Abstract
With the rapid development of digital medicine, drug therapy is gradually shifting from a traditional model centered on static information delivery to a dynamic management paradigm driven by software, data, and patient-centered care. In September 2023, the U.S. Food and Drug Administration (FDA) released the Prescription Drug Use-Related Software (PDURS) framework, providing a regulatory foundation for the regulation of software-generated end-user outputs that are directly related to prescription drug use and treated as part of drug labeling. From a digital medicine perspective, this review systematically summarizes the core concepts and technical features of the PDURS framework and examines its potential impacts on personalized medicine, drug safety, information transparency, medication use monitoring, and pharmaceutical innovation. The findings suggest that by transforming drug-use information into software-generated, patient-facing outputs that are subject to drug labeling requirements, PDURS supports patient-centered individualized medication decision-making, enhances adverse event monitoring and risk management, and facilitates the continuous generation and application of real-world data. Furthermore, the PDURS framework has profound implications for pharmaceutical companies, influencing research and development models, clinical trial efficiency, regulatory compliance, industry collaboration, and drug value creation, while also posing new requirements for medical education and global drug regulatory coordination. This review further highlights that strengthened international communication and institutional collaboration are essential to fully realize the potential of PDURS as a foundational infrastructure for digital medicine and to promote the healthy development of global drug digitalization and precision medication.
Keywords
1. Introduction
With the continuous development of digital health, digital technologies are increasingly embedded across the entire continuum of disease prevention, clinical decision-making, and health management.1,2 Within this broader context, digital health tools have emerged as key interfaces connecting patients, healthcare providers, pharmaceutical companies, and regulatory authorities. Importantly, however, the scope and functional emphasis of digital health tools vary across different stakeholders. From a public health and clinical care perspective, digital health tools typically include patient- and clinician-facing mobile applications, remote monitoring systems, wearable devices, and clinical decision support software. These tools are primarily designed to support medication guidance, efficacy monitoring, adverse event reporting, and patient engagement, with the overarching goal of improving treatment safety, transparency, and personalization. Such tools emphasize patient-centered care by leveraging real-time data and interactive interfaces to enhance medication management and health outcomes.1–4 In contrast, from the pharmaceutical industry perspective, digital health tools more commonly refer to software and digital systems integrated into drug development, regulatory compliance, and lifecycle management.5–7 These include digital platforms for clinical trial data collection and analysis, post-marketing safety surveillance systems, and software components that may be incorporated into drug labeling to support medication use and risk communication.5,7,8 In this context, digital health tools are closely aligned with pharmaceutical products, aiming to improve development efficiency, optimize risk management, and enhance drug value in real-world settings. Against the backdrop of the expanding role of digital health tools throughout the medication use continuum, the need to clearly define and regulate software directly related to prescription drug use has become increasingly apparent.
7
It is within this context that, in September 2023, multiple departments of the U.S. Food and Drug Administration (FDA) jointly released the framework for Prescription Drug Use-Related Software (PDURS, Docket Number: FDA-2023-D-2482),
9
providing an important regulatory foundation for the compliant integration of digital health tools into drug use and labeling. According to the FDA’s draft guidance, PDURS refers to software disseminated by or on behalf of a drug sponsor, where the software’s end-user output is considered part of prescription drug labeling (Box 1). Importantly, the FDA’s regulatory focus is not on the software itself, but on how its end-user output is categorized and regulated as required or promotional labeling. Following the release of this framework, it has sparked widespread attention and discussion among the U.S. public, pharmaceutical manufacturers, digital health researchers and producers, as well as pharmaceutical regulators from various countries.10,11 There is a broad consensus that this guideline ushers in a new era of digital health and pharmaceutical integration, marking a significant milestone in U.S. healthcare and global medical care. It has opened up new opportunities for integrating digital software into drug labeling.10,11Box 1. A plausible PDURS-type scenario: valproate pregnancy-risk outputs treated as labeling-like communications.
12
However, although the PDURS framework has attracted widespread attention in practice at the intersection of digital health and the pharmaceutical industry, existing academic research has not yet conducted systematic and multi-level analyses from an integrated perspective of digital medicine and drug regulation.13–15 Current studies on prescription drug use–related software primarily focus on individual-level medication behaviors or risk characteristics within specific populations, while lacking systematic discussion of PDURS as a software-driven regulatory infrastructure for medication use and its broader implications for public health, the pharmaceutical ecosystem, and transnational drug governance.14,15 In contrast to the relatively limited engagement within the academic literature, policy and industry stakeholders have increasingly begun to respond to the PDURS framework. For example, American Telemedicine Association (ATA) Action, an advocacy organization affiliated with the American Telemedicine Association, submitted formal comments on the FDA’s draft PDURS guidance expressing explicit support for the framework. ATA Action emphasized that PDURS provides an important regulatory pathway for appropriately integrating digital health software into prescription drug use and labeling, and highlighted its potential to advance patient-centered, digitally enabled medication management and telehealth practices. 16 Nevertheless, such policy-level feedback and practical expectations from industry stakeholders have not yet been sufficiently analyzed or theoretically synthesized in the academic literature, further underscoring the need for in-depth scholarly examination of the PDURS framework.
Against this backdrop, this article is structured as a policy review and commentary, aiming to provide a high-level regulatory analysis rather than an empirical study or clause-level regulatory research. To systematically characterize the potential global implications of the PDURS framework for digital drug labeling and related practices, this review adopts an international perspective to examine its effects on the public, pharmaceutical manufacturers, medical education, drug research, and pharmaceutical regulatory policies. A series of expert discussions were conducted involving drug researchers and pharmaceutical regulators from the United States, China, Spain, and other countries, with particular emphasis on the application of the PDURS framework outside the United States and its broader policy and institutional implications.
2. Impact on the public
The FDA’s release of the PDURS framework represents a significant step toward promoting the use of digital health technologies in drug administration, particularly the integration of software-generated end-user outputs into the drug labeling framework.
10
This framework provides pharmaceutical manufacturers with more flexibility and opportunities to launch digital health tools (such as mobile apps, software platforms, or smart devices) alongside prescription drugs and treat specific end-user outputs generated by digital health software as part of prescription drug labeling.9–11 The potential impacts of the PDURS framework on the public can be analyzed in the following aspects, see a summary in Figure 1. Summary of the effects of the PDURS framework on the public.
2.1. Enhancing personalized medicine
Under the PDURS framework, personalization is primarily enabled through software-generated end-user outputs—such as individualized dosing recommendations or risk alerts—that are treated as part of prescription drug labeling. The PDURS framework offers potential for personalized medicine, particularly in monitoring drug use, adjusting dosages, and managing side effects. By integrating digital software, patients can better understand how to adjust their medication use based on their individual health conditions and responses, thereby improving treatment outcomes and safety.17,18 In this context, Artificial Intelligence and Machine Learning (AI/ML) play a crucial role in enhancing the PDURS approach. These technologies enable more adaptive and intelligent systems that can analyze real-time patient data, predict risks (such as non-adherence or adverse reactions), and optimize treatment plans dynamically. As a result, AI/ML helps translate the vision of personalized, responsive medication management into practical digital solutions. 19 For example, Xue et al.20,21 used AI/ML models to predict warfarin blood concentrations in patients undergoing cardiac surgery based on longitudinal clinical data, and the results of this study demonstrated the potential of interactive digital labeling combined with AI/ML approaches in supporting personalized medication management.
2.2. Promoting drug safety
Digital software can track a patient’s medication use in real-time, helping reduce the risks of medication misuse or inappropriate use, especially for complex treatment regimens that require precise monitoring (such as cancer drugs or immunotherapies). Through software feedback, both patients and healthcare providers can adjust treatments in a timely manner, reducing the occurrence of adverse reactions.
2.3. Improving transparency and monitoring
Incorporating digital software into drug labeling enables a transition from traditionally static information delivery to a dynamic, data-driven medication management model, thereby substantially enhancing transparency throughout the treatment process.22,23 Under this approach, healthcare providers and patients are no longer limited to accessing basic indications and dosage instructions but can also obtain continuously updated information related to medication use, such as population-specific warnings, emerging trends in adverse event reports, and risk alerts derived from real-world data. 23
This mechanism is particularly important for newly approved drugs or medications with special usage requirements. For such therapies, safety and effectiveness information often continues to evolve after market entry. Digital drug labeling can serve as a rapid feedback and update channel, allowing clinicians to stay informed of the latest evidence and to adjust treatment strategies dynamically in clinical practice. At the same time, patients can gain a more intuitive understanding of changes in treatment response and potential risks through digital interfaces, which may improve treatment adherence and engagement.22,23
Furthermore, digital drug labeling can be integrated with electronic health record systems, clinical decision support tools, or patient monitoring technologies to enable continuous tracking and aggregation of key indicators, such as medication adherence, adverse event occurrence, and relevant biomarker changes. This software-enabled transparency and monitoring framework not only supports optimized clinical decision-making and efficacy assessment but also provides a more timely and systematic data foundation for post-marketing safety surveillance and regulatory decision-making.23,24
2.4. Fostering pharmaceutical innovation
The PDURS framework offers pharmaceutical developers an opportunity for innovation, enabling them to combine digital technologies with traditional drugs to create more valuable therapeutic tools. This could spur more interdisciplinary collaboration and drive the integration of the pharmaceutical industry with digital health technologies.
In summary, while the PDURS framework offers a promising pathway to integrate digital software outputs into drug labeling, thereby advancing more personalized and safer medication use, its successful implementation will require careful attention to regulatory, clinical, privacy, and patient-engagement challenges.
2.4.1. Regulatory challenges
While the framework holds great potential, it also brings regulatory and compliance challenges. For instance, how can the FDA ensure that these digital tools are adaptable and effective across different patient populations? How can patient privacy and data security be guaranteed? These concerns require the FDA to further clarify and refine relevant regulations.19,25
2.4.2. Impact on clinical practice
For doctors and clinical experts, PDURS provides a new way to assist in decision-making but may also introduce additional complexity. Healthcare providers will need training on how to effectively integrate these digital tools and understand how they influence a patient’s drug treatment plan.
2.4.3. Patient acceptance and engagement
The effectiveness of digital health tools often depends on patient acceptance and engagement. Some patients may be unfamiliar with or unwilling to rely on digital tools. Therefore, promotion and education will be key to the successful implementation of this framework. 26
3. Impact on the pharmaceutical industry
Similarly, for the pharmaceutical industry, the FDA’s release of the PDURS framework may have a profound impact, presenting both opportunities and challenges. The following is an analysis of the potential impacts of PDURS on the pharmaceutical industry.
3.1. Promoting innovation and enhancing drug toxicity monitoring
From an innovation perspective, the PDURS framework provides pharmaceutical companies with a new pathway to deeply integrate digital health technologies into drug development,10,11,27 particularly in the area of drug safety assessment and toxicity monitoring.28,29 By incorporating digital software as an integral component of medication use, pharmaceutical developers can continuously collect and analyze safety-related data throughout the drug lifecycle, thereby enabling more intelligent and refined safety management strategies. Within this framework, digital health applications, monitoring software, or smart devices can be deployed alongside drugs to enable real-time or near real-time monitoring of adverse events, medication adherence, and clinical or behavioral signals associated with toxicity. This approach facilitates the early identification of potential toxicity risks and supports targeted interventions and dose optimization for high-risk populations, ultimately improving the overall safety profile of drug therapies.28,29 Through such software-enabled safety monitoring, pharmaceutical companies can achieve product differentiation and enhance the real-world value of their drugs. However, the deep integration of digital software with drug toxicity monitoring also presents challenges. Building on the safety monitoring benefits outlined earlier, companies must increase investment in research and development and strengthen their technical and interdisciplinary capabilities to ensure regulatory compliance and successful market adoption. 30
3.2. Improving clinical trial and approval efficiency
Through digital software, pharmaceutical companies can more precisely track drug usage in clinical trials, obtaining higher-quality patient data. This not only helps better assess drug efficacy but may also accelerate the drug approval process. The FDA may grant faster approval to drugs that effectively incorporate digital software, as digital health tools can provide more accurate and real-time patient feedback and clinical data, reducing the duration and complexity of traditional clinical trials. However, integrating digital tools into trial design demands additional resources and validation to maintain effectiveness and compliance.
3.3. Promoting market access and increasing drug value
In the long term, integrating digital software into drug labels can increase the added value of pharmaceutical products, making them more attractive to the market. For example, the adaptability and personalization of digital treatments align with modern medical trends, attracting more hospitals and doctors. These value-added services can help pharmaceutical companies gain an advantage in a competitive market, particularly in treating complex diseases such as cancer and diabetes, where more refined treatment and management options are needed. However, in the short term, this requires pharmaceutical companies to not only have drug development capabilities but also the digital technology, data analysis, and privacy protection expertise. Some pharmaceutical companies may need to partner with external organizations to fill the technological gaps in this area.
3.4. Regulatory compliance and data security
Extending the privacy and compliance considerations from patient-facing use, PDURS imposes additional obligations on companies. The PDURS framework requires pharmaceutical companies to ensure not only the safety and efficacy of the drugs themselves but also the compliance, privacy protection, and data security of the associated digital health tools. Pharmaceutical companies will need to address more complex regulatory requirements to ensure the quality of the software, the security of patient data, and respect for patient privacy. This may necessitate additional compliance reviews, audits, and monitoring resources.19,25
If companies can effectively address these challenges, they will build a strong reputation in protecting patient privacy and data security, enhancing their brand credibility. This could become a key differentiating factor in future market competition.19,25
Building on the patient engagement benefits discussed earlier, digital integration can further enhance adherence and market performance for pharmaceutical products, though addressing acceptance challenges—particularly among older or tech-unfamiliar patients—remains essential.
In summary, the PDURS framework unlocks significant innovation and efficiency gains for the pharmaceutical industry, with the efficient integration of digital technologies into drug development emerging as a critical determinant of future success.10,15,31,32
4. Impact on medical education and drug research
The FDA’s release of the PDURS framework also has profound implications for medical education and drug research, particularly in enhancing clinical practice, fostering interdisciplinary collaboration, and driving drug development. Below is an analysis of the specific impacts of this framework on medical education and drug research.
4.1. Impact on medical education
The implementation of PDURS promotes the integration of digital health tools with traditional drug treatments, which will drive medical education systems to incorporate digital medicine technologies into curricula and training.18,27 Future medical students and practicing physicians will need to understand how to use and integrate digital tools to optimize drug therapy, monitor patient conditions, and adjust dosages.33,34 As a result, medical education institutions will place more emphasis on developing students’ capabilities in digital healthcare environments, including how to use digital health software to support personalized treatments, manage drug side effects, and improve patient adherence.33–35 However, medical education needs to keep pace with the rapid development of these new technologies, create appropriate teaching curricula, and train instructors to effectively teach students how to use these tools. This may increase the pressure on educational resources, requiring more technical support and training modules.
As drug development and treatment undergo a digital transformation, medical education will increasingly focus on interdisciplinary collaboration, particularly in the integration of medicine, data science, artificial intelligence, and digital health fields.35,36 Future medical education may include collaborative courses with computer science, data analytics, engineering, and other disciplines to cultivate medical professionals who can think and work across different fields. This interdisciplinary collaboration will help healthcare professionals better understand and apply emerging digital tools, improving the overall quality of medical services. However, implementing such collaboration requires educational institutions to offer more flexible and integrated course content, as well as a shift from traditional medical education models to avoid professional barriers and information gaps between disciplines.
The PDURS framework emphasizes the role of digital tools in assisting drug use, and medical education can enhance students’ and physicians’ clinical decision-making skills through simulation training (e.g., virtual reality, AI simulations).13,18 For example, training on adjusting drugs, monitoring patient reactions, and making treatment decisions using digital health applications can help physicians better address clinical challenges. This transformation may require significant adjustments to educational resources and assessment methods to ensure that students can proficiently use these new technologies in daily clinical practice, while avoiding the risks associated with over-reliance on technology.5,35
4.2. Impact on drug research
The PDURS framework facilitates the integration of digital health technologies into drug development, offering new directions for drug research.17,37 During the clinical trial phase, digital tools can monitor drug use and patient responses in real-time, providing more precise data. 37 This can not only improve the efficiency of clinical trials but also speed up the time to market for drugs. For example, by integrating patient data, pharmaceutical companies can identify potential issues with drugs more quickly, adjust treatment plans in a timely manner, and avoid safety or efficacy problems. However, introducing digital tools into drug development may add complexity, as researchers need to address issues related to verifying and standardizing the use of digital tools in clinical trials to ensure data reliability and consistency. Moreover, developing and integrating digital health tools may require substantial funding and cross-disciplinary technical support. I believe all this information would be more credible if supported by some examples of the previous studies related to the drug development process.
The introduction of the PDURS framework encourages drug development to focus more on personalized treatment.13,15,37 The integration of digital software with drugs allows for precise adjustments based on patients’ specific conditions, thus promoting the research and development of personalized drugs. In the future, drug development will increasingly focus on the integration of genomics, metabolomics, and other biomarkers with digital health tools to design drugs that better meet patients’ specific needs. The development of personalized drugs requires substantial data support and precise algorithms, which may increase the complexity and cost of research. Effectively integrating data from various sources and ensuring its validity and reproducibility are important challenges in drug research.
Digital tools provide data on drug usage after market release, helping drug development teams monitor the market performance and patient feedback of drugs in real time.7,32 This real-time data collection can help drug development teams identify the effects and side effects of drugs in different patient groups, thus optimizing drug formulations or treatment plans, extending the product lifecycle, and improving its market competitiveness.7,32 Post-market monitoring requires close collaboration between pharmaceutical companies, medical institutions, patients, and technology companies to ensure data accuracy and completeness. Data privacy and security concerns are also key issues to be addressed in post-market monitoring. By integrating digital health tools with drugs, researchers can obtain more clinical data in real-time, enabling more precise evaluation of drug efficacy and safety. For example, digital tools can monitor patients’ physiological responses, drug side effects, and other factors, helping researchers identify potential safety risks early and make adjustments.7,32 This provides stronger support for early drug evaluation and risk management. The diversity and complexity of digital tool data may present challenges for drug researchers in interpreting these data. Researchers need to have sufficient skills to handle these data and ensure that they comply with ethical and regulatory requirements.
In summary, the PDURS framework not only impacts the pharmaceutical industry but also brings profound changes to medical education and drug research. Medical education will place more emphasis on the integration of digital health tools, advancing interdisciplinary collaboration, and improving clinical decision-making skills. In drug research, the implementation of the PDURS framework will accelerate drug development processes, promote personalized treatment, improve drug safety and efficacy evaluation, and facilitate post-market monitoring and optimization. While the framework presents many opportunities, addressing challenges related to technology, compliance, and data security will be critical for medical education and drug research.
5. The potential impact of the PDURS framework on the digital health policies of various countries
Summary of potential impact of the PDURS framework on the digital health policies in various countries. #

Abbreviations: PDURS, Prescription Drug Use-Related Software; FDA, U.S. Food and Drug Administration; EMA, European Medicines Agency; EU, European Union; ePI, electronic Product Information; SmPC, Summary of Product Characteristics; PL, Package Leaflet; XML, Extensible Markup Language; XML-PM, XML Product Monograph; NDS, New Drug Submission; EUNDS, Extraordinary Use New Drug Submission; PMDA, Pharmaceuticals and Medical Devices Agency; HSA, Health Sciences Authority; PI, Prescribing Information; PIL, Patient Information Leaflet; MFDS, Ministry of Food and Drug Safety; NMPA, National Medical Products Administration; CDE, Center for Drug Evaluation; IFPMA, International Federation of Pharmaceutical Manufacturers & Associations.
#Notes. Some policies, such as the European Union’s Medical Device Regulation (MDR, EU 2017/745) and Germany’s DiGA (Digitale Gesundheitsanwendungen), although they fall within the broader policy framework of digital health, were not included in the scope of comparison because their regulatory focus and content differ substantially from that of the PDURS framework.
5.1. North America: A PDURS-centered regulatory pathway for digital drug labeling
In North America, the United States and Canada both exhibit relatively high levels of maturity in drug regulatory systems and digital health infrastructure; however, they have adopted distinct institutional approaches to the development of digital drug labeling. In the United States, the introduction of the Prescription Drug Use-Related Software (PDURS) framework represents a pioneering regulatory effort to incorporate software-generated end-user outputs directly related to prescription drug use into the drug labeling framework. 10 PDURS clearly distinguishes between the software itself and its end-user output, treating the latter as part of prescription drug labeling for regulatory purposes. This approach reflects a novel, output-centered regulatory pathway and provides a structured mechanism for the deep integration of digital health tools with drug labeling.
In contrast, Canada’s approach to digital drug labeling has primarily focused on the digitalization and structural standardization of statutory drug information. In recent years, Health Canada has advanced reforms to product monographs by introducing structured, XML-based electronic submission requirements, aimed at improving the readability, updatability, and interoperability of drug labeling information across digital systems. 39 This pathway emphasizes modernization at the level of labeling format and data architecture, rather than incorporating dynamic software-generated outputs directly into the scope of drug labeling regulation.
Overall, North America illustrates two representative institutional orientations in the evolution of digital drug labeling. The United States, through the PDURS framework, emphasizes regulatory oversight of software-driven medication-use outputs and their labeling status, while Canada prioritizes the development of a robust digital and structured foundation for drug labeling and product information. 44 Although both approaches share the overarching goal of enhancing the accessibility, safety, and effectiveness of medication information, they differ substantially in regulatory focus, scope, and implementation strategies. Together, these contrasting pathways offer valuable comparative insights for other jurisdictions seeking to develop regulatory frameworks for digital drug labeling.
5.2. Europe: Electronic product information–driven models of digital drug labeling
In Europe, the development of digital drug labeling has primarily progressed through the implementation of electronic product information and related digital health policies, rather than being regulated as an independent category of digital therapeutics. At present, software and digital tools associated with medicinal products that involve medical functions are typically regulated under the European Union’s Medical Device Regulation (MDR, EU 2017/745)45,46 and ePI (electronic Product Information) (The Health Policy Partnership). 37 However, the primary regulatory focus remains on the software as a medical device itself, rather than on drug labeling content.
At the national level, Germany has been at the forefront of advancing the digitalization of drug-related information. Although the DiGA (Digitale Gesundheitsanwendungen) program is mainly designed to provide reimbursement pathways for digital health applications rather than to directly regulate drug labeling, its institutional experience in electronic medication information, patient-facing interfaces, and digital health management offers a practical foundation and valuable policy reference for the future development of digital drug labeling. 47
Against this backdrop, the FDA’s PDURS framework offers European pharmaceutical companies a new policy reference by demonstrating how the deep integration of digital technologies with prescription drugs can enhance therapeutic effectiveness and improve the precision of personalized treatment. The PDURS approach—particularly its regulatory emphasis on treating software-generated outputs as components of drug labeling—may provide valuable insights for the European Medicines Agency (EMA) and national regulatory authorities as they consider the development of similar frameworks in the future. As relevant regulatory systems continue to evolve and mature, European pharmaceutical companies may gain broader policy support at the global level, thereby further promoting the international adoption of digital medicines and intelligent therapeutic tools.14,48
Additionally, many European countries (such as Germany, the UK, Switzerland) have a solid foundation in advancing precision medicine and medication adherence, and the PDURS framework could further improve the efficacy and safety of drugs through personalized treatment and smart monitoring. 48 However, Europe also faces challenges related to privacy and technological acceptance. For instance, European drug regulation and data privacy laws (such as GDPR) differ from those in the U.S., which may create complex compliance issues for multinational companies. 48 Ensuring cross-border data transfer complies with Europe’s strict data protection laws will be a significant challenge, particularly when dealing with patient data.14,48,49
5.3. Asia: Government-led and incremental pathways toward digital drug labeling
Across Asia, countries such as Singapore, Japan, South Korea, and China have pursued government-led and incremental approaches to the development of digital drug labeling. Although these jurisdictions differ in regulatory frameworks, technological readiness, and implementation pace, they generally share the objective of improving the accessibility of drug information, enhancing medication safety, and strengthening regulatory efficiency through the gradual digitalization of traditional drug labels and package inserts.
Singapore has been among the regional leaders in advancing digital drug labeling. The Health Sciences Authority (HSA) has implemented electronic labeling (e-labelling) for selected therapeutic products, enabling patients and healthcare professionals to access electronic versions of product information and patient leaflets via QR codes or online platforms. 41 This approach improves the timeliness and accessibility of drug information while maintaining regulatory compliance, and provides a practical model for the institutional adoption of digital drug labeling.
Japan has adopted a more transformative digitalization strategy. Since 2021, Japan has, in principle, eliminated paper package inserts accompanying medicinal products and replaced them with electronic access to drug information.40,50 This policy has significantly accelerated the comprehensive digitalization of drug labeling and enhanced the efficiency of information updates, while also contributing to environmental sustainability. 50 Japan’s experience represents one of the most advanced examples of institutionalized digital drug labeling in Asia.
South Korea remains at an exploratory stage, relying primarily on pilot-based implementation. Since 2022, the Ministry of Food and Drug Safety (MFDS) has launched e-labeling pilot programs focused on specific categories of medicinal products, particularly injectable drugs used in healthcare institutions. 42 Under defined conditions, electronic formats may substitute for traditional paper inserts. This cautious approach reflects South Korea’s regulatory strategy of validating the feasibility and safety of digital drug labeling through limited-scope pilots.
China has adopted a more cautious, phased reform strategy in the area of digital drug labeling. In recent years, the National Medical Products Administration (NMPA) has promoted initiatives such as pilot programs for age-friendly and accessible drug package inserts, aimed at improving layout design, readability, and digital formatting of drug information. 43 In parallel, technical guidelines have been introduced to standardize the format and content of electronic drug package inserts, thereby establishing an initial institutional and technical foundation for future digital drug labeling systems. Nevertheless, China’s current regulatory framework remains centered on legally approved paper-based labels and package inserts, and comprehensive digital drug labeling policies are still under development.
Overall, Asian countries have generally adopted incremental, pilot-driven reform strategies in the development of digital drug labeling, with a strong emphasis on government leadership, risk control, and regulatory stability. This approach differs substantially from the PDURS framework in the United States, which focuses on incorporating software-generated outputs into drug labeling regulation, and offers an alternative model for the evolution of digital drug labeling policies across diverse regulatory environments.
5.4. Other countries and regions
Developing and underdeveloped countries often face shortages in healthcare resources, and digital health tools within the PDURS framework can help improve drug management efficiency, especially in areas lacking healthcare professionals. Remote monitoring capabilities of digital medicines can help patients in these countries better adhere to treatment plans, reducing misdiagnoses and misuse. The global spread of the PDURS framework could drive international cooperation, helping developing countries gain access to advanced drug and digital health technologies, offering more treatment options through global supply chains.
However, many developing and underdeveloped countries face challenges in terms of healthcare infrastructure and technological support.51,52 The application of digital health tools may face challenges such as poor internet access and lack of technological devices. Additionally, funding shortages and technological gaps are key issues that need to be addressed for successful implementation of the PDURS framework in these regions.51,52
Comparative analysis of PDURS-related opportunities and challenges across regions.
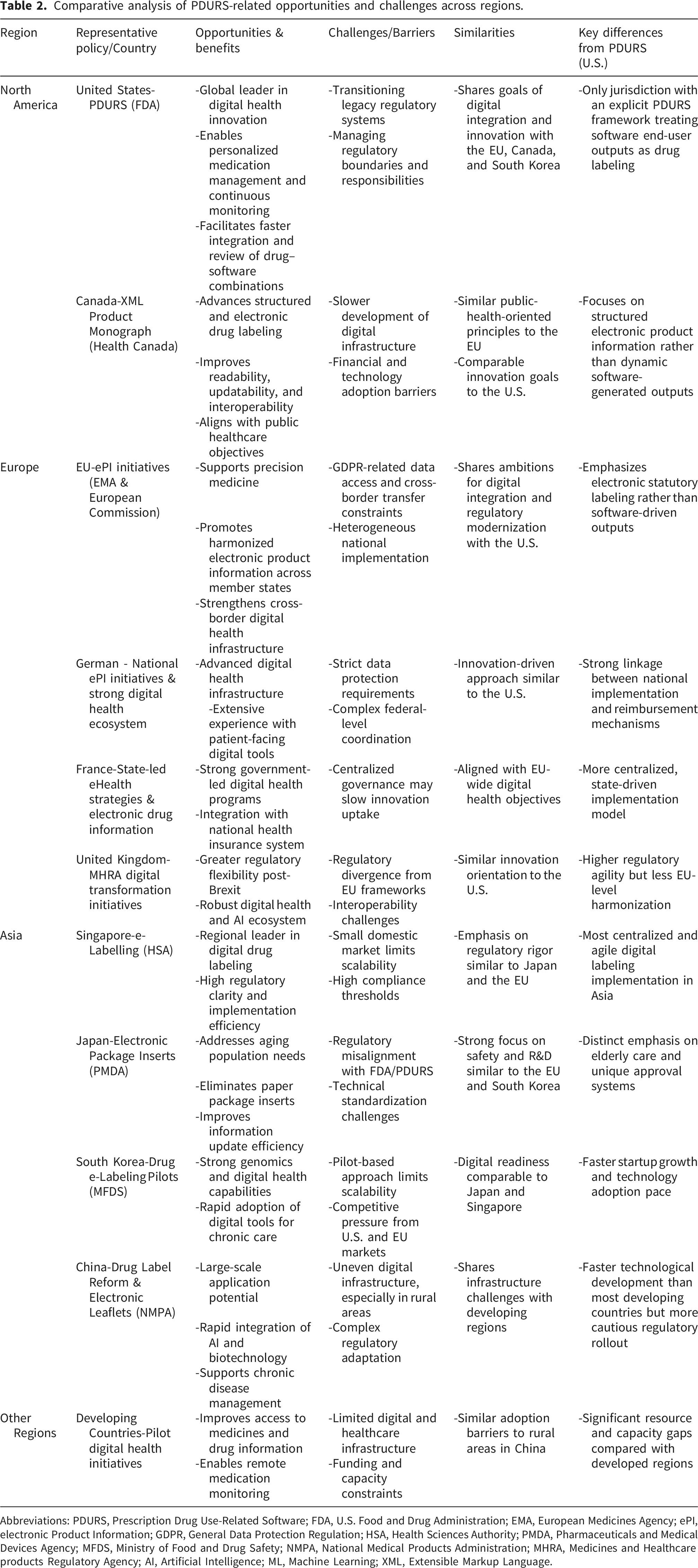
Abbreviations: PDURS, Prescription Drug Use-Related Software; FDA, U.S. Food and Drug Administration; EMA, European Medicines Agency; ePI, electronic Product Information; GDPR, General Data Protection Regulation; HSA, Health Sciences Authority; PMDA, Pharmaceuticals and Medical Devices Agency; MFDS, Ministry of Food and Drug Safety; NMPA, National Medical Products Administration; MHRA, Medicines and Healthcare products Regulatory Agency; AI, Artificial Intelligence; ML, Machine Learning; XML, Extensible Markup Language.
6. Conclusions
This policy review examined the evolution and emerging implications of the FDA Prescription Drug Use-Related Software (PDURS) framework. By adopting an output-centered regulatory approach in which certain sponsor-disseminated software-generated outputs are treated as prescription drug labeling, PDURS facilitates deeper integration of digital health technologies with pharmacotherapy. The framework provides new mechanisms for medication management and precision treatment, with potential to enhance therapeutic safety, improve drug development efficiency, and strengthen data-driven innovation in the pharmaceutical industry.
Internationally, PDURS offers a reference model for digital drug labeling governance. While the United States explicitly incorporates software-generated medication-use outputs into labeling regulation, other jurisdictions primarily advance digitalization through structured or electronic product information systems, and many regions remain in exploratory stages. Despite shared goals of improving medication information accessibility and safety, implementation faces technological, legal, and institutional challenges, including data governance constraints, regulatory coordination, infrastructure disparities, and accountability for software output updates. Continued international collaboration and technical development will be necessary to support sustainable expansion of PDURS-related approaches in precision medicine and chronic disease management.
Footnotes
Acknowledgements
We thank Dr. Rajeev K. Singla (West China Hospital, Sichuan University) for his valuable contributions to the discussion of the international impact of the PDURS framework.
Author contributions
B.S. conceived the project. B.S., Y.Z. designed the overall framework of the comments. Y.Z. drafted the initial manuscript. L.S., S.H., R.W., and J. W. critically reviewed the structure and content, providing substantial feedback and revision suggestions. H.Z. and A.U. contributed to literature research and assisted in refining the manuscript. X.L., R.W., A. C., D. F., S. W., and G. Z. provided further insights and revisions. All authors discussed the content, contributed to the interpretation of findings, and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (32570773, 32270690) and the Sichuan Science and Technology Program (2024YFHZ0205).
Declaration of conflicting interests
The authors declare no competing interests. In addition, all authors declare that no artificial intelligence tools were used for content generation in this manuscript; however, GPT-4.0 and GPT-5.2 were used solely for grammar correction and language editing.
