Abstract
Objective
The rapid global aging trend has led to a substantial increase in the prevalence of frailty among older adults. We developed a mobile app-based multidomain intervention (MI) program as part of a South Korean study to prevent the progression of frailty and aging-related diseases using a digital MI (SUPERAGING). We aim to evaluate the feasibility of the SUPERAGING app-based intervention in prefrail or frail older adults.
Methods
We will recruit 40 community-dwelling older adults aged 60 to 90 years classified as frail or prefrail according to the Modified Fried frailty criteria. Participants will be randomly assigned to intervention and control groups at a 1:1 ratio. The intervention group will receive a personalized program through the SUPERAGING digital platform, comprising four components: disease management, cognitive training, physical exercise and nutritional intervention for 16 weeks. The control group will receive standard lifestyle education only. The primary outcomes are adherence, retention, and recruitment rates. The main secondary outcomes are frailty, disability, cognitive function, physical performance, nutritional assessment, mood, quality of life, vascular risk factors, and occurrence of aging-related diseases. There will be an exploratory evaluation of biological aging markers.
Results
The intervention program will be considered feasible if the following success criteria are met: (a) a retention rate of 70% or higher, (b) an adherence rate of 70% or higher, and (c) a recruitment rate of 50% or higher.
Conclusions
The results will provide information on the applicability of a MI using a mobile app targeting older adults with prefrailty or frailty.
Trial registration
ClinicalTrials.gov identifier: NCT06891573. Registered on February 25, 2025.
Introduction
The global population is undergoing a rapid demographic transition characterized by aging with the number of individuals aged 60 years and older expected to rise from approximately 900 million in 2015 to 1.4 billion by 2030 and further to 2.1 billion by 2050. 1 According to National Statistics in Korea, the prevalence rate of older adults aged 65 and over in Korea was 14.3% in 2018 and is expected to double by 2028. 2 Increased life expectancy is primarily attributed to improved healthcare services, advances in medical technology, enhanced economic conditions, better nutritional status and lifestyle changes, as well as higher educational attainment all of which contribute to reduced mortality rates. 3 However, increased life expectancy does not necessarily mean a healthy life and aging is closely associated with an increased risk of frailty.4,5
Frailty is defined as a vulnerable condition characterized by a decline in overall physiological reserve and functional capacity due to aging, resulting in an impaired ability to maintain homeostasis and respond adequately to external stressors. 6 In the absence of a globally accepted gold standard, the most widely used frailty criteria were proposed by Fried et al., who operationalized it as the presence of at least three out of five phenotypic components indicating compromised energetics: weakness, slowness, exhaustion, low physical activity, and unintentional weight loss. 4 Previous studies have demonstrated that non-pharmacological interventions, such as physical activity and nutritional support, can have positive effects on frailty status, functional outcomes, and cognitive function.7–9 As frailty results from reaching a threshold of decline in different physiological systems, the approach to address frailty should act on multiple domains. 10
In recognition of this essential need for comprehensive, multi-faceted strategies to effectively combat frailty, multidomain intervention, which simultaneously encompasses various health factors such as physical activity, disease and metabolic disorder management and nutritional improvement, and so on, have recently gained attention as an effective approach for maintaining function and promoting health in older adults. 11
The Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) study conducted in Finland represented a landmark achievement in dementia prevention research, demonstrating that multidomain lifestyle interventions can effectively prevent cognitive decline in older adults at risk. 12 A recent post hoc analysis of the FINGER trial further demonstrated the effectiveness of multidomain lifestyle interventions in frail populations. In this study, a 2 year-intervention comprising dietary counseling, physical exercise, cognitive training, and vascular risk management significantly prevented the progression to phenotypic prefrailty and frailty. It even reversed existing frailty in community-dwelling older adults aged 60 to 77 years. The beneficial effects were sustained up to 7 years after the intervention, with physical activity identified as the most influential component. These findings provide robust evidence supporting the long-term impact of structured, multidomain strategies for frailty prevention and management among at-risk older adults. 13
The continuous advancement of digital technology has emerged as a promising solution to promote healthy aging in older adults. 14 Previous research has demonstrated that digital technology can positively impact multiple domains critical for aging well, including the maintenance of functional independence, prevention of accidents such as falls, enhancement of social connections, and management of mental health issues. 15 Digital platforms for multidomain interventions targeting dementia prevention in high-risk older adults have been successfully validated.
However, most existing digital multidomain interventions have primarily followed a fixed, uniform structure and lack the capacity for dynamic individualization during the intervention period. To address this gap, the present study introduces a digitally delivered, Information and Communication Technology (ICT)-based multidomain intervention that includes regular reassessments at 4-week intervals. Based on these reassessments, the intervention domains including disease management, cognitive training, physical activity and nutrition are individually reassigned and tailored to each participant's changing status. This adaptive, personalized approach represents a key innovation and a major strength of our study, distinguishing it from previous research.
In light of these findings, South Korean study to prevent the progression of frailty and aging-related diseases using a digital multidomain intervention (SUPERAGNIG) was designed to develop an ICT-based, digital, personalized multidomain intervention program with minimal visits, integrating disease management, cognitive training, physical activity, and nutritional support for prefrail or frail older adults. The feasibility and effectiveness of the intervention will be evaluated in real-world settings. Through this approach, we seek to propose a precision healthcare model for high-risk older adults and provide foundational data to inform the development of community-based intervention strategies to extend healthy life expectancy.
Methods
Study design
This feasibility pilot study will be conducted as a small-scale randomized controlled trial (RCT) to assess the feasibility of a mobile app-based multidomain intervention and obtain preliminary data on intervention effects. This study is a multicenter, outcome assessor-blinded, feasibility RCT with a two parallel-arm design. The multidomain intervention group is the experimental arm, while the control group is the comparator. The multidomain intervention period is 16 weeks. Participants will be enrolled in three hospitals across South Korea. Any important protocol amendments will be reported to the Institutional Review Boards (IRBs), updated in the registry, and communicated to all investigators and stakeholders. The protocol corresponds to version 1.2, finalized on December 17, 2024. The study is registered at www.ClinicalTrials.gov (registration number: NCT06891573). The study design adheres to the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) guidelines. 16
Participants
This study will involve 40 community-dwelling older adults who are classified as frail or prefrail according to the Modified Fried Frailty Phenotype criteria. 4 Individuals scoring 1 point or higher are classified as prefrail or frail. As the study includes individuals who are already in a prefrail or frail state, the intervention corresponds to a secondary prevention and care approach aimed at delaying the progression of frailty and maintaining functional capacity, rather than primary prevention. Participants will be recruited from three hospitals in South Korea using a combination of posters displayed in outpatient clinics and referrals from clinicians. Eligible participants will be recruited after applying the following inclusion criteria: (a) age of 60 to 90 years; (b) living in the community; (c) score 1 or higher on the Modified Fried Frailty Phenotype 4 ; (d) a standard version of Korean Mini-Mental State Examination, 2nd Edition (K-MMSE-2) 17 score equal to or above 1.5 standard deviations below the age- and education-adjusted norm; (e) ability to read and write; (f) ability to use a mobile app independently or with assistance; (g) having a reliable informant who can provide investigators with the requested information; and (h) provide written informed consent. The exclusion criteria are as follows: (a) major psychiatric illness; (b) dementia; (c) other neurodegenerative diseases such as Parkinson's disease; (d) malignancy within 5 years; (e) cardiac stent or revascularization within 1 year; (f) serious or unstable symptomatic cardiovascular disease; (g) other serious or unstable medical conditions; (h) significant laboratory abnormalities that may affect cognition; (i) inability to participate in exercise safely as judged by the study physician (e.g. unsteady gait, severe back or leg joint pain); (j) severe visual, hearing, or communication impairment; (k) any conditions preventing cooperation as judged by the study physician; and (l) concurrent participation in other intervention trials. Participants are permitted to continue their usual medications and healthcare visits; however, initiation of new structured cognitive or exercise programs outside of the study is discouraged during the trial. Additionally, mild cognitive impairment (MCI) will be diagnosed if a participant has complaints of cognitive decline reported either by the participant themselves or an informant, retains the ability to independently perform activities of daily living (ADL), and has a Z-score of <–1.0 on a delayed word or story recall test administered within the past year. Participants with MCI are classified as cognitive fraily. 18
Randomization
This feasibility RCT is designed to compare two groups, with participants randomly assigned to either the intervention or control group in a 1:1 ratio. Randomization will be stratified by the participating centers, sex, and frailty status as prefrail and frail groups, using a permuted block design generated through a Statistical Analysis System (SAS) macro program (SAS Institute, Cary, NC, USA) to ensure balanced allocation between the intervention and control groups. The allocation sequence will be generated using a SAS macro program and will be managed by an independent person who will not be directly involved in the research process. Although complete blinding is not possible due to the nature of the intervention, the secondary cognitive function efficacy assessment will be performed by an independent evaluator who is unaware of the participants’ group allocation.
Multidomain intervention
Participants in the intervention group will be provided with a mobile phone by the research team to be used throughout the 16-week intervention period. The multidomain intervention focuses on four components: disease management, cognitive training, exercise, and nutrition, all delivered via the SUPERAGING digital platform. The platform includes various content such as questionnaires, video materials, and neuroplasticity reflective games. Participants will have to access the platform on each intervention day, excluding holidays, over 16 weeks and perform personalized programs tailored to their assessment results for each component. Assessments for each component are conducted at 4-week intervals, when participants visit the clinic for re-evaluations. The intervention is designed to allow continuous adjustment of content based on individual changes. All staff delivering the intervention will be trained in the study protocol and must demonstrate competency in using the digital platform. All program results provided by the platform can be monitored in real-time through the administrator page.
Schedules for multidomain intervention group
Prior to the intervention, assessments of anthropometric measurements, medical history, cognitive function, physical function, and nutritional status will be conducted to identify frailty-related risk factors. Metabolic and nutritional status will be evaluated through blood tests and body composition analysis, while key metabolic and vascular risk factors, including hypertension, diabetes, dyslipidemia, obesity, abdominal obesity, kidney function, smoking, and alcohol consumption will be also assessed. Following randomization, participants will visit the clinic weekly for the first 2 weeks to participate in the intervention using the investigational medical device under the supervision of research nurses, and independently perform the intervention at home four times per week. From weeks 3 to 8, participants will visit the clinic once every 2 weeks; during visit weeks, they will perform one session under research nurse supervision and four sessions independently at home, and during non-visit weeks, they will perform five sessions independently. From weeks 9 to 16, clinic visits will occur once every 4 weeks, with one supervised session and four independent home sessions during visit weeks, and five independent sessions during non-visit weeks (Figure 1).

SUPERAGING protocol. Participants are classified into either the multidomain intervention group or the control group. The participants in the intervention group receives disease education programs three times per week, cognitive training and physical exercise five times per week, nutrition education once per week, and nutrition monitoring twice per week.
Disease management
Disease management will aim to improve disease awareness and self-management abilities among older adults through 27 customized educational videos (approximately 5 minutes each) filmed by specialists and quizzes targeting 14 chronic diseases that are risk factors for frailty. 18 Frailty management video will be included for all participants regardless of concurrent diseases. In addition, chronic diseases that participants already have or that are newly diagnosed after program initiation will be included in the intensive intervention targets, with disease status evaluated regularly every 4 weeks to adjust intervention contents. For participants with diseases management, educational contents lasting 5 minutes per session will be provided three times per week for 16 weeks, with algorithm-based personalized contents automatically provided according to individual disease types and quiz response results. Participants’ intervention engagement, including completion of quizzes after educational content viewing, will be evaluated as an adherence indicator, with individual progress monitored based on total content viewing and quiz completion rates. The educational content will be tailored to 14 chronic diseases with the structure and frequency of disease-specific sessions summarized in Table 1.
Contents of disease management intervention.

Cognitive training
The digital cognitive training program developed in this study specifically targets prospective memory, executive function, and selective attention, which are known to decline with normal aging due to frontal lobe dysfunction.19,20 Based on the previously validated program in South Korea, 21 we developed new digital modules specifically targeting prospective memory, executive function, and selective attention to address age-related decline in these domains. Cognitive training will be conducted five times a week for a total of 16 weeks. Time-based prospective memory training will comprise three stages: information registration, delayed recall after 3 hours, and recognition. A total of 80 tasks will be administered across three difficulty levels: simple word memory, daily conversation memory, and meaningful sentence memory. The difficulty of prospective memory training is adjusted differently depending on physical frailty and cognitive frailty. Serious game-based attention and executive function tasks will be provided daily, with difficulty levels adjusted according to participants’ performance among a total of 20 tasks consisting of difficulty A (advanced) and B (basic). The SUPERAGING-cognitive (SA-cog) test for cognitive function assessment will be conducted through the app at 4-week intervals and will be reflected in task composition and difficulty adjustment. The SA-cog test was developed as part of the SUPERAGING digital platform to monitor participants’ cognitive performance and adapt the difficulty of cognitive training tasks over time. It assesses memory, attention, and executive function, with a total score ranging from 0 to 100 (higher scores indicating better cognitive function). The memory component involves story recall tasks based on pictorial narratives, assessing immediate recall, delayed recall, and recognition. Two alternate forms (Form A and Form B) were developed to minimize practice effects during repeated assessments. Attention is evaluated through tasks measuring both selective and sustained attention, while executive function is measured by a digital Stroop-type task that assesses inhibitory control.
Physical Exercise
The physical fitness of the participants will be assessed with 30 seconds sit-to-stand test, 22 2 minutes stepping test, 23 and Timed Up and Go test. 24 Based on the measurement results, participants are classified into low, medium, and high physical fitness levels based on the national physical fitness index. 25 The personalized exercise protocol was developed in line with established exercise guidelines for older adults,25,26 taking into account physical fitness level, lifestyle factors (smoking and alcohol use), and cognitive status (MCI or cognitively normal). Based on the algorithm's output, 34 structured exercise program sets consisting of aerobic exercise, resistance exercise, balance exercise, and flexibility exercise will be automatically assigned to each participant according to their individual characteristics (Table 2). Prior to each exercise intervention, participants will be screened for exercise safety using the modified Physical Activity Readiness Questionnaire. 27 Each participant will exercise using a light stick, dumbbells, a chair, and their own body weight while watching a video on the app. The exercise intervention will be performed five times per week for 30 to 35 minutes per session. During the initial 2 weeks, participants will visit the clinic once weekly for guidance by research nurses, after which the proportion of self-directed exercise will be gradually increased over a total of 16 weeks. Personalized interventions will be adjusted through re-evaluations conducted at 4-week intervals.
Structured physical exercise program.

wk: week; min: minute; reps: repetitions; s: seconds.
Nutrition management
The nutrition intervention was developed based on evidence-based guidelines and validated tools for older adults, following the Nutrition Care Process (NCP) framework. 28 The nutrition intervention will include personalized education and assessment systems to address major nutritional risk factors in older adults. For nutritional intervention of participants, personalized nutrition solutions will be assigned based on the result of Nutrition Quotients for the Elderly, 29 Mini Nutrition Assessment score, 30 and Mediterranean-Dietary Approaches to Stop Hypertension Intervention for Neurodegenerative Delay diet (MIND diet) checklists. 31 The nutrition intervention will consist of weekly educational video viewing within 20 minutes, twice-weekly intake monitoring, and weekly motivational enhancement by a nutritionist. Intake monitoring will be conducted through speech recognition dietary surveys and MIND diet checklists, with result sheets to be provided after completion. Nutrition solutions will be reassigned in a personalized manner after re-evaluations every 4 weeks. Educational content will be divided into “Standard education material” and “personalized education material.” Standard education will consist of a total of 16 lectures and animations covering age-related physiological changes and practical strategies for maintaining a healthy diet. Personalized education will be selected according to each participant's assessment results and comprises 29 pieces of contents addressing disease-specific dietary management and symptom based nutritional strategies. If abnormalities are identified in specific indicators such as chronic disease status, hemoglobin or albumin levels, or inadequate intake of protein and calcium, targeted warning messages and educational content will be provided first. To enhance participant engagement, educational materials will be delivered in various formats, including animations, videos, and infographic-style card news. Detailed information regarding weekly topics, content types, and formats of both standard and personalized nutrition education sessions is presented in Table 3.
Contents of nutrition management session.

DASH: Dietary Approaches to Stop Hypertension; MIND: Mediterranean-DASH Intervention for Neurodegenerative Delay.
“Standard” category consists of 16 structured weekly sessions, each covering one topic per week. These sessions are provided uniformly to all participants throughout the 16-week intervention period.
“Personalized” category includes additional content that is selectively delivered based on individual nutritional assessments, health status, and specific needs of each participant.
Control condition
At baseline, participants in the control group will meet a study doctor, whom may prescribe them medication if necessary, and they will receive a booklet containing lifestyle guidelines for the prevention of frailty progression, based on recommendations from the Korean Academy of Family Medicine. 6
Outcome measures
The primary outcomes are retention rate, adherence rate, and recruitment rate. The intervention program will be determined feasible if the following success criteria are met 32 : (a) a minimum retention rate of 70% at week 16, (b) a minimum adherence to the intervention program of 70%, and (c) a minimum recruitment rate of 50%. The retention rate will be used to calculate the percentage of participants in each group who have not dropped out at the end of the study. The SUPERAGING app will be configured to allow administrators to view all participants’ data on the administration homepage. Since all interventions will be delivered through the app, adherence to the interventions will be automatically measured by the app. The recruitment rate is the enrollment rate among eligible participants who consent to trial participation.
The secondary outcome measures will assess changes in frailty, cognitive function, disability, physical function, quality of life, mood, nutrition, vascular risk factors, skeletal muscle mass, and the occurrence aging-related diseases and adverse events. Frailty will be assessed using the modified Fried Frailty Phenotype scale, 4 Korean Frailty Index for Primary Care, 33 and Korean version of Lubben Social Network Scale. 34 Cognitive function will be assessed using K-MMSE-2, 17 14-item cognitive subscale of the Alzheimer's Disease Assessment Scale (ADAS-cog14), 35 and Clinical Dementia Rating scale-Sum of Boxes. 36 The K-MMSE-2 provides three versions: brief version, standard version, and expanded version. The expanded version of the K-MMSE-2 is the standard version of the K-MMSE-2 with the addition of story memory and digit symbol coding tests. 17 In this study, we will use the standard version of the K-MMSE-2, which is similar to the original MMSE (range, 0–30). The ADAS-cog14 consists of word recall, naming, language, orientation, visuoconstruction, ideational praxis, Maze task, and digit cancellation task. 35 In this study, the parallel form of the word list from the ADAS-cog14 will be used for both baseline and post-intervention assessments. Other secondary outcome measures are summarized in Table 4.37–42 All secondary outcome measures were validated in Korean old adults.
Primary and secondary outcome measures.
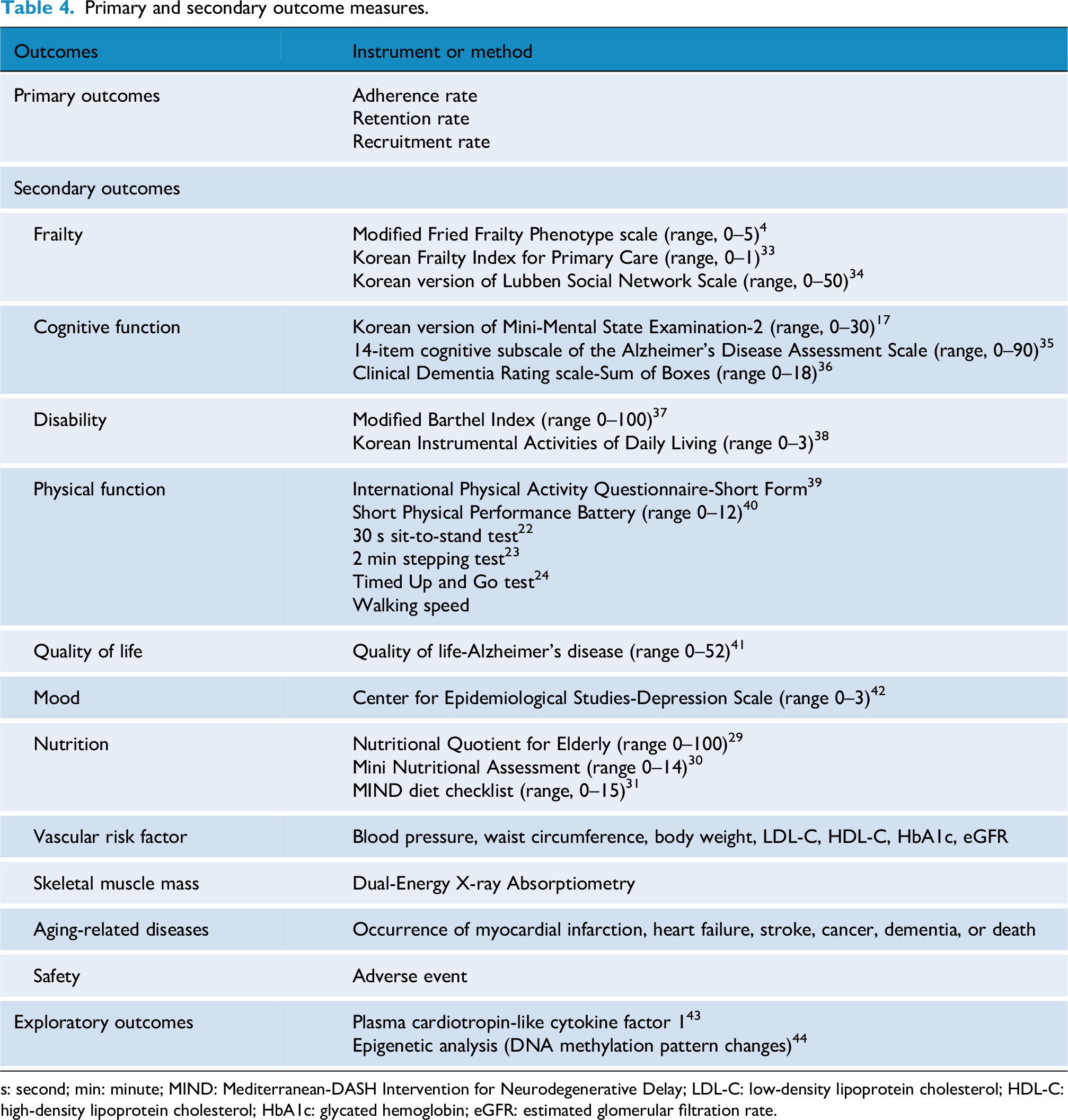
s: second; min: minute; MIND: Mediterranean-DASH Intervention for Neurodegenerative Delay; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; HbA1c: glycated hemoglobin; eGFR: estimated glomerular filtration rate.
Exploratory outcomes
Changes in plasma cardiotropin-like cytokine factor 1 levels after intervention will be assessed to investigate the biological effects of the intervention. 43 Additionally, the impact of the interventions on biological aging will be investigated through changes in DNA methylation patterns. 44
Study procedure
Prior to study initiation, training workshops on accurate study intervention procedures and assessment methods will be conducted for evaluators and related researchers who will perform the research intervention program. Study participants will be enrolled according to consent procedures and selection criteria. All intervention adherence will be automatically recorded through the app and collected in real-time for analysis. Study coordinators will monitor adherence in real-time, and adverse events will be evaluated by confirming their occurrence at each visit. Secondary and exploratory outcome measures will be assessed 4 weeks before intervention initiation and within 2 weeks after intervention completion. Participants who withdraw prematurely will be instructed to complete all endpoint assessments at early termination. The weekly training schedule and 4-week reassessment for tailored interventions are summarized in Figure 2.

Weekly education program and re-evaluation program. Participants in the intervention group will receive app-based training for prospective memory and frontal function, education videos for management of diseases that are risk factors for frailty, nutrition education videos, meal monitoring, and physical exercise consisting of aerobic exercise, resistance exercise, balance exercise, and flexibility exercise for 16 weeks. Personalized interventions will be applied based on the results of re-evaluations conducted every 4 weeks. Interventions and re-evaluations will be conducted through a mobile app.
Criteria for early discontinuation
Participants will be considered to have withdrawn from the study in the following cases: (a) when the investigator judges that continued participation in the clinical trial may be detrimental to the participant's health or welfare; (b) when the participant withdraws consent for study participation; (c) when the participant repeatedly violates protocol requirements and the investigator determines that discontinuation is necessary; (d) when the investigator determines that withdrawal is in the participant's best interest for any other reason; (e) when follow-up of the participant is not possible.
Data management
Study coordinators will complete data entry at each center. The data quality will be improved through automatic range examination of the data values. The data collected in this study will be monitored by an independent monitor in accordance with Good Clinical Practice (GCP) guidelines and clinical research protocols. Only authorized users will have access to the data system. Intervention-related data performed through the app will be automatically collected on the administrator page. The administrator page will be accessible only to authorized users. Deidentified individual participant data, data dictionaries, and statistical codes will not be publicly available but may be shared upon reasonable request, subject to IRB approval and data sharing agreements.
Sample size estimation
The sample size of 40 participants (20 per group) was determined based on feasibility and methodological guidelines for pilot randomized controlled trials, which recommend 12 to 20 participants per arm to estimate feasibility parameters and preliminary effect sizes.45,46 Therefore, a total of 40 participants was considered sufficient to evaluate feasibility and provide data for future sample size calculations.
Statistical analyses
Statistical analysis will be performed on a modified intention-to-treat population that include all randomized participants who have undergone a baseline assessment and at least one post-baseline assessment and who—if assigned to the intervention group—have engaged in the intervention program at least once. Auxiliary analysis will be performed using the Per Protocol population, which will separately classify participants who adhere to the study protocol. Baseline characteristics of participants will be presented as mean ± standard deviation or median (interquartile range) for continuous variables and frequency and percentage for categorical variables. Between-group comparisons will use t-test or Mann–Whitney U test for continuous variables according to normality, and Chi-squared test or Fisher's exact test for categorical variables. Intervention feasibility will be evaluated based on retention rate (≥70%), intervention adherence (≥70%), and recruitment rate (≥50%). Intervention adherence will be calculated through access records to exercise, cognitive training, nutrition, and disease management contents on the digital platform. Missing data patterns will be assessed to determine whether they are missing completely at random, missing at random (MAR), or missing not at random. If data are assumed to be MAR, multiple imputation using chained equations will be performed with an allowable dropout rate of up to 20% to ensure meaningful imputation of missing values. For continuous variables, predictive mean matching will be used. This method fits a linear regression model to the observed cases and uses predicted values to identify observed values with similar predictions, from which one is randomly selected for imputation. Pre- and post-intervention changes in secondary outcome measures will be analyzed using analysis of covariance (ANCOVA) with baseline values as covariates. Variables that significantly differ between the intervention and control groups in univariate analysis will also be considered as covariates in ANCOVA analysis. Safety will be evaluated in all participants who use the investigational medical device at least once. All statistical analyses will be performed using two-sided tests, with significance level set at p < 0.05.
Ethics and dissemination
This study will be conducted in accordance with the International Conference on Harmonization GCP Guideline. This study has been approved by the IRBs of all participating institutions: Inha University Hospital (IRB No. 2024-07-020), Ajou University Hospital (IRB No. AJOU-IRB-2024-448), and Ewha Womans University Seoul Hospital (IRB No. SEUMC 2024-07-057). Protocol modifications will be reported and approved by each institution's IRB. All potential participants will provide written informed consent and additional consent for collecting and using biological specimens facilitated by a study doctor before participating in the study. All participants’ names and privacy will be kept confidential throughout the research and dissemination processes, and they will be identified by the numbers assigned during the study. The results of this study will be disseminated through peer-reviewed academic journals, national and international conferences, and institutional reports. Additionally, key findings may be shared with relevant stakeholders and policymakers to support the development of community-based multidomain intervention strategies for older adults.
Discussion
Frailty refers to a vulnerable state characterized by an overall decline in physiological reserve due to aging, resulting in inadequate responses to external stressors. 6 It is recognized as a multidimensional health risk condition involving the complex interaction of functional decline, chronic diseases, cognitive impairment, physical inactivity and malnutrition, rather than a single disease entity. 47 Accordingly, the necessity of multidomain interventions that integrate various components such as exercise, cognitive training, nutritional management, and chronic disease management has been emphasized. 47 Furthermore, a prospective single-arm study involving hospitalized older adults with frailty evaluated the effects of a multidomain program incorporating cognitive stimulation, physical activity, frailty education, and nutritional counseling. 48 The intervention led to significant improvements in functional status, as well as reductions in hospital length of stay, healthcare costs, and readmission rates after discharge. These findings suggest that tailored multidomain interventions, which take into account an individual's functional and health status, may be effective in delaying the progression of frailty and improving clinical outcomes.
In the cognitive domain, the Sustainable Memory and Aging Risk Reduction Trial demonstrated the application of a personalized multidomain intervention. 49 This RCT targeted older adults who were at high risk of dementia and provided individualized interventions addressing various risk factors, including physical activity, sleep, and depressive symptoms, based on each participant's health status and risk profile. The intervention was dynamically adjusted through regular reassessments every 4 to 6 weeks, leading to significant improvements in cognitive function, dementia risk profiles, and quality of life. In the present study, we incorporated a similar structure using the digital cognitive assessment tool at 4-week intervals to monitor functional levels and adjust the content and difficulty of cognitive training tasks accordingly. In the physical activity domain, previous studies have shown that responses to exercise vary significantly depending on an individual's fitness level, physiological characteristics, and lifestyle, highlighting the importance of personalized exercise prescriptions.50,51 Reflecting this understanding, the American College of Sports Medicine launched the “Exercise is Medicine” initiative, which promotes the integration of individualized exercise prescriptions as part of medical treatment for patients with chronic conditions. 52 Such personalized approaches have been reported to enhance the effectiveness of exercise interventions and reduce non-responsiveness. 53 In the nutritional domain, the NCP model comprising systematic assessment, diagnosis, intervention planning, implementation, and re-evaluation enables tailored interventions based on the individual's nutritional status. 28 In this study, personalized nutritional educational contents for older adults were created based on this framework.
The evidence and key strategies derived from previous studies will be systematically incorporated into the content design of the SUPERAGING program. In the disease management domain, evidence-based educational modules will be developed to monitor chronic disease risk factors and promote disease-specific lifestyle modifications. In the cognitive domain, difficulty-adjusted training tasks will be implemented with regular reassessments to maximize cognitive function improvement. In the physical exercise domain, principles of personalized exercise prescription will be applied, taking into account individual fitness levels, physiological characteristics, and lifestyle factors. In the nutrition domain, the NCP model will be adopted to provide individualized nutrition education and dietary improvement strategies. By integrating these evidence-based intervention components across multiple domains and incorporating adjustments based on reassessments of participants’ health status and functional levels, SUPERAGING will be developed as a personalized, digital multidomain intervention program, and its feasibility and preliminary effectiveness will be evaluated. The intervention will be tailored to each participant's health status using an algorithm and dynamically adjusted through reassessments conducted every 4 weeks.
Limitations
This study has several limitations. First, the study focuses on evaluating short-term intervention effects, and thus, evidence on long-term effectiveness and sustainability is limited. Second, some older adults unfamiliar with smart devices may experience challenges in using the digital platform, potentially influencing intervention participation and outcomes. Third, a questionnaire assessing participants’ expectations about the intervention—which could help control for placebo effects—is not administered before the intervention in this study. Because participants are aware of their group allocation and the novelty of the digital platform, participants’ expectations regarding digital interventions may influence subjective outcomes such as engagement and self-reported behaviors. 54 The potential placebo effect should be taken into account when interpreting the findings. Fourth, in this study, the control group is not a waitlist control group. For ethical reasons, educational material on preventing frailty progression will be provided to the control group, which may itself induce behavioral changes and consequently influence outcomes and introduce bias.
Conclusion
This study will evaluate the feasibility of SUPERAGING, an ICT-based personalized multidomain intervention program integrating nutrition, physical activity, cognitive training, and chronic disease management for older adults with frailty or prefrailty. By providing individualized interventions based on periodic health reassessments, the program holds significant potential as a scalable and sustainable digital healthcare model for community settings. The findings of this study are expected to provide essential evidence to inform the development of evidence-based strategies for promoting health and extending healthy life expectancy among high-risk older populations.
Footnotes
Acknowledgments
The authors thank all researchers for their sincere cooperation. It is acknowledged that the statistical part of this paper was supported by the Biostatistics Center of Inha University Hospital, Incheon, Republic of Korea.
ORCID iD
Ethical considerations
This study has been approved by the Institutional Review Boards (IRBs) of Inha University Hospital (IRB No. 2024-07-020), Ajou University Hospital (IRB No. AJOU-IRB-2024-448), and Ewha Womans University Seoul Hospital (IRB No. SEUMC 2024-07-057).
Consent to participate
All participants of this feasibility trial provided written informed consent prior to participating in this study.
Consent for publication
Not applicable.
Contributorship
Soyoung Jung did resources, writing—original draft; Hae Jin Kang performed investigation, resources, writing—review and editing; Moonjung Choi did investigation, writing—review and editing; Jee Hyang Jeong and So Young Moon did investigation, writing—review and editing, supervision; Hang-Rai Kim, Soonoh Jung, Jiwoo Jung did software, data curation, app development, writing—review and editing; Yoo Kyoung Park and Seong Hye Choi performed supervision, funding, writing—review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Convergence Research Center Program (CRC22014-600) through the National Research Council of Science & Technology (NST), funded by the Ministry of Science and ICT, Republic of Korea. The funder has no role in the design of the study, data collection and analysis, site management, or manuscript preparation. This work was supported by the BK21 Plus Program “AgeTech-Service Convergence Major” through the National Research Foundation (NRF) funded by the Ministry of Education of Korea (No. 5120200313836).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SYM, JHJ, SHC, and YKP are shareholders of Rowan Inc. JJ, HRK, and SJ are employees of Rowan Inc. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data availability
The data of this study will be available upon reasonable request to the corresponding author.
