Abstract
Background
Multiple sclerosis (MS) is associated with significant impairment.
Objective
The objective of this article is to identify and compare clinical measures that can predict self-rated disability in patients with MS using the World Health Organization Disability Assessment Schedule II (WHODAS-II).
Methods
Patients with MS and healthy controls were consecutively recruited at one center. Patients were evaluated for cognitive function assessment, neurological status, perceived disability, mood, fatigue and disease duration. Controls underwent neuropsychological evaluation only. Data were analyzed using multivariate regression.
Results
WHODAS-II total score was predicted by fatigue (p < 0.001) and neurological status (p < 0.05). Student’s t test comparisons between published WHODAS-II normative data and the enrolled cohort of patients with MS showed significantly worse (p < 0.05) scores for patients on mobility, self-care, life activities, participation and total score domains, but not in cognition. Group differences between patients with MS (n = 61) and controls (n = 61) were significant in all cognitive measures except one verbal memory test subscale. Memory function correlated best with the social participation domain of the WHODAS-II.
Conclusions
Self-reported disability in patients with MS was most strongly influenced by fatigue and to a lesser extent by physical disability. Although cognitive function does affect self-assessment of disability, this is not captured by patients on the WHODAS-II cognitive domain.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory demyelinating degenerative disease of the central nervous system; it is the second most disabling neurological disease that affects young adults after traumatic brain injury. 1 Owing to the peculiar pathophysiology/unpredictability of the course of the disease, MS significantly limits patients’ autonomy, threatening their independence and self-respect. Patients’ health/disability status can be compromised not only by neurological symptoms that result in physical disability, but also by diverse symptomatic manifestations that can either be neuropsychiatric or neuropsychological. For example, anxiety, impaired mood, cognitive impairment and fatigue are very common features in MS and have been shown to significantly reduce patients’ quality of life (QoL) and contribute to disability. 2 Given the multifaceted nature of MS, accounting for the impact of all these features on patients’ health/disability would be very useful according to a biopsychosocial model: 1) to understand the real-life burden of MS in terms of activity limitations/participation restrictions; and 2) to compare burden in patients with MS with that of other health conditions. The World Health Organization Disability Assessment Schedule II (WHODAS-II) 3 is a useful measure to examine the role of various features of disease as contributors to disability. The WHODAS-II was designed as a disability assessment instrument based on the biopsychosocial conceptual framework of the International Classification of Functioning, Disability and Health (ICF). 4
The aim of this study was identification and comparison of potential correlates of patients’ perceived disability according to the ICF model, taking into account fatigue, mood, neurological status and cognitive functioning.
Methods
Participant demographics.
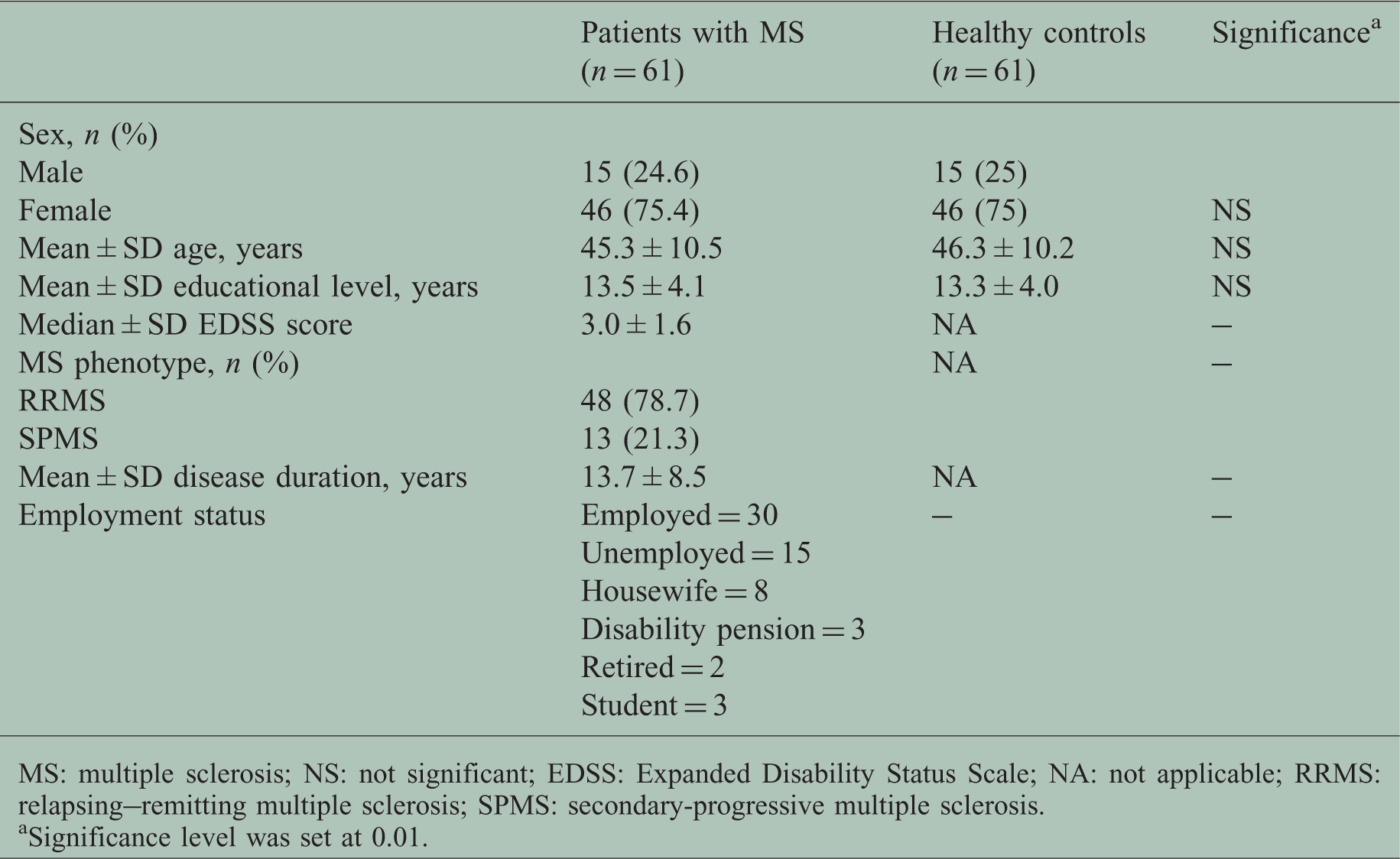
MS: multiple sclerosis; NS: not significant; EDSS: Expanded Disability Status Scale; NA: not applicable; RRMS: relapsing–remitting multiple sclerosis; SPMS: secondary-progressive multiple sclerosis.
Significance level was set at 0.01.
Measurement instruments
WHODAS-II
The self-administered version of the WHODAS-II 3 consists of 36 items that assess functioning and disability during the 30 days before testing. The validity, reliability and factor structure of the WHODAS-II has been evaluated in several conditions, such as systemic sclerosis 10 ankylosing spondylitis, 11 stroke, 12 psychosis and MS, 13 schizophrenia, 14 hearing loss, 15 rehabilitation patients 16 and back pain. 17 The WHODAS-II was also translated and validated for the Italian healthy population. 18 More recently, its validity has been confirmed with modern Rasch model analyses in MS. 19 The WHODAS-II covers six domains of functioning: cognition (understanding and communicating), mobility (moving and getting around), self-care (hygiene, dressing, eating and staying alone), getting along (interacting with other people), life activities (domestic responsibilities, leisure, work and school) and participation (joining in community activities). Seven scales can be derived from the six domains of functioning: understanding and communicating (six items), getting around (five items), self-care (four items), getting along with others (five items), life activities: household (four items), life activities: work/school (eight items) and participation in society (eight items). Response options for each item range from 1 (no difficulty) to 5 (extreme difficulty or cannot do). WHODAS-II scores for each scale are calculated by summing the recoded item responses and transforming them into a range from 0 to 100, where lower scores indicate lower levels of disability. A total score is calculated from all items of all the domains. In this study, we considered the life activities: work/school scale only for the computation of the total score and not independently, as only 30 patients were employed.
Predictor variables
Eight clinical variables were selected as potential contributors to WHODAS-II scores: age; gender; disease duration; physical disability, measured with the EDSS 19 as obtained by neurological examination; fatigue, measured with the Modified Fatigue Impact Scale (MFIS) 20 as completed by the patient; and mood, measured with the Chicago Multiscale Depression Inventory (CMDI) 21 mood subscale as completed by the patient. Cognitive function was measured with a battery of tests of verbal and visual memory, information processing speed, and executive function: immediate recall (sum across five learning trials), short-delay free recall, and long-delay free recall of the California Verbal Learning Test-II (CVLT-II); 22 10/36 Spatial Recall Test (10/36 SRT) total learning and delayed recall; 23 Paced Auditory Serial Addition Test; 24 Symbol Digit Modalities Test; 25 and correct sorts on the Delis-Kaplan Executive Function System 26 Sorting Test. Disease duration was measured from the time of diagnosis using McDonald’s criteria. 27
Statistical analysis
Mean WHODAS-II domain and total scores were compared with available Italian normative data reported in Federici et al. 9 Group differences between patients with MS and healthy controls were assessed with Student’s t test. The significance level for t tests was set at p < 0.05. Multivariate linear regression analyses (backward; p value to enter <0.05, p value to exit >0.10) were conducted to predict the WHODAS-II domains and total scale. Preliminary analyses did not identify collinearity between predictor variables. Three univariate outliers were excluded from the analyses. Variables relative to WHODAS-II scales 1 (understanding and communicating), 3 (self-care), 4 (getting along), 5 (life activities) and 6 (participation in society) underwent square root adjustment to address asymmetry levels below −1 and beyond +1. 28 Regression analyses were performed in two steps. In the first step, predictors into each clinical domain were entered into independent regression models for each scale of the WHODAS-II. In the second step, predictors retained in significant models (tested through an analysis of variance using a threshold of p < 0.05) were gathered together to predict each clinical outcome scale. Cohen’s ƒ,29,30 an appropriate measure for calculating local effect size within a multivariate regression model, was computed for each predictor retained in the second step in models that retained more than one predictor to better clarify/compare the role of each clinical domain in the prediction of self-rated disability scores in patients with MS. The variance inflation factor (VIF) was computed in order to detect the presence of multicollinearity (i.e. high correlation) among predictor variables.
Results
Of the initial 75 patients, 14 were excluded from the study. One patient was excluded because of past alcohol abuse, two for the occurrence of relapses, two for corticosteroid use in the six weeks before the study, three for missing answers in the questionnaires, five for receiving antidepressant medications, and one patient withdrew from the study owing to injury to the upper limb. Patients’ mean age ± SD was 45.3 ± 10.5 (range, 24–72) years, mean education was 13.5 ± 4.1 (range, 5–21) years, median Expanded Disability Status Scale (EDSS) score was 3.0 ± 1.6 (range, 1.0–8.0) and mean disease duration was 13.7 ± 8.5 (range, 2–40) years. There were 46 women (75.4%) and 15 men (24.6%). Forty-eight patients (78.7%) had relapsing–remitting MS, 11 (18%) had a secondary progressive and two (3.3%) had a primary progressive course. Healthy controls’ mean age ± SD was 46.3 ± 10.2 (range, 22–72) and mean ± SD education was 13.3 ± 4.0 (range, 5–18 years).
Group comparison of neuropsychological tests between patients with MS and healthy controls.

MS: multiple sclerosis; CVLT-II: California Verbal Learning Test-II; IR: immediate recall (sum across five trials); NS: not significant; SDFR: short-delay free recall; LDFR: long-delay free recall; SDMT: Symbol Digit Modalities Test; D-KEFS: Delis-Kaplan Executive Function System; 10/36 SRT: 10/36 Spatial Recall Test.
n differs from the value indicated in Table 1 owing to the removal of three univariate outliers.
Group comparison of WHODAS-II domain and total scores a between patients with MS and healthy controls.

Higher scores indicate worse impairment.
WHODAS-II: World Health Organization Disability Assessment Schedule II; MS: multiple sclerosis; NS: not significant.
n differs from the value indicated in Table 1 owing to the removal of three univariate outliers.
All significant p values indicate higher domain scores for the MS group, except for the getting along domain.
Variables independently predicting WHODAS-II domains and total scores in order of explained variance.

WHODAS-II: World Health Organization Disability Assessment Schedule II; MFIS: Modified Fatigue Impact Scale; 10/36 SRT: 10/36 Spatial Recall Test; CVLT-II: California Verbal Learning Test-II; SDFR: short-delay free recall; CMDI: Chicago Multiscale Depression Inventory; EDSS: Expanded Disability Status Scale; IR: immediate recall (sum across 5 trials); LDFR: long-delay free recall; SDR: short-delay recall.
Variables retained in final regression models predicting WHODAS-II domain and total scores and scales listed in order of local effect size.

WHODAS-II: World Health Organization Disability Assessment Schedule II; MFIS: Modified Fatigue Impact Scale; CVLT-II: California Verbal Learning Test-II; LDFR: long-delay free recall; SDFR: short-delay free recall; EDSS: Expanded Disability Status Scale; IR: immediate recall (sum across five trials); CMDI: Chicago Multiscale Depression Inventory; 10/36 SRT: 10/36 Spatial Recall Test; SDR: short-delay recall.
Discussion
This is the first study analyzing the clinical domains associated with self-perceived disability in patients with MS according to the ICF model, which describes disability in the context of biopsychosocial reciprocal interactions rather than simply physical function as evaluated traditionally with the EDSS in the MS field. The statistical method employed in the present study examined 12 important clinical variables to identify the relative impact of each on parameters of daily activities and participation as outlined by the ICF model based on self-report. As expected, the results showed that patients with MS experience strong limitations in the domain of mobility. Moreover, the impact of the disease on self-perceived disability was also observed in most of the other domains, including life activities, social participation and self-care, compared with healthy controls. The only exception was the cognition domain, where patients with MS reported scores similar to healthy controls (Table 3), even though they were clearly impaired on formal neuropsychological testing (Table 2). Measures of fatigue, neurological disability, cognitive function and mood accounted for a large percentage of the variance of the WHODAS-II scores. Interestingly, the measure of fatigue (MFIS) had the largest impact by far on all of the WHODAS-II domains (Table 4). As expected, the mobility scale showed the worse scores among all of the functioning scales and was significantly predicted by fatigue, neurological disability as measured by the EDSS and age. This is consistent with the knowledge that the EDSS primarily assesses mobility 31 and with a previous study showing that patient-reported mobility is strongly correlated with EDSS scores. 32 The models predicting the self-care and getting along scales accounted for a small percentage of variance predicted by fatigue and age, respectively. The regression model on the cognition scale of the WHODAS-II identified fatigue and verbal memory as significant predictors, both with a large effect size.
The getting along scale of the WHODAS-II showed better scores for patients with MS than healthy controls (Table 3). This may reflect problems with the domain structure of the WHODAS-II18,33,34 or may reflect a real difference and deserves further investigation. The life activities domain retained fatigue and CVLT-II, which was in line with previous studies that show how objective cognitive impairment negatively affects activities of daily living in MS. 2 The participation domain was strongly predicted by fatigue, mood and visual and verbal memory. The regression model of the total score reflected the strong relationship observed in the mobility domain, retaining both the MFIS and EDSS as significant predictors. However, when variables were inserted independently into the model to predict the total score, mood, visuospatial memory and age also were retained as significant predictors, echoing the impact of these variables on the life activities and participation domains.
In this study, we found that fatigue by far plays a major role in patients’ self-perception of disability. Among all predictors studied, self-reported fatigue had the greatest influence on self-perceived disability, even greater than physician-assessed physical disability. The MFIS was in fact retained in the majority of the domains and in total score as the predictor with the largest effect size. One possible interpretation of this result is that it may reflect a self-report bias. However, there is little evidence in the statistical literature that self-report bias could represent an issue in psychological research, and growing evidence suggests that the problem of common method variance is probably exaggerated (see Chan 35 and Conway and Lance 36 for review). Moreover, both MFIS and the WHODAS-II demonstrated good psychometric properties in several studies.9,10,12,13,16,19,37 The strong relationship between self-reported disability (and patient-reported outcome measures in general) and fatigue thus reflect the fact that fatigue represents a serious limitation to patients’ independence and appears to be the main contributor to disability from the patient’s perspective.
Moreover, age showed a significant impact on the mobility and the getting along domains of the WHODAS-II, showing a medium and small effect size, respectively. Finally, objective cognitive performance has a significant impact on several domains of everyday functioning evaluated by the WHODAS-II, not just the cognitive self-report domain, despite the relatively low albeit significant difference between the two groups in the neuropsychological measures. Specifically, cognitive performance plays a significant role in limiting patients’ social participation and daily activities. However, patients do not seem to be aware of it, as the objective difference between patients and healthy controls in objective cognitive measures is not captured by the self-reported cognitive domain of the WHODAS-II. This finding is consistent with several studies showing that MS patient self-report does not correlate with objective cognitive impairment and with a recent study showing that performance of patients with MS on an Internet-based task of everyday life can be predicted by objective cognitive performance and not by self-reported cognitive impairment.38,39 Moreover, it highlights that although the MS group in this study may have limited cognitive impairment, objective cognitive function has a significant impact on patients’ activities and social participation.
In conclusion, our results provide an important understanding of contributors to disability as perceived by patients with MS, replicating and extending what has been observed in previous studies exploring the relationship between QoL, physical disability, cognitive function, mood and fatigue.2,39 Furthermore, the coherent relationship between predictor variables and the domains of functioning in the WHODAS-II encourage further research with the WHODAS-II in MS.
There are some important limitations to this study. In addition to the aforementioned opportunity of a measurement bias, the sample size did not allow for the evaluation of all the possible variables that could explain construct variability in the WHODAS-II. The role of predictors, such as personality, social support and anxiety, has not been explored and can be an important area for future research in order to detect other variables potentially associated with the domains showing a negligible percentage of explained variance. Moreover, a larger sample size could allow assumptions about the associations between the WHODAS-II domains and specific cognitive domains assessed with the neuropsychological battery. Finally, the cross-sectional nature of this study could not capture the relevance of the selected variables relative to disease progression. Also, norms for the WHODAS-II are needed, as the published data from healthy controls were not appropriately matched. Further investigations in this direction are underway. This study also was limited to a single center in a large European city; therefore, additional research in patients with MS from different cultures and countries is needed.
Footnotes
Funding
This work was performed in the framework of the research project of the National Institute for Occupational, Safety and Prevention “Prevention of domestic and work accident in a sample of people affected by Multiple Sclerosis. Assessment methodology of neuromotor and neurocognitive disability in a prevention perspective” (grant B/33/DML/01). The statistical analysis was funded by Biogen.
Acknowledgments
Biogen provided funding for editorial support in the development of this paper; Lauren Nagy from Excel Scientific Solutions copyedited and styled the manuscript per journal requirements. Biogen reviewed and provided feedback on the paper to the authors. The authors had full editorial control of the paper, and provided their final approval of all content.
Conflict of interest
G.M. has served as a consultant for Biogen. D.C. is a full-time employee of Biogen. R.M. is a former employee of Biogen. O.A., C.I., V.P., C.C. and M.B. have nothing to declare. J.D. has served as a consultant for Biogen and Novartis Pharma. U.N. has served as a consultant for Biogen, Sanofi Aventis, Teva Pharmaceuticals, Novartis Pharma and Boehringer Ingelheim, and has received travel and research grants from Merck Serono, Novartis Pharma and Biogen.
