Abstract
Digital therapeutics (DTx), a burgeoning subset of digital health solutions, has garnered considerable attention in recent times. These cutting-edge therapeutic interventions employ diverse technologies, powered by software algorithms, to treat, manage, and prevent a wide array of diseases and disorders. Although DTx shows significant promise as an integral component of medical care, its widespread integration is still in the preliminary stages. This limited adoption can be largely attributed to the scarcity of comprehensive research that delves into DTx's scope, including its technological underpinnings, potential application areas, and challenges—namely, regulatory hurdles and modest physician uptake. This review aims to bridge this knowledge gap by offering an in-depth overview of DTx products’ value to both patients and clinicians. It evaluates the current state of maturity of DTx applications driven by digital technologies and investigates the obstacles that developers and regulators encounter in the market introduction phase.
Keywords
Introduction
Digital therapeutics (DTx) are an emerging type of medical therapy. The Digital Therapeutics Alliance (DTA) formally defines DTx as “
In recent research, DTx have been identified as a promising alternative to pharmacological treatments across various medical conditions, highlighting their potential to change current treatment models. For example, in the case of attention-deficit hyperactivity disorder (ADHD), DTx offer a new approach to management, with clinical evidence showing significant promise. 7 Additionally, DTx has been found to provide appealing alternatives in other domains, such as enhancing walking outcomes, 7 delivering cognitive behavioral therapy (CBT) for insomnia, and managing Parkinson's disease, 8 among the others. For some conditions, DTx could become a standalone alternative to pharmacological interventions, 9 while for others they could be used to complement medication,5,9 but in both cases the therapeutic effect should be clearly identifiable and measurable. Since DTx are considered one of the most innovative areas within digital health, startups around the world are working to develop and validate several DTx products, and regulatory bodies have also started to explore how to navigate regulatory pathways. 10
Despite early evidence of the potential of DTx and the variety of solutions that are rapidly entering the commercial markets of the United States and Europe, benefits and challenges are only recently being systematically studied by academics, regulators, and market analysts. In fact, while there is a proliferation of novel DTx solutions, their true potential is still in the early stages of exploration, so that manufacturers, healthcare providers (HCPs), patients, and regulators may not fully grasp their opportunities and challenges. Specifically, there are several points that remain under researched and contribute to a lack of comprehensive understanding of the DTx phenomenon, impacts, and challenges.
We identified four main areas that require further investigation (Figure 1). First, the current regulatory landscape for DTx is notably unclear and insufficient, lacking a definitive framework that ensures usability and sustained adoption. Secondly, uncertainties regarding patient safety still exist, which hinder the integration of DTx into clinical practice. Third, there is insufficient evidence supporting DTx effectiveness and clinical impact, which further limits the broader adoption of these solutions. All this also extends to the propensity of patients, HCPs, and payers in embracing these therapeutic approaches. Last, considering that we face a high variability of technological solutions that might enable DTx, we lack a comprehensive picture of the technologies that are currently used to enable such therapies as well as the manners in which they support therapeutic treatments in different cases. Consequently, all the clinical, regulatory, and organizational open points need to be addressed considering their unavoidable interrelations with the technological enablers that make DTx possible.

Key challenges to widespread adoption of DTx: four critical areas for further investigation.
Therefore, the ambiguity and uncertainty surrounding DTx suggests a lack of research that comprehensively analyzes this concept and its foundational building blocks, considering technological enablers, potential application fields, and the challenges that must be overcome to foster adoption. In response to this research gap, with this review paper, we aim to provide an overview of the value of DTx products for patients and clinicians, the maturity of DTx applications enabled by digital technologies, and the challenges that developers and regulators face during the go-to-market process. Specifically, we aim to address the following points: (1) What are the novel distinctive factors and peculiarities of DTx compared to other digital-enabled applications in the medical field? (2) What are the technologies involved in DTx interventions? (3) What are the challenges faced by manufacturers, HCPs, patients and regulators? (4) How ready is the current regulatory landscape in different countries to allow DTx's diffusion?
In so doing, this paper contributes to the DTx literature and community in the following ways:
Methods
In order to answer our research questions, in this paper we review articles from PubMed Central and ClinicalTrials.gov databases, as well as from Google Scholar search engine and global regulatory authority releases. Articles covered the period from 2015 to 2023. Databases were queried using the search terms that included a combination of (“digital therapeutics” OR “digital therapy”) AND (“technology” OR “challenge” OR “regulation”). Initially, we chose the keyword “technology,” as also discussed by Sverdlov et al. 10 to identify the most discussed technological enablers for the implementation of this treatment modality for patient care. Then, the keywords “challenge” and “regulation” were used to search for roadblocks during the go-to-market process as suggested by Patel and Butte. 5 In total, we analyzed 74 articles from journals and clinical trial reports, 13 sources from global authority releases (e.g. US Food and Drug Administration (FDA), The European Parliament) including DTA publications, and finally 1 DTx company report to support our findings.
Digital therapeutics: the state of the art
Key concepts and current applications of DTx
DTx are a type of product within the broader category of digital medicine, which is itself a subset of the larger field of digital health. Dang et al. 11 summarize these categories as described in Figure 2 and in the following paragraph.
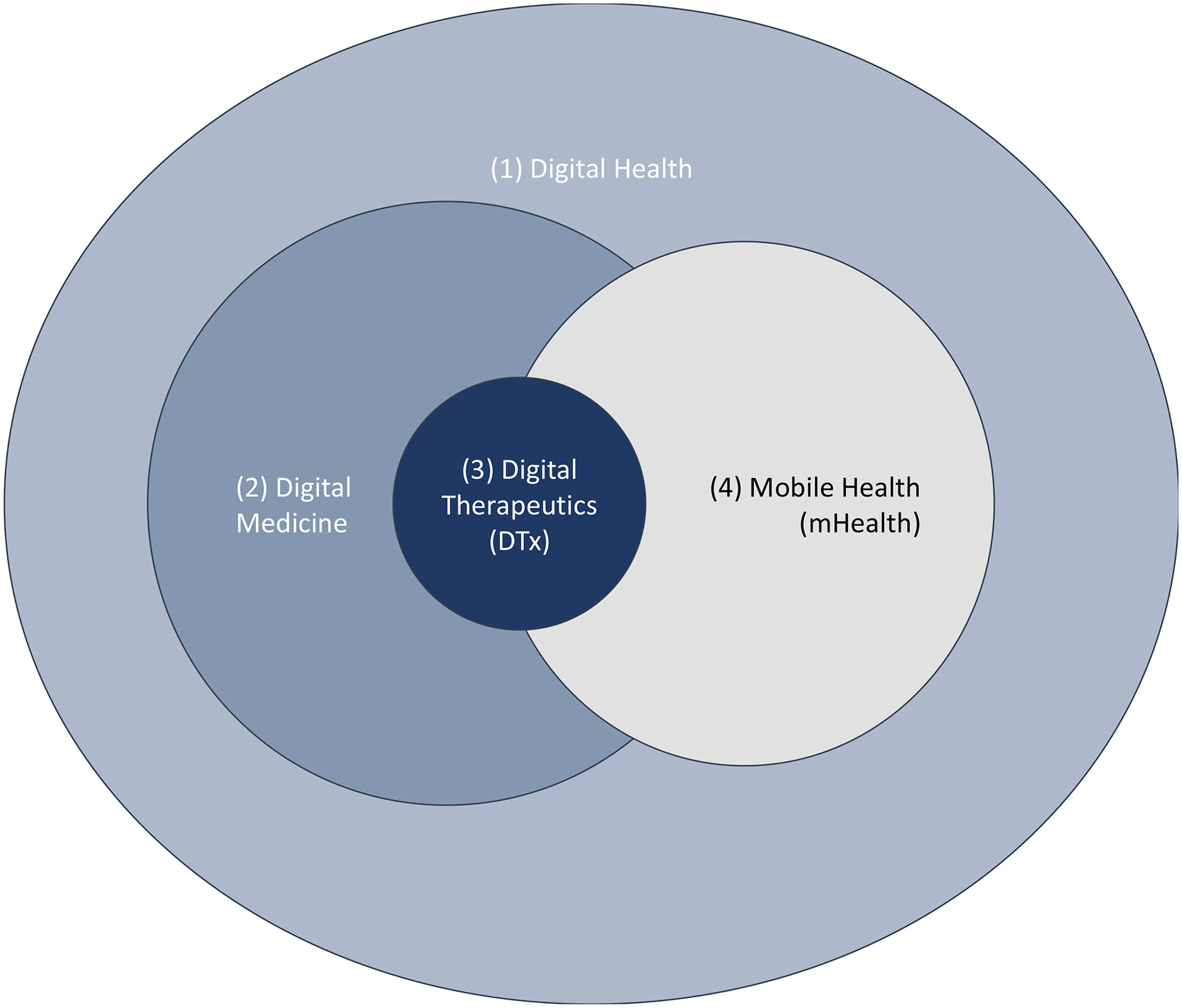
Definitions: digital health, digital medicine, digital therapeutics, and mHealth.
Digital health is a broad term that encompasses various technologies, platforms, and systems designed to involve individuals in activities related to their lifestyle, wellness, and overall health. Digital health entities are capable of capturing, storing, and transmitting health data to assist with clinical operations. These systems include health information technologies, telehealth systems, tools that leverage consumer health information, clinical care management software, and various other types of digital health solutions. Digital medicine, instead, is a term used to describe software or hardware products part of Digital Health that are backed by evidence and are intended to measure or intervene in human health. It includes digital diagnostics, digital biomarkers, remote patient monitoring devices, DTx, and other similar products. Finally, as previously defined, DTx refer to evidence-based therapeutic interventions that are used to prevent, manage, or treat medical disorders or diseases. Consequently, DTx appear as a specific subcategory of Digital Medicine, within the broader field of Digital Health.
DTx can be delivered in a variety of digital formats that are accessible to patients, including smartphone applications, video games, VR programs, and other similar platforms. Advancements in technology and a growing trend toward personalized and participatory approaches to health have led to significant progress in the field of DTx in recent years. These developments have spurred the growth of DTx solutions and improved their accessibility to individuals seeking personalized care options. 11
A part of DTx that are delivered through mobile technology can also be considered as falling within the adjacent field of mHealth.
As a consequence of their complementary but different scopes, digital health, digital medicine, and DTx have varying requirements for clinical evidence and regulatory oversight. These differences arise due to varying levels of claims and associated risk levels. 14 In fact, DTx tend to be more sophisticated digital health products, which are now more akin to medicines rather than simple applications or apps. Moreover, given that a key characteristic of DTx is the delivery of a well-defined and measurable therapeutic intervention, such interventions can be assessed both in experimental and real-world settings to determine its impact as a stand-alone effect.
Given these peculiarities, Fürstenau et al.
13
highlight dimensions that distinguish DTx from other healthcare technologies as below:
So far, DTx have evolved as a complement of existing interventions (e.g. use of DTx in addition to medication-assisted treatment in opioid use disorder),
15
and in the future, in some cases, they might even become a full alternative to some of them. Herein, DTx are becoming an effective tool to remotely collect real-time data from patients through wearables and other sensors,
8
use data analytics and AI to understand patient's behavior and condition and communicate these conditions to HCPs to create more informed care plans.16,17 These technology-enabled DTx services complement and add to the value of traditional healthcare delivery systems, and have the potential to deeply change not only patient experience,
11
but also patients’ behaviors and, ultimately, the effectiveness of healthcare interventions. At the same time, DTx address some of the current issues of traditional healthcare systems, including the shortage and the burden on HCPs.
18
For example, a hypertension DTx app could educate patients on their lifestyle modification, including correcting their lifestyle habits and measuring daily blood pressure values, which allows physicians not only to obtain better results but also to objectively evaluate behavioral changes that occur in the patient's home via app, thus saving time and adopting prompter decisions.
19
This app can ease the burden on HCPs who might currently not have enough time to provide sufficient lifestyle guidance for each patient's effective hypertension management. While this case is an example of individual patient-level use, real-world outcomes generated by DTx may also be used for optimizing outcomes at the population level. Indeed, data can be aggregated and used to track progress or compare outcomes based on patient disease state, level of acuity, age, and gender.2,20 This might generate more general benefits, as healthcare systems typically collect more data on process and output than on behaviors and outcomes.
To deliver these results, DTx can leverage a potentially large pool of enabling digital technologies. According to the study performed by Santoro et al., 4 the majority of DTx interventions (41.9%) are currently delivered through mHealth applications. This is followed by web-based systems (25.7%), videogames (8.8%), VR (4.4%), text messages (3.7%), and others. In addition, growing debate is considering the convergence between DTx and more advanced digital technologies, such as AI, analytics, and VR. Despite these initial efforts, the extent to which these technologies are integrated into DTx applications remains in the early stages.1,3,4 For instance, rather than relying on AI-enabled predictions, the majority of DTx interventions involve software using generic fixed algorithms that do not change dynamically from patient to patient and do not consider unique personalized patient symptom data. In the case of VR instead, key challenges relate to technical hurdles (i.e. discomfort for users) and ethical issues (i.e. users’ privacy due to the acquisition of users’ behavioral biometrics data), that play a key role in hindering the adoption rates of VR in this field.3,21
Thus, the next section aims to advance our understanding of the state of the art of the different technological enablers of DTx by offering an overview of their characteristics, applicability, and respective challenges.
Enabling technologies and tools for DTx applications
In this section, we start by discussing the state of mHealth applications, as they are currently widely adopted for DTx. 4 Second, we provide insights on AI integrations into DTx products, since this might lead to a customized and more tailored therapy regimen, as suggested by Palanica et al., 1 Finally, we discuss VR-enabled DTx applications due to their expanding role in this field. 21
Mobile applications are indeed used for a variety of tasks such as providing feedback to reinforce positive behavioral change (e.g. physical activity, food intake, 24 medication adherence, 25 and substance consumption 26 ), frequently adjust individually tailored programs, improve access to established therapies (e.g. CBT), and support ongoing connections with healthcare professionals through messaging features. 27 Moreover, as mHealth apps have greater flexibility, they become a more accessible, low-cost, and user-friendly alternative to traditional interventions.8,28 For instance, mobile DTx platform Clickotine, helps smokers quit using a customized plan and scientifically based strategies to overcome cravings as well as social support and direct access to quit aids.26,29
Moreover, app-based DTx interventions are effective for several common mental health problems such as improving depressive symptoms, anxiety symptoms, stress levels, general psychiatric distress, and quality of life. Although mental health apps are not meant to replace professional clinical services, the present findings highlight their potential to serve as a cost-effective, easily accessible, and low-intensity intervention for the millions of people worldwide who cannot receive standard psychological treatment. There is also huge potential for real-world data generation since apps allow continuous real-time data collection on the patient's phone, leading to the personalization of the care process and to the reduction of bias in healthcare databases and algorithms. 28
DTx relying on AI as a component of treatment can benefit from the “learning” feature typical of this technology that can help extract patterns from large datasets (e.g. data collected from a patient over time) and generate predictive models for the management and treatment of conditions, thus potentially increasing the effectiveness of digital interventions through a customized therapy regimen.5,30 In fact, since AI systems can constantly collect data from the patient and his/her environment (e.g. engagement and biometric data), they can be programmed to change the delivery of treatment in response to outputs of algorithms,1,30 and optimize data input to personalize outcomes. AI integration could further differentiate DTx from other forms of therapeutics, by enabling a more personalized form of healthcare that actively adapts to patients’ individual clinical needs, goals, and lifestyles. 1 For instance, an AI-powered chatbot (i.e. software that utilizes natural language processing (NLP) to create an interface that emulates human discourse, and elements of human relationships) can be used to provide CBT, medication adherence reminders, or online exercise and diet programs. 1
Table 1 presents some of the DTx applications that currently integrate AI. These product/company examples were selected from our DTx database.
Overview of various DTx products and their convergence with artificial intelligence.

CBT: cognitive behavioral therapy; IPT: interpersonal psychotherapy; IBS: irritable bowel syndrome.
While AI holds great promise for digital interventions, today AI-enabled DTx applications can be considered in the early phase due to a limited number of commercialized products and limited evidence from academic and clinical applications.1,30 Additionally, privacy and ethical concerns related to patient data acquisition, processing, and governance, and cultural resistance to AI-based decision-making are some of the limitations that remain to be addressed. 41 For instance, considering chatbot-based interventions, the lack of empathy and professional human approach is reported as an undesirable feature by some users. 42
One additional relevant drawback of AI systems is the need to create a representative, diverse dataset to build safe and efficient algorithms for AI-enabled DTx interventions. Indeed, the performance of healthcare algorithms is affected by social bias and other biases in health training datasets.43,44 Methods to recognize and remove such biases are paramount to ensuring the delivery of equitable healthcare and promoting transparency in AI-enabled DTx. To address this issue, developers, users, and regulators need to work together to leverage large and diverse datasets to improve accuracy while balancing privacy and regulatory requirements. Critically, clinicians should carefully limit overreliance on these systems in decision-making, understanding the limitations of AI-enabled DTx decision model interpretability. Special consideration is even more needed for those AI applications targeted directly to patients, which is mostly the case for DTx interventions, where patients may not be in the appropriate position to judge if the suggested action is reasonable. 43
Overview of various DTx products and their convergence with virtual reality.

In the realm of VR technical possibilities, potential further development with positive repercussions on DTx could come from the integration of VR with AI, that might further increase the “
This field is new and rapidly evolving, and as such its risks and benefits are continuing to be studied. Nearly every study that has been conducted on these technologies points out that more research is required to answer key questions, including effectiveness and safety.21,52
The role of regulation and the key challenges of DTx
In the evolving landscape of DTx, certain countries are making efforts to ease the integration of these medical solutions into the healthcare system, despite the regulatory challenges akin to those faced by traditional medical devices. Notably, the United States and Germany have emerged as frontrunners in this regard. In the United States, the FDA is actively working to simplify the regulatory process for Software as a Medical Device (SaMD). 61 This initiative might accelerate the adoption of DTx by streamlining the approval process for these technologies. At the same time, Germany has taken significant steps to incorporate digital health applications (DiGAs) into its healthcare system. 62 German doctors are now authorized to prescribe DiGAs to patients, who can then have these prescriptions covered by health insurance. This approach not only legitimizes the use of DTx but also enhances their accessibility to a broader patient population.
In this scenario, DTA is one of the global non-profit associations leading the discussion and development of harmonized pathways for the recognition and scalability of DTx at the local, national, and regional levels.
Nevertheless, despite these efforts, there is not a global regulatory framework for DTx. This absence may be attributed, in part, to the inherently dynamic nature of DTx themselves. As outlined in the previous section, DTx are exploiting new emerging technologies, undergoing constant evolution to leverage the latest technological advancements. This perpetual state of evolution presents a challenge in establishing a universally applicable regulation for DTx. Furthermore, distinguishing DTx from pharmaceuticals and traditional medical devices is their capacity for frequent updates and enhancements. This characteristic introduces an additional layer of complexity for regulatory bodies. The challenge lies in developing a regulatory mechanism that can effectively manage and monitor the rapid evolution inherent to DTx, ensuring both their efficacy and safety over time. 5
Indeed, it appears that the regulatory landscape is complex, evolving quickly, and different in each country.
17
This section aims to provide a comprehensive examination of this issue, beginning with an overview of the current regulatory landscape for DTx in the EU and the US. Subsequently, it delves into the challenges associated with the socio-ethical implications of DTx, highlighting the intricate balance between innovation and ethical considerations in the deployment of these technologies.
(a) The state of regulation in European Union (EU) and United States (US)
Regulatory framework in the EU
DTx are covered by EU regulation 2017/745 on medical devices, 63 which came into force in May 2021. The EU Medical Device Regulation contains no specific provisions for DTx and the approach to be taken by notified bodies concerning regulations applicable to DTx. 64 Although in some European countries, several procedures have been set up for the marketing authorization and reimbursement of DTx, the European regulatory system concerning DTx remains uncoordinated, and specific regulations aimed at evaluating DTx tools and ensuring the safety of the devices are lacking.65,66
Since the development and delivery features of DTx are similar to those of conventional drugs, evidence of efficacy and safety must be provided before they can be authorized for marketing. The evaluation of the efficacy and safety of DTx should be based on high-quality controlled clinical trials carried out according to the standards required by regulatory agencies.
65
Indeed, the lack of specific regulations to guarantee the safety and quality of these devices is a further obstacle to the development of DTx.17,64 In addition, considering that Europe is neither a single unified market nor does it have mutual recognition of certification, a parliamentary question titled “
Given that Germany has taken the lead on introducing many DTx products to the market since 2020 (enabling fast-track authorization under controlled conditions), its model could be considered a valid, useful template for other European countries to enable innovation and adoption of DTx. 67 For this reason, the next session is dedicated to the in-depth analysis of the regulation in Germany.
Regulatory framework in the EU—the case of Germany
In recent years, Germany has established itself as a pioneer in digital health innovation in Europe, 68 with 73 million German citizens covered by public health and DTx apps approved for reimbursement. The country that paved the way for DTx has inspired other European countries—that is, Belgium and France. 67
At the end of 2019, Germany implemented the Digital Healthcare Act (Digitales Versorgungsgesetz, DVG) which established a specific pathway for the reimbursement of digital health offerings. This act aimed to enhance transparency in the approval process for manufacturers, physicians, and patients in Germany. Under the fast-track process, DiGAs can be prescribed by doctors and reimbursed by health insurance companies. In order to gain access to the market, developers must register their CE-marked device (class I or IIA) at the Federal Institute for Drugs and Medical Devices (BfArM), and complete the compliance process of a DiGA with general requirements (e.g. app security, quality, functionality, data security, and data protection) and its positive healthcare effects (i.e. medical benefit or improvements of care structure and processes). To gain a permanent listing in the DiGA directory (DiGAV), the developer must submit such evidence and undergo review. As of November 2022, the approved DiGAs in Germany cover a wide range of medical fields including diabetes, obesity, depression, anxiety, insomnia, and back pain therapy. 62
Amid this environment in Germany, the prescription rate remained low.69,70 A study performed among rheumatologists in 2022 reports that only 7% have already prescribed a DiGA and 46% planned to do so. 69 This is aligned with another research held in 2021 among members of the German Respiratory Society, where 47.2% HCPs had prescribed or planned to prescribe DIGA. 70 This further highlights that, even in a positive environment like Germany's, the adoption of DTx remains very low.
Regulatory framework in the US
Currently, the US is the driving force in the world for the development and commercialization of DTx. 64 The FDA has issued many temporary policies to support digital health innovation during the Covid-19 pandemic, particularly guidance documents to expand the use of DTx, especially with regard to mental disorders like depression 71 and chronic ailments like diabetes. Today, FDA is leading in formally approving DTx, including the most recent and innovative interventions.
From a regulatory point of view, DTx products in the US are considered within the SaMD category. 61 Based on the DTx's intended use and level of risk, each product is subject to varying degrees of oversight, ranging from full 510(k) clearance by the FDA's Center for Devices and Radiological Health (CDRH) division to enforcement discretion.2,72 Beyond direct approval processes, the FDA has also recently established the Digital Health Center of Excellence (DHCoE) in September 2020 with the intent to speed the innovation of safe and effective digital technologies. 73 DHCoE acts as a central authority intended to manage and speed digital innovation (including SaMD, wearables, mobile health devices, AI, and ML that may be built into SaMD and medical applications).
While these incentives are helpful, the FDA and other regulatory bodies still need to adapt to the speed of technology and establish clear parameters for regulation and enforcement.
(b) Socio-ethical issues during the design and implementation stage of DTx.
The utilization of DTx is linked to several socio-ethical barriers among stakeholders, encompassing factors such as infrastructure limitations, health inequalities and bias, and liability issues. This section outlines the key socio-ethical issues that arise during the design and implementation stages of DTx.
74
A significant concern is the potential challenges posed by the digital divide (limited access to technology) and digital literacy (lack of technological literacy). These factors could have negative implications for the adoption of DTx, adherence, and diffusion. Existing literature underscores that digital literacy skills in the healthcare context are associated with various health behaviors, including maintaining a healthy diet, engaging in exercise, and managing sleep. Furthermore, associations have been observed between digital health literacy and factors such as the presence of chronic illness, perceived self-management skills, and an enhanced self-perceived understanding of health status, symptoms, and optional treatments. Therefore, fostering digital literacy and addressing concerns related to the digital divide can positively impact perceptions of DTx, subsequently increasing the intention to use DTx. 77
Furthermore, this situation raises liability concerns, posing the question of who ultimately holds responsibility for patients’ health: the AI software or the prescribing physician of the DTx system. This adds complexity to the issue of accountability. 76
(c) Other challenges
Given the wide content and applications of DTx, it is unlikely that there will be a one-size-fits-all approach for DTx regulations and ethics. Building on the previously discussed findings, our research has identified challenges beyond regulatory and ethics, including (1) the design of the clinical trials and evidence generation methods in DTx seeking regulatory approval6,78; (2) the limited evidence and a lack of understanding of efficacy related to the digital mode of delivery79,80; (3) adherence/compliance with DTx products and engagement support by HCPs.78,79
Other key solutions are to design DTx tools to increase adherence through both product features and to embed DTx treatment in the patient's care pathway. Key considerations on the patient's side include optimizing for user-friendliness, incorporating tailoring and personalization, integrating gamification elements, utilizing reminders, and ensuring patients comprehend the value of DTx for their health. Lastly, it is crucial to establish convenient methods aligned with existing practice operations and workflows to enable HCPs to monitor the usage of DTx.
Due to the relatively recent emergence of DTx, there has been limited research into approaches for introducing technologically advanced DTx solutions to the market, staying aligned with regulatory bodies for approvals and reimbursements, and promoting the adoption and adherence of DTx. Moreover, considering the therapeutic nature of these digital interventions, there is a need for additional research on the ethical implications specific to this field, beyond the considerations for general digital health products.
Figure 3 provides an overview of the main identified challenges.
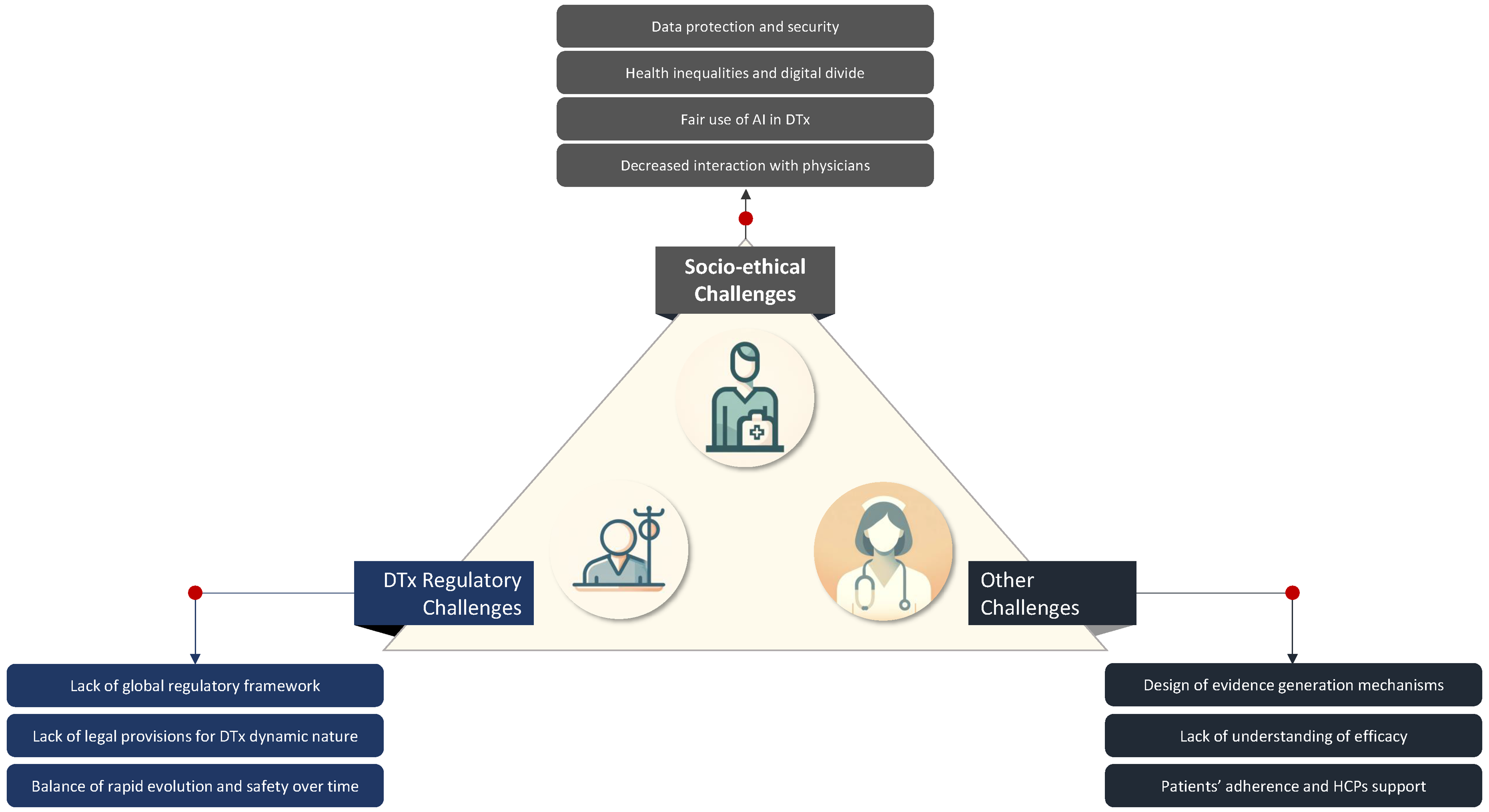
Overview of the main challenges of DTx clustered in regulatory, socio-ethical, and other challenges.
Discussion
The advent of DTx has promised to move healthcare to another and more contemporaneous level. DTx offer potential solutions for treatment gaps, enhancing patient engagement, and are a key tool for personalizing medicine to an unprecedented level. However, the path to integrate these digital solutions into healthcare systems is laden with challenges that need to be addressed, including uncoordinated, non-specific and evolving regulations, lack of a universally accepted definition, inadequate ad-hoc evidence of clinical efficacy, and data protection concerns. Until recently, research on the domain of DTx has focused on several relevant aspects that need to be addressed to foster the diffusion of this novel form of therapeutics. Studies have attempted to conceptualize and define DTx,1,2,5,6 provide early evidence of the effectiveness of DTx treatments in specific domains, thus investigating on their potential to evolve treatment models,5,7–9 and deep dived into the technological enablers of these digital therapies.4,5,21 Moreover, literature has explored various types of challenges surrounding the diffusion of DTx. Among these, regulatory uncertainty, socio-ethical concerns, and several concerns related to the effectiveness and acceptance of DTx by patients and HPCs. Notwithstanding the several streams of research that are focusing on DTx from these separate perspectives, evidence from this review paper shows that such concepts and challenges need to be examined and addressed with a comprehensive approach. Therefore, our review paper collects and organizes previous research with specific vertical focuses and aims to introduce managerial lenses to support the diffusion on DTx.
In particular this paper provides three main contributions. First, we provide a definition of DTx based on a synthesis of diverse definitions. Second, we offer an extensive discussion of new emerging technologies and their integration in this field. Third, we present a comprehensive overview of the challenges for DTx adoption. Table 3 provides an overview of the three main findings, their descriptions, and the contributions to the literature compared to the existing discourse on this topic.
Overview of findings and contribution.

First of all, one of the fundamental gaps in the discourse on DTx is the lack of a universally accepted definition. This is important not just from an academic standpoint, but also as a crucial aspect for regulating, evaluating, and reimbursing these products. Indeed, the DTx nature is not so obvious: from a functional perspective, they are in many instances similar to pharmaceuticals (with the active ingredient being a digital solution instead of a chemical/biotechnological entity) but from a technological perspective, they are generally embodied into medical devices. As a consequence, the ambivalent nature of DTx is not fully understood using the traditional categories of drugs and medical devices, leading to a heterogeneous perception of what they are. This lack of consensus can lead to confusion among stakeholders, including patients, HCPs, regulators, and payers. It is crucial that stakeholders, led by international health bodies, come together to establish a shared understanding of what constitutes DTx, which should reflect the dynamic nature of this emerging field and cover all aspects from intended use to functionality and software requirements.
Second, most of the technological discourse around DTx focuses on mobile apps that enable new DTx. While this aspect remains crucial, integrating advanced technologies is essential for developing new features. For instance, this paper identifies the expanding role of AI in DTx products and the use of VR to deliver DTx. These technologies offer unique opportunities but also complicate the adoption process of DTx. Indeed, the adoption of AI and VR introduces new regulatory and socio-ethical issues. In particular, AI-enabled DTx pose a challenge for regulatory bodies, which must determine how to regulate algorithms that primarily rely on learning from data and adaptation. While their application is currently limited, tools like general-purpose large language models (LLMs), medical AI models (such as Med-Gemini), and generative AI systems (GenAI) are attracting increasing attention in healthcare.83,84 Since ChatGPT's introduction in late 2022, GenAI has sparked both great enthusiasm and serious concerns. 85 To date, GenAI applications are mainly being evaluated for screening and diagnostics, such as predicting disease outcomes and improving diagnostic accuracy. GenAI also shows potential in care delivery processes and analyzing data for personalized medicine. Interest for the applicability of LLMs and GenAI is increasing also in the DTx field, but in general application remains limited and evidence of its use in health systems is lacking. Future research is expected to explore AI long-term impact, patient-centered outcomes, and scalability, as well as its impact on the treatment decisions and delivery. 86 As a result, discussions on regulating this field are expected to intensify, together with reflections on AI integration in DTx. Regarding VR, it is an essential technology to monitor, especially with the emergence of the Metaverse. While VR has not yet had a significant impact, it has the potential to be widely adopted in the coming years. This adoption could profoundly influence the field of DTx, creating new opportunities for innovation and patient care.
Third, this review identify a broad spectrum of challenges for DTx adoption in terms of regulatory, socio-ethical, the design of evidence-generation mechanisms, lack of understanding of efficacy, and patients’ adherence and HCPs support. Primarily, regulatory issues pose significant challenges to the development and adoption of DTx. The regulatory landscape for DTx is currently fragmented and evolving. In the EU, for instance, there is a lack of specific provisions for DTx, leading to an uncoordinated regulatory system. The closest regulation is the one on medical devices, but DTx are not explicitly mentioned, and this has slowed down most Member States’ adoption of ad-hoc regulatory pathways for DTx. Contrastingly, the US FDA has been more proactive, issuing guidance and even establishing the DHCoE to promote digital health innovation. Germany, too, has established a pioneering model for DTx regulation and reimbursement, which could serve as a blueprint for other countries. Nonetheless, regulatory harmonization across jurisdictions is required to facilitate the global expansion of DTx and ensure that patients worldwide can benefit from these innovative therapies. Moreover, the utilization of DTx is linked to several socio-ethical barriers among stakeholders, encompassing factors such as infrastructure limitations, health inequalities and bias, and liability issues. One of the significant concerns is that efficient DTx implementation fundamentally requires adequate health literacy and digital competency, such as the ability to use digital devices and understand information. Germany offers a relevant example. Despite establishing a pioneering model for DTx regulation and reimbursement, the prescription rate remains low. One possible reason is that HCPs and patients are not yet ready for this paradigm shift, in particular, due to patients having low health literacy and low confidence in using digital information for health decisions.87,88 Furthermore, the sensitive nature of health data collected and processed by DTx raises significant data protection and cybersecurity concerns. The potential for data breaches and cyberattacks necessitates robust data protection regulations and cybersecurity measures specifically tailored for DTx. Lastly, the absence of robust clinical evidence demonstrating the efficacy and safety of DTx is a further impediment to their adoption. The design of clinical trials for DTx needs to be tailored to their unique characteristics, with the choice of control and metrics for outcome measurement often needing to be customized on a case-by-case basis.
Overcoming these challenges will require a concerted effort from all stakeholders. For example, international health bodies could lead the charge in defining DTx and coordinating regulatory approaches, while developers could work closely with regulators to design suitable clinical trials and robust data protection measures. Meanwhile, HCPs could play a crucial role in promoting patient adherence to DTx, thus generating the real-world evidence needed to demonstrate their efficacy.
Limitations and future research
While this paper offers contributions to both academic research and practical applications, it is important to recognize its limitations. These limitations not only inform the evaluation of the findings presented in the paper but also pave the way for future research directions. The main limitations are divided into two groups: methodological limitations and uncovered areas of analysis.
First, from a methodological standpoint, as presented in section “Methods,” this paper is based on the analysis of 74 articles from journals and clinical trial reports, 13 sources from global authority releases, including DTA publications, and 1 DTx company report to support our findings. The selection process involved identifying relevant keywords from previous studies,5,10 which guided our research. While our analysis distinguishes itself from past research, we invite future studies to extend this review by incorporating new keywords that may reveal additional insights. Moreover, in the analysis of this paper, we adopted a manual review process. However, future researchers could implement more advanced clustering techniques, such as those based on NLP, to enhance the analysis and eventually identify additional findings and contributions.
Second, the paper identifies significant challenges for DTx, notably regulatory hurdles and the lack of robust clinical evidence. These challenges are particularly critical as they impact the adoption and efficacy of DTx. By highlighting these issues, this paper contributes to the academic discourse and opens new research avenues. Future research should delve deeper into these challenges, focusing on strategies to overcome regulatory barriers and enhance clinical evidence. Addressing these gaps will be crucial for advancing the field and ensuring the successful implementation of DTx solutions. Finally, this paper does not address the economic impact, whether positive or negative, of adopting DTx. We acknowledge the importance of this aspect and believe it is a crucial component that warrants detailed examination. Future research should investigate the economic implications of DTx adoption, considering factors such as cost-effectiveness, potential healthcare savings, and economic barriers to implementation. Understanding the economic impact will provide a more comprehensive view of DTx and support informed decision-making in healthcare policies and practices.
Conclusion
While there are great examples of technologically advanced DTx solutions emerging in the market, the road to widespread adoption is still paved with obstacles. To synthesize our findings and bridge the gap between DTx suppliers and patients, we propose five recommendations: First, we advocate for promoting a convergence of DTx definitions. International health bodies should lead the process of establishing a universally accepted definition that encompasses all aspects of these novel solutions, clarifying their hybrid nature. Second, there is a need to harmonize regulatory approaches. Regulatory bodies across different jurisdictions should collaborate to standardize regulations applicable to DTx, facilitating their global expansion and ensuring worldwide patient access. Corresponding reimbursement rules and models should also be considered. Third, we suggest designing tailored clinical trials. DTx developers should collaborate closely with regulators to develop customized methods for clinical trials that consider the unique characteristics of DTx. This entails creating customized controls and metrics for outcome measurement to accurately assess efficacy and safety, consistent with future DTx regulations. Fourthly, there is a crucial need to establish ad-hoc data protection and cybersecurity measures. Given the sensitive nature of health data collected and processed by DTx, developers must prioritize robust data protection and cybersecurity safeguards. Regulatory bodies should provide clear guidelines and requirements in this area. Finally, promoting HCPs engagement and patient adoption and adherence is essential. HCPs should receive training and encouragement to promote access to DTx and encourage patient adoption and adherence. Their active involvement can bolster patient confidence in DTx and generate real-world evidence necessary to demonstrate efficacy.
Footnotes
List of Abbreviations
Contributorship
Paper conceptualization and review: P.A., A.G. Literature review: I.P. Manuscript writing: P.A., I.P., L.M.D.R., L.D., S.M. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded through SDA Bocconi's LIFT Lab, which is supported by AB MEDICA SPA, BRACCO IMAGING SPA, FOND. E.A. FIERA INTERN. MILANO, FONDAZIONE CARIPLO, FONDAZIONE ENPAM, INTESA SANPAOLO FORMAZIONE SPA, INTESA SANPAOLO INNOVATION CENTER SPA and MEDTRONIC ITALIA SPA. This research was also co-funded by a SDA Bocconi internal research grant focused on Digital Therapeutics.
