Abstract
Objectives
This study is the first to develop and evaluate a machine learning (ML) model for predicting pelvic adhesions based on ultrasound features, utilizing the SHapley Additive Explanations (SHAP) framework for interpretability analysis.
Methods
This prospective study included 220 patients who underwent laparoscopic surgery and preoperative ultrasound assessments at our hospital between April 2023 and June 2024. Patients were randomly assigned to training and validation sets. A Least Absolute Shrinkage and Selection Operator regression was used to identify independent risk factors, followed by incorporation into an Extreme Gradient Boosting prediction model. Model performance was evaluated using receiver operating characteristic (ROC) curves, calibration plots, and a decision curve analysis.
Results
The included patients were randomly divided into a training set and a validation set in a 7:3 ratio. The final model included four predictors—obstructed ovarian activity, surgical history, endometriosis, and gynecological inflammation—and demonstrated strong discriminatory performance, with an area under the ROC curve of 0.869 and 0.846 in the training and validation sets, respectively. The ML model demonstrated a sensitivity of 0.946 and a specificity of 0.597 in the training set, while in the validation set, it achieved a sensitivity of 1.000 and a specificity of 0.600. Calibration analyses showed good agreement between predicted and observed outcomes. The model exhibited high clinical utility. SHAP analysis revealed that endometriosis contributed most significantly to the predictions, followed by surgical history, obstructed ovarian activity, and gynecological inflammation.
Conclusions
The interpretable ML model developed in this study demonstrates strong predictive performance for assessing the risk of pelvic adhesions in patients prior to surgery. It can be utilized to accurately identify high-risk patients before the procedure, enabling the implementation of appropriate measures during surgery to reduce the occurrence of postoperative pelvic adhesions.
Background
Pelvic adhesion is the most common complication following abdominal or pelvic surgery. Unlike other postoperative conditions, such as wound infections or anastomotic leaks, most adhesions remain clinically silent. Nonetheless, they can lead to infertility and chronic pelvic pain by forming between the fallopian tubes, uterus, and ovaries, thereby disrupting normal reproductive function.1–3
A key challenge in managing pelvic adhesions lies in the uncertainty surrounding their biological mechanisms. Adhesion formation is primarily driven by inflammatory responses, fibrous tissue deposition, and an imbalance between coagulation and fibrinolysis. Elevated levels of plasminogen activator inhibitor (PAI)-1 and the tissue-type plasminogen activator/PAI complex (tPA/PAI) may indicate increased risk. In cases where fibrinolysis is impaired, the temporary fibrin matrix persists, promoting neovascularization and, ultimately, the formation of stable adhesions.4,5 Although minimally invasive surgery may reduce adhesion incidence and associated morbidity and mortality, 6 effective strategies for preventing postoperative adhesions are lacking. 7 Thus, early identification of high-risk individuals is essential for implementing timely preventive measures.
Among diagnostic tools, the visceral slide test, introduced by Sigel et al. 8 in 1991, is among the most widely used methods to detect intra-abdominal adhesions and prevent surgical injury. This technique assesses the mobility of intra-abdominal viscera during spontaneous or forced respiration or manual manipulation. A negative sliding sign may indicate adhesion to the abdominal wall. However, results across studies vary substantially due to differences in anatomical landmarks (e.g. umbilical versus periumbilical regions), with reported sensitivity and specificity ranging from 17–100% and 32–100%, respectively.9–12
Machine learning (ML) has emerged as a powerful pattern recognition and knowledge extraction approach. Unlike traditional statistical methods, ML models can uncover latent patterns in data and often exhibit stronger generalization capabilities.13,14 Among them, Extreme Gradient Boosting (XGBoost) is a highly effective algorithm that uses a second-order Taylor expansion to optimize the loss function and incorporates regularization to prevent overfitting. Techniques such as the synthetic minority oversampling technique combined with an edited nearest neighbor are often applied during preprocessing to address class imbalance. 15 Wang et al. 16 developed and validated an XGBoost prediction model for predicting adhesive small bowel obstruction by integrating deep learning-based computed tomography imaging features with clinical electronic health records. This model achieved AUCs of up to 0.915 in the training set and 0.912 in the validation set.
One major limitation of ML, however, is its lack of interpretability. 17 SHapley Additive Explanations (SHAP), based on cooperative game theory, provides both local and global explanations by estimating each feature's contribution to model predictions, thereby addressing the so-called “black-box” problem.
In conclusion, this study aimed to integrate ultrasound-based sliding sign assessments and clinical features to develop a risk prediction model for pelvic adhesions using the XGBoost algorithm. Additionally, we applied the SHAP framework to enhance model interpretability by quantifying the relative importance of each predictive feature. Our goal was to create a clinically useful, interpretable tool to support the early identification of high-risk patients and improve surgical planning and patient outcomes.
Methods
Patients
In this prospective study, adult women who underwent gynecological laparoscopic surgery at the Second Affiliated Hospital of Fujian Medical University between April 2023 and June 2024 were enrolled.
Inclusion criteria were: (1) age between 18 and 60 years; (2) regular menstrual cycles; and (3) absence of serious systemic diseases.
Exclusion criteria were: (1) acute genital infections, malignancies, or adnexal lesions; (2) pregnant or menopausal; (3) comorbidities such as hypertension, coronary heart disease, or diabetes; (4) emergency surgery; and (5) absence of sexual history.
This study was approved by the Ethics Committee of the Second Affiliated Hospital of Fujian Medical University [IRB-2023-26]. All participants had fully understood the relevant details of the study and signed the written informed consent form prior to their enrollment in the study and undergoing ultrasound examination. This study was conducted adhering to the principles of the Declaration of Helsinki.
Sample calculation
This study aims to build a predictive model for pelvic adhesion. The overall incidence rate of all participants ultimately enrolled in the study is 0.605. According to the sample size calculation formula proposed by Riley et al., 18 we have calculated that our research requires a minimum of 197 participants to be enrolled.
Preoperative ultrasonography examination
Ultrasound equipment
Ultrasound examinations were conducted using GE Voluson E8 and E10 machines (GE Healthcare, Chicago, IL, USA), equipped with RIC 5-9-D transvaginal probes (5–9 MHz) and C1-6 abdominal probes (1–6 MHz).
Assessment procedures.
Transabdominal color Doppler ultrasound examination
Patients lay in the supine position with the lower abdomen exposed. After applying an ultrasound coupling agent, the C1-6 probe was positioned on the abdomen to assess the sliding motion of intra-abdominal organs during quiet and deep breathing. A positive finding was defined as a spontaneous visceral sliding distance of <1 cm or amplitude of <2 cm during deep inspiration.
Transvaginal color Doppler ultrasound examination
Patients were placed in the lithotomy position. The RIC 5-9-D transvaginal probe covered with a disposable sterile cover was slowly inserted into the vagina to examine the cervix and vaginal fornix. The probe was rotated to examine the uterus and adnexa. Pressure was applied to assess ovarian adhesions to adjacent organs, such as the broad ligament, uterosacral ligament, rectum, bladder, and peritoneum. If the relative mobility between the ovary and adjacent organs is restricted, it suggests the presence of obstructed ovarian activity; otherwise, the result for obstructed ovarian activity is negative. The Pouch of Douglas was evaluated for pelvic fluid, and the “sliding sign” was dynamically assessed. For anteverted uteri, cervical pressure was used to observe anterior rectal wall-cervix/vaginal posterior wall sliding (positive if present); for retroverted uteri, posterior uterine wall probe pressure was used to observe anterior rectal wall-posterior uterine wall sliding (positive if present). The examination was continued in the anterior and posterior pelvic compartments.
The anterior compartment includes the bladder, vesicouterine space, and ureters. The posterior compartment includes the uterosacral ligaments, rectovaginal septum, posterior vaginal fornix, anterior rectal wall, rectosigmoid junction, and anterior rectosigmoid wall. The vesicouterine space was assessed using the sliding sign technique, a transvaginal probe at the anterior vaginal fornix sliding between the posterior wall of the bladder and the anterior wall of the uterus to determine the presence of a sliding sign.
In summary, the present study will collect the following sonographic indicators: obstructed ovarian activity, obstruction of bowel movement beneath the abdominal wall, negative sliding sign between the anterior rectal wall and the uterus, sliding sign of the interstitial region between the bladder and the uterus, and pelvic encapsulated effusion.
Data collection
Pelvic adhesions were diagnosed intraoperatively using the modified American Fertility Society (AFS) scoring system, 19 with scores >1 indicating adhesion presence.
Demographic and clinical data, including age, body mass index (BMI), history of abdominal surgery, endometriosis, chronic pelvic inflammation, and other relevant variables were collected before surgery (Table 1).
Comparison of baseline features between training and validation sets.

BMI: body mass index; SD: standard deviation.
Selection of interpretability tools
The current mainstream interpretability tools include SHAP, LIME, and permutation importance. A core advantage of SHAP lies in its adherence to the axiomatic framework of Shapley values, which allows for the assignment of unique and fair weights to features. This computation is independent of feature order, sample distribution, or model structure, ensuring mathematical consistency in both local and global interpretations. In contrast, LIME employs a local linear approximation approach, relying on data perturbation generation strategies, which results in a lack of global interpretability consistency. Although permutation importance is easy to implement, it fails to quantify the local contributions of features and is prone to evaluation biases when features exhibit multicollinearity. Therefore, this study selects SHAP as the interpretability tool for ML models.
Statistical analysis
Statistical analyses were conducted using R version 4.2.2 (R Core Team, Vienna, Austria) and Python version 1.2.1. In this study, the K-Nearest Neighbors imputation method was employed to correct and fill in both outliers and missing values. Normally distributed, continuous data are presented as means ± standard deviations, and related comparisons between two samples were performed using the t-test. Non-normally distributed continuous data are expressed as medians (Q1, Q3), and related intergroup comparisons were assessed using the Mann–Whitney U test. Categorical data are presented as frequencies (percentages), and related intergroup comparisons were performed using the chi-squared test.
The predictive performances of the ML models in the training and validation sets were evaluated using receiver operating characteristic (ROC) curves, calibration curves, and a decision curve analysis (DCA). The models were assessed based on the area under the curve (AUC), sensitivity, and specificity. SHAP analysis was employed to assess the contributions of different variables to the risk prediction. P-values < 0.05 were considered statistically significant.
Results
Basic characteristics
A total of 220 patients were included in this study and randomly assigned to the training and validation sets in a 7:3 ratio using a random number table (Supplement 1). Among all participants, 133 (60.45%) were diagnosed with pelvic adhesions, while 87 (39.55%) were not (Table 1). No significant differences in baseline variables were observed between the training and validation groups, indicating comparability between cohorts.
ML model construction
Predictive features with non-zero coefficients were identified using the Least Absolute Shrinkage and Selection Operator (LASSO) regression model. As the regularization parameter (λ) increased, the number of retained variables decreased progressively. The optimal penalty value was determined using 10-fold cross-validation, selecting the model corresponding to the minimum cross-validation error plus one standard error (λ + 1). Based on this criterion, four key predictive variables were retained: obstructed ovarian activity, surgical history, endometriosis, and gynecological inflammation (Figure 1).

(A) Least Absolute Shrinkage and Selection Operator (LASSO) coefficient distribution for eight risk factors. (B) Cross-validation curve.
The ML model was developed using the scikit-learn Python library (version 1.2.1) and the XGBoost package (version 1.7.3). During preprocessing, continuous variables were scaled using the MinMaxScaler method, and categorical variables were encoded using OneHotEncoder, both from scikit-learn.
With four-fold cross-validation, hyperparameter tuning for the XGBoost model was performed using Scikit-learn's GridSearchCV. The hyperparameters selected for optimization included learningrate, gamma, maxdepth, subsample, minchildweight, and nestimators. These steps were crucial for enhancing the model's performance and predictive capabilities.
ML-model predictive-performance evaluation
ROC curve analysis was used to evaluate the predictive performance of the XGBoost model for pelvic adhesion risk (Figure 2). The AUC, sensitivity, and specificity were 0.869, 0.946, and 0.597 for the training set and 0.846, 1.000, and 0.6000 for the validation set, respectively (Table 2). No significant difference was observed between the training and validation groups (D = 0.388, P = 0.698).

Performance receiver operating characteristic curves of the XGBoost model for predicting pelvic adhesions in the training and validation sets.
XGBoost machine learning model accuracy for predicting pelvic adhesion risk.
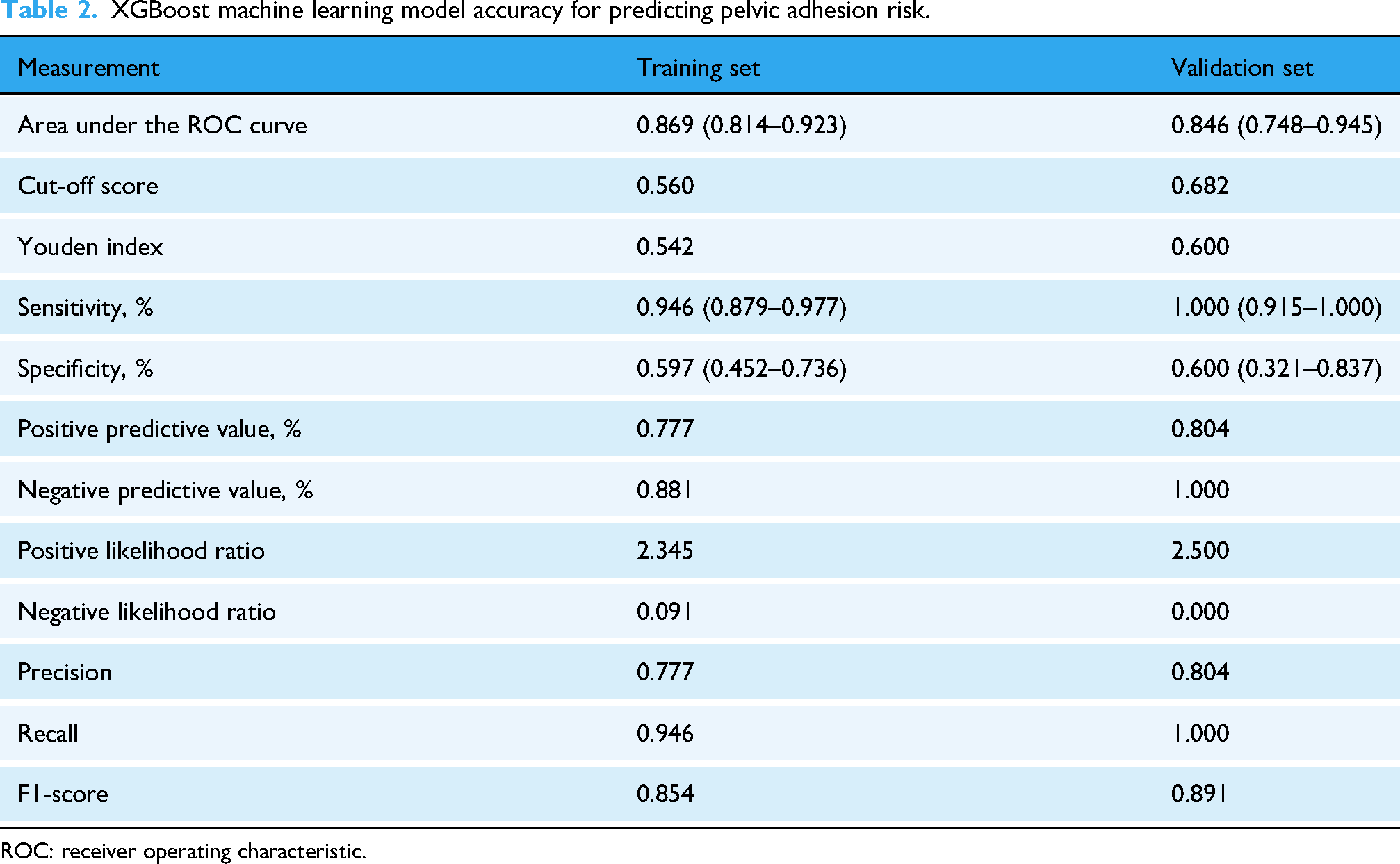
ROC: receiver operating characteristic.
Calibration curves were used to assess the alignment between the probabilities of pelvic adhesions predicted by the XGBoost model in the training and validation sets and the actual probabilities (Figure 3). The XGBoost model yielded predictions close to the ideal diagnostic outcomes for patients in the training and validation sets, demonstrating a high calibration level for pelvic adhesions. The calibration curve demonstrates that beyond a 10% clinical threshold the model-predicted probabilities closely approximate the observed event rates, indicating reliable decision-making at this cutoff and enabling avoidance of unnecessary laparoscopy while optimally balancing sensitivity and specificity.

Performance calibration curves of the XGBoost model for predicting pelvic adhesions in the (A) training and (B) validation sets.
DCA results showed a higher net benefit when the XGBoost model operated in the probability range of 10–96% for the training set and 10–88% for the validation set. These results suggested that the decision curve model had a broad range of probability domains for patients in the training and validation sets, indicating strong clinical utility and significant clinical value (Figure 4).
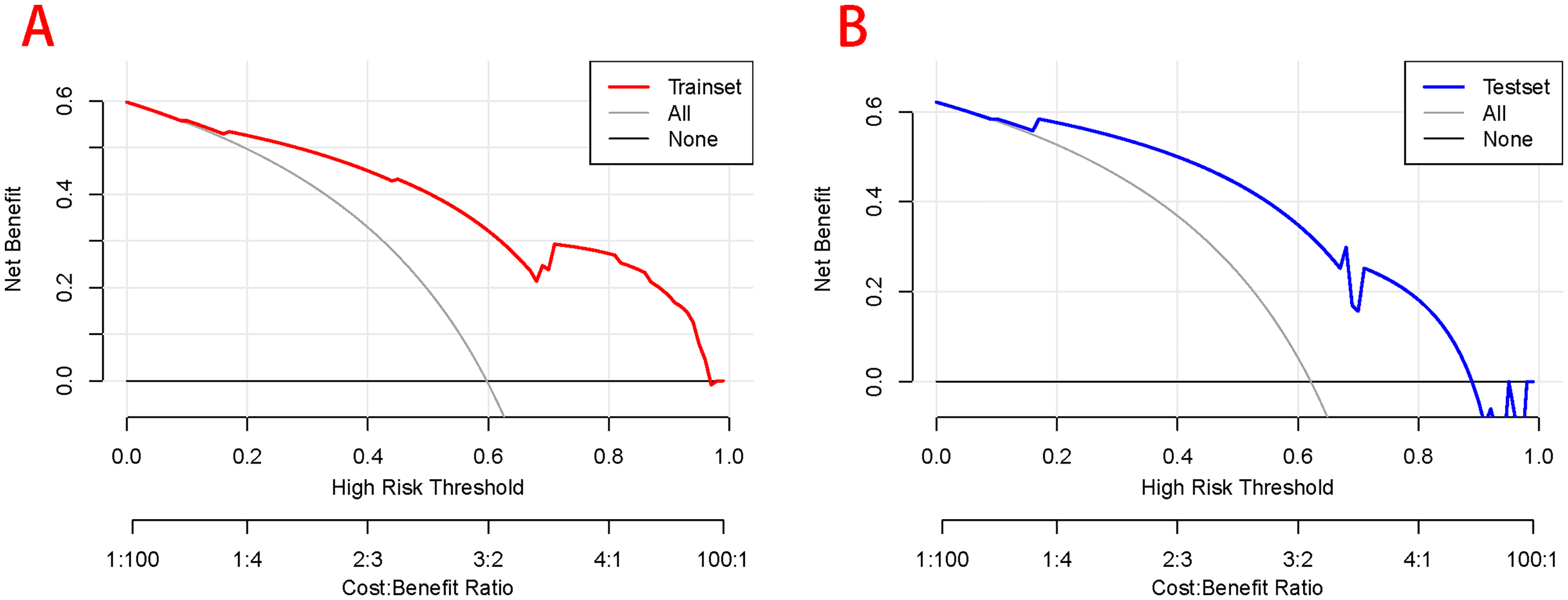
Decision curve analysis for the XGBoost model for predicting pelvic adhesions in the (A) training and (B) validation sets.
SHAP analysis
SHAP analysis is a versatile method for explaining individual predictions and providing global interpretability. Grounded in rich game theory principles, SHAP estimates feature importance by utilizing the concept of optimal integration based on Shapley values. Figure 5 shows the four key features evaluated using the mean absolute SHAP values, sorted using the absolute values of the mean SHAP values, reflecting the importance of each feature to the overall model predictions. Feature ranking (y-axis) indicates the importance of the predictive model. The SHAP values (x-axis) are a unified metric showing the impact of specific features in the model.

Importance of the predictor variables selected by XGBoost.
The top-ranked features, according to their mean absolute SHAP values, were as follows: endometriosis, surgical history, obstructed ovarian activity, and gynecological inflammation. SHAP analysis reveals that endometriosis exerts the strongest positive influence on model output: its presence raises the predicted score by an average of 0.54, flagging a sharp rise in the likelihood of pelvic adhesions. Previous pelvic surgery ranks second, contributing roughly 0.23. A history of gynecologic inflammation adds 0.17, whereas impaired ovarian activity provides a modest increment of 0.095.
SHAP force plots provide an intuitive visualization of how each feature contributes to individual predictions. A key advantage of SHAP for global interpretability lies in its ability to quantify feature importance and reveal how each variable influences the model's output. Additionally, SHAP values are consistently distributed across the feature space, which helps build confidence in the model's predictions.
We further applied SHAP summary plots to interpret the overall behavior of the baseline XGBoost model (Figure 6). The ability to visually depict the model decisions by mapping the cumulative SHAP values for each prediction is crucial. The x-axis represents the model's output, which, in this case, was the probability of pelvic adhesions. The graph is centered on the x-axis at the expected value. The y-axis lists the model's features in descending order of importance. Each observation prediction is represented by a colored line determined by its corresponding prediction value. The solid and dashed lines represent correctly and incorrectly classified observations, respectively. From the bottom to the top of the graph, each feature's SHAP value is added to the model's baseline value, illustrating how each feature contributes to the overall prediction and how the predictions vary in the decision-making process.

Decision plot for the XGBoost model using the SHAP method.
SHAP was also employed to provide local interpretability for individual predictions. The SHAP values quantify how each feature contributes to a specific patient's risk estimate, offering transparent and individualized explanations of the model's output.
To illustrate this, two representative cases were visualized to highlight the model's interpretability: (1) a patient without pelvic adhesions but with obstructed ovarian activity, endometriosis, no surgical history, and no gynecological inflammation; (2) a patient with pelvic adhesions, obstructed ovarian activity, endometriosis, or surgical history but no gynecological inflammation (Figure 7).
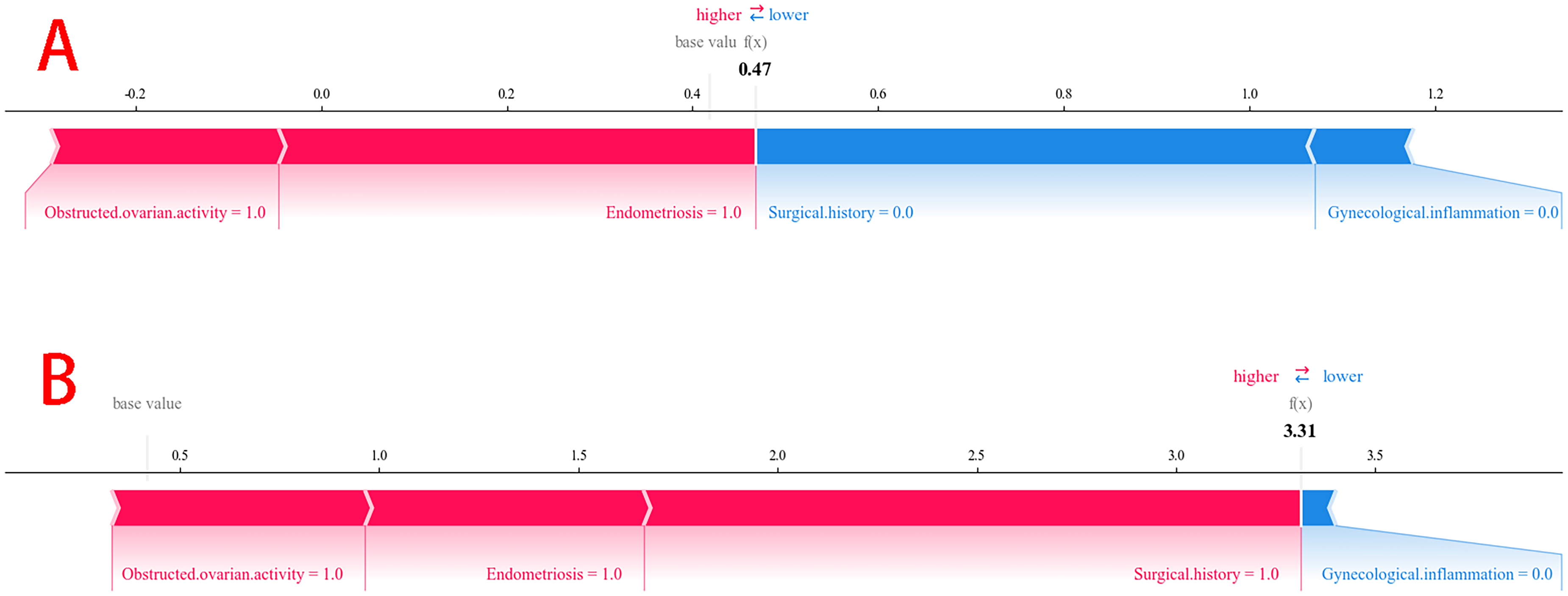
Two representative cases correctly predicted as negative for pelvic adhesion (patient A) and positive for pelvic adhesion (patient B) were individually visualized using the SHAP method.
Multi-threshold diagnostic performance evaluation
To reconcile sensitivity with specificity, we applied a clinical-weighting protocol that identified 0.750 as the optimal cut-off in the training set. Table 3 shows that, at this threshold, the XGBoost model achieved a specificity of 85.7% while sensitivity declined to 76.8%.
Accuracy of the different cut-off scores in estimating the risk of pelvic adhesions.

Discussion
In this study, we developed and evaluated an ultrasound-based interpretable XGBoost ML model to assess the risk of pelvic adhesions preoperatively. A LASSO regression analysis identified endometriosis, surgical history, obstructed ovarian activity, and gynecological inflammation as key predictive factors. These variables were used to construct the XGBoost model.
The model demonstrated strong predictive performance in both the training and validation sets, with AUCs of 0.869 (0.814–0.923) and 0.846 (0.748–0.945), respectively. Sensitivity values were 0.946 and 1.000 for the training and validation sets, respectively, indicating high accuracy in identifying negative cases. This finding may be attributed to the dynamic assessment of bilateral ovarian activity enabled by transvaginal ultrasound. Regrettably, the model displayed high sensitivity but low specificity in both training and validation sets, implying a considerable false positive rate that may lead to overtreatment or unnecessary operations. Preoperative screening demands a choice between missing disease and overcalling disease. Missed pelvic adhesions markedly elevate the risk of intraoperative injury to bowel, bladder, or uterus and increase reoperation rates. A false positive only adds a prophylactic adhesion barrier or an extra 5–10 min of adhesiolysis, with far lower morbidity and mortality. Therefore, for high-risk cohorts such as patients undergoing repeat pelvic surgery or endometriosis surgery, we propose a diagnostic threshold of 0.56 to maximize sensitivity. In resource sufficient settings that are sensitive to false positives, for example primary surgery or asymptomatic infertility, a cutoff of 0.75 rebalances sensitivity and specificity. At present, investigations that harness machine-learning models to predict peritoneal adhesions remain scarce. Wang et al. integrated deep-learning signatures extracted from CT images with clinical electronic health-record data to develop and validate a predictive model for adhesive small-bowel obstruction. Nevertheless, the model was constructed retrospectively from emergency-department cases and relies on static CT imaging, which cannot supply real-time dynamic information; these constraints may hinder its broader clinical dissemination.
To enhance model transparency, we incorporated the SHAP interpretability analysis. This method enables the identification of the relative contribution of each feature and illustrates how predictions evolve within the model's decision-making framework. Endometriosis exhibited the highest impact on pelvic adhesion risk among the four predictive variables. This can be explained by the cyclical activity of ectopic endometrial tissue and associated inflammatory responses, which cause repeated damage and tissue repair. During this process, fibroblasts are excessively activated, producing large amounts of collagen, ultimately leading to fibrosis and adhesion formation.20,21
Pelvic adhesions in patients with endometriosis, especially in those with moderate-to-severe disease, have a high prevalence. Pelvic adhesions are nearly universal in these cases and may contribute to infertility, a prevalent complication of endometriosis.22,23 Endometriosis significantly increases the risk of infertility, which may be closely associated with the presence of pelvic adhesions. 24 Abdominal surgery inevitably causes mechanical damage to the peritoneum, pelvic organs, and the surrounding tissues. Local inflammatory reactions are initiated following tissue injury, releasing several inflammatory mediators (e.g. interleukins and tumor necrosis factor) and fibrosis-related factors. Inflammatory reactions are significant triggers of adhesion formation. As surgical wounds heal, the inflammatory response may promote the deposition of fibrous proteins and adhesion formation. Notably, pelvic adhesions significantly increase in patients who have undergone multiple surgeries compared with those who underwent only one.25,26
In this study, obstructed ovarian activity detected via transvaginal ultrasonography was also a key predictive factor. This imaging modality allows real-time visualization of pelvic anatomy and ovarian mobility, aiding early detection of adhesion-related signs. Gynecological inflammation may lead to inflammatory reactions in pelvic organs, including the uterus, ovaries, and fallopian tubes, causing adhesions and scar formation between pelvic tissues and increasing the risk of pelvic adhesions. 27
Our results suggest that applying SHAP explanations to the XGBoost ML pelvic adhesion prediction model enhances its interpretability. This approach empowers clinicians to better understand and trust the model's outputs by providing detailed insight into the contribution of each risk factor. The use of global decision tree analyses and individual-level explanations may further support clinical decision-making in high-risk scenarios.
Despite the valuable insights obtained, this study has several limitations. First, the single-center design and modest sample size may introduce selection bias; external validity awaits confirmation in larger multicenter cohorts. Second, the high sensitivity but only moderate specificity raises concern for overfitting, mandating prospective real-world evaluation before clinical deployment. Third, the limited range of imaging and laboratory variables may omit established or emerging risk factors for pelvic adhesions. Future work should expand the sample size, incorporate additional clinical dimensions, and identify stronger predictors to refine model performance. Integration into ultrasound workstations or web-based risk calculators represents a logical next step to enhance clinical utility and accessibility.
Conclusions
We developed an ultrasound-based interpretable ML model to assess the risk of pelvic adhesions in female patients preoperatively. The model demonstrated excellent predictive performance and benefited from enhanced global and local interpretability through the SHAP method. These findings support its potential as a noninvasive, clinically relevant tool for early diagnosis of pelvic adhesions and as a valuable resource for personalized medical decision-making.
Supplemental Material
sj-doc-1-dhj-10.1177_20552076261416797 - Supplemental material for An ultrasound-based machine learning model for predicting pelvic adhesions: A SHAP-enhanced XGBoost approach
Supplemental material, sj-doc-1-dhj-10.1177_20552076261416797 for An ultrasound-based machine learning model for predicting pelvic adhesions: A SHAP-enhanced XGBoost approach by Yanyan Huang, Shanshan Su, Jiemin Chen, Xiaoqian Zhang, Kailing Tan and Qiuling Guo in DIGITAL HEALTH
Footnotes
Acknowledgements
This research is supported by the Science and Technology Bureau of Quanzhou.
Abbreviations
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Quanzhou Maternity and Children's Hospital (2024NY068).
Consent for publication
The study was reviewed and approved by the senior authors’ institutional review board and approved for publication.
Author contributions
S.S.: conceptualization; H.Z.: methodology; J.C.: validation; X.Z.: formal analysis; X.Z.: investigation; S.S.: resources; H.Z.: data curation; S.S.: writing—original draft; H.Z.: writing—review & editing; S.S.: visualization; Q.G.: supervision, project administration, and funding acquisition. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Science and Technology Bureau of Quanzhou [2024NY068].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets used and analyzed in the current study are available from the corresponding author, [Q.G.], upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
