Abstract
Diabetic optic neuropathy (DON) is an increasingly recognized, distinct neurodegenerative complication of diabetes and a significant independent cause of vision loss. Its diagnosis is challenging due to heterogeneity, tool limits, and lack of biomarkers, leading to underdiagnosis and delayed intervention. This review aims to provide a comprehensive overview of DON, focusing on its pathophysiological mechanisms, current diagnostic challenges, and the emerging role of artificial intelligence (AI) as a transformative tool for enabling earlier detection and personalized management. This review provides a narrative synthesis of the literature on DON, covering clinical manifestations and multifactorial pathophysiology involving metabolic, vascular, inflammatory, and neurodegenerative pathways. Based on the foundational success in application of AI in diabetic retinopathy (DR), the translational application of machine learning and deep learning algorithms is systematically explored, covering key areas such as optic nerve head segmentation, disease classification, differential diagnosis, predictive analytics, and the discovery of novel imaging biomarkers through radiomics. AI demonstrates significant potential in quantifying subtle structural signs of DON and integrating multimodal data to overcome current diagnostic limitations. The transition from AI models in DR to those for DON represents a shift from detecting microvascular lesions to identifying neurodegenerative changes. Future directions hinge on developing explainable AI for clinical trust and leveraging longitudinal data for predictive modeling of disease progression. The integration of sophisticated AI tools into clinical practice is poised to shift the management of DON from reactive intervention to proactive, precision-based care, ultimately improving visual outcomes for the vast global diabetic population.
Keywords
Introduction
Diabetic optic neuropathy (DON) is a serious and increasingly recognized complication of diabetes mellitus, involving direct damage to the optic nerve due to prolonged hyperglycemia. 1 While diabetic retinopathy (DR) has traditionally been the main focus of diabetic eye disease, DON represents a distinct metabolism-driven neurodegenerative process that is a significant independent cause of vision loss. 2 The global burden of diabetes, affecting over 500 million people, 3 establishes a vast population at risk for DON. Its prevalence is strongly linked to diabetes duration and poor glycemic control, 1 though it can occur even without detectable retinal vascular changes. This underlines its unique pathophysiology and suggests that many patients may be affected before traditional DR becomes apparent.2,4,5 As a leading cause of irreversible visual impairment, DON profoundly reduces quality of life and imposes a significant socioeconomic burden on healthcare systems. 6
The significance of DON lies in its direct injury to the neural visual pathway.7,8 It is characterized by the progressive degeneration of retinal ganglion cells and their axons, which form the optic nerve. This leads to debilitating visual deficits such as reduced contrast sensitivity, impaired color vision, and visual field loss—often while standard measures of central acuity remain normal.7,9 This damage results from a complex interplay of microvascular ischemia, oxidative stress, chronic inflammation, and direct metabolic toxicity to neurons,10,11 highlighting its role as a parallel complication to microvasculopathy. Despite its clinical impact, DON is frequently underdiagnosed due to major challenges and delays in clinical identification.6,12 Early-stage DON is often asymptomatic and invisible on routine fundus examination. Although advanced imaging like Optical Coherence Tomography (OCT) can quantify structural loss, such as thinning of the retinal nerve fiber layer (RNFL) and ganglion cell complex, these changes are typically detectable only after irreversible neuronal loss has occurred. 13 This diagnostic lag means opportunities for early intervention are often missed. Additionally, there is a lack of standardized diagnostic and grading criteria, and clinicians must differentiate DON from other optic neuropathies, such as glaucoma or compressive lesions, further complicating and delaying diagnosis.
Artificial intelligence (AI), particularly through its subfields of machine learning and deep learning, has revolutionized the field of ophthalmology. 14 By utilizing sophisticated algorithms, especially convolutional neural networks (CNNs), AI offers unprecedented capabilities in automated image analysis. 15 This has been most successfully demonstrated in the screening and diagnosis of DR, where AI systems can now identify referable lesions—such as microaneurysms, hemorrhages, and exudates—with high accuracy, classify disease severity according to standardized scales,16,17 and even predict the risk of future progression to sight-threatening stages. 18 This technology has transitioned from research to clinical practice, with the United States Food and Drug Administration (FDA)-approved systems enabling large-scale, efficient screening that improves access to care, particularly in underserved regions.19,20
However, while the application of AI in retinal vascular disease is well-established, its exploration in DON remains significantly underexplored and represents a critical frontier in ophthalmic AI. This gap exists for several key reasons: the diagnostic focus has historically been on the more overt microvascular changes of DR; the structural signs of DON (like optic disc (OD) pallor and nerve fiber layer thinning) are often more subtle and require different analytical approaches; and there is a relative scarcity of large, well-annotated datasets specifically for DON compared to the vast repositories of fundus images available for DR. Consequently, the development of robust AI models capable of detecting early axonal loss, differentiating DON from other optic neuropathies like glaucoma, or identifying novel imaging biomarkers from OCT scans is still in its nascent stages. Bridging this gap holds immense promise, as AI-driven tools could unlock the ability to diagnose DON earlier, monitor its progression more objectively, and ultimately develop new quantitative indicators for a condition that currently relies heavily on subjective clinical assessment.
This review aims to provide a comprehensive overview of DON, focusing on its pathophysiological mechanisms, current diagnostic challenges, and emerging tools including AI that may enable earlier detection. This review will also highlight the necessity of establishing clear diagnostic criteria and integrating multimodal data to improve clinical outcomes for diabetic patients worldwide.
Clinical manifestations and pathophysiological mechanisms
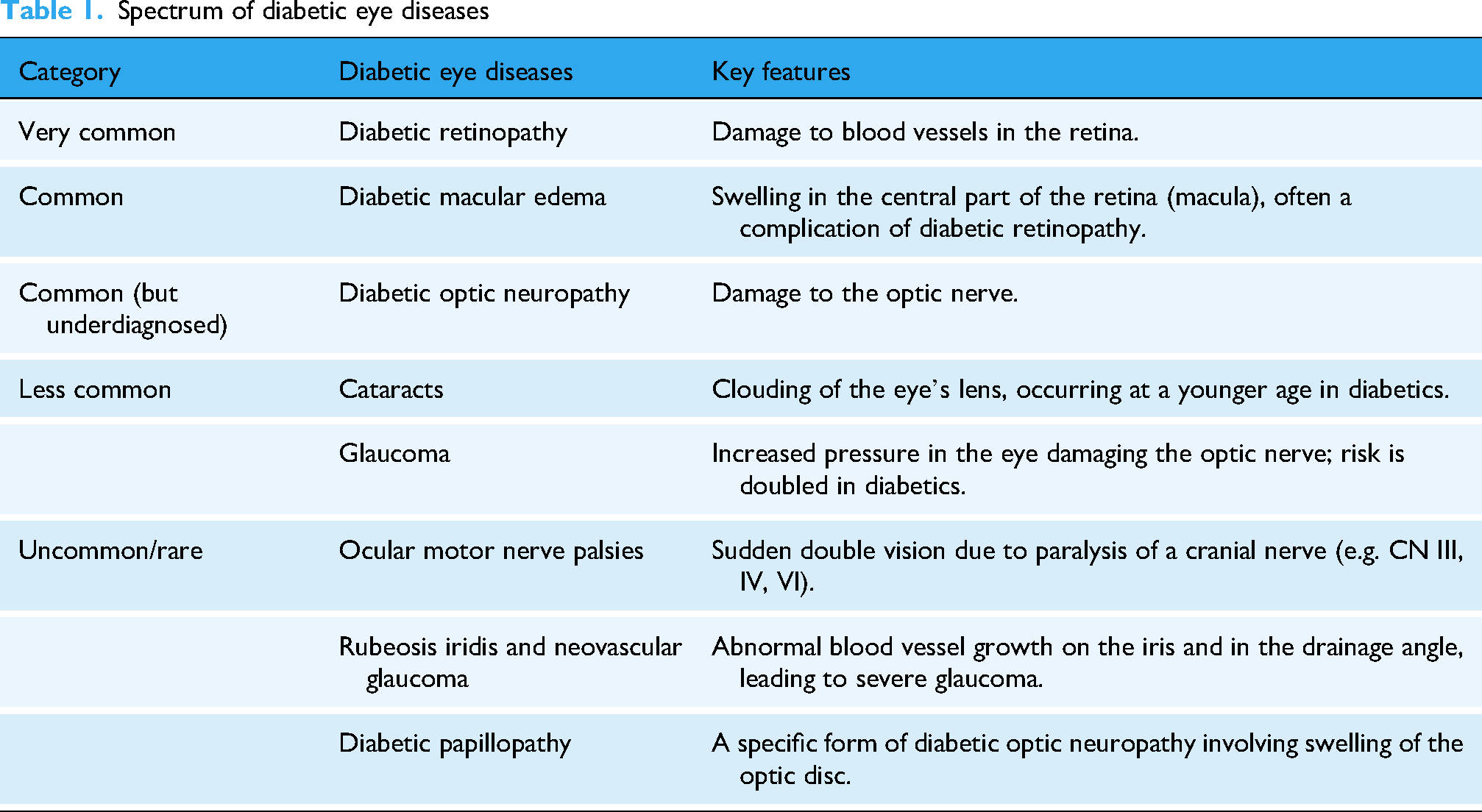
Diabetes mellitus, a group of metabolic diseases characterized by elevated blood sugar levels, progressively damages systems throughout the body, with the eyes being particularly vulnerable; this leads to a spectrum of diabetic eye diseases (Table 1), such as primarily DR, common complication diabetic macular edema,21–29 and DON.25,26,30–32
Spectrum of diabetic eye diseases
Clinically, DON manifests through a range of visual impairments that often develop insidiously. Patients typically report gradual, painless vision loss that may not be fully correctable with refractive aids, alongside reduced contrast sensitivity—making it difficult to distinguish objects in low-light conditions or fog.33,34 Dyschromatopsia, particularly affecting the blue–yellow axis, is another common feature, 33 where colors appear desaturated or washed out. Visual field defects frequently occur, often presenting as altitudinal loss (affecting the upper or lower half of the visual field), arcuate scotomas, or generalized constriction. 35 In cases of asymmetric involvement, a relative afferent pupillary defect may be observed. 36 Ophthalmoscopic examination may reveal OD pallor, reflecting axonal loss; thinning of the RNFL; venous sheathing indicating perivascular inflammation; and in acute/subacute forms such as diabetic papillopathy, mild OD edema.29,36 It is important to note that DON can present independently of DR, underscoring its distinct pathophysiology and clinical significance.
DON can be classified into several subtypes based on underlying mechanisms and clinical features (Table 2). Diabetic papillopathy is attributed to microvascular dysfunction and impaired autoregulation at the OD, resulting in mild edema and hyperemia, often with relatively preserved vision. 37 Anterior ischemic optic neuropathy, in the context of diabetes, represents an acute ischemic event causing sudden vision loss and disc swelling, with diabetes being a significant risk factor due to its impact on microvascular health.6,37 The most prevalent form, however, is chronic progressive optic neuropathy, which results from prolonged metabolic and oxidative insult, leading to gradual axonal degeneration and optic atrophy.38,39 DON can also be categorized based on the presence or absence of concomitant DR, highlighting that neural damage can occur independently of visible retinal vascular changes, thus necessitating a high index of clinical suspicion.13,40–42
Classification of diabetic optic neuropathy.

The pathogenesis of DON is multifactorial (Figure 1). As a secondary complication of diabetes mellitus, DON arises not as an isolated disorder but as a direct consequence of systemic metabolic dysregulation. 1 This metabolic insult initiates a cascade of oxidative stress, vascular dysfunction, and neuroinflammation. 43 Central to this process is chronic hyperglycemia, which plays an essential role by activating abnormal biochemical pathways that disrupt both neuronal and vascular homeostasis. The resulting dysfunction of the neurovascular unit, an integrated complex comprising neurons, glia (astrocytes and microglia), and vascular cells (endothelial cells and pericytes), represents a critical final pathway in the development of optic nerve damage.44–46 Diabetes disrupts this unit through pericyte loss, endothelial damage, glial activation, and chronic inflammation, impairing hemodynamics and neurotrophic support. 43 Notably, dysfunction of the neurovascular unit in its early stage is predominantly functional and reversible. At this point, the primary manifestation is impaired vascular autoregulation, occurring in the absence of overt structural damage to neurons or axons. However, persistent insult driven by chronic hyperglycemia can convert this functional disorder into irreversible structural damage. This pathological transition is mediated by several hyperglycemia-activated metabolic pathways, including the polyol pathway, the formation of advanced glycation end-products, and increased hexosamine flux. These pathways converge to generate oxidative stress and propagate inflammatory signaling, ultimately disrupting the integrity of the neurovascular unit.38,47,48

Proposed pathogenesis of diabetic optic neuropathy.
A pivotal mediator in this cascade is the sustained activation of protein kinase C (PKC).48–50 Hyperglycemia-induced diacylglycerol overproduction triggers PKC, which then orchestrates multiple aspects of DON pathology. First, PKC alters vascular function by inhibiting endothelial nitric oxide synthase and promoting endothelin-1, leading to reduced perfusion and ischemia. Second, it upregulates pathogenic factors such as vascular endothelial growth factor (VEGF)—increasing vascular permeability and disrupting the blood-retinal barrier—and modulates insulin-like growth factor-1 and transforming growth factor-beta, fostering fibrosis and aberrant remodeling. Third, PKC activation amplifies inflammatory and oxidative stress by stimulating proinflammatory cytokines (e.g. tumor necrosis factor-α, interleukin-1 beta) via transcription factors like nuclear factor kappa-B, and by exacerbating mitochondrial dysfunction. This culminates in reactive oxygen species overproduction, energy failure, and the direct activation of apoptotic pathways in retinal ganglion cells and their axons.48,51–53
From a clinical perspective, optic nerve damage in DON is closely linked to systemic metabolic control. Glycated hemoglobin A1c (HbA1c) serves as a marker of glycemic regulation, reflecting average blood glucose levels over the preceding two to three months. Chronic exposure to elevated HbA1c is clinically significant, as prolonged hyperglycemia may lead to irreversible optic nerve damage, even if glycemic control is subsequently achieved. Supporting this, a recent study demonstrated significant correlations between reduced radial peripapillary capillary density and multiple metabolic parameters, including HbA1c, urinary microalbuminuria, blood urea, and the albumin-to-creatinine ratio. Among these, HbA1c and blood urea emerged as the most significant determinants for radial peripapillary capillary impairment. 54 Beyond steady-state glycemic control, emerging evidence highlights the role of glucose variability and HbA1c variability as independent risk factors. These fluctuations may exacerbate diabetic microvascular complications through pathways involving oxidative stress, endothelial dysfunction, and chronic inflammation.55,56 Such dynamic metabolic disturbances likely contribute further to the pathogenesis of optic nerve damage in DON.
Thus, while microvascular ischemia contributes to acute injury, DON is increasingly recognized as a metabolism-initiated neurodegenerative disorder driven by metabolic toxicity and neuroglial dysfunction, with PKC serving as a key signaling hub integrating these insults. This complex pathophysiology results in heterogeneous and often insidious clinical presentations, posing significant diagnostic challenges. The subtle progression of DON underscores the critical need for early diagnostic strategies capable of detecting subclinical neurovascular and neuronal compromise before irreversible visual field loss occurs, shifting the therapeutic paradigm from vascular management alone to neuroprotective interventions.
Challenge in current clinical diagnosis
The clinical diagnosis of DON remains significantly challenging, leading to frequent underdiagnosis, delayed intervention, and an incomplete understanding of its natural history. These difficulties stem from the disease's heterogeneous presentations, the limitations of existing diagnostic tools, and the absence of definitive biomarkers.
Differential diagnosis among DON types
One of the primary challenges lies in the difficulty of diagnosing different type of DON.57,58 The clinical spectrum of DON ranges from acute ischemic events, such as DP or anterior ischemic optic neuropathy, to chronic, insidious neurodegeneration. Each subtype demands a distinct diagnostic approach, yet they often share overlapping features.59–61 For instance, OD edema—a hallmark of acute presentations like papillopathy—may be mild and transient, easily overlooked during routine examination.62,63 Conversely, the more common chronic progressive form presents with subtle, slowly evolving signs like OD pallor or RNFL thinning, which are nonspecific and may be attributed to other optic neuropathies, particularly glaucoma. 26 This overlap complicates differential diagnosis, as DON must be distinguished from compressive, inflammatory, hereditary, and other acquired optic neuropathies, a process that often necessitates additional, costly investigations such as neuroimaging or functional tests.12,36,64 The absence of universally accepted diagnostic criteria or a standardized classification system further exacerbates this problem, leading to heterogeneity in clinical practice and research.
Compounding this issue is the strong diagnostic lag inherent in DON. The neurodegenerative process begins long before clinical symptoms become apparent or detectable on conventional imaging. By the time patients report visual complaints such as reduced contrast sensitivity or minor field defects, a substantial proportion of retinal ganglion cells may have already been irreversibly lost. This lag is partly due to the brain's compensatory mechanisms, which mask early functional deficits. Consequently, the window for early intervention—potentially the most effective period for neuroprotective strategies—is often missed. This delay transforms management from prevention to damage mitigation, significantly impacting long-term visual outcomes.13,65–68
The limitations of current examination methods, particularly imaging technologies (Table 3), represent another critical barrier. While tools like OCT have revolutionized objective assessment by quantifying structural loss (e.g. RNFL and ganglion cell complex thickness), they primarily detect atrophy only after significant neural tissue loss has occurred.69,70 This structural approach fails to capture earlier, potentially reversible phases of the disease characterized by metabolic dysfunction, inflammation, or microvascular compromise. OCT angiography (OCTA), though promising for visualizing capillary dropout in the peripapillary network, lacks standardized quantitative thresholds for DON and is influenced by media opacities and artifacts. 71 Functional tests, such as visual field perimetry, are subjective, variable, and often insensitive to early neural loss.72–74 Moreover, techniques like fluorescein angiography are invasive and provide limited specific information about neural tissue health. 75 Thus, while each tool offers valuable insights, none alone provides a comprehensive picture of the disease's early evolution or severity. Currently, despite not universally adopted, some clinicians and researchers grade the severity of chronic DON based on structural and functional criteria,40,76,77 similar to DR.
Key diagnostic tests in diabetic optic neuropathy.

Finally, the lack of sensitive and specific biomarkers for DON remains a major obstacle. Unlike other diabetic complications, such as nephropathy (e.g. albuminuria) or DR (e.g. microaneurysms), DON lacks a validated biomarker for early detection, progression monitoring, or therapeutic response. Blood-based biomarkers reflecting neural injury (e.g. neurofilament light chain, NGF/BDNF, VEGF) or systemic oxidative stress (e.g. MDA, GSH/SOD, 8-OHdG, NFE2L2, PON-1, GGT) and inflammation (e.g. hs-CRP, tumor necrosis factor-α, IL-6, Homocysteine, AGEs, sRAGE, MCP-1, nuclear factor kappa-B, Adiponectin, GSK-3β, and miRNAs like miR-146a, miR-155 and miR-126) remain exploratory and nonspecific.78,79 Imaging biomarkers beyond layer thickness, such as textural analysis, functional connectivity, or metabolic profiling, are areas of undergoing research but have not yet entered routine clinical practice.80–83 Without such tools, clinicians are left relying on late-stage structural changes, perpetuating diagnostic delays and hindering the development of targeted therapies.
Distinguishing DON from other diabetic eye complications
Currently, a central clinical challenge in diagnosing DON lies in its significant phenotypic overlap with other common optic neuropathies, most notably glaucoma, but also optic neuritis and compressive neuropathies.
Glaucoma represents the most critical and common differential diagnosis for chronic progressive DON, as both present as painless, progressive optic neuropathies characterized by OD cupping and RNFL thinning. However, key distinguishing features exist. In glaucoma, neuroretinal rim thinning typically follows a pattern that violates the ISNT rule (where the rim width is normally largest in the Inferior, then Superior, Nasal, and Temporal quadrants), showing preferential loss in the superior and inferior poles, leading to vertical elongation of the cup. In contrast, DON more often presents with diffuse pallor and a more generalized RNFL thinning that tends to respect the ISNT rule. Fundus examination in DON may also reveal venous sheathing or features of concurrent DR. Furthermore, a notable clinical clue is that visual acuity in DON can fluctuate with blood glucose levels, a feature absent in glaucoma.84,85
Optic neuritis is distinguished by its typical acute or subacute presentation, including vision loss associated with orbital pain exacerbated by eye movements, reduced color vision, and a relative afferent pupillary defect. Magnetic resonance imaging (MRI) is often definitive in the acute phase (within three months), showing optic nerve contrast enhancement and T2 signal hyperintensity—findings absent in DON. While both conditions can lead to RNFL atrophy in the chronic phase, the history is pivotal: optic neuritis follows an acute inflammatory event, whereas DON atrophy develops insidiously. 86
Compressive optic neuropathies cause progressive visual loss through direct mechanical compression and are frequently asymmetric or unilateral. They may produce distinct patterns of quadrantic visual field defects corresponding to the site of compression. In contrast, DON typically manifests with altitudinal loss, arcuate scotomas, generalized depression, or central scotomas. Neuroimaging (MRI or computed tomography/CT) is the gold standard for excluding a compressive lesion, as it directly visualizes mass effects on the anterior visual pathway. 87
Therefore, the current diagnostic paradigm for DON is hampered by its clinical variability, significant lag between pathological onset and detection, reliance on imperfect tools that prioritize late-stage structural changes, and the absence of validated biomarkers. Overcoming these challenges requires a multifaceted strategy integrating advanced imaging analytics, functional testing refinements, and the discovery of novel molecular or imaging biomarkers to enable earlier, more precise diagnosis and improve patient outcomes. Under such circumstances, AI and computer algorithms emerge as promising tools, offering the potential to translate the complex pathophysiology of DON into actionable intelligent diagnostic strategies.
Current applications of AI and computer algorithms
AI in DR: the foundation
The application of AI in ophthalmology is most advanced and successfully translated into clinical practice in the domain of DR (Supplemental Table S1), where it has received regulatory approval (e.g. the first FDA-approved autonomous system, IDx-DR) for autonomous screening. These systems are predominantly built upon CNNs, a class of deep learning models exceptionally proficient at deconstructing and analyzing pixel-level data within images. The process involves training these algorithms on hundreds of thousands of fundus photographs that have been meticulously graded by panels of human retinal experts. Through this training, the AI learns to identify the presence of DR-referable lesions—such as microaneurysms, dot-blot hemorrhages, and hard exudates—with a high degree of accuracy.20,88 For instance, the Singapore I Vascular Assessment software can identify microvascular lesions that significantly associated with the progression of DR through quantifying retinal vascular parameters, such as venular fractal dimension and central retinal vein equivalent values. 18 AEYE Diagnostic Screening is the first FDA-approved autonomous AI tool for DR screening. It is compatible with both portable handheld retinal cameras and conventional tabletop fundus cameras, enabling point-of-care screening to ensure regular monitoring of patients with diabetes. 89
Beyond mere detection, these systems can classify disease severity according to standardized scales like the International Clinical DR Severity Scale or the more granular Early Treatment DR Study scale.16,18 For example, the Multi-scale Spatial-aware Transformer Network, which comprises a spatial-aware module, a multiscale encoder, and a cross-fusion classifier. It employs a multiple instance learning strategy to accurately diagnose and grade DR. 90 The EyeArt AI Automated DR Detection System is an FDA-cleared cloud-based retinal diagnostic software device that can accurately detect both more-than-mild DR and vision-threatening DR by analyzing digital color fundus photographs of patient eyes for signs of DR without physician oversight or need for dilation in most individuals. 16
This automation is transformative for public health, enabling high-volume, cost-effective screening that can be deployed in remote and underserved areas with a shortage of ophthalmologists, AI-powered DR screening stands as a powerful tool for reducing the global burden of preventable blindness.
The translational leap to DON
The remarkable success of AI in detecting and grading DR has established a powerful foundation and provided the technical blueprint for its application to DON. However, this translation represents a significant paradigm shift, as the diagnostic task is fundamentally different. While AI in DR focuses on identifying hemorrhagic, exudative, and proliferative vascular lesions,91,92 its primary goal in DON is to detect far more subtle signs of neurodegeneration.29,36,93 Specifically, the structural and functional changes affecting the optic nerve and the inner retinal neural layers. This requires a move from analyzing the retinal vasculature to quantifying the neuronal architecture, and some of the AI systems have the ability to meet certain requirements. For image segmentation, representative architectures including U-shaped Network (U-Net) and fully convolutional networks are deployed to achieve high-precision delineation of critical ocular anatomical structures. These models are distinguished by their end-to-end learning capability and superior segmentation accuracy, yet their clinical translation is impeded by the imperative for large-scale and annotated training datasets. 94 For disease classification and recognition, CNNs and support vector machines (SVMs) serve as primary tools for DON severity grading and differential diagnosis. Notably, CNNs enable automated feature learning from raw imaging data but mandate substantial datasets to mitigate overfitting and ensure generalizability. And SVMs show strong performance in situations with small sample sizes but are hindered by the need for time-consuming manual feature engineering.94,95 For feature extraction and regression, models such as Random Forests and Linear Regression are leveraged to predict continuous clinical variables, with the key advantage of enhanced interpretability. However, their predictive performance depends on the initial feature engineering processes. For multimodal fusion, Multiinput CNNs and Graph Neural Networks integrate the datasets spanning OCT, OCTA, Visual Evoked Potential and functional MRI (fMRI) to facilitate holistic DON assessment. By capitalizing on complementary information, these models exhibit great clinical potential. Nevertheless, their implementation is challenged by inherent model complexity and practical hurdles pertaining to multimodal data acquisition standardization. 96 Table 4 summarizes common algorithm models and their potential applications in DON.
Common algorithm models and their potential applications in diabetic optic neuropathy.
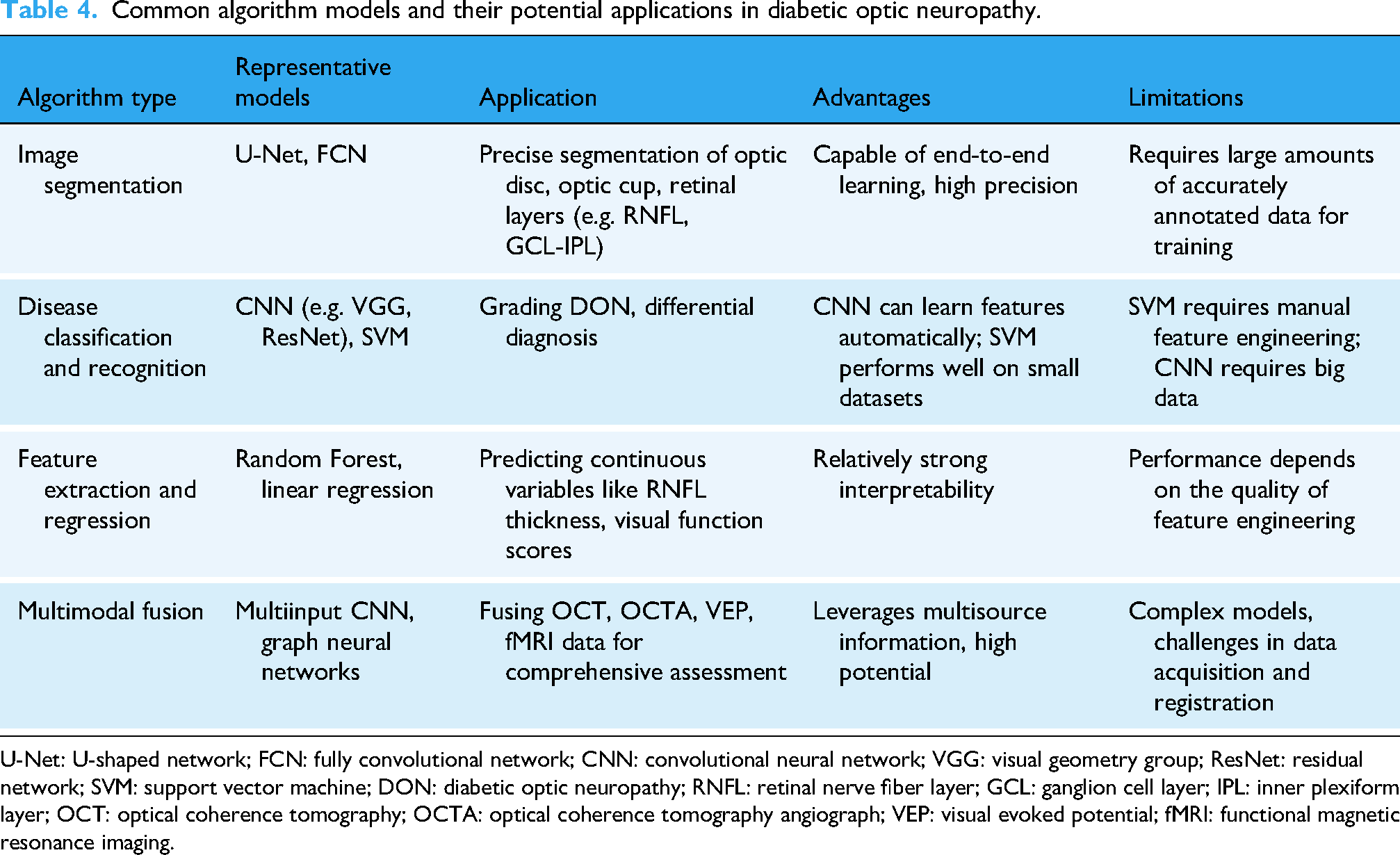
U-Net: U-shaped network; FCN: fully convolutional network; CNN: convolutional neural network; VGG: visual geometry group; ResNet: residual network; SVM: support vector machine; DON: diabetic optic neuropathy; RNFL: retinal nerve fiber layer; GCL: ganglion cell layer; IPL: inner plexiform layer; OCT: optical coherence tomography; OCTA: optical coherence tomography angiograph; VEP: visual evoked potential; fMRI: functional magnetic resonance imaging.
The initial and most critical step in this quantitative analysis is the precise delineation of ocular structures. Recent advancements in OD and optic cup (OC) segmentation have leveraged sophisticated adaptations of deep learning architectures. M-Net establishes a robust U-Net-based foundation, employing a one-stage, multi-label system that integrates a multi-scale input layer, side-output layers, a multilabel loss function, and a polar transformation to segment the OD and OC jointly. 97 Building upon this foundation, the Coarse-to-Fine Transformer Network represents a significant evolution by hybridizing the U-Net architecture with a Transformer module. This integration, augmented by a Multi-Scale Dense Skip Connection module, substantially boosts segmentation accuracy. 98 Further pushing the boundaries of this architectural shift, ONH Detection Transformer (ODFormer) is a dedicated optic nerve head detection network based entirely on the Swin Transformer. It introduces two key innovations: a lightweight bidirectional feature recalibrator and a multiscale context aggregator, which work in concert to refine feature maps and capture global contextual information, thereby achieving state-of-the-art performance. 99 Collectively, these models illustrate a clear trajectory from enhanced CNNs to hybrid and finally pure Transformer architectures, all aimed at delivering more precise and reliable OD or optic nerve head measurements. In the context of DON, these models perform several essential functions: they precisely define the boundaries of the OD and OC to calculate the cup-to-disc ratio—a key metric for differentiating DON from glaucoma 100 —and they automatically segment and measure the thickness of the RNFL and the ganglion cell complex from OCT scans. This provides highly reproducible, objective quantitative data on neural loss, offering a sensitivity far greater than qualitative clinical assessment.
Beyond segmentation, AI is directly applied to disease detection and classification as a diagnostic support tool.101,102 CNNs—such as Residual Network (ResNet), Visual Geometry Group, and Inception—are trained to analyze both fundus photographs and OCT scans.103–106 These models learn to identify direct signs of DON from fundus images, including OD pallor, RNFL defect patterns (e.g. localized wedge-shaped defects), and venous sheathing. More powerfully, when applied to entire 3D OCT volumes, CNNs can classify eyes as healthy or affected by DON by identifying complex, often imperceptible patterns of thinning and textural changes across layers. A crucial application is their ability to differentiate DON from other optic neuropathies, particularly glaucoma, by recognizing the distinct patterns and topography of RNFL loss characteristic of each disease.
Perhaps the most promising and advanced application of AI in DON is in the realm of predictive analytics. Using Recurrent Neural Networks or hybrid deep learning models integrated with traditional survival analysis techniques, algorithms can analyze longitudinal data from sequential OCT scans. By modeling the individual rate of RNFL thinning over time, these systems can predict which patients are at the highest risk for rapid disease progression. This enables a shift toward personalized medicine, allowing clinicians to tailor more aggressive monitoring and treatment plans for high-risk individuals.
Finally, AI excels at the discovery of novel imaging biomarkers through a process known as radiomics.107–109 An OCT-omics prediction model, including 11 key features screened using the Mann–Whitney U test, utilizes logistic regression, SVM, and backpropagation neural network classifiers to assess anti-VEGF treatment response in patients with diabetic macular edema. 110 An unsupervised deep learning architecture for OCT images enables unbiased, data-driven biomarker discovery. It identifies subclinical markers, including known and novel ones, with no prior domain knowledge. The novel one showed positive correlation with visual acuity and were validated via clinical outcomes (visual acuity, lesion activity, retinal morphology) using correlation and machine learning regression. 111 By extracting hundreds of quantitative texture features (e.g. entropy, contrast, fractal dimension) from the RNFL and ganglion cell complex on OCT scans, AI can detect subvisual changes that represent the earliest signs of axonal dysfunction. These alterations may be identified while the disease is still in a potentially reversible stage, often long before clinically significant thinning, which is more likely to reflect irreversible structure damage, becomes apparent. The power of AI lies in its ability to identify which specific combination of these radiomic features best correlates with a functional deficit (e.g. on visual field testing) or, most importantly, predicts future atrophy. This paves the way for the validation of novel, highly sensitive imaging biomarkers that could revolutionize the early diagnosis and management of DON.
Current challenges and future directions in AI for DON
These advancements of AI signify a shift from qualitative assessment to quantitative analysis of optic nerve. AI starts to address the key problems in preclinical diagnosis of DON by precise segmentation, objective detection and so on. However, the translation to clinical practice and patient management still faces lots of hurdles that must be addressed to realize its full clinical potential.
Current dataset limitations
One primary limitation is data scarcity. Unlike DR, for which large, publicly available, well-annotated datasets exist, curated datasets for DON are severely limited. This scarcity originates from several key obstacles: the low diagnostic rate of DON, heterogeneity in imaging protocols across clinical centers, the high cost and expertise required for multimodal data acquisition and interpretation, and a lack of consensus labels for definitive diagnosis and staging. Consequently, the available data is often insufficient in both volume and quality. This data paucity forms a critical bottleneck, as it directly restricts the training, generalizability and robustness of AI models. Models trained on small, unrepresentative datasets are prone to overfitting, exhibit reduced sensitivity and specificity for early-stage disease, and often perform poorly when applied to diverse populations or imaging equipment not seen during development.
Translational limitations
Another limitation is translational challenge of certain experimental models to patients with long-standing or poorly controlled diabetes. In these clinically relevant populations, the applicability of such models faces substantial challenges. Notably, these models are inherently limited in their ability to recapitulate the full chronicity and cumulative metabolic burden characteristic of human disease. They also lack the granularity to distinguish between varying degrees of disease severity and progression, thereby failing to capture the heterogeneity observed in clinical practice.
Furthermore, while these models may be sensitive to the pathophysiological features typical of early neuropathy, this sensitivity is often diminished in the context of advanced disease. In patients with long-term metabolic dysregulation, the cumulative and severe tissue alterations that define chronic diabetic complications can obscure the more subtle signs of early optic nerve damage. Consequently, experimental findings derived from models of early or uncomplicated disease may not directly translate to the complex, multimorbid reality of patients with poorly controlled or long-standing diabetes, underscoring the need for cautious interpretation when applying such data to these populations.
Potential strategies
To overcome the significant data scarcity and heterogeneity challenges in DON AI research, a focused technological approach is essential. Promising strategies are emerging to build more robust and generalizable models. Foremost among these is federated learning, a decentralized framework that enables collaborative model development across multiple institutions without centralizing sensitive patient data. By training algorithms locally and aggregating only model updates, it effectively pools heterogeneous datasets while rigorously preserving patient privacy and adhering to data governance regulations. This directly addresses the core bottleneck of acquiring large-scale, diverse training data.
Complementing collaborative learning, synthetic data generation offers a powerful method for data augmentation. Advanced generative models, such as generative adversarial networks or diffusion models, can produce high-fidelity, annotated images of the optic nerve that mimic the appearance of true DON pathology. These synthetic datasets can artificially expand limited training cohorts, mitigating overfitting and enhancing a model's ability to generalize to rare or subtle presentations. Furthermore, to ensure models trained in one environment perform reliably in another, domain adaptation and generalization techniques are critical. These algorithms are specifically designed to minimize “domain shift"—the performance drop caused by differences in imaging devices, acquisition protocols, or patient demographics between sites. By aligning feature distributions or learning domain-invariant representations, these methods enhance model robustness and transferability across the varied landscape of clinical practice.
Integration of emerging quantitative biomarkers for AI-based detection
The true power of AI will be unlocked by fusing these diverse data streams. Beyond traditional structural parameters, several emerging imaging biomarkers show promise for early DON detection and can serve as endpoints for AI models, including Structural, Vascular, and Functional–Structural Correlation. 112 Besides OCT, advanced imaging techniques provide a richer dataset for AI algorithms to analyze. OCTA can noninvasively quantify vascular perfusion around the optic nerve.113,114 Diffusion Tensor Imaging (DTI), an MRI technique, can assess the microstructural integrity and organization of the optic nerve fibers themselves by measuring water diffusion. 115 fMRI can reveal altered functional connectivity within the entire visual pathway and the brain.82,116 A key future direction is the development of sophisticated AI architectures—such as multiinput CNNs or graph neural networks—capable of integrating these multimodal imaging data (OCT, OCTA, DTI, fMRI) with clinical variables (e.g. HbA1c, diabetes duration) to build a comprehensive, sensitive, and specific assessment system for DON.
AI-driven radiomics and multimodal integration will validate these biomarkers as robust endpoints for AI-based DON detection. By prioritizing these emerging parameters, AI models can shift DON diagnosis from late-stage atrophy detection to early identification of disease onset. Specifically, AI has the potential to integrate and interpret an array of emerging indicators that surpass the sensitivity of conventional clinical metrics. Firstly, unearthing earlier, more subtle signs of DON by analyzing textural and microstructural changes in the RNFL and ganglion cell layer on OCT that precede overt thinning. Secondly, finding patterns in blood flow derived from OCTA, such as reduced peripapillary capillary density. It is more dynamic that may reveal impaired perfusion states earlier than static tests. Thirdly, detecting specific functional changes that correlate with nerve damage, by integrating data from visual field testing and other psychophysical tests. AI excels at modeling the complex relationship between structure and function, which can serve as a specific signature for DON. Finally, the most transformative application of AI will be in longitudinal analysis and predictive modeling. By applying AI to sequential imaging data from multiple patient visits, it becomes possible to dynamically track subtle progression patterns that are invisible to the human eye. This will enable the development of models that can predict an individual's risk of DON onset or their specific rate of progression, shifting the management paradigm from a one-size-fits-all approach to true early intervention and personalized management. This could fundamentally change patient outcomes by enabling preventative therapies to be deployed before significant vision loss occurs. Table 5 summarizes the potential clinical applications and the associated techniques of AI in the management of DON.
Clinical potential applications of AI in diabetic optic neuropathy.

AI: artificial intelligence; RNFL: retinal nerve fiber layer; OCT: optical coherence tomography; GCC: ganglion cell complex; DON: diabetic optic neuropathy; CNNS: convolutional neural networks.
Proposed diagnostic workflow of DON
The current diagnostic reliance on parameters like RNFL thickness measured by OCT presents a fundamental problem: these metrics often only detect damage after significant and irreversible neural loss has already occurred. This diagnostic lag underscores the critical need for novel, computer algorithm-based quantitative indicators that can identify the earliest stages of disease. Therefore, the proposed diagnostic workflow for DON would integrate AI into a multistep clinical pathway to enable earlier and more precise diagnosis (Figure 2). It begins with the initial assessment of high-risk diabetic patients using standard ophthalmic exams, OCT, and visual fields. For complex or early-stage cases, the workflow incorporates an AI-augmented analysis. Here, a secure platform aggregates de-identified, multimodal data—including advanced imaging like OCTA and clinical history—for processing by specialized AI models. These models perform multimodal integration, extracting and quantifying subtle structural, vascular, and functional–structural correlation biomarkers that are imperceptible to conventional analysis. The AI generates a comprehensive decision-support report, providing a probability score, visualizing abnormalities, and offering differential diagnostic guidance. This assists clinicians in confirming DON, staging the disease, and ruling out mimics like glaucoma. Finally, the system facilitates personalized management and enables AI-powered longitudinal monitoring. By tracking micro-changes over time, it can detect progression earlier than standard methods and holds the potential to predict individual disease trajectories, shifting management toward proactive, personalized intervention before significant vision loss occurs.

Diagnostic workflow of diabetic optic neuropathy.
Transparency and validation of future AI models
For successful clinical adoption, medical AI must transcend the “black box” problem. 117 Developing explainable AI (XAI) models is paramount.118,119 Techniques like Gradient-weighted Class Activation Mapping (Grad-CAM) can generate heatmaps that visually indicate which regions of an image (e.g. a specific sector of the neuroretinal rim or a particular texture pattern in the RNFL) most influenced the algorithm's decision. This transparency is dual-purpose: it not only enhances clinician trust and facilitates integration into clinical workflow but also, in reverse, can discover new, biologically significant imaging biomarkers. 120 By highlighting previously overlooked features that are highly predictive of disease, XAI can directly contribute to a better understanding of DON pathophysiology.
The clinical translation of AI's transformative potential for DON diagnosis hinges on addressing three pivotal challenges through forward-looking innovations: ensuring explainability, managing cross-domain variability, and establishing rigorous model validation. For XAI, moving beyond current heatmap-based interpretability, future models could leverage causal reasoning frameworks to map AI-derived imaging features to specific pathophysiological processes in DON. This could involve integrating biological prior knowledge into model architecture to provide biologically plausible explanations. Regarding cross-domain variability, the next frontier lies in developing adaptive AI systems that can dynamically adjust to new imaging protocols, patient populations, or clinical settings without retraining. This might involve meta-learning approaches where models are pretrained on diverse multimodal datasets (OCT, OCTA, and fMRI) to acquire generalizable feature representations. For model validation, future frameworks should move beyond traditional evaluation metrics to incorporate clinical utility and longitudinal predictive power. This could include prospective, multicenter trials that assess whether AI-assisted diagnosis improves patient-centered outcomes. Collectively, these advancements could transform AI from an auxiliary diagnostic tool to an integral component of personalized DON management.
Therefore, AI will evolve progressively with the discovery of new biomarkers and the growth of the volume of dataset. The ultimate goal is to develop explainable and sophisticated AI architectures which can integrate multimodal imaging data with clinical variables. Successfully developed AI systems promise to improve diagnostic efficiency, validate novel biomarkers, refine our understanding of disease progression and ultimately achieve a personalized and precise management paradigm for DON.
Conclusion
While AI applications in DON are still in a more developmental stage compared to DR, the potential is immense. Current research is focused on moving beyond simple replication of human grading to extracting entirely new, quantifiable insights from ocular imaging data. The future lies in developing integrated, multimodal AI systems that can detect DON at its earliest stages, predict its individual course, and uncover novel biomarkers, ultimately enabling a shift from reactive treatment to proactive, precision-based management of this sight-threatening complication.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261435864 - Supplemental material for Advances and challenges from pathological mechanisms to intelligent quantified diagnosis in diabetic optic neuropathy
Supplemental material, sj-docx-1-dhj-10.1177_20552076261435864 for Advances and challenges from pathological mechanisms to intelligent quantified diagnosis in diabetic optic neuropathy by Bingying Liu, Jian Lian, Yuhan Liu, Wenwen Xu, Qiang Zhu and Wanzhen Jiao in DIGITAL HEALTH
Footnotes
Author contributions
Bingying Liu and Jian Lian contributed equally to this study. Bingying Liu: conceptualization, investigation and writing—original draft; Jian Lian: software, methodology and writing—review and editing; Yuhan Liu: investigation, formal analysis and writing—original draft; Wenwen Xu: formal analysis, funding acquisition and writing—review and editing; Qiang Zhu: formal analysis, funding acquisition and writing—review and editing; Wanzhen Jiao: conceptualization, supervision, project administration and writing—review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shandong Provincial Natural Science Foundation General Project (Grant No. ZR2023MH087) and Key Research and Development Program of Shandong Province (Grant No. 2025KJHZ045).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
