical disability in multiple sclerosis (MS).1 Patients report that walking is the most valuable function to preserve and the most challenging aspect of having MS.2,3 Approximately half of MS clinical relapses are manifested by gait changes.4 The transition from relapsing–remitting MS to secondary progressive MS is commonly accompanied by a slowing of walking speed.
The current standard method for assessing gait in MS clinical care and research is the Timed 25-Foot Walk (T25FW).5–8 Because this test typically requires an in-person clinic or research visit, its utility is constrained by periodic sampling. Within-subject variability of the T25FW is particularly high in patients with slower walking speeds;9 furthermore, the short distance of the test leads to floor and ceiling effects.
Continuous outcome measures of gait enable the objective assessment of changes over time without the constraints of an in-clinic visit. In addition, continuous sampling enables the evaluation of variables that confound periodic measurement (such as environment, time of day, amount of sleep, and temperature). Wearable sensors, including accelerometers, are emerging as useful tools for remote monitoring of gait in MS and can distinguish among patients at various disability levels.10,11 However, patients must remember to wear and recharge wearable sensors. Additional limitations of wearable sensors include poor signal-to-noise and environmental heterogeneity (e.g. location and activity). For example, level of exercise can introduce considerable variability within and between users. In contrast, in-home passive monitoring better controls for environment and activity. In one clinical application, non-worn infrared body motion sensors were used to assess in-home walking speed and variability in an aging healthy population, and demonstrated potential as an early predictor of mild cognitive impairment.12
A touchless shelf-top sensor using infrared technology to detect body movement without the use of wearables or carried sensors was developed to assess walking speed in real-time in the homes of people with MS. The goal of this study was to investigate the clinical utility of continuous in-home walking speed measures to assess gait in adults with MS.
Materials and methods
Participant characteristics
Six female patients with MS (four with relapsing–remitting MS, two with secondary progressive MS, mean age 56.2 (SD 9.4) years, median Expanded Disability Status Scale (EDSS) 3.5, EDSS range 2.0–6.5) were recruited from the Massachusetts General Hospital MS Clinic (Table 1). Inclusion criteria for the study were: (a) a diagnosis of MS of any subtype; (b) aged 18–70 years; (c) EDSS 0–6.5, inclusive; (d) able to ambulate within the home; (e) residing in a home without co-habitants; and (f) residing in a home with wireless internet access. Exclusion criteria included any significant medical or neurological condition other than MS. Patients were consecutively screened for eligibility and approached for participation at clinic visits. All participants provided prior written informed consent and followed the institutional review board-approved study protocol.
Baseline participant demographics.

DD: disease duration; EDSS: Expanded Disability Status Scale; T25FW: Timed 25-Foot Walk; 6MW: 6-Minute Walk.
aUser 4 reported using a walker always when outside of the home and sometimes when inside the home.
bUser 6 reported using walker sometimes when outside of the home.
Ambulatory assist devices were used when determining T25FW and 6MW as indicated. User 6 achieved a lower EDSS without the use of an ambulatory assist device.
Study design and data collection
At study commencement, a consumer tablet computer fitted with a commercial consumer electronics three-dimensional spatial sensor (Microsoft Kinect, version 2) was placed in each participant’s home and connected to the home wireless internet. Each such ambient measurement system (AMS) (Echo5D, Atlas5D, Cambridge, MA, USA) was initially placed on a shelf at approximately waist height in a room where participants reported walking frequently. Each AMS was equipped with proprietary mobility-tracking software. Successful use of the Kinect Version 2 to assess motor function in-clinic has been reported in the past.13,14
The AMSs in this study utilize infrared light to detect body movement passively without the collection of identifiable photography or video. Users are not required to wear or recharge any device or wearable. The deployed AMSs acquired movement metrics continuously and autonomously for 8 months from patient homes. The AMSs used in this study did not have the capability to distinguish a person of interest from other individuals. Participants therefore had the option of pausing data collection as needed; for example, when additional occupants were visiting the home. Data collection was constrained to an approximate volume of 5 × 5×3 m3 in front of the sensor. Movement data were measured at a rate of 10 frames per second and transmitted from each AMS via an encrypted connection to a secure cloud-based web service (Amazon Web Services, Seattle, WA, USA). Using custom algorithms as previously reported,15 movement data were analyzed to yield summary metrics of average walking speed (AvWS) and peak walking speed (PWS). These metrics are calculated hourly and could be evaluated immediately after calculation by study investigators via a secure web portal.
Floor plans were collected for each room where the sensors were located and included information such as obstructions of walking space to account for potential differences in movement metrics. Rooms were categorized as obstructed or unobstructed; an unobstructed walking space was defined as an area where a person could walk in at least one straight line from one side of the sensor field of view to the other without encountering obstacles.
At the end of an initial 2-month period, each AMS was moved from the original location (location one) to a new location (location two) in the participant’s home. At the end of a subsequent 2-month time period (4 months from baseline), each AMS was returned from location two back to location one, and remained at location one for an additional 4 months. Total data collection time was 6 months in location one and 2 months in location two. For the purposes of analyzing the difference between obstructed and unobstructed locations, only data from the first 4 months were included, so that the total analysis time in each location was approximately equal (2 months at each location). At the end of the study, the AMSs were removed from the homes. One participant withdrew from the study following 4 months of data collection because the participant relocated where the AMS could not be serviced if necessary or retrieved.
To evaluate concurrent reliability of gait outcomes, in the case of one participant (user 1) a second AMS was placed in the same room as another AMS, but at a different position and orientation (Figure 1). This configuration provided simultaneous, overlapping data collection from two independent vantage points for a period of 3 months.

Floor plan for user 1 showing the locations of the overlapping ambient measurement systems (AMSs) (locations 1 and 3) and the location of the non-overlapping AMS (location 2).
Data post-processing
Each AMS sampled location (the person’s distance and angle from the AMS) and stance (whether the person was standing or sitting) at a rate of 10 times per second whenever a human was detected within the range of the sensor. The body detection algorithm rejects movement of smaller moving animals (such as pets) with morphological features inconsistent with human bodies. Instantaneous walking speed was calculated for every frame of data, and AvWS during a given epoch, or time interval, was calculated by dividing the cumulative total distance traveled within the AMS’s field of view by the total time elapsed during that epoch. Samples in which: (a) the participant was seated, or (b) when the participant’s instantaneous walking speed fell below a minimum cut-off interpreted as ‘standing still’ (chosen to be 0.1 m/s) were excluded from walking speed calculations.
PWS was calculated by smoothing the instantaneous walking speeds with a median filter (to remove outliers due to measurement noise), then selecting the highest smoothed walking speed within each hour-long epoch.
Clinical outcome measurements
Each participant completed four in-clinic study visits that included the acquisition of clinical outcomes and patient-reported outcome measures. Study visits occurred at baseline (just prior to sensor placement in the home); at 2 months (at the time of sensor relocation); at 4 months (at the time of sensor relocation back to its original location); and at 8 months (at study conclusion).
At each study visit, a certified EDSS16 neurologist performed the EDSS evaluation. Other clinical outcomes included T25FW and 6-Minute Walk (6MW).17 Patient-reported outcome measures included the MS Walking Scale (MSWS-12),18 5-item MS Fatigue Impact Scale (MFIS),19 NeuroQoL,20 and a subjective measure of walking ability (1–10 scale).
Time series analysis
AvWSs were calculated for hourly, daily, and weekly epochs from AMS data points, as described above. Means and standard deviations were calculated for each movement metric per participant. Data points greater than or equal to three standard deviations from each participant’s mean were excluded from analysis as spurious. Data points reported by participants as being generated by the presence in the home of someone other than the participant (e.g. guest or worker) were excluded from analysis as contaminated. In total, less than 3% of data points were excluded for each participant. Movement metrics were aggregated corresponding to hourly increments of time of day from 7:00 am to 12:00 am. Only one participant was employed outside the home.
Statistical analysis
Spearman correlation was used to correlate in-home continuous gait outcomes with each other, and with baseline standard in-clinic outcome measures. The Wilcoxon signed-rank test was used to compare gait outcomes by AMS location in the home. SPSS version 20 was used for statistical analysis.
Results
Movement metrics
Daily AvWS ranged from 0.33 m/s to 0.96 m/s across all participants during the 8-month study duration (Table 2). Daily PWS ranged from 0.89 m/s to 1.51 m/s. Mean total within-participant coefficient of variation (COV) for daily AvWS and PWS were 10.75% and 10.93%, respectively. Daily AvWS and PWS demonstrated a strong correlation with each other with a trend towards statistical significance when the AMSs were in locations that captured unobstructed walking areas (ρ = 0.77,
Movement outcomes.
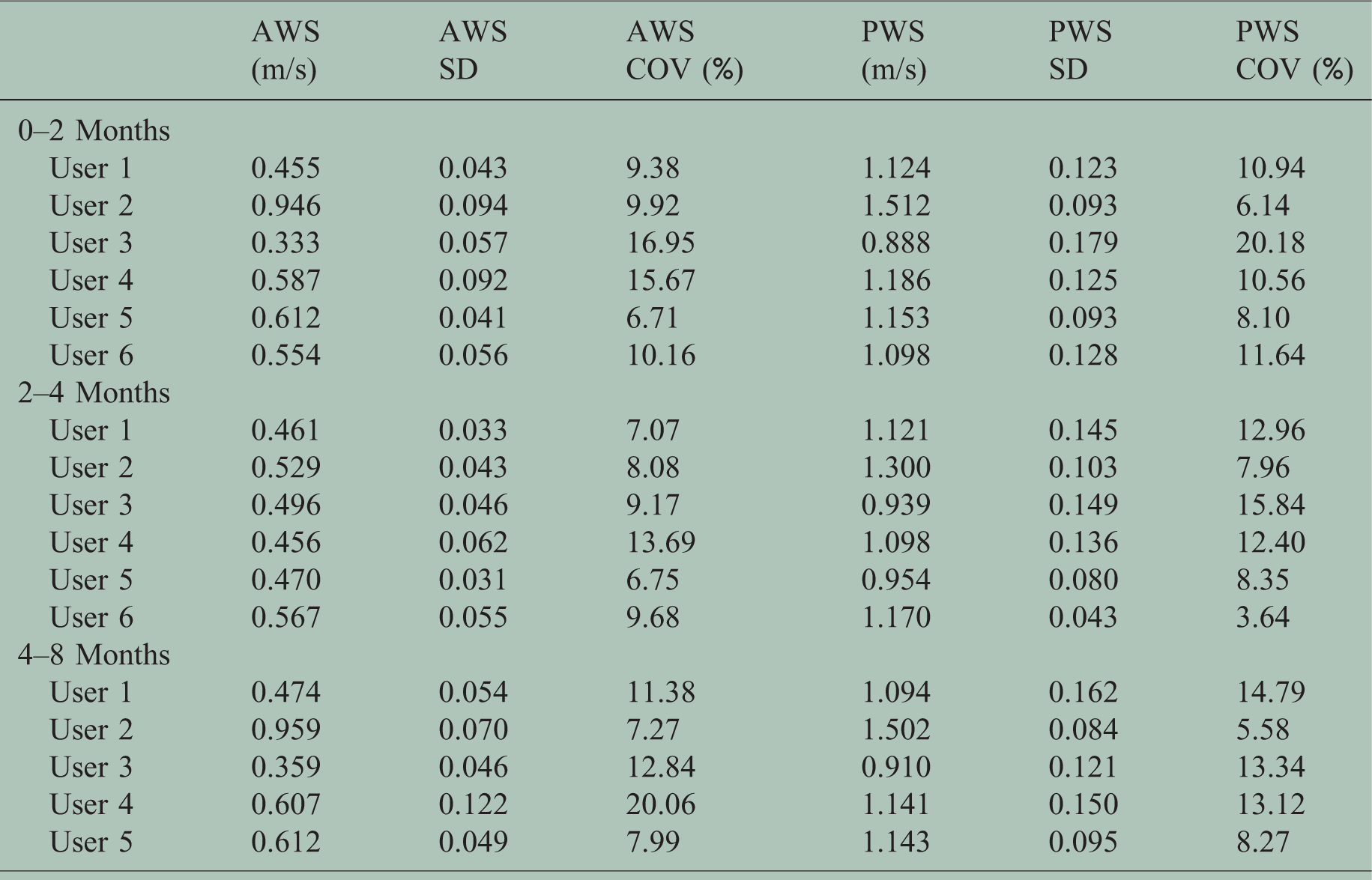
Averages of daily average walking speed (AvWS) and daily peak walking speed (PWS) are displayed for each participant across each displayed time epoch.
SD: standard deviation; COV: coefficient of variation.
Compared to unobstructed walking areas, AvWS was slower in obstructed walking areas (
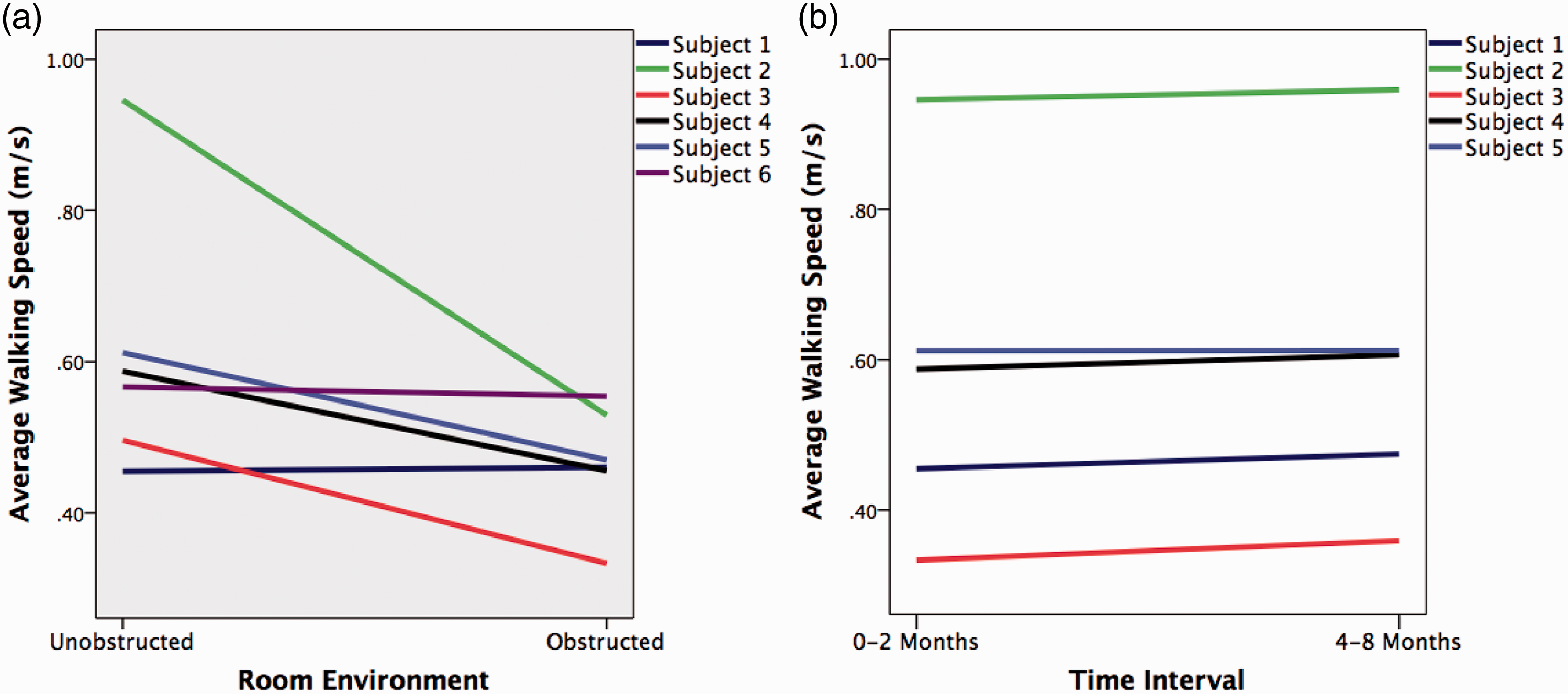
(a) Average daily walking speed for users 1–6 (0–4 months of data) separated by obstructed and unobstructed walking areas. (b) Measurement of average daily speed for users 1–5 taken from the same location at different epochs (0–2 months and 4–8 months).
Analysis of AMS movement outcomes during the first 2 months of AMS placement, when compared with baseline in-clinic disability and ambulatory outcomes, revealed a strong relationship between movement outcome variability and in-clinic established outcomes. AvWS COV correlated significantly with EDSS (rs = 0.841,
Across the 8-month study duration, AvWS and PWS increased over the course of the morning and early afternoon; peaked in mid-afternoon; then decreased in late afternoon and early evening (Figure 3(a) and (c)). When individual users were separated out, some users did not demonstrate as dramatic a change over the daytime hours (Figure 3(b) and (d)). There was no difference in AvWS or PWS between weekdays and weekend days for any participant.

Average and peak walking speed for six participants, displayed in aggregate (a and b) and for individual participants (c and d) over waking hours. Error bars represent 95% confidence intervals.
The two AMSs that collected simultaneous movement data within overlapping fields of view demonstrated a high degree of agreement based on the raw instantaneous velocities (Figure 4(a)). Daily AvWS from the two AMSs demonstrated near-perfect correlation as averaged over the hour, day, and week (r = 0.920, (a) Instantaneous walking speed from two different ambient measurement systems (AMSs) with overlapping field of view during a 20-second period of time when user 1 was moving in front of the AMSs. (b) Overlapping daily average walking speed for the same AMSs.
Discussion
Ambulation represents a function commonly affected in MS, and an important source of clinical disability. In-clinic outcomes to quantify walking ability focus on speed (T25FW) and endurance (6MW). While these outcomes provide valuable information and can be followed over time, they are limited by periodic sampling, which may introduce sampling bias. Furthermore, these outcomes limit the setting within which ambulation is assessed. Wearable sensors allow continuous measurement, but at the expense of introducing noise to the data, stemming from variable and unpredictable environments.
This study describes an AMS that addresses some of these shortcomings using infrared technology to quantify body movement in MS. The AMS can be placed in a patient’s home and does not require the use of wearables or carried sensors. In this study, we demonstrated the ability to deploy such AMSs in patient homes and acquire walking speed continuously over a period of 8 months.
We correlated the continuous in-home measures with gold standard outcome measures used in both research and clinical practice for MS. Several in-home to in-clinic relationships demonstrated moderate to strong correlation. Interestingly, walking speed variability demonstrated a stronger correlation with clinical disability than did absolute measures of in-home walking speed. The ‘good days’ and ‘bad days’ that many people with MS disability describe may be reflected in walking speed variability quantified here. There was a strong correlation between AvWS and the MFIS. Future studies may consider more frequent rating of fatigue to correlate with measures of gait speed. In a previous study evaluating an aging population and their transition to mild cognitive impairment, walking speed variability also correlated with clinical outcomes.12
Other in-home to in-clinic relationships correlated imperfectly. There are several potential explanations for the imperfect correlations. First, the sample size of this pilot study was small; larger studies will be required fully to assess agreement between various measurements and each of their sensitivities to change. Second, it is possible that the two types of measures – in-clinic and in-home – may assess different clinically relevant features of ambulation.
The results of this study suggest that environment strongly impacts walking speed. We demonstrated that differences in walking speed could be detected in different rooms or hallways of the same home. The size of the room, length of open walkways, and presence of walking obstructions such as furniture can alter walking speed. In patients with stable neurological function, after moving the AMS to a different location in the home, we found that AvWS and PWS decreased in rooms that contained more walking obstructions. However, after the planned 2-month break, when sensors were returned to their original locations, measurements were consistent with the initial 2-month data collection. This suggests that for evaluation in the individual user, the placement of the AMS should remain in consistent locations, and optimally in rooms where the user walks frequently and has adequate unobstructed walking space.
We observed a low day-to-day COV (∼10%) for in-home walking speed outcomes (AvWS and PWS). Low variability is desirable when assessing an individual patient’s ambulation in the home setting over time. The participants in this study remained clinically stable over the duration of the study, without clinical relapses and without sustained progression of disability, so that the effect of a relapse on ambulation could not be determined. Future studies should assess these in-home measures’ sensitivity to change in clinical scenarios such as clinical relapse or disease progression. Clinical relapses and progression of disability represent short-term and long-term scenarios in which in-home information could supplement or corroborate other clinical information. With the AMS, it is possible for in-home walking speed to be remotely assessed in real time in order to corroborate a patient’s subjective complaints suggestive of a clinical relapse. In the long-term, walking speed can be assessed over months to years during clinic encounters for patients who complain of progressive gait decline. Measurements from the AMS may have the potential to be a reliable predictor of transition to progressive MS.
When assessing walking speed over the course of daytime and evening hours, we hypothesized that walking speed would decrease over the course of the afternoon hours, as many people with MS complain of fatigue during these times of day. However, walking speed tended to peak in early afternoon hours, then slowly decrease over late afternoon and evening hours, which is a later time and less abrupt than hypothesized. These preliminary results will require replication in a larger study.
There are several limitations of the current study that should be addressed with future studies. First, there is no community monitoring of ambulation in this study. Coupling the in-home monitoring with wearable sensors could provide a more comprehensive, complementary, continuous evaluation of gait. In its current iteration, the algorithms we describe for body motion detection require solo-living situations in order to avoid contamination from people other than the subject of interest. Advanced algorithms are being developed for multi-user technology intended to enable and distinguish data collection for just the person of interest, even when other people (such as family members) are co-located within the field of view. This will allow a significant expansion of the number of participants in future studies.
In conclusion, AMS non-wearable technology provides reliable and continuous in-home assessment of walking speed in MS. This study demonstrates feasibility for the development of new continuous outcome measures to assess gait in MS and in other neurological diseases.
Footnotes
Conflicts of interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VMJS has received consulting fees from Atlas5D; JSV, RAW and ZMK are employees of Atlas5D and are Atlas5D stockholders. WG and GAP are past employees of Biogen and are or were Biogen stockholders. ECK has received research grants from Atlas5D, Biogen, EMD Serono and Roche; and consulting fees from Acorda, Atlas5D, Biogen, Celgene, EMD Serono, Genentech and Shire. AWR and KRP have nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was funded by a grant from Biogen via Atlas5D.
