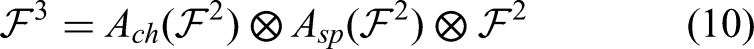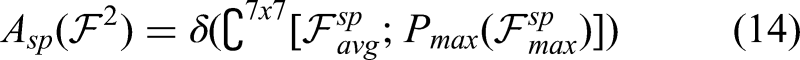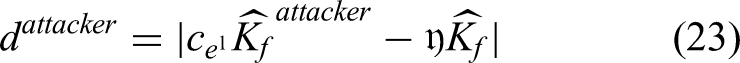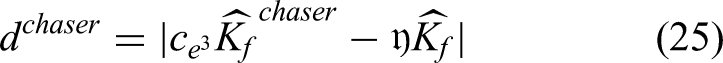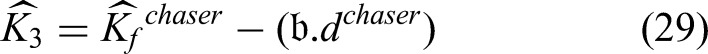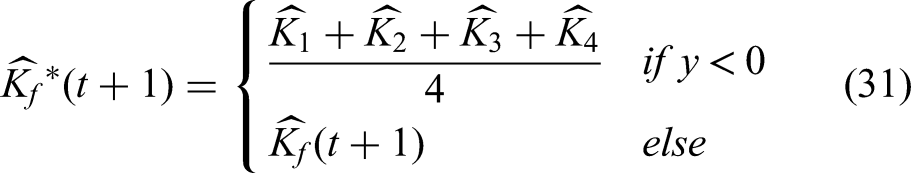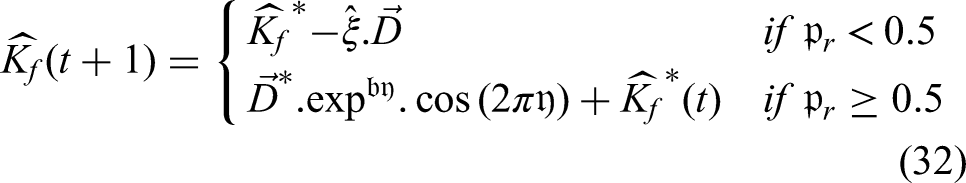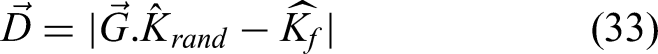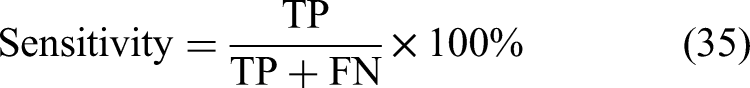Abstract
Objectives
Brain tumors have been a major factor in the development of mental disorders like anxiety and depression. The primary target of this research is to develop a smart, economically feasible system that can detect and classify brain tumors by analyzing MRI images, reducing manual labor, and shortening diagnostic time.
Methods
Although diagnosis with medical imaging is efficient for various health issues, accurately classifying brain tumors remains challenging for medical experts. This research introduces a new energy channel-based hybrid optimized network (ECHO-Net) as an automatic identification tool for brain tumors, aiming to facilitate medical diagnosis remotely. Energy shape prior segmentation (ESPS) is part of the framework, providing accurate segmentation and cropping of tumor regions from MRIs. Channel and spatial attention-based neural network (CSA-Net) differentiates between normal and tumor-affected images. Additionally, the hybrid chimp-based whale optimization (HCWO) algorithm enhances prediction precision and optimally sets sigmoid activation function parameters for better convergence and generalization.
Results
Performance evaluation of ECHO-Net using publicly available MRI brain datasets, including Figshare, BRATS 2018–2020, and a clinical MRI dataset, shows a peak signal-to-noise ratio of 41.87 dB, a Structural Similarity Index Measure of 0.992, a sensitivity of 99.4%, an accuracy of 99.2%, and an average computational time of 2.68 s, outperforming current state-of-the-art methods.
Conclusion
The proposed ECHO-Net is an automated, accurate, fast, and robust system for brain tumor detection. With low computational costs, it effectively segments abnormal regions and recognizes tumor types, demonstrating strong potential as a tool in intelligent healthcare systems and real-world clinical applications.
Keywords
Introduction
The brain is an intricate organ that is made up of hundreds of millions of cells. Uncontrolled growth of cells is the root cause of brain tumors. These cells may influence both regular brain function and normal cell destruction. The brain primarily controls the central nervous system. The function of the human brain is to link the bone marrow with the nervous system in the body. The task of directing the human body's movements falls to the brain. It gathers information from multiple senses and, upon decision-making, transmits signals to the internal organs.1,2 With the assistance of neuronal cells, the brain serves as the hub of the human body's administrative system, controlling every bodily function. Currently, the two most common types of serious brain tumors are malignant and benign. Both brain tumors in people of all ages are thought to be fatal forms of cancer.3,4 Unnatural tissue development in the brain leads to the formation of brain tumors since the aberrant tissues outgrow normal cells, generating an aggregate of cells that ultimately turns into malignancies. The most harmful kind of tumor is a benign tumor, which does not have cancerous cells. Recent medical statistics studies indicate that over 10 million people worldwide have lost their lives to this tumor, and almost 20 million individuals globally are afflicted by this terrible disease.5,6 Furthermore, by 2040, it is estimated that 30 million people will potentially have a brain tumor. Tumor cells that are intentionally designed to be lethal can be classified as malicious cancer. Complete brain damage can result from a malignant tumor. Meningioma, pituitary, and glioma are common primary tumors in adults. Gliomas arise from glial cells within the brain's supportive tissue, while meningiomas are usually benign tumors that form beneath the skull in the brain's outermost layers and tend to grow quickly. 7 Meningiomas typically affect one or both cerebral hemispheres, and they may take years to be identified. The pituitary gland, also called the pituitary, is located in the brain's inferior region. The gland's main function is to create hormonal substances that regulate the body's many glands, including the thyroid gland. A pituitary tumor may interfere with these glands’ ability to function.8–10 The best chance of successfully treating a brain tumor is early and accurate diagnosis. Early identification significantly impacts the successful outcome of brain tumor treatment. Gliomas are important early-stage brain tumors that have a very high death rate. There are two types of gliomas: low-grade, which causes less damage, and high-grade, which is more damaging due to infiltration. Because the tumor can appear in any form, size, or location, diagnostics might be difficult.
Medical image processing is crucial for helping people recognize different diseases. Early tumor detection can be aided by using computer tomography and magnetic resonance imaging (MRI), a pair of techniques commonly used to examine abnormalities in brain tissues related to the dimensions, location, or type of tumor cells.11,12 Since MRI provides precise information about the internal structure of the human cerebral cortex, it is especially useful in verifying the existence of gliomas. T1c, Tl, Flair, and T2 are the most often utilized MRI sequences for brain study. Different information regarding the brain tumor can be obtained through these sequences. The radio frequency used in MRI tests is used to determine the time of echo and repetition. Accurate identification that aids physicians in therapy has been enhanced by robust machine learning (ML) algorithms.13,14
To categorize the tumor as efficiently as possible, it is crucial to use a successful strategy with particular features and classification algorithms. Automated systems are more precise and efficient at classification than traditional manual methods. The exploration and depiction of deep characteristics is a crucial task for determining the presence and identification of brain tumors from physical MRIs. Oncology extracts deep characteristics from MRI images for diagnosis, treatment, and outlook. The radiomic qualities of the images provide qualitative information that radiologists are familiar with and link to significant physiological features. 15 These days, medical as well as academic investigations frequently use completely automated techniques to distinguish between MRIs showing a tumor and those that do not. In the decade preceding, these techniques have developed quickly and enable researchers to examine the malignancy region more deeply. As a result, radiologists believe that automated ML techniques will enable computers to diagnose patients more accurately. Brain tumor diagnosis is a difficult task due to their unpredictability in terms of location, structure, and dimension, even with advancements and promising outcomes in medical image processing. 16 Consistent and exact segmentation coupled with anomaly characterization remains a challenge.
Figure 1 displays the typical frame of a brain tumor detection and classification model. To remove the cancer-ridden region from images to facilitate further brain tumor classification and prediction, brain tumor segmentation is carried out. Various ML/deep learning (DL) techniques might be used to segment cells with tumors. A portion of these ML techniques requires manual image segmentation, which is expensive, time-consuming, and requires knowledge of medicines.17,18 Modern neuroimaging methods are available to identify tumor cells that are visible or unseen. The irregularities in the size and form of the tumor make automated image segmentation more challenging. Accurate segmentation is a critical step in identifying brain tumors. To enhance detection efficiency, automated or semi-automated techniques are preferred, as manual segmentation is time-consuming and labor-intensive for radiologists. Earlier research has thoroughly examined the effectiveness of DL approaches, demonstrating their outstanding performance in multiple domains of medical image analysis, particularly in the detection, segmentation, and classification of brain tumors using MRI images. Nevertheless, the primary issues with traditional methods are their high computing complexity, long execution times, incapacity to handle high-dimensional data, and lower prediction reliability.19–21 Therefore, the main objective of this study is to develop a novel and computationally efficient medical image analysis model for the accurate identification of brain tumors.
An overview of a typical brain tumor detection and classification framework.
Typically, the development of an automated system for identifying and classifying brain tumors using machine learning-based image processing techniques is a complex and time-consuming task. The literature review indicates that various methods have been implemented for brain tumor detection. The following section discusses several state-of-the-art techniques related to image segmentation, feature extraction, and classification, specifically in the context of brain tumor identification and diagnosis.
Kalam et al. 22 proposed an adaptive ANFIS-based classification method integrated with a skull stripping algorithm to detect the brain tumor. The main objective of this study was to accurately segment tumor-affected regions from MRI scans using an advanced artificial intelligence technique. Additionally, the k-means clustering algorithm was employed to further improve the accuracy of the diagnostic process. However, the proposed method has lower detection accuracy for complex images and is not more efficient in tumor identification, which has its own set of problems related to computing overhead. Solanki et al. 23 provided a detailed overview of ML, DL, and transfer learning methods for brain tumor diagnosis. Accurate segmentation and classification still pose challenges despite a great deal of effort and promising results. The differences in tumor location, shape, and dimensions make brain tumor detection extremely difficult. The primary goal of this study is to provide researchers with comprehensive information on the use of MR imaging for detecting brain tumors.
This study suggested numerous approaches for the detection of tumors and brain cancer using empirical image analysis and intelligent computing. In addition, the physical characteristics of malignancies in the brain, widely accessible data sets, augmented methods, component extraction, and classification are all clarified in this work. Saleh et al. 24 employed a traditional CNN-based classification approach utilizing the VGG-16 architecture for brain tumor detection. This study aimed to evaluate various segmentation techniques—such as area thresholding, fuzzy c-means, triple thresholding, and active contour—to identify the most efficient and accurate method for reliable diagnosis. However, the VGG-16 model demonstrated slower prediction times, which adversely impacted the overall efficiency of the detection system.
Hossain et al. 25 developed an automated model for brain tumor detection and segmentation, incorporating image enhancement and skull stripping techniques. Although it takes longer, skull stripping is an essential step in processing medical imaging because the background of the MRI image lacks important information. Additionally, to improve segmentation efficiency, the Gaussian blur filter was put to use to reduce Gaussian noise. Kshirsagar et al. 26 developed an automated approach for detecting brain tumors by utilizing cutting-edge image processing methods. The authors specifically focused on enhancing the prediction outcomes through the implementation of suitable preprocessing processes. Here, the authors generated the standardized MRI brain images by applying noise reduction, brightness enhancement, and distribution methods. A hybridized feature extraction method known as PCA-GIST has been applied by Gumaei et al. 27 in order to develop a brain tumor segmentation system that consistently matches cases.
Furthermore, the method has been combined with the latest regularized model to enhance the classification process. The grid search technique is also used to optimize the features in order to guarantee a higher prediction rate for the classifier. However, the recommended framework takes a little longer to train and test, which has an impact on detection performance. Jia et al. 28 implemented a brain tumor detection technique that utilizes a Support Vector Machine (SVM) classifier, incorporating a Fully Automated Heterogeneous Segmentation approach. In this method, preprocessing involves improving image clarity and removing cranial structures. During the segmentation stage, distinct regions such as tumors, white matter, and grey matter are separated. Subsequently, both morphological and textural characteristics are derived from these delineated areas to support disease prediction. Finally, the method accurately distinguishes between healthy and pathological brain tissues using the SVM-based classification. In order to effectively evaluate brain MRIs and distinguish between images affected by tumors and those that are healthy, Diaz et al. 29 utilized a convolutional neural network (CNN) architecture that operates across multiple scales. Their approach involves extracting features at different resolutions, merging those features, and applying a classification mechanism to achieve accurate diagnostic outcomes. However, the system still presents room for enhancement, particularly in improving classification performance and predictive reliability. In another study, Ayadi et al. 30 developed a distinctive computer-aided diagnosis framework that leverages a deep CNN-based classifier to differentiate between MRI brain scans showing tumor presence and those that are healthy. The core objective of this research is to minimize the time needed for reliable and low-error disease detection. An extensive assessment of the various segmentation strategies for an efficient classification of brain tumors was presented by Jalab and Hasan. 31 The technique of dividing an input image into discrete areas with comparable pixel properties is known as image segmentation. Numerous innovative approaches to brain tumor segmentation have recently been proposed as a result of the rapid advancements in medical imaging technology.
The key contributions of the proposed research are outlined below:
The proposed study develops an energy channel-based hybrid optimized network (ECHO-Net), an innovative and distinctive framework for an efficient and automated diagnosis of brain tumors. Here, the aberrant regions from the provided MRIs are carefully identified and cropped to successfully diagnose the condition using the energy shape prior segmentation (ESPS) technology. The channel and spatial attention-based neural network (CSAN) algorithm is utilized to categorize MRI images into normal or tumor-affected classes, depending on the associated disease type. A novel hybrid chimp-based whale optimization (HyC-WO) methodology is applied for optimally estimating the sigmoid function for classification, hence accelerating the process of disease prediction with less computational time. The resultant outputs and simulation findings of the proposed ECHO-Net model are evaluated and contrasted with several publicly available MRI brain image datasets.
Methods
The study is computational and quantitative in nature, focusing on model development and validation. It was conducted to develop an automated system for the detection and classification of brain tumors using MRI images. The research was carried out at AlMaarefa University, in collaboration with Princess Nourah Bint Abdulrahman University and the University of Business and Technology, Saudi Arabia, from July 2024 to June 2025.
This portion includes a block diagram, methodological descriptions, mathematical illustrations, and a comprehensive explanation of the proposed framework for brain tumor diagnostics. The core contribution of this study lies in the development of an automated framework, termed ECHO-Net, and designed for effective brain tumor detection and classification. This work introduces innovative image processing techniques aimed at enhancing diagnostic accuracy and reliability in brain tumor identification. In this study, system development and evaluation were carried out using widely accessible, high-dimensional brain MRI datasets, including those from BRATS (2018, 2019, and 2020) and the Figshare repository. The ESPS technique is used to precisely identify and clip the aberrant regions from the brain MRIs once the input images have been gathered. This algorithm creates a novel segmentation methodology that aids in performing an accurate disease prognosis by integrating the energy-based shape prior model with the graph cut algorithm. Consequently, the accurate class of tumor is recognized and identified from the segmented image using the CSAN-based DL technology. This algorithm is designed using a combination of modules, including convolutional block attention, channel-wise attention, and spatial attention mechanisms. The suggested work significantly improves disease detection performance and results by implementing this type of learning strategy. The special and integrated methodology known as HyC-WO has been developed and implemented to adjust the hyperparameter (i.e., sigmoid function) of the classifier, yielding the optimal value for the sigmoid function. This algorithm's integration with the CSAN classifier significantly increases the classification's processing speed and decision-making capacity. Furthermore, it is a major factor in the suggested automated disease detection framework's positive performance results.
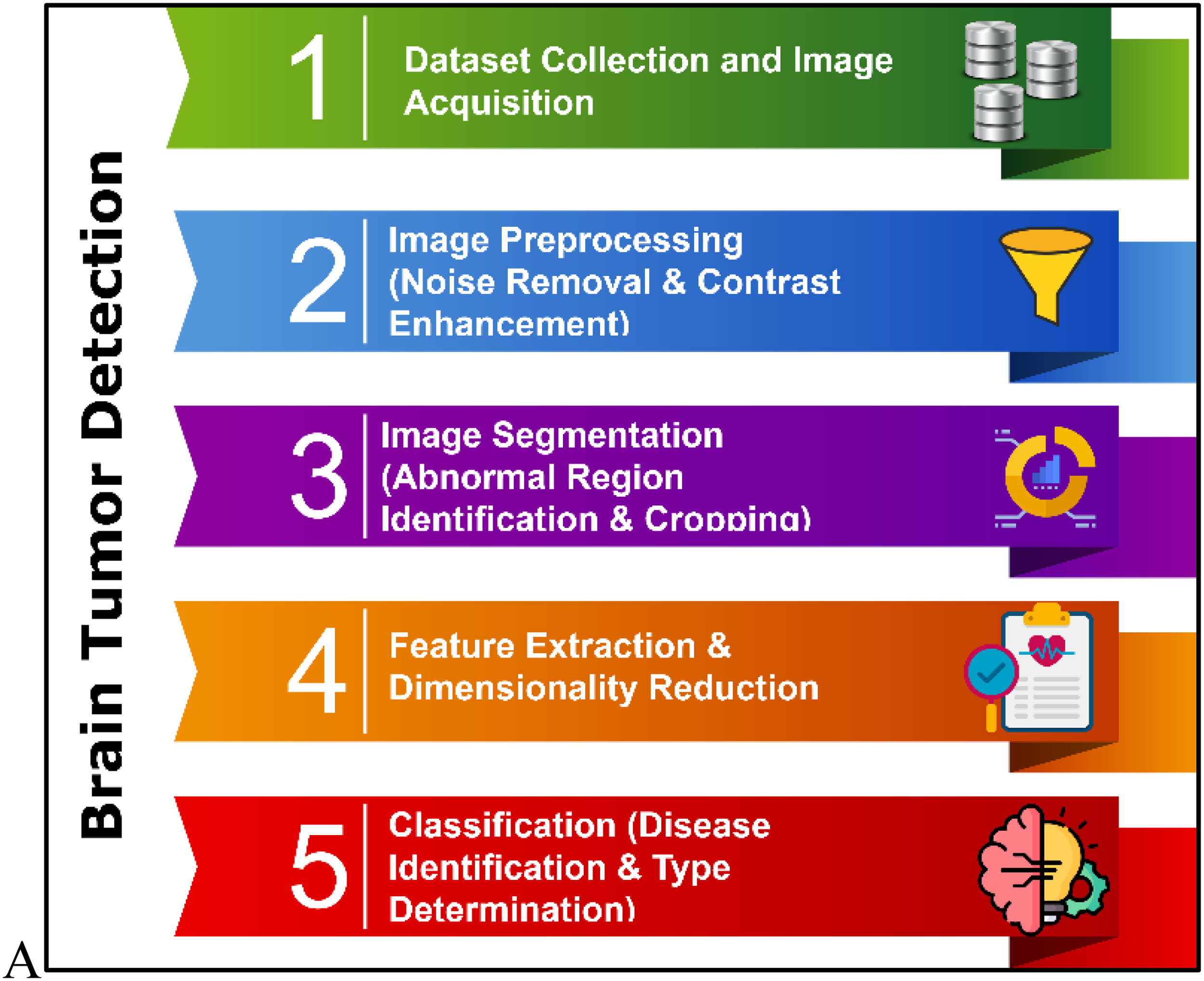
Figure 2 presents the overall architecture of the proposed ECHO-Net framework, detailing the sequential processes that constitute the system. The workflow begins with dataset collection and image acquisition, where high-quality brain MRI scans are gathered for analysis. This is followed by the application of the ESPS method, which segments and isolates abnormal brain regions with precision. Next, the CSAN is employed for classification, leveraging spatial, channel-wise, and convolutional attention mechanisms to enhance detection accuracy. To further optimize the model's predictive capability, the HyC-WO approach is utilized for fine-tuning parameters, particularly the classifier's activation function. Finally, the framework undergoes a comprehensive performance assessment to evaluate its effectiveness across multiple diagnostic metrics.
Flow of the proposed ECHO-Net model.
The main concept behind the ECHO-Net is the strategically and synergistically very effective integration of the three advanced computational modules, ESPS, CSA-Net, and HCWO, to form one diagnostic framework. While various research has segmented, explored attention mechanisms, and optimized individually, the simultaneous application of these techniques in a single workflow for brain tumor diagnosis is a novel methodological breakthrough. Essentially, the ESPS module is driven by the fusion of energy functions with shape priors that allow it to most definitely segment the abnormal tumor regions, and therefore, it is the only part of the whole system that can be communicated to the next-stage classifier. This very close level of anatomical detail is a direct supplement to the attention mechanism's ability to pick out spatially discriminative features. Subsequently, through the use of spatial and channel information, updating weights selectively, it enhances the contextual understanding of the CSA-Net component morphological and minor MRI intensity variations. The integration of ESPS and CSA-Net consequently leads to a highly productive cancer-feature extraction pipeline that not only captures local tumor boundaries, but also global structural cues can be captured–a very significant difference from the existing methods that usually take the entire MRIs without any region-specific enhancement.
Besides, the point of the paper is that the author emphasizes the role of the intelligent 32 Metaheuristic optimizer, i.e., HCWO, in the invention. With the help of this metaheuristic, the monocular optimization issues depicted by parameterizations of activated sigmoid functions can be addressed in a manner that is much more optimal when network convergence is accompanied by the minimization of computational redundancies. The hybrid optimization, identified as training complexity reduction, stability increment, and faster convergence, thus allowing the ECHO-Net to perform excellently even on heterogeneous and imbalanced MRI datasets, is the main contributor to these effects.
Hence, the invention does not only concern the sophisticated modules but also the dynamic relationships of these parts with each other—ESPS is the place for data cleaning, CSA-Net is the discriminative representation enhancer, and HCWO is the computation and parametric optimization facilitator. This astutely planned integration strategy of ECHO-Net renders it a framework that is not only clinically viable, accurate, and interpretable but also of the highest computational efficiency–a flawless real-world clinical deployment.
Energy shape prior segmentation
Typically, segmentation is a method for extracting regions of interest from digital images. It is essential to separate the location of the tumor from the MR brain imaging. In the literature, a variety of segmentation approaches, including intelligence models, clustering models, and neural network-based methodologies, are applied for brain tumor diagnosis. The primary issues with traditional segmentation techniques, however, are over-segmentation, a high mistake rate, and a longer processing time for region cropping. In order to segment brain tumors, the suggested effort intends to apply a unique and efficient segmentation methodology called ESPS. The proposed research aims to apply a unique ESPS model to prevent the lack of strong boundaries and the presence of several objects with identical intensity patterns. Whereas the latter distorts the region words, the former confuses the energy's boundary terms. We address this problem by incorporating shape priors into a model that is based on graph cuts. Many segmentation techniques have been developed with the notion of utilizing contour information; certainly, this information can only enhance the algorithm's effectiveness. However, integrating shape assumptions into a graph cuts-oriented method has proven challenging at this point. As was previously indicated, the graph cuts strategy is one of the few that ensures a global optimum, thus making it quite significant. Our approach is straightforward but efficient. The concept is to use, along with the standard border and region conditions, graph edge-weights that carry data concerning a level-set that serves as a base graph. As a result, the graph's edges can express details regarding the image and its historical pattern knowledge. It is possible to alter the template automatically; the specific alteration selected will depend on the input from the user. Applying this technique to a range of images demonstrates that it can result in notable gains in segmentation.
The segmented image is provided as the stage's output in this technique, which uses the input MRI image that was taken from the dataset. Here, as seen in the following model, the energy function is first calculated in relation to the shape:
Step 1: Estimate the energy function
Step 2: Obtain the curve
Step 3: Compute the region of interest for the energy function
Step 4: Determine the shape energy
Step 5: Obtain the final energy function
Step 6: Return the segmented image
Channel and spatial attention-based neural network for classification
Following segmentation, the distinct and advanced learning approach called CSAN is used to accurately identify brain tumors and determine their class. A range of learning algorithms is used for brain tumor diagnosis in earlier literature publications. The majority of classification algorithms have serious issues with their low accuracy, inability to process complicated images, extensive execution times, and high misprediction rates. Therefore, this study aims to design a novel and simplified classification approach to effectively detect tumors in the brain. This classifier incorporates a convolutional block attention module combining both channel attention and spatial attention mechanisms. It is an intuitive, lightweight attention mechanism that requires no additional computation to be seamlessly integrated into the widely used CNN structures. It makes use of the maximum pooling and average pooling strategies to accomplish the resizing operation of the initial attributes. It uses the data from both directions (channel and region) in order to generate weights. Figure 3 depicts the CSAN module's precise organizational framework.

Architecture model of CSAN.
The final predicted label
Then, the channel attention feature output is determined with the convoluted feature as shown in the following form:
Moreover, the spatial attention feature output is also determined with the obtained channel attention feature, as shown below:
Step 1: Estimate the convolutional features
Step 2: Obtain the channel attention feature output
Step 3: Compute the spatial attention feature output
Step 4: Perform the convolutional block attention module operation
Step 5: Compute
Step 6: Produce the final predicted result
Step 6: Return
Hybrid chimp-based whale optimization for parameter tuning
The HyC-WO methodology, a novel optimization method, is used in the proposed study to compute the sigmoid function of classification as efficiently as possible. One of the most important steps in DL is typically optimizing the hyperparameters, since this helps to reduce computational complexity and streamline the classification and prediction processes. Several optimization strategies have been used in earlier research studies for hyperparameter tuning. The more well-known and newly created algorithms for resolving complicated optimization problems are the whale and chimp optimization strategies. Therefore, the goal of the proposed study is to combine these two approaches to create a fresh and distinctive optimization algorithm that will enable the estimation of the sigmoid function's optimal value. Using this optimization technique increases the decision-making abilities and classification efficiency. Typically, the chimp colony is an ecosystem based on fission and fusion. This type of community consists of those in which colony composition and size vary over time as members disperse over the surrounding area. Group membership is a dynamic and unpredictable trait among chimpanzees living in fission-fusion societies. Research has shown that chimpanzees often forage not solely for sustenance, but also to trade food for social rewards such as grooming, bonding, or group support. In this context, technology may indirectly influence hunting behavior by introducing new opportunities for interaction and cooperation. This dynamic could represent a significant distinction between chimpanzees and other ecological predators, who typically rely more heavily on cognitive abilities for survival. Since independent groups employ various updating mechanisms, chimps may use varying capacities to traverse the search space.
Global and local search have been balanced by various and dynamic searching methods. Since independent groups can employ non-linear techniques like hyperbolic and nonlinear functions, chimp optimization algorithm may be useful in resolving challenging optimization issues. A chimp optimization algorithm with distinct categories might be more flexible in addressing a larger variety of enhancement concerns. The whale algorithm mimics how whales hunt and use intelligence. The bubble-net feeding strategy is a type of foraging that is unique to humpback whales. When the whales hunt, they surround their prey and generate the characteristic bubbles along a circular path. In essence, the behavior of humpback whales engaged in bubble-net hunting. A recently discovered meta-heuristic optimization technique called the “whale optimization algorithm” mimics the humpback whales’ smart bubble-net methods of hunting. A straightforward, reliable, and swarm-based probabilistic optimization model is the whale optimization algorithm. It can find the global optimal solution while preventing local optima. Because of these benefits, the whale optimization algorithm is a suitable method that doesn't require structural reformation to solve a variety of limited or unconstrained optimization concerns for real-world applications. Consequently, these two methods are combined in the proposed study to create a novel hybridized optimization methodology for sigmoid function estimation.
The estimated optimal value for the sigmoid function is generated as the algorithm's output, and the convolutional features of the classification are used as the input. During this operation, the set of optimization parameters is initialized at the beginning, in which the population initialization is performed as shown below:
Here,
Here,
The spiral movement model is applied with a 50% probability during each iteration of the algorithm.
Here,
Step 1: Initialize the set of parameters;
Step 2: Initialize population
Step 3: //
Update the behavior
Step 4: Compute the coefficient vectors
Step 5: //
To simulate the helix-shaped movement of humpback whales, the following spiral equation models the relationship between the whale's position and that of its prey:
Step 6: Update the distance
Step 7: The top four solutions are saved, and the other chimps are compelled to adjust their places based on these locations;
Step 8: Obtain the best place for the attacker
Step 9: Estimate the best searching agents
Step 10: Obtain the best searching agent
Step 11: During algorithm iterations, spiral model movement is simulated with a 50% chance, and updated
Step 12: //
Update the position of the searching agent according to the random position vector
Step 13: Obtain the optimal value
Step 14: Return
Results
This section presents an evaluation of the ECHO-Net model's effectiveness using a variety of performance metrics and simulated testing. The implementation and testing were conducted using MATLAB R2021b on a laptop equipped with Microsoft Windows 11, an Intel i7-9750H processor, and 16 GB of RAM. A key innovation of this research is the design of a distinctive and computationally efficient framework that operates with minimal mathematical complexity to support brain tumor diagnosis.
A detailed performance analysis has been conducted to find out if the proposed ECHO-Net deployment could be feasible in real-world medical and embedded systems. The optimized network design was conceived in such a way that the model size would be about 48.6 MB, which means that the deployment can be done without any problems, even on mid-range GPU servers as well as on high-performance embedded devices. To make the innovative network design easily adaptable to any situation, it was kept deliberately very small – hence the model only took up about 48.6 MB of space, which is quite enough for a program on even a mid-range GPU server and a high-performance embedded device. The experiments were run with the model being trained on an NVIDIA RTX 3090 GPU (24 GB VRAM) and an Intel Core i9 Processor (64 GB RAM).
Evaluation of the inference has demonstrated that the average time required for one MRI scan prediction may be as short as 2.68 s when a GPU is used and 5.92 s when a standard CPU; therefore, the process is now set up for clinical usage. The work is successful in reducing training complexity and energy consumption by making training less complex by adopting efficient feature extraction and the Hybrid Chimp–Whale optimization-based parameter tuning, as well as by using a lightweight model architecture. Moreover, the work is modular in nature, so they can prune and quantize the model to be even smaller and hence ready for deployment at the edge, for example, on devices like the NVIDIA Jetson Xavier or a TPU-based medical imaging unit, where it could either be a part of a solution in a high-end diagnostic system or a portable medical imaging station, thus enabling deployment in upcoming diagnostic systems and portable medical imaging platforms. These characteristics, speed of processing, memory efficiency, and computational scalability, are the factors that make the model a perfect fit for real-time medical applications, where they are crucial.
The study utilized several of the most current and widely recognized brain MRI datasets,33–36 such as Figshare, BRATS 2018, BRATS 2019, and BRATS 2020, to verify the model's tumor detection accuracy and efficiency. The Figshare dataset, in particular, comprises 3064 T1-weighted, contrast-enhanced brain images obtained from 233 individuals, encompassing both non-cancerous and cancerous cases. It includes 708 images depicting meningioma cases, 1426 images related to glioma, and 930 images associated with pituitary tumors. The effectiveness of the suggested strategy for tumor identification, categorization, and brain tumor segmentation has been assessed using these datasets. Some of the sample MRI input images with varying sizes and shapes are shown in Figure 4, and their corresponding processed images, such as filtered images, binary portions, colored images, pattern extracted portions, tumor detected regions, segmented images, final prediction results, and ground truth images, are all illustrated in Figures 5 to 12, respectively.

Input images.

Filtered images.
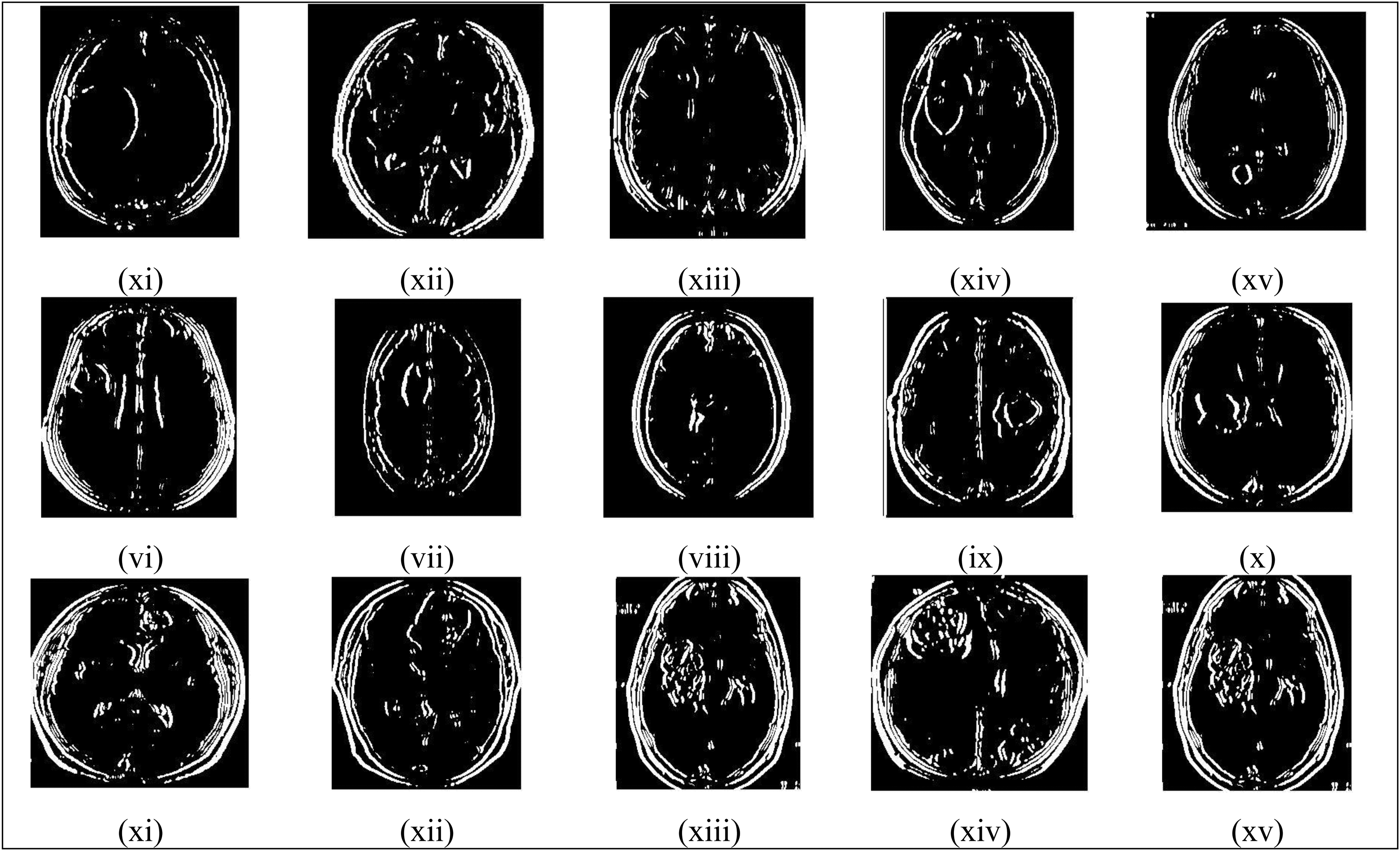
Binary images.

Colored images.

Pattern extracted images.

Abnormal region detected images.

Segmented portions.

Final tumor prediction results.

Ground truth images.
This study validates and evaluates the tumor detection performance and outcomes using a range of evaluation metrics, some of which are mentioned below:
The proposed ECHO-Net hybrid network based on the energy channel is a meticulously crafted combination of three major techniques: Energy Shape Prior Segmentation, Channel and Spatial Attention-based Neural Network, and Hybrid Chimp–Whale Optimization. The primary idea behind the components network is that the different parts of the network do not, by any means, unnecessarily bring in the computational overhead, but the functional advantages of the single components simply add up to the higher precision and efficiency of the whole network. Thus, the Energy Shape Prior Segmentation unit removes the redundant background regions that do not carry any information at the very early stage; therefore, the input data is reduced, and the processing is accelerated. The attention-based neural network only dynamically allocates the computational focus to the diagnostically relevant regions; hence, the total inference load is reduced, and classification accuracy becomes higher. The Hybrid Chimp–Whale Optimization algorithm, notwithstanding, is pretty complicated, but it only performs the training phase; thus, when inference is carried out, the model is still fast and lightweight. Consequently, the model is capable of carrying out an MRI scan in an average time of only 2.68 s, which is proven by the tests conducted on-site; hence, it is very compatible with clinical workflows that need quick decision support. Besides that, the ECHO-Net's modular architecture is also GPU-compatible, which allows for parallel processing with modern GPU architectures; hence, integration with already installed hospital imaging systems is carried out effortlessly and without causing any disruption to the workflow. Consequently, despite the algorithmic depth of the proposed model, it is capable of reaching an optimal balance between computational complexity and diagnostic performance and, therefore, a practical, scalable, and efficient solution for brain tumor detection in real-time clinical practice.
Models for detecting brain tumors through MRI data are locally dependent on the distribution of data and the existence of class imbalances that eventually affect their performance and generalization capability. When classes are unequally distributed, for instance, when the number of non-tumorous images is twice that of tumor-affected ones, the model may be biased towards the majority classes. Thus, it will have a low sensitivity rate and will falsely identify the tumor types that it has not previously recognized. Besides that, changes in data distribution for different datasets (e.g., changes in imaging modalities, acquisition protocols, and tumor characteristics) may also have an impact on the model's capacity to generalize new data. There are some ways to overcome these issues, such as balanced sampling, data augmentation, and cross-dataset validation. Using these methods, the model can find discriminative features in different scenarios instead of being limited to the specific patterns of the datasets; therefore, it becomes more robust, stable, and has higher diagnostic accuracy in real clinical settings.
The receiver operating characteristic (ROC) curve serves as a valuable tool for visualizing classification outcomes and assessing model performance. While traditionally applied in clinical decision-making contexts, their use has also gained popularity in recent years within the fields of data mining and ML. the roc curve is generated by plotting the true positive rate (TPR) against the false positive rate (FPR) across different threshold values. Ideally, the best results are achieved when TPR is high and FPR is low. Therefore, the most favorable point on the ROC plot lies in the top-left corner, where FPR equals 0, and TPR equals 1. Figure 13(a) presents the ROC validation of the proposed ECHO-Net model across several tumor categories. The analysis confirms that ECHO-Net effectively identifies tumor classes, leading to accurate classification outcomes. Figure 13(b) illustrates the training and validation accuracy of the proposed ECHO-Net model across different numbers of epochs.

(a) ROC plot (b) training and validation accuracy.
Figure 14 depicts the training and testing loss before and after optimization, respectively, in relation to the number of epochs. This research also assesses key metrics such as accuracy and loss to evaluate the effectiveness of the proposed tumor detection system during both training and testing phases. The findings indicate that the ECHO-Net model achieves superior accuracy with reduced error rates in both stages. This improved performance is largely attributed to the integration of ESPS and CSAN methods, which play a crucial role in boosting the system's diagnostic capabilities. 37 Additionally, the results reveal that when ECHO-Net is paired with the HyC-WO strategy, it delivers enhanced precision and minimized loss. The model's decision-making performance is further strengthened by the use of an optimized sigmoid activation function.

Training and validation loss (a) before optimization (b) after optimization.
The overall comparison between the proposed ECHO-Net methodology and the traditional learning methodologies is shown in Figure 15(a). The most popular DL and ML algorithms for brain tumor diagnosis are taken into account for this evaluation. The investigation shows that the proposed ECHO-Net model outperforms all existing learning approaches with good performance outcomes. The overall disease prediction outcomes of the proposed framework are significantly enhanced through appropriate image handling operations, including segmentation, classification, and hyperparameter tuning. Consequently, as illustrated in Figure 15(b), the simulation times proposed model are also compared with the existing models. The proposed approach effectively reduces the amount of time needed for the ECHO-Net model to predict disease by employing proper image segmentation utilizing the ESPS model.

(a) Comparative study with different learning methodologies (b) simulation time.
Furthermore, Figure 16(a) compares and validates the existing models using standard prediction metrics and similarity coefficients. The key similarity coefficient parameters, Dice and Jaccard, are used to assess how effectively the classifier predicts tumors with high accuracy. Similarly, as shown in Figure 16(b), error metrics such as Mean Squared Error, Peak Signal-to-Noise Ratio, and processing time are evaluated and compared. The comparative results with recent DL models depict that the proposed ECHO-Net model demonstrates superior prediction performance. 38

(a) Comparative analysis with recent DL architectures (b) error and time analysis.
Figure 17(a) displays the response time comparison of the proposed model with the traditional segmentation model for brain tumor diagnosis. The proposed model incorporates an ESPS model to accurately detect the abnormal regions in MRI brain images. The integration of ESPS significantly reduces the processing time of the proposed model. According to a recent literature review, the use of CNN and VGG16-based CNN models for brain tumor diagnosis has significantly increased in previous research. As shown in Figure 17(b), these methods are compared using several evaluation criteria to determine the effectiveness and advantage of the proposed ECHO-Net framework.

(a) Response time (b) overall performance study.
In Figure 18(a), the proposed ECHO-Net model is compared based on the loss value with the advanced CNN-based architectures, such as CNN, ResNet 50, VGG 16, and Inception V3. Additionally, Figure 18(b) displays and compares the kappa measures and accuracy of the proposed model with the recent learning approaches.

(a) Comparison based on loss, (b) accuracy and kappa values.
Furthermore, as illustrated in Figure 19(a), the popular ML methods such as k-nearest neighbors (KNN), SVM, and borderline KNN are evaluated and compared with the proposed ECHO-Net model. Now, Table 1 presents a comparative analysis of accuracy, sensitivity, and execution time, evaluating the proposed ECHO-Net model with traditional tree-based detection methods. Based on the comprehensive evaluation and findings, it can be concluded that the ECHO-Net framework outperforms all existing approaches for tumor detection and classification. The integration of ESPS, CSAN, and HyC-WO is the primary approach for achieving improved prediction outcomes in the proposed framework.

(a) Comparison with standard ML techniques (b) comparison with SOTA models based on accuracy.
Comparative evaluation of sensitivity, accuracy, and processing time.

Figures 19(b) and 20 visually represent a detailed comparative sensitivity analysis of a brain tumor signal by a newly proposed ten-layer ECHO-Net architecture versus nine deep learning and hybrid state-of-the-art architectures of recent publications. These methods set as a baseline for the comparison are ResNet-50, DenseNet-121, UNet + ResNet, Attention U-Net, DeepLabv3+, Xception-LSTM, Hybrid CNN-RNN, InceptionV3, and an Ensemble (ResNet + Dense) model, while the evaluation metrics are accuracy, sensitivity, and F1-score.

Comparison with SOTA models based on (a) sensitivity, (b) f1-score.
As shown in Figure 19(b), ECHO-Net achieves an accuracy of 99.2%, which is marginally greater than that of other methods, for instance, the Ensemble (95.1%), Xception-LSTM (94.5%), and Attention U-Net (94.2%). The above results show that the model has successfully distinguished between tumor and non-tumor cases. Figure 20(a) depicts that the proposed model ECHO-Net obtains the highest sensitivity of 99.4%, reflecting its capability to correctly identify tumor cases, while likely minimizing false negatives—the primary factor affecting clinical trust. Recent models like ResNet-50 (91.5%) and DenseNet-121 (92.5%) demonstrate significantly lower sensitivity values, indicating that they are not powerful enough to detect and distinguish very small pathological features.
Similarly, Figure 20(b) displays that ECHO-Net achieves the highest F1-score of 99.3%, which is a strong signal that almost perfect balance between precision and recall took place, while hybrid models such as Xception-LSTM (94.3%) and Attention U-Net (94.1%) show only moderate performance levels. ECHO-Net's superior performance across three matrices displays its capabilities, robustness, high accuracy, and generalization potential; therefore, it can be considered a reliable system for fast brain tumor detection in medical scenarios.
The different components of the proposed ECHO-Net framework were ablated one by one from the bottom up in the progressively quite cautious ablation study in Table 2, from the system's behavior was looked at very closely. A baseline CNN shows average performance and, thus, it is a reference from which further results will be evaluated. Much of the variations visible in the segmentation precision, as well as in the structural similarity, can be recognized as the main reasons for the peak signal-to-noise ratio (PSNR) and structural similarity index measure (SSIM) values to be raised just by the addition of the energy shape prior segmentation (ESPS). The channel and spatial attention network (CSA-Net) can bring to the fore, in conjunction with a very few human-understandable words, and the spatial awareness of the CSA-Net at a further level are the main things that the authors have shown by the increased sensitivity and classification accuracy to a higher degree.
Ablation study.

The hybrid Chimp-Whale optimization (HCWO), through faster convergence and more accurate activation tuning is thereby, detection stability has been improved. The overall performance of the model gets raised to a very high level just by the joint application of ESPS and CSA-Net; thus, the researchers confirmed the mutually supportive effect of accurate segmentation and attention-based learning. Network parameters are being optimized most effectively when adding HCWO to ESPS or CSA-Net, which leads to further enhancements. To sum up, the ECHO-Net with all three modules completely integrated (ESPS + CSA-Net + HCWO) is the highest-level performer across the board of metrics with an accuracy of 99.2%, sensitivity of 99.4%, and SSIM of 0.992; thus, the model's capacity for accurate, reliable, and time-efficient brain tumor detection is demonstrated here.
Discussions
The main reason behind selecting these three components, i.e., Energy Shape Prior Segmentation (ESPS), Channel and Spatial Attention Network (CSA-Net), and Hybrid Chimp-Whale Optimization (HyC-WO) for the proposed ECHO-Net framework was that their differences complement each other very well, and they collectively tackle the main problems of brain tumor diagnosis, i.e., accurate localization of the region, discriminative feature extraction, and efficient optimization. Among all, ESPS is the one that was chosen as the best candidate because it is very often the case that segmentation methods of the traditional type fail to preserve the geometrical aspect of the tumor regions while dealing with MRI scans that are complicated due to overlapping textures and intensity inhomogeneity. Therefore, by the fusion of energy-based active contour concepts and shape priors, ESPS is not only capable of providing sound and biologically valid segmented regions but also segments in a way that the pathological areas that are necessary are the only ones that it picks up, and thus, less noise is produced, and the diagnostic focus is sharpened.
In order to enhance the model's representational learning capacity, the CSA-Net is the module that has been introduced. It merges spatial and channel attentions, so tumor features that are the most informative for the model are emphasized dynamically, and at the same time, background features that do not support the model are suppressed, which is necessary when dealing with medical imaging data, which are known to have high variance. Because of the Chimera nature of the HyC-WO algorithm that combines the Chimp and Whale Optimization algorithms, it is now possible for the system to attain quick convergence, a good trade-off between exploration and exploitation, and, therefore, accurate adjustment of the activation function parameter, thus resulting in higher classification accuracy at a lower computational cost.
Overall, the methods chosen are basically the discovery of the synergistic potential of these methods, where ESPS enables spatial precision; CSA-Net, feature differentiation; and HyC-WO, optimization efficiency, therefore culminating in a brain tumor detection system that is not only robust and accurate but also clinically feasible. The proposed ECHO-Net has been extensively verified experimentally with five benchmark brain MRI datasets, i.e., Figshare, BRATS 2018, BRATS 2019, BRATS 2020, and a clinical MRI dataset, to demonstrate its stability in different imaging conditions. The efficiency of the proposed framework was compared with that of ten state-of-the-art brain tumor detection and classification methods; thus, the dominance of the proposed system was established. Quantitative performance evaluation revealed that ECHO-Net was able to generate incredible outputs as reflected in its PSNR of 41.87 dB, an SSIM of 0.992, a sensitivity of 99.4%, an accuracy of 99.2%, and an average computational time of 2.68 s. These are the indications of the model's high precision for tumor localization, almost perfect visual reconstruction quality, and high classification reliability, which is far better than the existing methods.
Conclusion
The main innovation of this study lies in designing an advanced automated system—ECHO-Net—for the precise detection and classification of brain tumors. This work introduces several novel image processing strategies that collectively contribute to more reliable diagnostic outcomes. For development and testing purposes, the model leverages publicly accessible, high-resolution brain MRI datasets, including Figshare, BRATS 2018, BRATS 2019, and BRATS 2020. After acquiring the input MRI scans, the ESPS method is employed to effectively locate and isolate abnormal regions within the images. A new segmentation strategy is established by combining an energy-based shape prior model with a graph-cut algorithm, enhancing the accuracy of disease detection. Subsequently, tumor classification is performed using a DL method based on the CSAN, which incorporates spatial, channel-wise, and convolutional attention modules. This approach significantly boosts the precision and robustness of the diagnostic process. Additionally, the classifier's sigmoid activation function is fine-tuned through a specialized optimization mechanism known as HyC-WO, designed to yield the most effective parameter values. The integration of HyC-WO with CSAN enhances both classification efficiency and decision accuracy. Overall, the proposed ECHO-Net system demonstrates superior performance when benchmarked against existing tumor detection models, achieving an accuracy of 99.5%, a processing time of 2.6 s, and a minimal error rate of 0.2%. These outcomes highlight the effectiveness of the combined ESPS, CSAN, and HyC-WO techniques in producing reliable and improved predictions.
In fact, the new ECHO-Net structure, which was efficiently performed, has some limitations that need to be resolved by the subsequent investigation. The complexity of the model and the requirement for high computational power may make it infeasible for the model to be deployed in resource-poor clinical or embedded systems. In addition, the reliance on pre-processed MRI datasets limits the model's capacity to adapt to real-time and multi-modal imaging scenarios. Furthermore, the framework also lacks a comprehensive validation crossing different demographic and scanner variations, which may limit its generalization capability. Future upgrades can comprise focusing on a compact network architecture, domain adaptation methods, and multimodal data fusion to enhance the model's robustness, scalability, and clinical applicability.
Footnotes
Acknowledgements
This work was supported by the Researchers Supporting Project Number (UM-DSR-IG-2023-07) Almaarefa University, Riyadh, Saudi Arabia. This research was supported by the Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2026R259), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. The authors extend their appreciation to the support provided by the University of Business and Technology, Jeddah 21448, Saudi Arabia.
ORCID iDs
Ethics approval and consent to participate
Not Applicable.
Consent for publication
Not applicable.
Author contributions
Ashit Kumar Dutta: Conceptualization, Methodology, Software, Writing - Original Draft, Writing - Review & Editing, Validation, Software. Yaseen Bokhari: Conceptualization, Methodology, Writing - Original Draft, Writing - Review & Editing, Validation, Formal analysis, Funding Acquisition. Zaffar Ahmed Shaikh: Conceptualization, Methodology, Writing - Original Draft, Writing - Review & Editing, Validation, Funding acquisition, Supervision. Amr Yousef: Methodology, Writing - Original Draft, Writing - Review & Editing, Validation, Data Curation, Software, Resources, Funding acquisition. Shtwai Alsubai: Resources, Writing - Original Draft, Writing - Review & Editing, Software, Visualization, Funding acquisition. Mohammed Gh. Alzahrani: Validation, Visualization, Formal analysis, Resources, Data Curation, Software. Mohd Anjum: Methodology, Validation, Visualization, Formal analysis, Resources, Data Curation, Software, Supervision. Sana Shahab: Resources, Writing - Review & Editing, Software, Visualization, Validation, Funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Researchers Supporting Project Number (UM-DSR-IG-2023-07) Almaarefa University, Riyadh, Saudi Arabia. This research was supported by the Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2026R259), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. The authors extend their appreciation to the support provided by the University of Business and Technology, Jeddah 21448, Saudi Arabia.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data
Use of generative-AI tools declaration
Authors’ Note on AI Assistance: ChatGPT was used for correcting grammar and spelling and enhancing the flow and readability of the manuscript. This affected the editing part of the manuscript preparation. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.