Abstract
Objective
To assess the feasibility, acceptability, and preliminary effects of circadian rhythm for sleep (CRS), a mobile digital therapeutic that delivers closed-loop, wearable- and light-sensor–driven circadian coaching for insomnia.
Methods
Six-week, single-arm feasibility study in adults with short-term or chronic insomnia. CRS provided daily recommendations emphasizing stable wake-up time, morning light exposure, and daytime activity. Feasibility and acceptability outcomes were assessed (completion, passive-sensor data capture/adherence, satisfaction), and the primary clinical outcome (exploratory) was change in Insomnia Severity Index (ISI) from baseline to Week 6; the key secondary clinical outcome (exploratory) was Pittsburgh Sleep Quality Index (PSQI). Objective sleep–wake metrics from wearable device were explored.
Results
Twenty-three participants were enrolled; 20 completed the program (87.0%), and 16 comprised the prespecified analysis set based on data-fidelity criteria. Among these 16 participants, valid passive sensor data from the wearable and light sensors were captured on 88.6% of study days. ISI significantly improved from baseline to Week 6 (median 21.0 → 14.0; p < .001 by within-subject analysis), and PSQI improved (mean 10.9 → 7.7; p < .001; partial η2 ≈ 0.50). Objective wearable metrics (total sleep time, time-in-bed, sleep onset, wake time) did not change significantly over time in this short pilot. Satisfaction was favorable (mean 37.9/45). No adverse events occurred.
Conclusions
CRS was feasible and acceptable, and was associated with within-subject improvements in subjective insomnia symptoms in this single-arm feasibility study; however, because there was no control group, these findings are preliminary and hypothesis-generating, supporting further evaluation in larger randomized controlled trials.
Trial registry name
Clinical Research Information Service
URL
Trial registration number
KCT0010801
Keywords
Introduction
Insomnia is one of the most common health-related complaints in modern society, characterized by difficulty in initiating or maintaining sleep.1–3 It affects an estimated 10–30% of the adults, with approximately 10% meeting the criteria for chronic insomnia disorder.2,4 Beyond sleep difficulties and daytime impairment, insomnia is associated with increased risks for depression, anxiety, cardiovascular disease, and diminished quality of life.2,5–9
Cognitive Behavioral Therapy for Insomnia (CBT-I) is recommended as the first-line treatment for chronic insomnia disorder by major clinical practice guidelines, including the American College of Physicians guideline. 10 CBT-I protocols typically involve cognitive restructuring (e.g. modifying dysfunctional beliefs) and implementing behavioral strategies such as sleep restriction, stimulus control, sleep hygiene education, and relaxation techniques. 10 In particular, the sleep restriction and stimulus control components, which require patients to maintain consistent bedtimes and fixed wake-up times, may indirectly promote circadian rhythm stabilization by enforcing regular sleep–wake schedules.10,11 However, CBT-I does not directly target circadian misalignment nor systematically incorporate environmental synchronizers such as light exposure, which are critical for regulating biological rhythms. 12
Moreover, many individuals with insomnia show signs of circadian misalignment, including delayed sleep phases, irregular sleep–wake patterns, and insufficient morning light exposure.13,14 These patterns are especially prevalent modern environments dominated by artificial lighting and screen use. Circadian rhythm disruption is increasingly recognized as a key etiological factor in sleep disorders, mood dysregulation, and metabolic diseases.15–19
Chronotherapy—the use of behavioral and environmental strategies to restore circadian alignment—offers a promising complementary approach to treating insomnia. 11 However, structured chronotherapeutic interventions remain underutilized in routine clinical practice. 20
Digital therapeutics (DTx), defined as evidence-based software tools to prevent, manage, or treat medical conditions, provide a scalable solution to this gap. 21 The circadian rhythm for sleep (CRS) app was developed as a mobile DTx that integrates wearable data to promote a consistent and healthy sleep–wake schedule, optimally timed light exposure, and morning activity. By targeting underlying biological timing mechanisms, CRS aims to improve sleep outcomes through circadian realignment. By leveraging real-time data from commercial wearable devices, CRS delivers adaptive and personalized feedback, highlighting its potential as a chronotherapeutic sleep intervention. This study outlines the conceptual foundation and feasibility findings of the CRS app. We report user engagement, changes in subjective and objective sleep parameters, and examine the potential of wearable-driven chronotherapy as an accessible intervention for insomnia.
Conceptual framework
Unlike standard CBT-I–based or sleep-hygiene–focused digital treatments, CRS targets circadian misalignment—an important contributor in a substantial subset of individuals with insomnia—through adaptive, wearable- and light-sensor–driven feedback on daily behavior.13,22 The development of the CRS app is grounded in an integrated framework combining circadian neuroscience, behavioral psychology, and digital intervention strategies. Central to this conceptualization is the recognition that many cases of insomnia are not solely the result of cognitive or behavioral dysfunction but are closely linked to circadian misalignment.13,22 In modern society, delayed sleep phase tendencies, inconsistent daily routines, and insufficient morning light exposure disrupt the synchronization of the internal circadian system with environmental time cues. 22 CRS was therefore designed to provide daily behavioral guidance to reestablish these temporal anchors—primarily through morning activity, consistent wake-up times, and appropriately timed light exposure. In practice, CRS translates this framework into concrete daily recommendations—for example, advancing or stabilizing wake-up times, increasing early-day outdoor light exposure, and reducing irregular late-night activity—with targets that are dynamically adjusted to each user's recent sleep–wake history. From a chronobiological perspective, the app draws on evidence that early-day light and activity serve as powerful zeitgebers—external cues that reset the biological clock. 23 The suprachiasmatic nucleus (SCN), located in the hypothalamus, is the central pacemaker of the circadian system and regulates rhythms in sleep, hormone release, and body temperature. 24 Morning light, particularly in the blue-light spectrum, stimulates intrinsically photosensitive retinal ganglion cells, which signal the SCN and facilitate phase advancement. 25 This biological principle forms the basis for the app's recommendation to increase light exposure immediately upon waking. In particular, the app applies this principle using a mathematical model that calculates and recommends the optimal timing of light exposure. 26
The CRS framework incorporates empirically supported behaviour-change techniques—such as self-monitoring, performance feedback, goal setting, and reinforcement—that are consistent with Social Cognitive Theory and the broader behaviour-change literature. 27 These techniques are implemented in CRS through features such as real-time progress monitoring, automated motivational prompts, and intuitive data visualizations, which are intended to support self-efficacy and sustained adherence to circadian-aligned routines.28,29
In line with best practices in digital therapeutics,30,31 the design of CRS emphasizes low user burden, incorporating passive data collection via wearable devices and smartphone sensors to reduce friction and maximize adherence. The platform dynamically adapts to each individual's circadian profiles and behavioral patterns, delivering personalized interventions informed by continuous sleep–wake data. Importantly, CRS functions as a closed-loop system, where users’ behavioral data continuously shape the following day's circadian recommendations—effectively operating as a real-time, adaptive chronotherapy engine.32,33
To make this closed-loop architecture explicit as a prediction model–based intervention, CRS incorporates a supervised machine-learning component that estimates each day's probability of insomnia-like sleep disruption from multiday patterns of sleep, activity, heart-rate, and light exposure. We prespecified the outcome (insomnia-like sleep disruption), predictor domains (wearable- and smartphone-derived metrics summarized over 2-, 4-, and 7-day windows), modeling approach (a LightGBM-based binary classifier), and internal validation strategy; these elements are detailed in the CRS algorithm for personalized feedback section and are central to how CRS personalizes feedback for each user.
From a systems perspective, CRS passively collects multiday streams of sleep, activity, heart-rate, and ambient light data from commercial wearables and smartphone sensors, preprocesses these signals on a secure cloud server, and feeds them into a pretrained prediction model that summarizes them into a daily sleep health forecast and circadian rhythm indicators; these indicators, in turn, determine which coaching messages, alerts, and behavioral targets are delivered on a given day (elaborated in the overview of CRS and CRS algorithm for personalized feedback sections).
Overview of CRS
CRS is a nonprescription digital therapeutic developed to improve sleep health in individuals with chronic insomnia. The overall system architecture of CRS is illustrated in Figure 1. It operates by collecting digital phenotypes from the study-provided commercial wrist-worn wearable device (brand masked) (sleep, activity, and heart-rate metrics) and from the smartphone's built-in light sensor (ambient light exposure), which are stored on a secure cloud server and analyzed using a pretrained machine learning model to monitor, visualize, and provide feedback on sleep health status. At the initial setup, users wear the study-provided commercial wrist-worn wearable device (brand masked) (the only device compatible with this feasibility version of CRS), install the CRS app on their Android smartphone, and complete a brief onboarding process that includes a short questionnaire, synchronization between the wearable device and app, and granting permission for passive sensing via the phone's light sensor. Thereafter, each morning when the app is opened, digital phenotype data from the previous day and current day are automatically transmitted to the server.

System architecture of the circadian rhythm for sleep (CRS) digital therapeutic note. This flowchart illustrates the system architecture of the CRS digital therapeutic. Data collected from wearable devices and smartphone ambient light sensors are integrated and processed by an AI-driven algorithm that interprets real-time behavioral and environmental inputs. Based on this analysis, the CRS app provides a daily sleep health forecast (0–100%) and personalized feedback across four domains: sleep, light exposure, physical activity, and circadian rhythm stability. These outputs inform individualized behavioral recommendations—particularly targeting wake-up time, morning activity, and early-day light exposure—to promote behavioral change, improve sleep quality, and enhance circadian alignment.
The feedback delivered by the CRS app primarily includes (1) sleep health forecast and (2) circadian rhythm scores. The circadian rhythm score consists of subcomponents reflecting sleep health, light exposure, physical activity, and circadian regularity. Personalized coaching comments are generated based on score-specific thresholds. If a score falls below a predefined critical threshold, the app triggers a phone alert—such as sound or vibration notifications—along with targeted recommendations to improve the corresponding aspect of circadian health.
Through this feedback and alert mechanism, the application aims to encourage users to voluntarily improve their sleep health by gradually aligning and optimizing their own circadian rhythms. In operational terms, a typical CRS cycle proceeds through the following steps: (1) passive acquisition of wearable-derived sleep, activity, heart-rate, and smartphone light-sensor data, (2) secure cloud transmission and preprocessing, (3) model-based estimation of the “daily sleep health forecast” and “domain-specific z-scores,” (4) translation of these outputs into rule-based feedback messages and alerts, and (5) user-level behavioral adjustments that in turn shape subsequent days’ input data (Figure 1).
The collected data are then analyzed to assess patterns of sleep health and circadian rhythm. From the commercial wrist-worn wearable device (brand masked), CRS ingests device-generated summaries for sleep (total sleep time (TST), time in bed, sleep efficiency, nap duration), activity (step counts within predefined morning, afternoon, evening, and night windows), and heart rate (minute-level heart rate and resting heart rate), while from the smartphone light sensor it ingests ambient light intensity sampled every 5 min during self-reported wake time and aggregated into circadian-relevant windows.
Based on these analyses, the system generates daily sleep health and circadian rhythm scores and delivers personalized feedback. Since users are only required to review the feedback provided, no additional training or complex interaction with the app is necessary.
Building on this mechanistic rationale, CRS focuses on five core behavioral strategies: (1) encouraging consistent and, when appropriate, earlier wake-up times; (2) increasing exposure to natural light during morning hours; (3) reducing exposure to bright or artificial light in the evening and nighttime; (4) minimizing late-night physical activity; and (5) promoting daytime activity, especially in the morning.
CRS algorithm for personalized feedback
As part of the CRS development, a binary machine learning model predicting wearable-derived insomnia-like sleep disruption (“insomnia-like” vs “non–insomnia-like” nights) was constructed based on two datasets: a 21-day observational dataset from 30 adults with self-reported insomnia symptoms and longitudinal data from a clinical cohort of 112 individuals diagnosed with major depressive disorder or bipolar disorder (i.e. mood disorders). 34
On average, each participant contributed approximately 149 days of data collected during euthymic (mood-stable) periods. Input features were derived from wearable devices and smartphone sensors, including metrics related to sleep, physical activity, heart rate, and light exposure.
As shown in Table 1, circadian features were extracted and aggregated over 2-, 4-, and 7-day windows to capture rhythm stability. For algorithm training, we defined an insomnia-like sleep disruption label as sleep efficiency <85% or TST <300 min persisting for ≥3 consecutive days, focusing on short and/or inefficient sleep sustained over several nights rather than isolated single-night disturbances; this operational rule was selected to be feasible and robust for use with longitudinal wearable data and does not represent a full clinical diagnosis of insomnia disorder.
Wearable-derived variables and definitions.
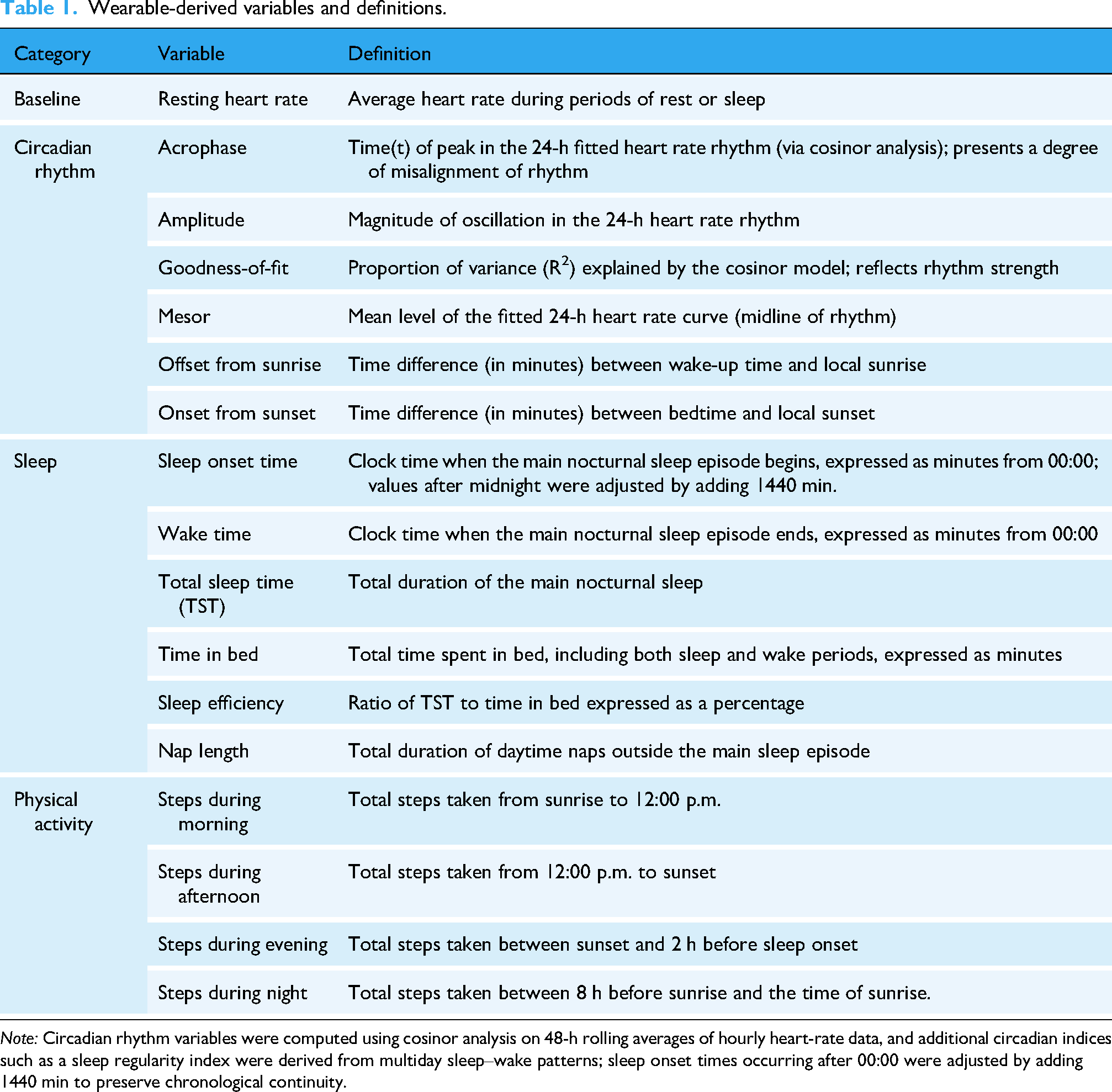
Note: Circadian rhythm variables were computed using cosinor analysis on 48-h rolling averages of hourly heart-rate data, and additional circadian indices such as a sleep regularity index were derived from multiday sleep–wake patterns; sleep onset times occurring after 00:00 were adjusted by adding 1440 min to preserve chronological continuity.
To more effectively capture circadian rhythm stability, all circadian-related features derived from the four passive data sources (heart rate, step counts, light exposure, and sleep logs) were aggregated across 2-, 4-, and 7-day windows. Extending each variable to multiple temporal perspectives allowed the model to incorporate short-, mid-, and longer-term behavioral patterns. This approach is supported by prior digital phenotyping research, 34 which shows that accumulated behavioral changes over several days may be more informative for predicting near-future states than single-day snapshots. Multiday aggregation also enabled computation of stability metrics such as mean values, variability, and temporal gradients, providing a more robust representation of circadian rhythm regularity. Following this rationale, we applied 2-, 4-, and 7-day windows to capture short-range, intermediate-range, and weekly rhythm stability, and these aggregated features were then used as predictors to represent both immediate and accumulated circadian patterns in the model.
This rule-based definition was used to operationalize an “insomnia-like” prediction outcome tailored to longitudinal wearable data and is related to, but not identical with, a full clinical diagnosis of insomnia disorder. Predictors thus comprised prespecified domains (sleep, activity, heart-rate, and light exposure) and their circadian aggregates over short- (two-day), mid- (four-day), and longer-term (seven-day) windows, allowing the model to capture both acute deterioration and more persistent rhythm instability.
The model was trained using LightGBM, with hyperparameters optimized via grid search and cross-validation. To avoid information leakage, we applied grouped cross-validation at the participant level and time-aware feature windows, restricting parameter estimation to training folds. Class imbalance was addressed using class-weighted loss, and the decision threshold was fixed at 0.5 across all splits (see Supplemental Table S3).
A concise model card, detailing the model family, version, hyperparameters, feature definitions, and training/validation scheme, is provided in Supplemental Table S3. Internal validation on the development dataset (independent of the feasibility cohort) demonstrated strong performance, with sensitivity of 98.4%, specificity of 90.0%, and an AUC of 0.944; the corresponding confusion matrix is reported in Supplemental Table S4.
The finalized model was then integrated into the CRS app to compute a daily sleep health forecast. The sleep health forecast is expressed as a percentage-based probability score (0–100%) representing the model-derived probability that the individual's recent three-day sleep pattern does not meet the above insomnia-like criterion (i.e. persistent short/inefficient sleep), based on wearable and smartphone data. This daily probability score integrates multiday sleep, activity, heart-rate, and light-exposure features into a single indicator of sustained sleep health, rather than the probability of fulfilling International Classification of Sleep Disorders, 3rd edition (ICSD-3) diagnostic criteria for insomnia disorder, and is updated once per day using data up to and including the most recent night without using any future information.
To support individualized feedback, the app additionally computed domain z-scores for sleep, activity, and light exposure from each participant's rolling prior two-week observations, standardized using reference-population parameters (mean and dispersion) estimated during algorithm development. Circadian amplitude, acrophase (the timing of the peak of the fitted 24-h rhythm), and mesor (the midline/mean level of the fitted rhythm) were estimated via cosinor analysis of 48-h heart-rate data to assess rhythm stability.35,36
Behavioral feedback was triggered according to pre-specified decision rules based on reference-population norms. Specifically, the CRS scoring system standardizes the daily sleep health forecast score and each domain score using the mean of the reference population used to develop the algorithm. Alerts were triggered when a participant's daily sleep health forecast score or any domain score fell below the reference-population mean (i.e. below-average relative to the reference distribution), prompting targeted coaching messages (e.g. stabilizing wake time and increasing morning light exposure). Conversely, when scores remained at or above the reference mean, the app delivered reinforcing feedback emphasizing maintenance of circadian-aligned routines. In all cases, the system mapped detected patterns to predefined coaching templates and, when indicated, delivered push notifications to the user.
Furthermore, the system recommended individualized optimal wake-up and light exposure times based on estimated dim-light melatonin onset (DLMO). 26 Further details of the CRS app are available in Supplemental Note 1. Key elements of the prediction model (input domains, feature windows, label definition, validation scheme, leakage control, and outputs) are summarized here and are described in greater detail in the full model card provided in Supplemental Table S3.
Methods
Study design and participants
This study employed a six-week, single-arm feasibility design to evaluate the usability, acceptability, and preliminary efficacy of the CRS app in adults with insomnia. The six-week duration was selected to balance participant burden with the need to observe behavioral entrainment across multiple weekly cycles in this first feasibility study. We acknowledge that longer intervention periods and extended follow-up may be required to detect durable and measurable circadian phase shifts. Participants were recruited from January to March 2024 through social media platforms and community bulletin boards.
Eligible participants were adults aged 19 years or older who met the diagnostic criteria for short-term or chronic insomnia according to the ICSD-3, as determined during a board-certified psychiatrist-administered diagnostic interview at the screening visit, and had an Insomnia Severity Index (ISI) score of 8 or greater.
Additional inclusion criteria included ownership of an Android smartphone with OS version 7.0 or higher and willingness to comply with the study protocol.
Exclusion criteria were as follows: (1) diagnosis of other sleep disorders (e.g. obstructive sleep apnea, restless legs syndrome); (2) insomnia secondary to serious medical conditions (e.g. cardiovascular disease, malignancy, severe renal or hepatic dysfunction, endocrine or gastrointestinal disease, pulmonary disease); (3) insomnia caused by medications (e.g. chronic stimulant use) or lifestyle (e.g. excessive caffeine, smoking, alcohol); (4) psychiatric disorders contraindicating sleep restriction (e.g. psychosis, bipolar disorder); (5) PHQ-9 score ≥ 10; (6) irregular work schedules (e.g. night or rotating shifts); (7) history of severe neurological disorders (e.g. head trauma, seizure disorders); (8) visual, auditory, or cognitive impairments preventing app use; (9) inability to operate smartphones or wearable devices; (10) inability to complete daily in-app sleep satisfaction surveys; (11) pregnancy or breastfeeding; (12) other conditions deemed inappropriate for participation by the investigator.
Given the exploratory nature of the study, no formal sample size calculation was performed. A target enrollment of 25 participants was established, aligning with recommendations for feasibility trials. 37
Study procedure
At enrollment, all participants installed the CRS mobile application on their Android smartphones. The CRS mobile application evaluated in this study was developed by Hucircadian (company name), and “CRS” is used throughout this manuscript to refer to the intervention. Each participant was also provided with a commercial wrist-worn wearable device (brand masked), a consumer-grade wearable device with acceptable accuracy for measuring sleep parameters 38 and heart rate. 39 Participants were instructed to wear the device continuously throughout the six-week intervention period. Standardized instructions were provided for both the mobile app and wearable device.
During setup, participants completed brief app-based screening questionnaires to evaluate eligibility and characterize baseline sleep and mental health status, with the specific instruments and assessment schedule described in the outcomes and assessment schedule section and Table 2.
The assessment items in the study.

Note: ✓: Assessment conducted at the corresponding time point; PSQI: Pittsburgh Sleep Quality Index; CSM: Composite Scale of Morningness; ESS: Epworth Sleepiness Scale; ISI: Insomnia Severity Index; WHOQOL-BREF: World Health Organization Quality of Life—Brief version.
The CRS app was linked to each participant's assigned commercial wrist-worn wearable device (brand masked), enabling continuous collection of device-generated summaries for sleep (TST, time in bed, sleep efficiency, nap duration), activity (step counts in predefined morning, afternoon, evening, and night windows), and heart rate (minute-level heart rate and resting heart rate); we relied on manufacturer-processed sleep staging rather than raw accelerometer or photoplethysmography signals.
In parallel, the CRS app passively collected ambient light exposure data via the smartphone's built-in light sensor, sampled every 5 min during self-reported wake time and aggregated into circadian-relevant windows (morning, afternoon, evening, night) before being passed to the algorithm.
All data from the CRS app and wearable device were securely transmitted to a central server and processed in real time using a proprietary machine learning algorithm. This algorithm analyzed individual sleep–wake and circadian patterns and, as detailed in the CRS algorithm for personalized feedback, produced the daily sleep health forecast and domain-specific z-scores that were translated into personalized behavioral recommendations to improve circadian alignment and sleep quality.
Each participant received individualized recommendations, including suggested wake-up times and optimal periods for light exposure in the morning, which were prominently displayed on the app's home screen. This interface also provided a nightly sleep score and actionable daily tips.
Additionally, the app featured a “circadian coaching” page that offered tailored guidance based on users’ adherence to behavioral targets across subdomains such as sleep quality, light exposure, activity regularity, and circadian phase stability. Users could also view comparative rankings relative to the broader CRS user base and monitor longitudinal trends in their sleep–wake cycles, activity levels, and intervention adherence through interactive visualizations. Participants were encouraged to iteratively modify their daily routines based on this feedback to promote alignment with their intrinsic circadian rhythms and thereby enhance sleep quality and overall well-being.
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki and received approval from the Institutional Review Board of Korea University Anam Hospital (Approval No. 2023AN0558). All participants provided written informed consent before enrollment. Participants who completed the study were reimbursed 50,000 KRW (approximately USD 35) for each in-person visit and 10,000 KRW (approximately USD 7) for the telephone assessment.
Outcomes and assessment schedule
Outcome assessments were conducted at four time points: baseline, Week 2 (in-person), Week 4 (telephone), and Week 6 (in-person). As summarized in Table 2, insomnia severity and related patient-reported outcomes were administered repeatedly across these visits, including the Pittsburgh Sleep Quality Index (PSQI), Insomnia Severity Index (ISI), Epworth Sleepiness Scale (ESS), Composite Scale of Morningness (CSM), and World Health Organization Quality of Life-Brief (WHOQOL-BREF). In contrast, app-based screening items for other sleep disorders (e.g. obstructive sleep apnea, restless legs syndrome), night-shift work, and psychiatric comorbidities such as depression and anxiety were collected at baseline only. Table 2 therefore integrates both screening and outcome measures into a single overview of the assessment schedule. The assessment protocol was designed to evaluate the intervention's clinical efficacy, feasibility, and safety, and at baseline participants also provided sociodemographic information, clinical history, and current medication use.
The primary outcome was change in the ISI from baseline to Week 6. The PSQI was designated as a key secondary outcome. Both questionnaires were administered at baseline, Week 2, and Week 6 to evaluate insomnia severity and subjective sleep quality.
Secondary outcomes included both objective and additional subjective measures. Objective sleep–wake data were continuously collected via a wearable device throughout the six-week intervention, including time in bed, sleep onset time, wake time, and TST (definitions in Table 1). Subjective secondary outcomes included the ESS for daytime sleepiness, the CSM for chronotype assessment, and the WHOQOL-BREF for quality of life.
To evaluate feasibility and acceptability, we analyzed average adherence to wearable device usage and responses from a user satisfaction survey. Daily adherence (%) was calculated as the number of 10-min heart rate recordings per day divided by 144 (the total possible recordings per day), multiplied by 100. The average adherence rate was derived by averaging daily values, excluding the baseline and final visit days due to missing data.
For transparency, we prespecified an analysis set based on data-fidelity criteria (completion of the Week-6 assessment, average wearable device adherence ≥70% during the intervention window (excluding baseline and final-visit days), and successful delivery of circadian feedback). Adherence is therefore summarized for completers and for the prespecified analysis set; participants who discontinued early were not included in adherence summaries due to truncated observation windows.
At the final visit, participants completed a satisfaction survey evaluating overall satisfaction, perceived usefulness, device comfort, and willingness to participate in similar future programs. Items were rated on a 5-point Likert scale (1 = not at all to 5 = very much); the total score (nine items) ranges from 9 to 45, with higher scores indicating greater satisfaction. In this feasibility version, we did not capture detailed in-app engagement logs (e.g. whether specific feedback messages were opened, viewed, or acted upon); therefore, engagement with feedback could not be quantified beyond overall program completion and device adherence.
Safety assessments included monitoring of adverse events (AEs), physical examinations, and vital signs. At weeks 2, 4, and 6, study staff asked participants the following standardized question: “Since your last visit, have you experienced any discomfort, new symptoms, or health problems that you think might be related to using the CRS app or wearable device?” Any reported issues were followed by open-ended probing and classified as AEs, serious AEs (SAEs), device-related events, or non-AE discomfort, in line with our protocol (outcomes and assessment schedule section). Physical examinations and vital signs (blood pressure, pulse rate, and body temperature) were conducted at baseline, Week 2, and Week 6.
Statistical analysis
All analyses were conducted using SPSS version 29. Descriptive statistics were calculated for demographic characteristics, baseline clinical variables, and feasibility indicators. For wearable-derived data, values were averaged over three consecutive two-week periods (Week 1–2, Week 3–4, Week 5–6) to ensure stable estimates.
Primary and secondary outcome variables—specifically, the primary ISI change and the key secondary PSQI change—were analyzed using repeated measures ANOVA (RM-ANOVA) to evaluate within-subject changes across time points (baseline, Week 2, and Week 6). To control the family-wise Type I error across outcomes, we prespecified a hierarchical testing strategy: ISI (α = 0.05, two-sided) followed, conditional on significance, by PSQI (α = 0.05). Other outcomes (ESS, CSM, WHOQOL-BREF, wearable-derived sleep–wake metrics, and safety variables) were treated as exploratory and are presented with nominal p-values without multiplicity adjustment. The Shapiro–Wilk test was used to assess normality, and Mauchly's test was used to assess sphericity. When the assumption of sphericity was violated, Greenhouse–Geisser corrections were applied. If parametric assumptions were not met, the Friedman test was used as a nonparametric alternative.
Main effects in RM-ANOVA were interpreted based on estimated marginal means, and Bonferroni-adjusted post hoc comparisons were conducted when significant effects were observed. Effect sizes were reported as partial eta squared (η2) for RM-ANOVA and Kendall's W for Friedman tests. Partial eta squared values of .01, .06, and .14 were interpreted as small, medium, and large effects, respectively. 40 Kendall's W values were interpreted as small (.1–.3), moderate (.3–.5), or large (>.5) effect sizes. 41
For safety indicators (e.g. vital signs), either RM-ANOVA or the Friedman test was used to evaluate time effects. Categorical safety outcomes were summarized as frequencies.
All statistical tests were two-tailed, and a p-value < .05 was considered statistically significant; α was set to 0.05 for the primary outcome and, if the hierarchical criterion was met, for the key secondary outcome. Missing data were handled using pairwise deletion.
Results
Participant flow and characteristics
A total of 23 participants were enrolled between February and March 2024. Of these, three participants discontinued the study—two due to low compliance (i.e. early discontinuation because core study procedures such as wearing/synchronizing the wearable device and engaging with the CRS app were not maintained despite reminders and troubleshooting) and one due to voluntary withdrawal. Additionally, four participants completed the study but were excluded from the prespecified analysis set based on data-fidelity criteria: three due to insufficient compliance with wearing the wearable device (average adherence <70%; 63.08%, 65.34%, and 34.17%), and one due to a retrospectively identified failure in delivering circadian feedback during participation. Since the core therapeutic component of CRS depends on feedback derived from wearable data, the final analysis was ultimately based on 16 participants.
Baseline demographic and clinical characteristics of the final sample are presented in Table 3. Among the 16 participants, 10 (62.5%) were female. The mean age was 51.56 years (SD = 10.79 years).
Baseline characteristics of the participants (N = 16).

Notes: PSQI: Pittsburgh Sleep Quality Index; CSM: Composite Scale of Morningness; ESS: Epworth Sleepiness Scale; ISI: Insomnia Severity Index; WHOQOL-BREF: World Health Organization Quality of Life—Brief version; PHQ-9: Patient Health Questionnaire-9.
Values are presented as mean (standard deviation) for continuous variables and frequency (percentage) for categorical variables.
At baseline, participants reported a mean ISI score of 20.38 (SD = 3.57), a PSQI score of 11.00 (SD = 2.71), and a CSM score of 31.69 (SD = 6.83). The mean score on the ESS was 8.44 (SD = 4.13), and the WHOQOL-BREF mean score was 61.40 (SD = 6.88). Depressive symptom severity, measured by the PHQ-9, had a mean of 5.94 (SD = 3.57).
Seven participants (43.8%) reported at least one comorbid medical condition. The reported conditions include reflux laryngitis, allergic rhinitis, hypertension, hyperlipidemia, hypothyroidism, lumbar disc herniation, chronic gastritis and osteoporosis. Four participants (25.0%) were taking medications at baseline. These included antihypertensive agents, lipid-lowering agents, a thyroid hormone replacement, and a gastrointestinal agent. Among these, one participant was receiving benzodiazepine (alprazolam, bromazepam), nonbenzodiazepine sedative-hypnotic (eszopiclone), antidepressants (escitalopram, trazodone).
Regarding lifestyle factors, one participant (6.3%) was a current smoker, five (31.3%) reported current alcohol consumption, and nine (56.3%) reported current caffeine use.
For lifestyle factors (smoking, drinking, and caffeine consumption), categories were defined as: Never (no lifetime use), Past (former use but not current at baseline), and Present (current use at baseline). Alcohol and caffeine status were collected as self-reported categorical variables (never/former/current) without applying a quantitative cutoff (e.g. number of drinks or mg/day).
Feasibility and acceptability outcomes
Among participants who completed the intervention (n = 20; excluding the three discontinuations), the mean compliance rate for wearable device data collection was 86.63% (SD = 15.21%; range, 34.17%–97.97%). For the final analysis sample (n = 16), the mean compliance rate was 88.64% (SD = 6.95%; range, 75.35%–97.97%). These adherence estimates are reported for completers and for the prespecified analysis set; participants who discontinued early were not included in adherence calculations because their observation windows were truncated.
The mean satisfaction score among all completers was 37.90 (SD = 4.60; range, 29–45) (possible range: 9–45), while the mean satisfaction score among those included in the final analysis was 37.88 (SD = 3.63; range, 33–44) (possible range: 9–45).
Safety outcomes
Using the standardized AE question at weeks 2, 4, and 6, no participants reported any discomfort, new symptoms, or health problems related to the CRS app or wearable device. No AEs occurred during the study period. Consequently, no device-related AEs, SAEs, serious adverse device effects, or unanticipated adverse device effects were reported, and no participants discontinued due to safety concerns. No additional physical examination issues were reported across the study period; therefore, no statistical testing was performed. For participants who completed the full study, no significant changes were observed in systolic blood pressure, diastolic blood pressure, pulse rate, or body temperature across time points (all p > .234).
Exploratory clinical outcomes
Significant improvements in the primary outcome (ISI) and the key secondary outcome (PSQI) were observed across the intervention (Table 4, Figure 2). The main effect of time revealed statistically significant improvements (ISI: Friedman χ2(2) = 28.00, Kendall's W = .875, p < .001; PSQI: F(2, 30) = 15.26, p < .001, partial η2 = .504). Consistent with the prespecified hierarchical testing strategy, PSQI was formally evaluated only after ISI reached significance. Post-hoc comparisons showed significant improvements from baseline to Week 6 for both ISI (p < .001) and PSQI (p < .001), indicating reduced insomnia severity and enhanced subjective sleep quality (Supplemental Table 2). However, although scores improved, the absolute values at Week 6 remained relatively high, suggesting residual insomnia symptoms and incomplete normalization of subjective sleep quality in some participants.

Changes in subjective sleep and functioning measures across time points note. Lines represent changes across three time points. Mean values with standard deviation are plotted for variables analyzed using repeated measures ANOVA (PSQI, CSM, ESS), and median values with interquartile range are plotted for variables analyzed using the Friedman test (ISI, WHOQOL-BREF). Bonferroni-adjusted p-values were used for post hoc comparisons.
Changes in subjective sleep and functioning measures from baseline to Week 6.

Notes: W0: baseline; W2: Week 2; W6: Week 6; PSQI: Pittsburgh Sleep Quality Index; CSM: Composite Scale of Morningness; ESS: Epworth Sleepiness Scale; ISI: Insomnia Severity Index; WHOQOL-BREF: World Health Organization Quality of Life—Brief version.
For variables analyzed with repeated measures ANOVA (PSQI, CSM, ESS), values are presented as mean (standard deviation).
For variables analyzed with the Friedman test (ISI, WHOQOL-BREF), values are presented as median with interquartile range (Q1, Q3).
Primary endpoint.
Key secondary endpoint; formally tested only if ISI reached significance (hierarchical testing).
Outcomes other than ISI and PSQI are exploratory.
p-values are nominal.
Given the single-arm feasibility design without a control group, these within-subject changes should be interpreted as preliminary and require confirmation in adequately powered randomized controlled follow-up studies.
For subjective secondary outcomes, a significant main effect of time was observed for CSM (F(2, 30) = 4.132, p = .026, partial η2 = .216) and ESS (F(2, 30) = 5.223, p = .011, partial η2 = .258), with effect sizes in the moderate-to-large range; these analyses were exploratory, and p-values are reported as nominal. Pairwise comparisons with Bonferroni correction indicated that changes in CSM were not statistically significant between any time points (p > .05). However, a nominal decrease in ESS scores was observed from baseline to Week 2 (p = .039), suggesting an improvement in daytime sleepiness. WHOQOL-BREF scores showed no significant change over time (Friedman χ2(2) = 2.375, Kendall's W = .074, p = .305) (exploratory; nominal p-values).
Unless otherwise specified, outcomes other than ISI (primary) and PSQI (key secondary) were treated as exploratory, and their p-values are nominal.
Exploratory wearable-derived outcomes
Objective sleep parameters derived from wearable device, including TST, time-in-bed, sleep onset time, wake time, showed no statistically significant changes over time (all p > .05). Time-course trends are illustrated in Figure 3 and summary statistics are presented in Supplemental Table S1.

Changes in wearable-derived objective sleep parameters across time points note. Plots show mean values with 95% confidence intervals, aggregated across three consecutive two-week periods during the intervention. Objective measures are exploratory outcomes; values and any p-values are nominal.
Discussion
This pilot study introduces CRS as a novel, nonpharmacological, app-based digital therapeutic targeting insomnia by facilitating circadian realignment. Unlike existing digital interventions—primarily focused on CBT-I, sleep hygiene, or mindfulness practices 42 —CRS uniquely incorporates continuous, individualized circadian feedback derived from wearable data which prevents subjectivity bias. 43 Insomnia interventions focusing on direct and sustained correction of circadian misalignment remain uncommon. Prior circadian-focused approaches have typically relied on light therapy as an adjunctive therapy44–46 rather than leveraging continuous, individualized monitoring of circadian patterns to guide adaptive recommendations.
The conceptual foundation of CRS is consistent with established chronobiological principles, wherein early wake times, morning light exposure, and physical activity act as potential zeitgebers to advance circadian phase. Modern lifestyle factors such as delayed bedtimes and artificial evening light exposure have been strongly implicated in circadian misalignment and chronic insomnia. CRS aims to counteract these disruptions by offering personalized guidance rather than providing static menus of generalized advice.
Subjective outcomes
Participants using CRS showed significant improvements in subjective insomnia severity and sleep quality. The primary endpoint (ISI) improved significantly over six weeks, and the key secondary endpoint (PSQI) also improved under the prespecified hierarchical testing plan. Although ESS and CSM trends indicated reduced daytime sleepiness and a shift toward morningness, these were exploratory analyses and are reported with nominal p-values; post-hoc pairwise differences did not consistently retain significance after correction, and thus should be interpreted cautiously. Nevertheless, despite improvement, the absolute ISI and PSQI values at Week 6 remained relatively elevated, indicating that a meaningful level of symptom burden persisted.
Because this was a single-arm feasibility trial without a control group, these within-subject changes should be interpreted cautiously and regarded as preliminary and hypothesis-generating rather than definitive evidence of treatment efficacy. Overall, CRS was associated with within-subject improvements in subjective insomnia-related outcomes in this feasibility study; however, efficacy and the magnitude of benefit should be confirmed in controlled randomized trials.
Objective outcomes
Objective wearable-derived sleep parameters (e.g. TST, time in bed, sleep onset time, and wake time) did not show statistically significant changes over the six-week intervention, although small directional trends were observed (Figure 3; Supplemental Table S1). This pattern is consistent with prior insomnia research, including CBT-I studies, where improvements in subjective outcomes may not be accompanied by robust changes in objective sleep metrics. One plausible explanation is that the daily feedback on sleep patterns may have reduced sleep misperception, thereby improving subjective insomnia symptoms even in the absence of measurable physiological change. Given the feasibility design, limited sample size, and short intervention duration, these exploratory objective findings should be interpreted cautiously and reevaluated in larger controlled trials with longer follow-up. Importantly, the absence of significant changes in wearable-derived sleep–wake parameters over six weeks does not necessarily indicate a lack of circadian phase shifting; rather, the intervention window may have been too short and/or the selected consumer wearable metrics may be insufficiently sensitive to capture circadian phase changes. Although CRS generated personalized recommendations using an estimated DLMO, we did not validate these estimates against biological circadian markers in this feasibility trial. Future trials should incorporate more direct and sensitive objective measures of circadian timing, such as research-grade actigraphy (with concurrent light exposure monitoring), salivary melatonin-based DLMO assessment, and—when feasible—EEG/PSG-based sleep recordings, to quantify circadian phase and sleep physiology more precisely.
Feasibility, acceptability and safety outcomes
Feasibility results were encouraging. Nearly 90% (87.0%) of participants completed the program, adherence to wearable use was high, and user satisfaction was favorable. No AEs were reported. However, occasional technical disruptions such as device-app desynchronization highlight the need for improved system robustness. Future development should incorporate more granular engagement and fidelity metrics, such as whether feedback messages were opened and followed, wake-time adherence to prescribed targets, and compliance with light-exposure recommendations, to enhance intervention fidelity. However, acceptability assessments did not specifically quantify smartphone-related burden from passive sensing (e.g. battery drain or overheating), which should be systematically evaluated in future studies.
Limitations and future directions
This study's limitations include its single-arm design, small sample size, and lack of randomization, precluding causal inference. In particular, without a control arm we cannot rule out the possibility that the observed improvements in ISI and PSQI reflect placebo responses, regression to the mean (given that participants were enrolled based on elevated baseline symptoms), Hawthorne effects, or natural fluctuation in symptoms rather than specific effects of CRS. Therefore, the observed improvements in patient-reported outcomes should be interpreted cautiously, as they may partly reflect changes in sleep perception or reporting rather than definitive physiological sleep improvement.
Technical issues and dropouts further reduced analytic power. Moreover, wearable devices, while ecologically valid, may lack sensitivity for detecting circadian phase shifts compared to laboratory-based assessments. In addition, a six-week intervention may be insufficient to yield detectable changes in circadian phase proxies derived from consumer wearables, and longer-duration trials with follow-up assessments are warranted to evaluate whether CRS induces sustained circadian realignment. We did not formally assess smartphone-level burden related to passive sensing (e.g. battery drain, overheating, or increased charging frequency) using standardized questionnaires or objective device metrics; therefore, the acceptability profile may be incompletely characterized. Future trials should prospectively measure these factors alongside usability and satisfaction outcomes to provide a more comprehensive evaluation of acceptability.
Generalizability is also constrained by the inclusion of adults only (≥19 years) and the exclusion of individuals with irregular work schedules (e.g. night or rotating shifts); moreover, older adults may have been underrepresented in this convenience sample. Moreover, because one of the training datasets was a clinical cohort of patients with major depressive disorder or bipolar disorder, the model's performance and risk thresholds may not fully generalize to individuals with insomnia who do not have comorbid mood disorders.
Finally, because only ISI was designated as the primary endpoint and multiplicity control was implemented via hierarchical testing (with PSQI as a key secondary), inferences for other outcomes—as well as any efficacy-related conclusions drawn from this feasibility trial—are exploratory and hypothesis-generating and should be confirmed in larger randomized controlled trials.
Future research should employ randomized controlled trials with larger, more diverse populations, extended follow-up, and multimodal circadian assessments. Tailoring interventions to chronotype, insomnia subtype, or illness duration may further optimize outcomes. Enhancing the clarity, frequency, or motivational framing of circadian feedback may also improve behavioral adherence and therapeutic efficacy. More granular research design that better supports the mechanism of CRS application such as tracking user adherence to the feedback may also add reliability and value of CRS.
To strengthen the generalizability of the insomnia-like sleep disruption prediction model, we plan external validation in independent cohorts that were not used for model development, ideally across multiple sites and with broader demographic and clinical diversity (e.g. wider age range, balanced sex distribution, varying insomnia subtypes, and comorbidity profiles). External validation will quantify both discrimination and calibration, and we will report subgroup-specific performance (e.g. by age group, sex, and relevant clinical strata) to assess fairness and identify potential performance gaps. We also acknowledge potential sources of bias in the current work, including convenience-sampling–related demographic skew in the feasibility cohort and spectrum bias introduced by incorporating a mood-disorder clinical cohort during model development; therefore, thresholds and/or model recalibration may be required when deploying CRS in insomnia populations without mood disorders. Although we used participant-level grouped cross-validation and time-aware feature windows to reduce leakage and mitigate overfitting risk, overfitting remains possible given the feature set and development data characteristics; hence, we will prioritize prospective external validation, model recalibration (and, if needed, updating with additional diverse data), and routine postdeployment performance monitoring.
Conclusion
This study provides initial evidence that CRS is feasible and acceptable, and was associated with within-subject improvements in subjective insomnia-related outcomes in a single-arm feasibility study. Because this study lacked a control group, these findings are preliminary and hypothesis-generating; randomized controlled trials are required to establish efficacy and quantify effect sizes. By promoting consistent wake times and morning activity, CRS has the potential to facilitate behavioral phase advancement and improve subjective sleep outcomes. These findings support the promise of circadian-targeted digital therapeutics in clinical practice and underscore the role of behavioral entrainment as a core mechanism in sleep health. Notably, this study represents one of the first digital therapeutics efforts to directly target circadian rhythm misalignment as a primary mechanism, marking a meaningful step forward in the development of personalized chronotherapeutic intervention for insomnia.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261430230 - Supplemental material for The Circadian Rhythm for Sleep digital therapeutic for insomnia: Conceptual background and single-arm feasibility study
Supplemental material, sj-docx-1-dhj-10.1177_20552076261430230 for The Circadian Rhythm for Sleep digital therapeutic for insomnia: Conceptual background and single-arm feasibility study by Minhee Seo, Soohyun Park, Jaegwon Jeong, Yerim Nam, Eunbi Lee, Yujin Lee, Ji Won Yeom, Chul-Hyun Cho, Leen Kim, Jung-Been Lee and Heon-Jeong Lee in DIGITAL HEALTH
Footnotes
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki and received approval from the Institutional Review Board of Korea University Anam Hospital (Approval No. 2023AN0558). Participants who completed the study were reimbursed 50,000 KRW (approximately USD 35) for each in-person visit and 10,000 KRW (approximately USD 7) for the telephone assessment.
Consent to participate
All participants provided written informed consent before enrollment.
Consent for publication
Not applicable.
CRediT authorship contribution
Minhee Seo: formal analysis, data curation, and writing—original draft; Soohyun Park: formal analysis, data curation, writing—original draft, and visualization; Jaegwon Jeong: investigation, data curation, and writing—review and editing; Yerim Nam: investigation and data curation; Yujin Lee: investigation and data curation; Ji Won Yeom: investigation, writing—review and editing; Chul-Hyun Cho: supervision, and writing—review and editing; Leen Kim: conceptualization and supervision; Jung-Been Lee: methodology, software, writing—review and editing, and supervision; Heon-Jeong Lee: conceptualization, methodology, investigation, writing—review and editing, supervision, and funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF), funded by the Korean government (MSIT) (No. RS-2024-00440371), and by the Technology Innovation Program for SMEs funded by the Korea Technology and Information Promotion Agency for SMEs (TIPA) under the Ministry of SMEs and Startups (Grant No. S3200157).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Leen Kim and Dr. Heon-Jeong Lee are co-founders and shareholders of Hucircadian, the company that developed the CRS application evaluated in this study. All other authors declare no financial or non-financial competing interests.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. Due to privacy and ethical considerations, individual-level data cannot be made publicly available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
