Abstract
Objective
To systematically evaluate the effect of digital intervention on improving routine vaccination in the elderly and to conduct a comparative analysis of different intervention modalities using network meta-analysis (NMA).
Methods
PubMed, Web of Science, The Cochrane Library, Embase, Scopus, CINAHL, and WanFang Data were searched for randomized controlled trials (RCTs) using digital interventions to promote vaccination in older populations from inception to 15 June 2024. We performed a final update of the literature search in May 2025; no additional eligible studies were identified. Two researchers independently screened the literature, extracted data, and assessed the risk of bias in the included studies, and an NMA was performed using RevMan 5.4 and R Studio, PROSPERO Registration Number: CRD42024527483.
Results
Eleven RCTs were included. The traditional meta-analysis demonstrated a small but statistically significant increase in influenza vaccination rates (RR = 1.01, 95% CI [1.01, 1.01], P < 0.00001), accompanied by substantial heterogeneity (I2 = 86%). Pneumococcal vaccine uptake was significantly enhanced (RR = 1.11, 95% CI [1.03, 1.18], P < 0.01), with moderate heterogeneity (I2 = 46%). The single study on the herpes zoster vaccine reported a statistically significant effect, whereas COVID-19 vaccine reminder interventions showed no significant efficacy. In the NMA, video-based interventions ranked first based on the surface under the cumulative ranking curve, but all pairwise comparisons between different intervention modes crossed the null value.
Conclusion
Digital interventions show a significant, yet highly heterogeneous, positive impact on vaccination rates in older adults. While video-based education showed the highest ranking probability, the current evidence is insufficient to conclude that any specific digital modality is statistically superior to others. Due to the limited included studies, the findings need to be supplemented by more high-quality studies. Future research should focus on newer digital technologies to help the older population keep up with the “digital intelligence era.”
Introduction
As the global population continues to age, ensuring the health of older adults has become a pressing issue in public health. Vaccination serves as an effective means to prevent infectious diseases, reduce disease burden, and improve quality of life, playing a vital role in achieving the goal of “healthy aging.” However, in practice, vaccination coverage among older adults, particularly for routinely recommended but underutilized vaccines such as pneumococcal and influenza vaccines, has remained consistently low. 1 Multiple factors influence vaccine uptake, including cognitive biases, barriers to information access, insufficient health beliefs, and lack of systemic support. 2 Therefore, innovative and scalable intervention strategies are urgently needed to enhance vaccine acceptance and uptake among the elderly.
With the rapid advancement of digital health technologies, implementing health behaviour interventions through information and communication technology (ICT) has become a global trend. Recent research highlights that digital health tools are increasingly being integrated into immunization programs across diverse regions to enhance coverage and communication. 3 The World Health Organization defines digital health as “a field of knowledge and practice associated with the development and use of digital technologies to improve health, including smart devices, digitally engaged consumers, and health-related services using the Internet of Things.” 4 Correspondingly, digital interventions refer to treatment, management, and prevention efforts based on digital health formats and technologies. These interventions typically include those delivered via web-based or computer platforms (e.g. websites and online forums), mobile devices (e.g. mobile applications and SMS services), or any electronic medical device (e.g. wearable health monitoring equipment). 5
Although some studies have indicated that digital interventions can improve vaccination uptake in certain populations, including influenza vaccination among pregnant women, 6 promotion of adult HPV and pneumococcal vaccines, 7 interventions targeting adolescents and young adults 8 as well as those directed at parents and caretakers of children in the context of COVID-19 vaccination, 9 evidence among older adults remains inconsistent. 10 A systematic review suggested that ICT-based interventions may be effective in increasing vaccination uptake among older adults in high-income countries; however, it was limited to a qualitative synthesis and did not distinguish between types of interventions or conduct a meta-analysis to synthesize effect sizes. 11 Odone et al. offered important practice-based insights by conducting expert interviews on digital support for vaccination, but their scope was geographically limited to Europe. 3 Crucially, older adults face unique barriers to digital access—such as low technology acceptance, limited digital literacy, and poor accessibility. 12 This “digital divide” may contribute to heterogeneity in intervention outcomes, further limiting the generalizability and equity of such interventions. To address these critical evidence gaps, it is essential to employ network meta-analysis (NMA) to consolidate global evidence, provide broader geographic inclusion, and rank the comparative efficacy of different digital intervention types.
Given the current research questions and the practical barriers faced by older adults, this study aims to systematically compare the relative effectiveness of common digital interventions on vaccination uptake among older adults. We incorporated a range of common digital intervention formats—including text messaging (SMS), video-based education, email reminders, and electronic health record (EHR) portal access—and quantitatively evaluated their effectiveness in increasing vaccination uptake among older adults to provide an evidence-based foundation for designing tailored interventions suitable for older populations, thereby promoting the digital transformation and equity of public health strategies.
Methods
This systematic review and NMA was reported in accordance with the PRISMA guidelines 13 (see Appendix A) and was prospectively registered in the PROSPERO database (Registration Number: CRD42024527483).
Data sources and searches
This study was conducted from January 2025 to May 2025. A comprehensive literature search was conducted across the following electronic databases from inception to May 2025: PubMed, Web of Science, The Cochrane Library, EMBASE, Scopus, CINAHL, and WanFang Data. In addition to electronic databases, supplementary searches for gray literature were conducted, including targeted manual searches of major public health organization websites (e.g. WHO and CDC), clinical trial registries (ClinicalTrials.gov, WHO ICTRP). No additional eligible studies were identified. Reference lists of included studies and relevant reviews were manually screened to identify potentially eligible studies. The search strategy was developed based on four conceptual domains: (1) Population (older adults, aged ≥60 years); (2) Intervention (digital interventions, including but not limited to text messaging, mobile apps, patient portals, email reminders, video education, social networks, and virtual reality tools); (3) Outcomes (vaccination uptake and related behavioral indicators); (4) Study design (randomized controlled trials (RCTs) or controlled clinical trials). Controlled vocabulary (e.g. MeSH terms) and free-text terms were used in combination. Keywords within each domain were combined using the Boolean operator “OR,” and domains were connected using “AND.” The detailed search strategy is provided in Appendix B.1.
Inclusion criteria
We defined the target trials according to the population, interventions, comparators, outcomes, study design (PICOS) selection criteria.
Population
Older adults aged 60 years or above. The age threshold of ≥60 years was selected based on internationally recognized definitions of older adults. The World Health Organization identifies individuals aged 60 years or above as older adults in its World Report on Ageing and Health and subsequent global strategies on ageing and immunization.14,15 This cut-point is also widely used in vaccine-related clinical and behavioral research, where older adults are commonly defined as those aged ≥60 years. 16 Adopting this threshold ensures consistency with international guidelines and enhances comparability across studies.
Interventions
Digital health interventions aimed at improving vaccine uptake, such as text/email reminders, EHR-based decision aids, and multimedia education (e.g. videos).
Comparisons
Usual care (e.g. standard physician recommendation without structured support) or no intervention (i.e. blank control with no active component).
Outcomes
The primary outcome was the uptake of at least one commonly recommended vaccine in older adults, including influenza, pneumococcal, herpes zoster, or COVID-19 vaccines. Secondary outcomes included vaccination intention, knowledge, and attitudes.
Study design
Only RCTs or quasi-experimental trials were included. Reviews, commentaries, editorials, letters, and guidelines were excluded.
Data selection and extraction
The literature was independently screened, data were extracted, and the results were cross-verified by two researchers. Discrepancies were resolved through discussion or, if necessary, by consulting a third reviewer. The screening process consisted of an initial review of titles, followed by assessments of abstracts and full texts to determine study eligibility after the exclusion of irrelevant records. Studies were excluded if they lacked accessible full texts, contained insufficient or unextractable data, or focused on atypical vaccination practices in specific subpopulations (e.g. administration of Haemophilus influenzae type b or tetanus vaccines to post-splenectomy patients).
The extracted data included: (1) basic study characteristics (e.g. title, first author, publication year, and study location); (2) baseline characteristics of the study population (e.g. sample size and mean age); (3) intervention details (e.g. vaccine type and intervention method); (4) key domains of risk of bias assessment; and (5) outcome indicators and corresponding measurement data.
Quality appraisal
Two researchers independently evaluated the risk of bias in the included studies and cross-checked the results. The risk of bias assessment was conducted using the Risk of Bias 2 (RoB 2) tool, as recommended in the Cochrane Handbook for Systematic Reviews of Interventions. 17 This assessment evaluates six domains of bias: selection bias (including random sequence generation and allocation concealment), performance bias (referring to the blinding of participants and personnel), detection bias (pertaining to the blind assessment of outcomes), attrition bias (related to the completeness of outcome data), reporting bias (concerning selective reporting of outcomes), and other bias.
Statistical analyses
NMA was conducted using RevMan 5.4 and R Studio. (1) A comparative matrix was constructed to extract the number of vaccination events (event) and sample size (n) from each study for both the experimental and control groups. The intervention measures (treat1) and control measures (treat2), along with the study names (study), were clearly defined. All direct comparison data were consolidated into a network structure to perform a multi-intervention comparative analysis. The odds ratio (OR) and/or risk ratio (RR) were used as effect size measures, and a random-effects model was employed for the analysis. (2) The network consistency was assessed using the node-splitting method to evaluate the consistency between direct and indirect evidence. The size of the nodes represents the number of participants, while the lines between the nodes indicate comparative studies between different interventions. The thickness of the lines reflects the number of studies comparing the two interventions; thicker lines indicate a greater number of studies and stronger evidence supporting the comparison. (3) Based on the analysis results, the surface under the cumulative ranking curve (SUCRA) values for each intervention were calculated to estimate their probabilities of being the best intervention. A rankogram was created to display the probability distribution of each intervention across various rankings. Statistical analyses were primarily conducted using the netmeta package, with visualizations produced using ggplot2 and functions from the netmeta package. (4) Sensitivity analysis was performed using a leave-one-out method with the metafor package in R to examine the influence of individual studies on the overall estimates. A significance level of P < 0.05 was established. (5) The certainty of evidence for the network estimates was judged using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework.
Results
Literature screening process and results
The initial review obtained 2234 studies from the electronic database and 257 trial registries. After deleting duplicate studies, 1664 records were screened, and the eligibility of 95 full texts was evaluated, of which 9 reports could not be retrieved. The exclusion reasons for 75 full-text articles included 40 due to wrong population (e.g. non-older adult groups such as providers or special populations), 26 due to inappropriate study design (e.g. non-digital interventions and non-randomized designs), 8 due to insufficient or unextractable data, and 1 due to wrong vaccine type (specifically, tetanus vaccine). Supplementary searches of gray literature sources (e.g. Google Scholar, which yielded high duplication, and OpenGrey which yielded no relevant studies) identified no additional eligible reports. Finally, 11 RCTs met the inclusion criteria.18–28 The funnel plot indicated that no significant publication bias was detected (Figure B.1 in the supplemental materials). The literature screening process and results are shown in Figure 1.

Flow chart of literature screening.
Basic characteristics and quality evaluation of the included studies
This study retrieved as many publicly published studies as possible and finally included 11 studies. Five studies were conducted in the United States,20–22 three in Europe (France/Monaco, 23 Denmark, 25 and the United Kingdom 28 ), and in addition, one each was from Australia, 18 China 19 and Thailand. 24 Among the 11 studies, a total of 9 promoted the influenza vaccine,18–20,23–26 and the studies also focused on the pneumococcal vaccine,23,26,28 zoster vaccine 21 and the COVID-19 vaccine. 22 All studies reported the results of vaccination rates, which were the main research indicators.
The intervention methods used in the included studies were diverse. Two studies19,24 utilized video technology for intervention, three studies18,23,27sent SMS reminders, three studies via emails,20,21,25 and five studies20–22,27,28 sent information through the health system, namely the patient portal. These intervention measures usually combine health education, reminders and auxiliary decision-making elements that help with personalized analysis. Among the seven studies that adopted the reminder strategy, one 21 included a health education component (i.e. information investigating the importance and safety of vaccines), four18,20,22,26 only provided vaccine-related information to remind patients of vaccination, and one 23 reminded patients to communicate vaccine information with doctors. A national study in Denmark 25 adopted a variety of reminder strategies, including simple vaccination reminders, health education reminders emphasizing the health benefits of vaccines, and repeatedly sending reminders to strengthen the willingness to be vaccinated. The basic characteristics of the included studies are shown in Table B.1 in the supplemental materials.
The overall quality of the literature included in this study was high, but the research design of some studies was not rigorous enough, among which only four studies19–22 explained the method of generating random sequences. Due to limitations in the implementation of digital interventions, investigator blinding was not possible in most studies,18,19,21,23,24 but as subsequent data collection and analysis were based on electronic medical records, the risk of information bias was negligible. Data on loss to follow-up were interpreted in all the studies. The evaluation results of bias risk are shown in Figure B.2 in the supplemental materials.
Results of network analysis
Network-related diagram
The analysis (Figure 2) formed six pairs of comparisons across four digital intervention methods: SMS reminders, email reminders, video education and electronic portal reminders. The sample sizes of each scheme group were 2612, 345877, 1577, and 35989, respectively.

Network of available comparisons. Note: “N/A” indicates that comparisons of an intervention with itself are not applicable.
Intervention ranking chart
In this study, based on 11 RCTs, the intervention methods included SMS reminders, health education videos, push notifications from electronic medical record portals, email, etc. The analysis results showed a potential differential effect across intervention measures.
According to the ranking results of SUCRA values (Figure 3), the SUCRA value of the health education video intervention was the highest, suggesting it possessed the highest probability of being the most effective modality. The Rankogram graph further corroborated that this intervention method had the highest ranking probability among all comparisons. SMS reminders and email push notifications came second, while the effect of medical record portal reminders was relatively weak.

SUCRA plot and ranking heatmap. Note: “N/A” indicates that comparisons of an intervention with itself are not applicable.
Pooled estimates of vaccine uptake across digital interventions
Figure 4 presents a heatmap of pairwise comparisons between different digital interventions in improving vaccine uptake among older adults. The effect sizes were expressed as log OR with corresponding 95% confidence intervals. Negative values indicated that the column treatment was favored over the row comparator. Among all interventions, video-based education demonstrated the best numerical trend, showing moderately better performance compared to SMS reminders (OR = −0.05, 95% CI [−1.11, 0.92]), email (−0.23 [−1.20, 0.74]), medical portals (−0.31 [−1.13, 0.44]), Not applicable [NA] (−0.52 [−1.29, 0.15]), and usual care (−0.40 [−1.49, 0.62]). Crucially, however, the confidence intervals across all comparisons crossed the null value.
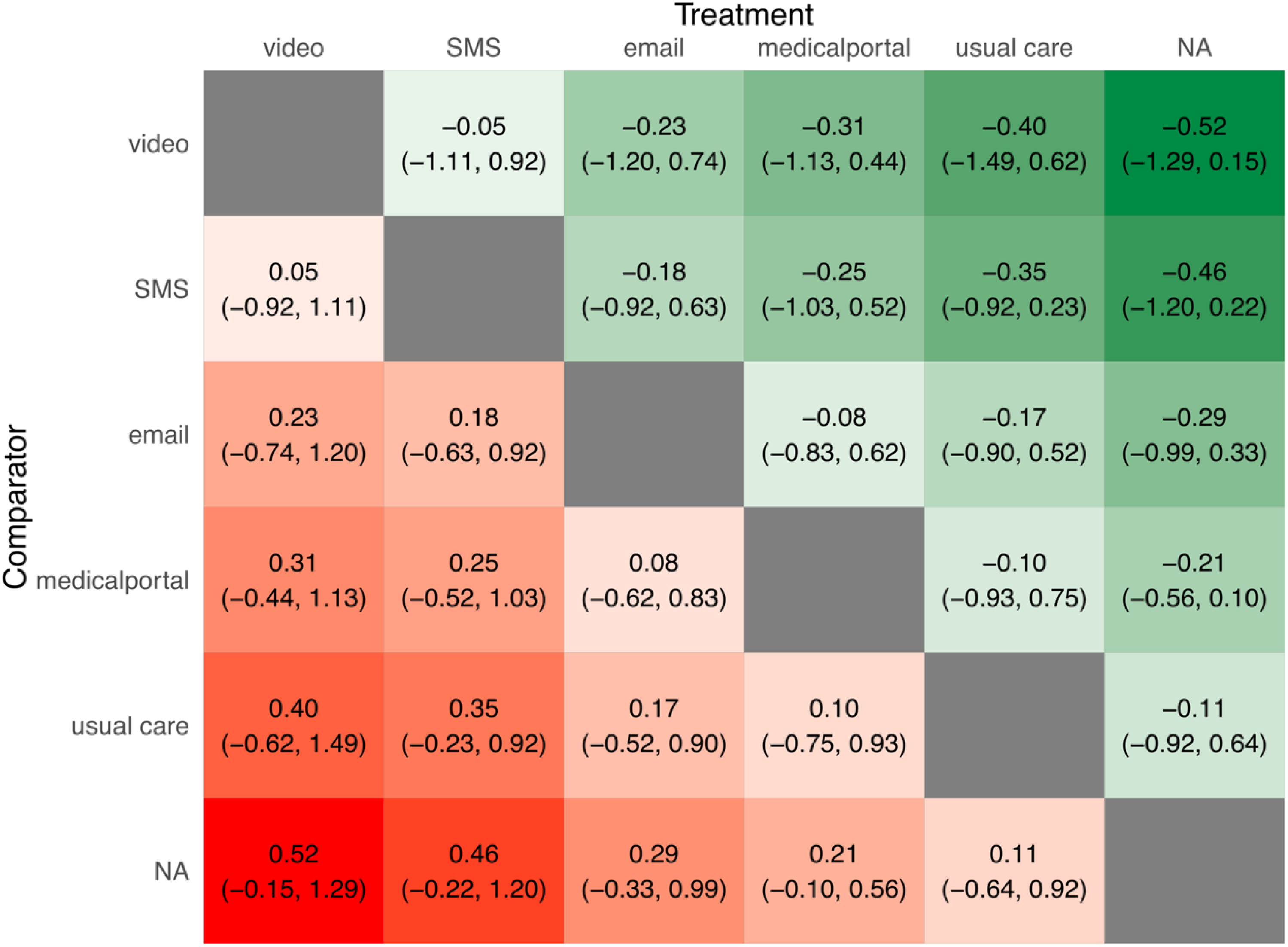
Pooled hazard ratios comparing different interventions. Note: “N/A” indicates that comparisons of an intervention with itself are not applicable.
Meta subgroup analysis of different types of vaccines
Subgroup analyses were performed for the influenza vaccine, pneumococcal vaccine, zoster vaccine, and COVID-19 vaccine (Figure 5). For the influenza vaccination rate, the intervention group had a slight increase compared with the control group (RR = 1.01, 95% CI [1.01, 1.01], P < 0.00001), but the heterogeneity was substantial (I2 = 86%). The three studies of the pneumococcal vaccine showed that the intervention was highly effective, resulting in a statistically significant increase in the vaccination rate (RR = 1.11, 95% CI [1.03, 1.18], P < 0.01), with moderate heterogeneity (I2 = 46%). Only one study investigated the herpes zoster vaccine, and the intervention effect was statistically significant. However, the COVID-19 vaccine reminder intervention did not show statistically significant efficacy.

Meta-analysis of vaccine-type subgroups.
Sensitivity analysis
The sensitivity analysis (Figure B.3-a in the supplemental materials) confirmed stability of the small pooled effect (RR = 1.01). While the exclusion of certain studies, such as Jiang et al., 19 significantly affected the RR, the general reliability of the analysis suggested that the influenza vaccine's effectiveness was consistent across different contexts. The sensitivity analysis (Figure B.3-b in the supplemental materials) for the pneumococcal vaccine also indicated a stable overall effect (RR = 1.11). The analysis revealed that heterogeneity was primarily driven by the inclusion of the study by Loo et al., 26 as removing it reduced I2 to 0%. Additionally, excluding the study by Harari et al. 28 resulted in a higher P-value that exceeded the statistical significance threshold, highlighting its critical role in the overall effect.
Certainty of evidence assessment results
The certainty of evidence for the network estimates was judged using the GRADE framework. The evidence for the influenza vaccine subgroup was rated as Low, primarily due to serious inconsistency (I2 = 86%) reflecting heterogeneous effects across the included interventions. Conversely, the efficacy of digital interventions for the pneumococcal vaccine was supported by Moderate certainty evidence. For the NMA comparisons, the certainty of evidence for the relative effectiveness between all digital intervention modalities was predominantly judged as Low. The primary and most critical factor contributing to this substantial downgrade was imprecision, as the confidence intervals for all pairwise estimates consistently crossed the null value, severely limiting the statistical confidence in concluding the superiority of any specific digital strategy.
Discussion
This systematic review and meta-analysis found that digital interventions exerted a statistically significant positive effect on vaccination uptake among older adults. However, the observed effect was minimal overall with notable variation across vaccine types (e.g. influenza: RR = 1.01; pneumococcal vaccine: RR = 1.11) and accompanied by a very high level of heterogeneity.
A substantial level of heterogeneity stemmed from diverse potential sources. Firstly, variations in vaccine types constitute a significant source of heterogeneity. Given that knowledge, confidence, attitude and affordability subsequently influence vaccination willingness and rates, 29 different vaccines may lead to varying vaccination responses, affecting individuals’ vaccination preferences. 30 Secondly, this heterogeneity may also be attributed to multiple social factors, 31 including regional variations, policy diversity in vaccination strategies, and cultural disparities. These factors collectively influence vaccination rates, consequently resulting in digital intervention effectiveness across different geographical contexts. For example, sensitivity analysis highlighted the absence of a free influenza vaccination policy in the research by Jiang et al. 19 as a critical factor. Lastly, variations in research methodologies, including demographic characteristics and specific intervention implementation strategies, contribute to substantive disparities. For instance, some studies, such as Johansen et al., 25 incorporated a typical email reminder within their “usual care” control condition, whereas others 23 utilized conventional interventions involving direct knowledge transfer methods, including face-to-face consultations and informational materials. This wide divergence in non-digital components impacts the baseline comparison. The minimal effect observed in the influenza subgroup may be attributed to the technological barriers. For older adults, the perceived ease of use and perceived usefulness of digital tools can limit the adherence and effectiveness of interventions, as they face significant technological hurdles. 32 Therefore, future practice should not treat digital interventions as a one-size-fits-all tool, but rather design and evaluate them according to the specific context of the target vaccine.
NMA revealed that the video intervention exhibits the most favorable numerical trend among the evaluated digital strategies. Educational videos can improve attitudes, thereby bolstering participants’ vaccination confidence and significantly enhancing influenza vaccine uptake, with particularly pronounced effects observed among individuals with lower educational levels. 24 By integrating images and text, this format is easily comprehensible. Thus, videos can facilitate the sustainable implementation of influenza vaccination programs among low- and middle-income elderly populations through the enhancement of knowledge and attitudes.33,34 Jiang et al. 19 reported consistent findings that participants exhibited greater emotional resonance and enhanced comprehension and retention of the content, alongside an increase in vaccination motivation. This emphasizes that optimizing information transmission, enhancing knowledge and attitudes, and increasing participation flexibility strategically position video interventions as an effective approach to improving vaccination rates and stimulating motivation among the elderly. From the perspective of older adults, this phenomenon may be linked to their unique physiological and psychological characteristics. Many older adults experience issues such as age-related cognitive decline 35 and visual impairments. 36 This numerical trend can be better understood through the lens of the technology acceptance model. 32 The combination of visual and auditory cues in video format makes information more accessible to older adults, thereby improving their attention, retention, and attitudes towards vaccination.
However, in existing research, older adults are often passive recipients of digital interventions. While conventional digital communication technologies are easily accessible and offer immediacy, their potential is limited by this passive communication. Although passive reception reduces cognitive burden on older adults, fully leveraging digital interventions requires focusing on and enhancing their active role in vaccination decision-making. 37 Digital technologies should not only serve as information delivery but, more importantly, as tools to raise proactive health management and support the active decision-making process. 38 Emerging interventions, such as chatbots, 39 have demonstrated that stage-tailored online interventions positively impact influenza vaccination among older adults. These interactive approaches are particularly significant for promoting greater participation and enhancing vaccination willingness. 38 Furthermore, “Pay-it-forward,” 40 a recent innovative intervention model, promotes pro-social behavior and enhances older adults’ initiative regarding their health, effectively improving vaccine confidence and significantly increasing vaccination rates among the target population. Although this approach does not utilize digital technology, its focus on social interaction and support provides insights into motivating older adults to actively participate in health decisions. As suggested by emerging evidence, future research lies not only in standalone interventions but also in the integration of digital health solutions into broader immunization ecosystems. 41 This integration, particularly as artificial intelligence technologies advance, warrants further exploration to develop more intelligent solutions to effectively address older adults’ cognitive and technical challenges in information access and processing through personalized guidance.
Several limitations of this study should be acknowledged. First, the included studies exhibited considerable heterogeneity in vaccine types, intervention modalities, study settings, methodological designs, and participant characteristics (eg, health literacy), which may compromise the robustness of the effect estimates and limit the generalizability of the findings. Second, most included studies lacked systematic evaluation of process-related variables such as vaccine-related knowledge, attitudes, and user experience, thereby limiting a deeper understanding of the intervention mechanisms. Moreover, a general lack of comprehensive evaluation regarding implementation costs, adherence, and long-term sustainability makes it difficult to assess the real-world feasibility and cost-effectiveness of the interventions. These limitations highlight existing gaps in outcome selection, reporting standards, and clarity of intervention pathways in the current research landscape.
Conclusions
This study, through a systematic review and NMA, demonstrated the potential of digital interventions in improving vaccination uptake among older adults, with video-based approaches showing the most favorable numerical trend. However, challenges such as high heterogeneity, limited data, and the digital divide underscore the need to broaden the scope of implementation evaluation, conduct large-scale RCTs, and explore intelligent, adaptive intervention models. These findings provide scientific evidence for the development of targeted vaccination strategies in older populations and contribute to the advancement of healthy aging.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261416313 - Supplemental material for Digital interventions to promote vaccine uptake among older adults: A systematic review and network meta-analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076261416313 for Digital interventions to promote vaccine uptake among older adults: A systematic review and network meta-analysis by Shuhui Shang, Xiaolong Wang, Enming Zhang, Yumeng Zhang, Yuhan Li and Qiong Fang in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076261416313 - Supplemental material for Digital interventions to promote vaccine uptake among older adults: A systematic review and network meta-analysis
Supplemental material, sj-docx-2-dhj-10.1177_20552076261416313 for Digital interventions to promote vaccine uptake among older adults: A systematic review and network meta-analysis by Shuhui Shang, Xiaolong Wang, Enming Zhang, Yumeng Zhang, Yuhan Li and Qiong Fang in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors are grateful to Yanyan Liu for her insightful methodological advice and support during this research.
Ethical considerations
There are no human participants in this article, and informed consent is not required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanghai Jiao Tong University Nursing Discipline Talent Team Development Project (Grant Number AJ1-0204-00-0115).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Most of the data generated or analyzed during this study are presented in the Results section of the article. Additional data related to the original literature screening and extraction process are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
