Abstract
Digital technologies, such as mHealth interventions and integrated data management tools, are increasingly being developed and implemented to support patients and health care providers in low-resource, high tuberculosis (TB)-burden countries in initiating and proceeding through the TB care cascade (e.g., screening, testing, diagnosis, treatment). Yet, given the proliferation of these tools, there exists a need to synthesize what technologies are being used and where, as well as build a comprehensive understanding of their respective functionality and implementation considerations. The objectives of this systematic scoping review were: (1) to systematically identify literature on digital technologies for supporting the TB cascade in high TB-burden countries; and (2) to describe the facilitators and barriers to technology implementation. Four databases were systematically searched for published literature using a search hedge of terms related to TB, technology, and implementation. Two independent reviewers conducted screening of retrieved literature, data extraction, and data analysis. Eighteen digital technologies were identified, with 10 classified as backbone technologies and eight as add-in technologies. Three key implementation domains were identified: (1) Interoperability and Integration, (2) Digital Infrastructure, and (3) User Experience. Backbone technologies showed higher integration rates with National TB Programs and were more likely to be sustainably implemented. Key barriers to technology implementation included connectivity issues, inadequate user training, and complex multistakeholder integration processes. Included sources described how implementation success was influenced by the interplay between systems-level, technology-level, and user-level factors. Future research should prioritize implementation science approaches to facilitate technology adoption and use to support the TB care cascade.
Keywords
Introduction
Tuberculosis (TB) is a critical public health issue, and is one of the top ten leading causes of death globally. 1 In 2023, an estimated 10.6 million new cases and 1.25 million deaths were reported, representing the highest burden since monitoring began in 1995. 2 Disruptions in the healthcare system, primarily triggered by the COVID-19 pandemic, led to reduced accessibility of healthcare services, shortages in the healthcare sector, and delays in screening and treatment, significantly contributing toward the recent rise in TB incidence.3–5 Despite advances in TB care, particularly with the development of new digital health technologies, there exists an urgent need to understand and address implementation challenges across the TB care cascade to reduce patient attrition.
The TB care cascade framework evaluates patient outcomes across sequential stages of health system engagement, providing a comprehensive approach to assess quality of care and identify intervention points. 6 The TB care cascade includes four distinct steps, initiated by a patient recognizing symptoms and seeking health care services or through passive or active case-finding 7 : (1) screening, which is the first interaction between the patient and a health care provider and involves assessing symptoms using a set of screening questions; (2) testing, using a WHO recommended diagnostic tool to detect active TB, including testing of sputum samples with GeneXpert MTP/RIF assay, sputum microscopy, or a chest x-ray; (3) diagnosis, involving the evaluation of test results by a health care provider, confirmation of active TB, and decision to initiate treatment; and (4) treatment, namely the initiation of a treatment regimen for active TB. 6 In high TB-burden countries, substantial losses occur at each cascade stage, with studies showing significant patient attrition across the care pathway from initial screening and diagnosis through to treatment completion and posttreatment monitoring.6,8 These losses reflect complex systemic challenges including inadequate health infrastructure, geographic barriers to care access, prolonged treatment regimens, and insufficient resources for patient monitoring and support. As such, examining TB care across multiple stages of the cascade can highlight both barriers and opportunities for the development of effective and comprehensive TB care strategies.
Digital technologies, such as mHealth interventions and integrated data management tools, are increasingly being developed and implemented to support TB care for individuals in high TB-burden countries in initiating and proceeding through the TB care cascade.9–11 The World Health Organization's Digital Health for TB strategy recognizes the potential of information and communication technologies to transform TB prevention, diagnosis, treatment, and care. 12 These technologies encompass a diverse range of interventions, from comprehensive health information systems and electronic medical records to targeted mobile health applications and digital adherence technologies.13–15 However, though numerous digital tools have been deployed to improve TB case notification and management, many are concentrated on treatment adherence rather than upstream case detection.10,14,16 Given that patient attrition occurs at multiple stages across the TB care cascade, there is a need to further examine the implementation and use of TB technologies to support linkages of patient data across the care cascade. Further, the excitement surrounding groundbreaking digital health technologies must be balanced with a pragmatic understanding of the challenges associated with their implementation.
Indeed, despite their potential, digital technologies often face significant barriers to implementation and adoption. Shared challenges include insufficient infrastructure, lack of digital literacy among healthcare workers, and fragmented health information systems.11,17,18 In Ethiopia, for example, healthcare facilities frequently lack the necessary equipment and trained personnel to implement digital health interventions effectively. 19 Cost-effectiveness is another key concern; while some technologies reduce costs by minimizing the need for in-person supervision, upfront investment and maintenance costs can be prohibitive. 20 Enabling factors include strong stakeholder engagement, policy support, and adaptive implementation strategies. Linked digital technologies are particularly effective as they can be used across different phases of a disease care cascade to improve comprehensive care and patient outcomes. 21 Countries that have successfully integrated digital health solutions, such as India's use of Ni-kshay, highlight the importance of embedding technologies within existing healthcare structures and ensuring alignment with provider and patient needs.16,22–24 Moreover, digital interventions may be most effective when tailored to specific health system constraints and sociocultural contexts.25,26
Although much of the current research focuses on the technology supporting treatment adherence rather than the earlier stages of the TB care cascade, such as case finding and diagnosis, few studies examine their implementation in real-world settings, with limited understanding of how contextual factors shape outcomes.27,28 Yet, given the proliferation of these tools, there exists a need to synthesize what technologies are being used and where, as well as build a comprehensive understanding of their respective functionality and implementation considerations. The aim of this review was to examine technological interventions used to support at least two stages of the TB care cascade in high TB-burden countries, with a focus on backbone and add-in digital technologies that are relevant for programmatic workflow processes. The specific objectives were: (1) to systematically identify literature on digital technologies for supporting the TB care cascade in high TB-burden countries; and (2) to describe the facilitators and barriers to technology implementation.
Methods
Study context
This study is situated within a broader project conducted by a multidisciplinary team at the University of Waterloo, Canada that is designing and piloting a technological intervention to support TB care in Nigeria and other high TB-burden countries. The review team has collective expertise in public health, global health, TB, implementation science, and knowledge syntheses. This review was designed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews guidelines (see S1 Appendix). The protocol was registered on 18 July 2023, in PROSPERO (Registration #: CRD42023443377).
Search strategy
The overarching search strategy involved a systematic search of the published (peer-reviewed) literature to identify technological interventions being used, followed by a focused unpublished (“grey”) literature search to provide detailed information about each identified intervention. Additionally, to add rigor and to validate the search, the review team consulted with experts in this field to generate a preliminary list of TB technologies in use within high TB-burden countries.
On 2 June 2023, the following four databases were searched for peer-reviewed primary research articles published from the year 2000 to present: Web of Science™, Scopus®, PubMed®, and CINAHL (via EBSCOHost®). No restrictions to language or type of record were applied, except for Scopus® given the size of the database (e.g., search will be restricted to English or French); and CINAHL® (e.g., search will be limited to academic journals). A search hedge consisting of terms related to TB, technology, and implementation was developed and refined in consultation with a University of Waterloo health sciences research librarian (Table 1). The search hedge was initially developed for PubMed®, then adapted for each database (see S2 Appendix for complete search hedges). Additionally, the following journals were hand-searched to explore the sensitivity of the search hedge: The Lancet Global Health, The Journal of Global Health, The Lancet Infectious Diseases, The Journal of Clinical Tuberculosis and Other Mycobacterial Diseases, The International Journal of Tuberculosis and Lung Disease, SAGE Digital Health, GALE Telemedicine and e-Health and Health Policy and Technology. All citations from database searches were exported in .ris format and uploaded into COVIDENCE™ web-based software for automatic de-duplication and eligibility screening.
Sample search hedge and constituent terms developed for the PubMed® database and subsequently adapted to other databases.

An updated database search was conducted on 7 February 2025, to retrieve any additional, relevant literature published within the calendar year prior to publication, from 3 June 2023, through to and including 7 February 2025.
In addition to published literature, a targeted unpublished (“grey”) literature search was conducted to identify relevant documents (e.g., reports, policy briefs, working papers) available in the public domain. This search focused on geographic locations where the review team had a priori knowledge of TB technologies being implemented, as well as specific technologies identified in the published literature, in order to retrieve additional implementation information if available. The search also aimed to fill gaps in the database searches by identifying technologies not reported in published literature. Searches used a combination of TB, technology, and implementation terms within country- or region-specific databases (e.g., African Journals OnLine, AJOL); websites for TB-specific organizations (StopTB, The Union, TB Alliance); and within Google Incognito (using an abbreviated search hedge and focused on specific technologies identified through published literature). Unpublished literature searches and retrieved records were tracked and organized in an Excel® spreadsheet (see S3 Appendix and S4 Appendix).
Article selection
To be included, retrieved records from the database searches had to be primary research articles that reported on user experience and/or implementation of a technology used to support at least two points of the TB care cascade for patients with active TB in a high TB-burden country (Table 2). Technologies that supported multiple points of the TB care cascade were included to identify technologies that promoted sustained continuity of care for TB and addressed multiple care cascade outcomes. 29 High TB-burden countries were determined according to the WHO 2021 global list (see S5 Appendix). 30
Specific inclusion and exclusion eligibility criteria used for article selection and screening.
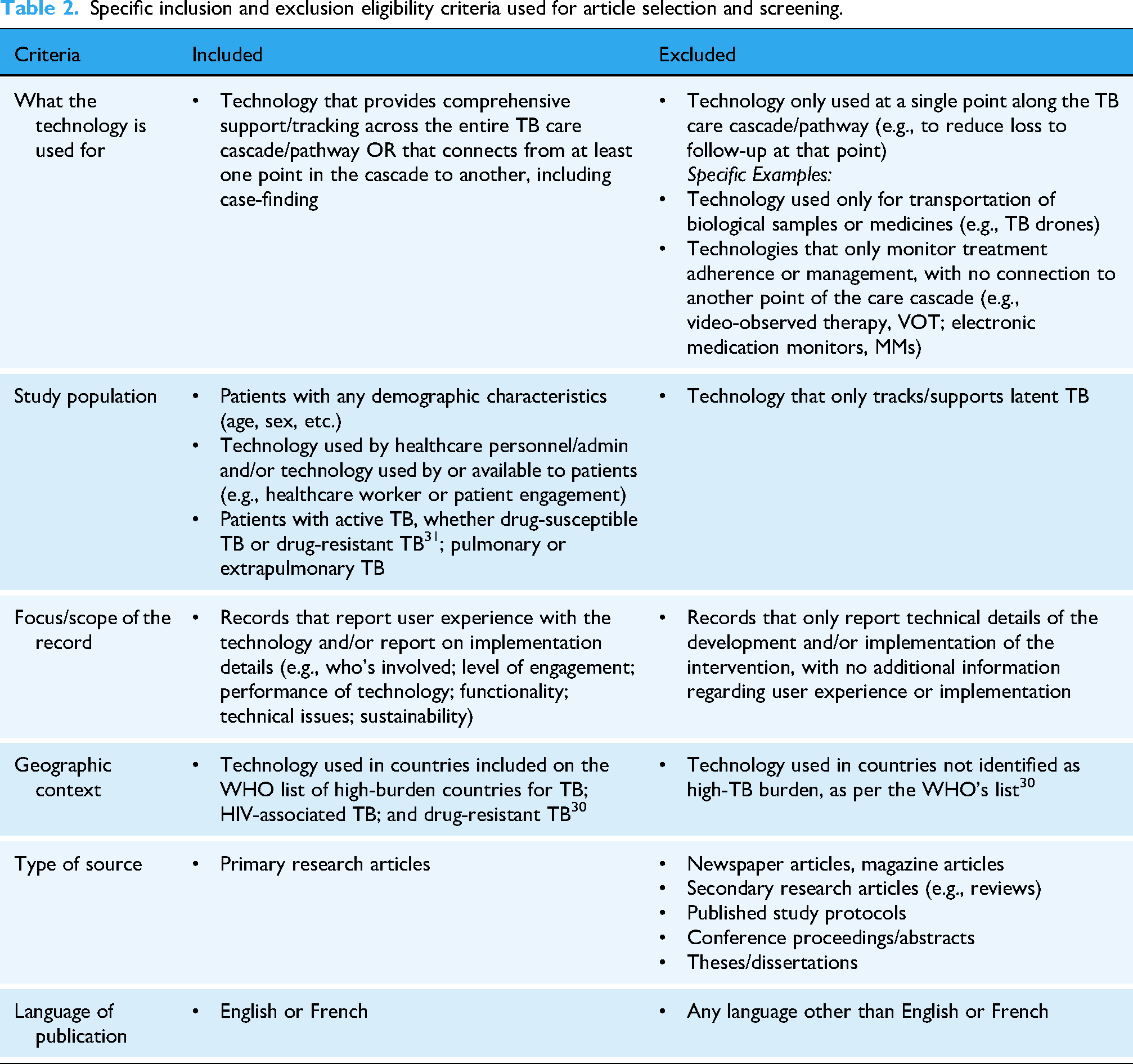
Two independent reviewers (LJB and MB) conducted Level 1 (title/abstract) and Level 2 (full text) screening according to eligibility criteria using COVIDENCE™. At the full-text stage, to meet the criterion regarding implementation, a published article needed to report at least two sentences of implementation information in the results or discussion section. Conflicts between reviewers with respect to inclusion/exclusion at both stages was discussed and resolved by consensus. A Cohen's Kappa coefficient of 0.54 and 0.71 for Levels 1 and 2 screening indicated moderate and substantial agreement, respectively (proportionate agreement of 96.7% and 85.9%). Records meeting all inclusion criteria at the full-text stage proceeded to data extraction and analysis.
Data extraction and analysis
Data from published articles were extracted by two independent reviewers (LJB and MB) into a piloted data extraction form in COVIDENCE™ (see S4 Appendix). Data extraction domains included citation details; basic information about the record (e.g., methodology, research question/objectives); and detailed information about the technology (e.g., functionality, utility) and its implementation (e.g., scale, duration of use, strengths/limitations), as reported in the results or discussion sections of included sources. An adapted form was used to retrieve implementation information from unpublished literature sources.
Descriptive statistics were calculated to quantify the extent and range of the literature (e.g., geographically, methodologically, temporally). A content analysis was conducted by one reviewer and thoroughly examined by two additional team members to identify and characterize the facilitators and barriers to effective technology implementation. This analysis generated three broad domains and six mechanisms that supported technology implementation across the included literature. NVivo Release 1.7.1® software was used for organization and retrieval of codes and coded excerpts.
Results
Extent and range of the literature
A total of 4387 records were retrieved from database searches, with a final total of 2475 records after automatic de-duplication. Following two-stage screening, 40 published articles met the eligibility criteria and were included in the review. No additional articles were retrieved from the hand search. Eleven unpublished records were retrieved and integrated into analyses (see Figure 1; see S3 Appendix for a list of included unpublished records).
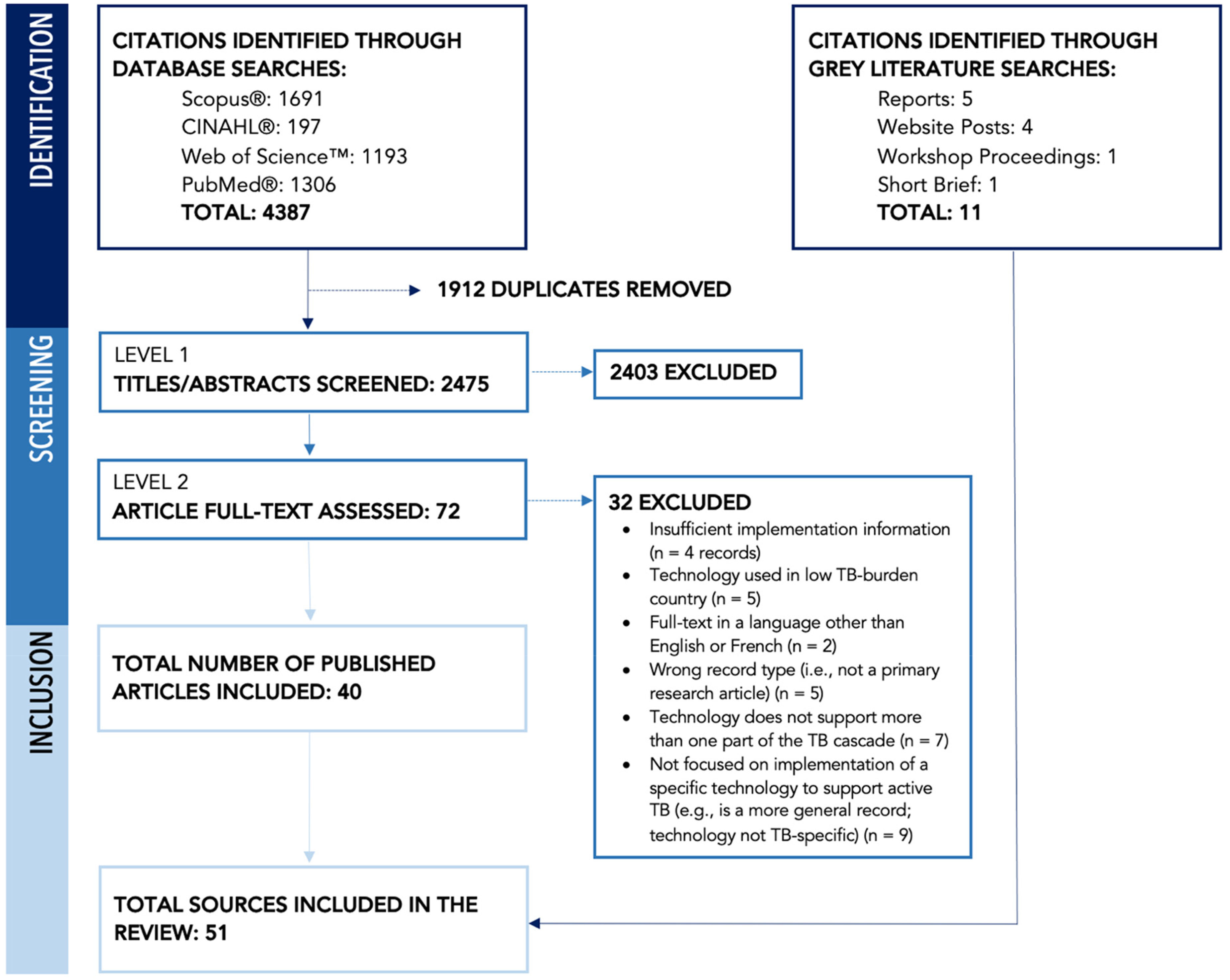
PRISMA flow diagram, indicating retrieval and screening of published and grey literature.
The countries with the most published literature meeting the study criteria were Uganda (n = 7; many among the same authorship group) and India (n = 6). A number of included published and unpublished sources were global in scope or focused on implementation of a specific technology across multiple country contexts (n = 5; 9.8%). The remainder of included literature focused on a specific technology in a single country context and taken together, reflected a broad geographic range across high TB-burden countries (see Table 3). While this review focused on high TB-burden countries, we noted in the screening process that there were very few eligible papers reporting technologies used in low TB-prevalence countries.32,33 The majority of published literature used a mixed methods (n = 16 articles; 40.0%) or quantitative study design (n = 12; 30.0%) to examine implementation outcomes and experiences.
Descriptive characteristics of included literature (published and grey) and technologies (n = 51).

Source focuses on three or more countries.
Denominator of 51 included sources.
Published literature only (n = 40).
Descriptive study; needs assessment.
Denominator of 20 technologies, including both backbone and “add-in.” Within the results, the total number of technologies described is 18 technologies, as the three identified EMR systems share many characteristics with one another.
Though there are three EMR technologies included in the list, there were shared characteristics across the three identified systems, and therefore they were combined into one category within the results.
Includes two specific technologies, Open Data Kit (ODK) and Mobile App for Tuberculosis Screening (MAT), that were discussed in more detail across multiple included sources, and then a catch-all “Other mHealth applications or phone-based technologies” group considered as one add-in technology in the n = 20 total technologies count because they were each only discussed in one included source.
Includes 1 specific technology, GxAlert, that was discussed in detail across multiple included sources, and then a catch-all ‘Other Tools for SMS Notifications’ group considered as one add-in technology in the n = 20 total technologies.
Values do not sum to n = 18 (100%) as some technologies were implemented at multiple scales, with different implementation strategies, so are captured in more than one category.
Profiling digital technologies in use across high TB-burden countries
Across the published and grey literature, 18 technologies that supported at least two or more stages of the TB care cascade in high TB-burden countries were identified (see Table 4). These technologies were categorized as providing comprehensive patient tracking and information management across the entire TB care cascade, and with the ability to have multiple other platforms or tools integrated into them (e.g., “backbone” technologies) (n = 10); or as tools that could be integrated into a backbone to support two or more stages of the TB care cascade (e.g., “add-in” technologies) (n = 8). Two additional technologies (SITB in Indonesia; NODIS in Myanmar) were identified through expert consultations; however, as there was limited evaluation or implementation information on these technologies available in English, these technologies were not formally considered part of the review (see S6 Appendix for additional technologies identified but with insufficient implementation information available to meet this study's eligibility criteria).
Backbone and add-in technologies identified in the review.

Citations of backbone and add-in technologies are referring to shared insights across sources for each technology. See S3 Appendix for more details.
Source received through expert consultations and not available in the public domain.
Profile of backbone technologies
In this review, 10 backbone technologies were reported (see Table 4). Backbone technologies often included health information systems implemented at a national or multinational level (Ni-kshay; e-TB Manager; TIER.net; SISTB; DHIS2; ITIS; TIBU). In comparison to add-in technologies, backbone technologies were more commonly integrated into countries’ National TB Programs (NTPs). These backbone technologies facilitated the flow of information across different levels of the health care system, and supported patients through various phases of the TB care cascade (TIER.net; SISTB). Some of these technologies (e.g., e-TB Manager; EMRs; DHIS2) were widely used across numerous high-TB burden countries and leveraged for comprehensive case management and tracking across the TB care cascade. By collecting, reporting, and tracking patient data, backbone technologies were also frequently used for TB surveillance (TIER.net; SISTB; TBIMS; EMR; DHIS2; ETL). By aggregating and improving access to data, backbone technologies were further used to generate data visualization outputs (e.g., indicators, charts, tables) and improve access to quality data for decision-making (SISTB; ITIS; ETL). Some technologies also sent information and notifications to both patients and providers (Ni-kshay; ETL; TIBU). For instance, while Ni-kshay is an integrated case management and reporting system that can broadly be used by both private and public practitioners, both TIBU and ETL can be used to send SMS messages to streamline communication. More specifically, TIBU not only sends reminders to patients but can be also used to disseminate health education materials to train healthcare workers and patients, which can further facilitate comprehensive TB management and care.
Profile of add-in technologies
Aligned with backbone technology functionality, “add-in” technologies were also used for data collection and TB screening processes (ODK; Other Digital Data Collection Tools; Other Tools for SMS Notifications; Other mHealth Applications or Phone-Based Technologies; MATS). For instance, patient screening and referrals are conducted using the MATS mobile screening app, while updating diagnostic test outcomes is conducted via the web portal. Since the launch of the MATS add-in program in Nigeria, there was a noticeable increase in the efficiency of referrals between facilities and community-based entities screening for TB. Further, some technologies allowed data to be collected and uploaded in real time to facilitate active case finding (Other Digital Data Collection Tools; MATS; ODK). For instance, ODK supported data collection and aggregate reporting with high data quality. However, with the exception of the MATs screening tool, all other add-in technologies reported in the literature were most frequently used for TB testing, diagnosis, and treatment. Some technologies offered the ability to track patient data and testing status. For instance, one technology used in Uganda enabled digital fingerprinting to identify and track patients between their homes and clinics, which could be used for contact tracing (Other Tools for SMS Notifications). Further, GxAlert, used in Mozambique and Uganda, relies on cloud-based technology to transmit test results for both users (e.g., lab technicians, nurses, TB personnel) and patients.
Domains underpinning implementation facilitators and barriers
To highlight facilitators and barriers shaping the implementation of both backbone and add-in technologies, three main domains that impact technology implementation were identified: (1) Interoperability and Integration; (2) Digital Infrastructure; and (3) User Experience. Examples supporting these findings were drawn from the identified backbone and add-in technologies to examine the different factors that impact the implementation of digital technologies aimed at addressing the TB care cascade (see Figure 2 and Table 5).

Summary of three main domains and six corresponding mechanisms used to guide the analysis of implementation factors and considerations.
Key considerations related to identified domains and mechanisms across included digital technologies.

Implementation barriers and facilitators of various digital tools are further shaped by the intended end user of the technology (e.g., TB program managers, healthcare providers, data managers in health systems).
Interoperability and integration: with National TB Programs
Interoperability and integration with NTPs was frequently highlighted as an important mechanism for the implementation of digital health technologies to address different phases of the TB care cascade. Backbone technologies were more commonly integrated into countries’ NTPs, with 8 of the 10 identified technologies (80%) reported to be implemented at the national scale. The use and implementation of these backbone technologies was also typically ongoing. Generally, collaboration with or adoption of a technology by NTPs facilitated effective implementation and sustained use of technologies. For example, the ITIS (Philippines), TIBU (Kenya), and Ni-kshay (India) technologies were considered “in house” programs that exist under the NTPs of these specific countries. However, it was noted that technology integration within an NTP may also act as a barrier in some contexts, as NTP priorities and limited budgets may make training and implementation challenging (e-TB Manager). By comparison, add-in technologies were not as commonly integrated into NTPs. However, e-Chasqui, used primarily in Peru, was identified as an add-in technology that was implemented by the NTP, and well-integrated into the broader public health structure. Similarly, GxAlert, used in Uganda and Mozambique, was integrated by NTPs, and installed on Xpert testing machines.
Interoperability and integration: with other digital technologies
In addition to integration with NTPs, the interoperability of some backbone technologies with other existing platforms shaped their implementation and use. For instance, the integration of multiple backbone technologies with the DHIS2 system facilitated improved decision-making and data accuracy, as well as effective and timely reporting to country NTPs (EMR; ETL). The DHIS2 system, which is primarily used across Africa, was interoperable with other digital tools like e-TB Manager, which is a backbone system used across multiple countries. In turn, e-TB Manager enabled real-time access to patient information and facilitated information sharing across providers. Similarly, TIBU, used primarily in Kenya, allowed for immediate feedback to facilities regarding case holding and other patient-monitoring parameters. By linking to the Laboratory Management Information System and the Ministry of Health District Health Information System, TIBU facilitated efficient flow of data among clinicians, policy makers, and patients. In addition, the integration of the TB and HIV EMR systems supported comprehensive care for both of these comorbidities and improved decision making among health providers (EMR). However, integration between backbone technologies and other platforms was complicated when these processes were time consuming, required sustained multistakeholder engagement or collaboration, or when data transfers between technologies were complex (Ni-kshay; TBIMS; EMR).
Though not as commonly reported on as backbone technologies, some add-in technologies relied on integration with other platforms. Of note, some of the add-in tools that integrated into backbone technologies tended to be short-term innovations (ODK; Other Digital Data Collection Tools), which meant they were implemented either in pilot studies or for a given study period. However, some of these technologies aligned with existing initiatives and were also considered to be effective in supporting efficient reporting and communication (e-Chasqui; Other mHealth Applications for Phone-Based Technologies; MATS). For instance, e-Chasqui, used in Peru, was a module within the EMR backbone technology and was used for communicating test results between the National Reference Laboratory, district laboratories, and health centers. In addition, rollout of the MATS technology, used in Nigeria, was supported by the Global Fund under a TB Public Private Mix grant, and was designed to align with the national TB algorithm to provide easy screening and diagnosis of TB cases by private health care providers. However, integration challenges were also highlighted among add-in technologies, as the GxAlert technology experienced some initial challenges with interoperability with the GeneXpert testing device, and lacked integration with other diagnostic platforms.
Digital infrastructure: technology features
The tools and features offered by different technologies also influenced the implementation of digital technologies and facilitated ease of use (SISTB; DHIS2). For instance, DHIS2 had a widely accessible reporting portal, while technologies like TIBU, EMR, and ITIS were shown to have user friendly interfaces that promoted broad usage across providers. Further, SISTB, a technology used in Brazil, had three registration tools that integrated into a single source, which was shown to be effective in improving the registration of patient data. TIER.net and TIBU both reduced the dependency on and replaced the use of paper-based registers, effectively generating reports and substantially reducing workloads. Similarly, DHIS2 included flexible data entry tools that could be customized to replicate paper forms, further facilitating data validation and data quality. In addition, some technologies collected various data that contributed to more efficient tracking and reporting (DHIS2; TIBU; ETL). For instance, TIBU captured patient-level data that included demographic characteristics, laboratory results, and treatment outcomes data. Finally, strong data security was also noted as a key facilitator for implementation among backbone technologies. For instance, ITIS, used in the Philippines, provided a centralized and secured data storage, which included data recovery and a failsafe/net secure policy. In addition, data integrity for TIBU was supported through limiting data access to certain users and data encryption.
Aligned with reporting on backbone systems, add-in technologies that were simple and easy to use also facilitated effective implementation (ODK; MATS). For instance, the GxAlert technology, used in Uganda and Mozambique, was able to generate reports and modify SMS alert messages, and also had the ability to adapt the user interface to a specific context (i.e., translate into a local language). In contrast, when challenges were identified with a tool's ability to support reporting and record keeping, there were often mixed experiences, with some users viewing these technologies as an inadequate replacement for in-person services (Other Tools for SMS Notifications; Other Digital Data Collection Tools). Further, when comparing the add-in technology to paper-based registries, security concerns were raised by patients and technology users for some add-in technologies (ODK; GxAlert). These security concerns and preferences for paper-based registries could be viewed as a barrier to technology adoption and implementation.
Digital infrastructure: technology infrastructure
Digital infrastructure and resources also had a direct impact on the implementation of backbone technologies. While ITIS had capacity for printable forms (including screening forms, treatment cards, laboratory forms, and TB registers), TIBU stored data in the cloud server, which allowed any lost data to be easily restored. TIBU also had a tablet for digital data entry, bundled with internet data and airtime, to work around infrastructure or connectivity challenges (DHIS2; TIBU). Further, provided as a service in the cloud and licensed under an open-source license, users could start using the DHIS2 system immediately and there was no need for them to install and maintain the software themselves (DHIS2). ITIS, used in the Philippines, could be used on the Android operating system and was also a hybrid software, with capacity for both online and offline usage depending on the internet connection. ITIS also provided tool-top guides and ease of access for laptops, desktops, tablets, and smart phones. Similarly, when ETL was implemented in Tanzania, the program procured laptops for district coordinators to enter facility-level data for all facilities. In facilities that lacked the hardware or infrastructure to enter data, all the data were compiled and shared with the district, where case-based data were then entered into ETL. However, one of the most commonly cited barriers to implementation of these backbone technologies were related to device connectivity (Ni-kshay; e-TB Manager; ITIS; ETL; MATS; TIBU). Similarly, the mobile devices used for TIBU needed to be transported to a location with connectivity, which delayed real-time data entry and sharing. In addition, some technologies were labeled as complicated or nonuser-friendly (Ni-kshay), while others required significant financial investment, which included both start-up and ongoing costs like power, connectivity, and IT support (EMR). For example, TIER.net required uniformity in computer equipment across districts in order for central IT support from higher levels of the health system.
Aligned with findings from backbone technologies, system resources and technological infrastructure also facilitated effective implementation of add-in technologies. For instance, similar to the ITIS backbone technology, MATS, used in Nigeria, was compatible with Android mobile devices for full functionality. Further, the MATS program provided real time information through a web interface, and all information could be captured and viewed on the application by the private providers and DOTS providers in referral facilities. Importantly, both internet accessibility and data quality were reported as key facilitators among multiple add-in technologies (Other Digital Data Collection Tools; MATS; e-Chasqui; ODK; GxAlert; Other Tools for SMS Notifications; Other mhealth Applications or Phone-Based Technologies). In some cases, network and connectivity issues exacerbated existing reporting challenges at different phases of the TB care cascade or created new issues with screening and reporting on add-in technologies. As an alternative to unreliable network infrastructure, implementers of the ODK technology highlighted the use of backup power sources and an offline format for the databases to address any network challenges. Similarly, data were reported real time on MATS and the application also had an offline mode to hold screened data until the device could connect to its linked network for transmission.
User experience: integration among users
Beyond the interoperability of backbone technologies with the NTP and existing digital health systems, the integration of these technologies among diverse stakeholders facilitated implementation. Indeed, technologies that were perceived as user-friendly, efficient, and reliable were shown to facilitate implementation (e-TB Manager; EMR; TIER.net; DHIS2). For instance, TIBU, used in Kenya, requires basic computer training for the use of the technology in the field. As a result of training and professional development opportunities, the use of TIBU boosted field staff morale and encouraged them to frequently update data by visiting peripheral health facilities. More frequent visits subsequently had positive impacts on peripheral healthcare worker performance and improved case holding of patients starting TB treatment. In addition, the incorporation of user-feedback contributed to effective implementation. In the Philippines, ITIS relied heavily on a “user-centered design,” with actors at various levels of the health system contributing to the iterative design of the technology. However, broad adoption of a backbone technology could be challenged when individual- and organization-level changes were required among users to support technology implementation. For example, the implementation of the TIER.net technology highlighted a need to prioritize “change management,” and an organizational culture that promoted the adoption of digital technologies over paper-based systems.
In alignment with backbone technologies, some add-in technologies integrated with users’ standard workflow and mirrored a familiar paper-based format to positively facilitate implementation. Familiarity of the technology used by the ODK technology was highlighted as beneficial, as it included tablets and mobile devices. In addition, user perceptions also influenced the implementation of add-in technologies. For instance, the e-Chasqui and the GxAlert technologies maintained high user satisfaction, while the ODK technology was further perceived by users as both time saving and easy to use. Users also perceived ODK to be more secure than paper-based systems, and a feasible alternative to paper-based systems of data collection and aggregate reporting. However, full integration of add-in technologies into existing workflow proved to be a challenge for some technologies. For instance, although the MATS add-in tool required an Android mobile device to be used for screening, not all health care providers used these devices, which constrained the adoption and coverage of this technology across health care facilities and users. Overall, ongoing evaluation of a technology's ability to integrate into existing workflows combined with training on a technology's use and benefits facilitated sustained adoption among users.
User experience: user training
One of the key factors associated with the implementation of digital health technologies to strengthen outcomes from the TB care cascade was comprehensive training opportunities for users. Training and computer literacy were frequently highlighted as necessary for implementation across backbone technologies (e-TB Manager; TBIMS; EMR; Ni-kshay; SISTB), with some technologies requiring significant data management skills and instruction for users (EMR). Training was not only necessary for introduction to certain technologies but was also important for prolonged systems management. To facilitate implementation, a stepwise implementation of the EMR technology allowed users to adapt to the digital technology from paper-based systems, while ongoing stakeholder engagement ensured sustainability. Further, hundreds of DOTS providers and data clerks from high-volume public and private facilities were trained and mentored to use the ETL technology. Similarly, the ITIS technology included an electronic manual, which facilitated ease of use for both online and offline versions of the technology. However, some user-related training challenges were reported as a barrier to technology implementation (TIBU; Ni-kshay). For example, there were challenges with handling the physical devices (tablets) in early stages of TIBU implementation, as some staff mishandled the tablets (sometimes exacerbated by bad road or weather conditions) when they were transported to the peripheral health facilities. While some add-in technologies also noted the importance of supervision, data mentoring, and partnerships between different stakeholders as facilitators for technology implementation (GxAlert; Other Digital Data Collection Tools), these factors were not emphasized when compared to the comprehensive training required for the backbone technologies.
Discussion
This review mapped three key domains and six mechanisms that directly impacted the implementation of digital health platforms aimed at addressing the TB care cascade in high TB-burden countries. Previous reviews have documented the digital TB technologies that are being used globally.13,28,33 This research builds from these existing syntheses to more closely examine the implementation of these technologies, including salient facilitators and barriers. We identified a total of 18 technologies that supported at least two stages of the TB care cascade, with 10 classified as backbone technologies and eight as add-in technologies. The three domains included Interoperability and Integration, Digital Infrastructure, and User Experience, and these encompass broader systems-level, technology-level, and individual user-level factors that collectively determine implementation success. This multilevel approach aligns with established frameworks for understanding implementation processes, where success depends on complex interactions between intervention characteristics, organizational contexts, and individual factors. 84
Generally, there was less information about add-in technologies when compared to backbone systems, which reflects the nature of add-in technologies as more commonly short-term innovations or pilot projects. Mechanisms related to integration with NTPs and user training had less information available for add-in technologies, representing an opportunity for further research and reflection. Importantly, some mechanisms could be linked to both facilitators and barriers. For instance, regarding integration among users and user trust in systems, some technologies experienced positive perceptions while others struggled to integrate into existing workflows, highlighting the importance of contextual factors in implementation outcomes.
Implementation domains
The Interoperability and Integration domain emerged as fundamental to technology success, consistent with findings from other digital health implementation studies.85–87 Backbone technologies demonstrated higher rates of integration with NTPs, suggesting that comprehensive integration strategies are essential for sustained implementation. However, integration between backbone technologies and other platforms became complex when processes were time-consuming, required multistakeholder engagement, or involved complex data transfers between technologies. Country-specific experiences illustrate these challenges. India's Ni-kshay platform integrates with GeneXpert and TruNat systems, but challenges remain in leveraging data from multiple sources into a central repository.39,51 Similarly, Nigeria operates dual platforms with e-TB Manager and DHIS2, requiring data extraction and import between systems.41,51,88 Ethiopia adopted DHIS2 for TB notification but maintains manual registers at facilities, creating integration bottlenecks.51,89 These findings reflect broader challenges documented in health systems integration efforts, where technical complexity sometimes intersects with organizational and policy barriers.18,90,91
The Digital Infrastructure domain highlighted critical technology-related factors. Technologies perceived as user-friendly, efficient, and reliable facilitated implementation, while connectivity and network issues represented the most commonly cited barriers to backbone technology implementation. Infrastructure challenges are widespread across high-burden countries. Ethiopia's health extension workers reported lack of properly functioning tablets, connectivity, and power supply as major barriers, with poor network coverage and inconsistent airtime recharging among primary infrastructure challenges. 92 India's Ni-kshay platform faces difficulties scaling real-time data collection from GeneXperts and TrueNAT devices, with laboratory information systems limited to reference laboratories. 51 Multiple countries including Bangladesh, Ethiopia, Ghana, Malawi, Mozambique, and Nigeria lack adequate training, analytical skills, and resources to effectively utilize TB diagnostic connectivity platforms.93,94 These infrastructure challenges are particularly acute in low-resource settings where digital health interventions for TB are most needed, 95 underscoring the fundamental importance of technological infrastructure as foundational before more sophisticated interventions can even be attempted.
User Experience factors proved equally critical, with successful technologies demonstrating effective integration among diverse stakeholders and incorporation of user feedback. Training and computer literacy requirements were frequently emphasized for backbone technologies, which often demanded significant data management skills and comprehensive user instruction. Training gaps are evident across high-burden countries. Ethiopia's health extension workers reported poor quality of training, follow-up, and supervision as barriers hindering electronic community health information system use, with parallel recording using manual and electronic systems creating significant burden. 92 Studies across South Africa, Ethiopia and Nigeria highlight that digital literacy appears to be the most important factor for enhancing system use and user satisfaction, with specific user training identified as essential.96,97 This emphasis on user-centered approaches reflects growing recognition that healthcare providers’ attitudes and capacity significantly influence the success of digital health interventions. 17
Critical gaps and implementation challenges
An important finding of this review was the lack of comprehensive technologies that track and support patients across the entire TB care cascade. This finding represents a major challenge for achieving seamless patient care and optimal outcomes, as most technologies addressed specific cascade components rather than providing integrated support across the full pathway from screening through to posttreatment monitoring. This fragmentation mirrors broader challenges in TB care delivery, where different stages of the cascade are often managed by different systems and providers, similar to other TB care cascade analyses and represents a critical area for future technology development.8,98–100
Our review also highlighted a concerning gap in implementation documentation within published peer-reviewed literature. Generally, TB digital technologies are behind in overall discussions on efficiencies in data management and changes in healthcare management, especially as AI is increasingly used in healthcare contexts. Much of the available literature focused on outcomes rather than implementation processes, creating what might be characterized as an implementation “black box.” In the race to demonstrate outcomes of interventions, there is a pressing need to understand how and why certain outcomes are produced. This knowledge gap significantly hampers efforts to understand how successful outcomes are achieved and limits the ability to replicate successes in new contexts. This challenge extends beyond TB technology to other complex global health interventions, where understanding what implementation actually looks like—how it happens, who is involved, and what contextual factors matter—remains poorly documented.75,101
The dynamic nature of many digital health platforms presents additional implementation considerations. Technologies like Ni-kshay undergo frequent updates, requiring continuous adaptation and user training. This evolution challenges traditional implementation frameworks that assume relatively stable interventions and highlights the need for adaptive implementation strategies that can accommodate technological change. The tendency of government programs to focus on showcasing best practices while minimizing discussion of implementation challenges and barriers further compounds this problem. Indeed, understanding failure modes and implementation difficulties is crucial for future technology development and deployment strategies. Overall, these findings emphasize the importance of moving beyond an exclusive focus on health system capacity to also consider user experiences, integration, and training in recognition that a digital technology is only as effective as its users.
Limitations and future research directions
There are several limitations in interpreting these findings. Technologies supporting only one component of the TB care cascade were excluded from analysis, potentially missing valuable implementation insights or contextual understanding. This review also focused on TB-specific technologies, rather than disease surveillance or information systems at large. Further, while AI tools may offer potential for streamlining work processes and advancements in data management, these tools were not part of the current discussion on digital technologies. Our focus on high TB-burden countries, while appropriate for the research question, may limit generalizability to other settings. Additionally, the exclusion of latent TB technologies may have overlooked relevant implementation lessons. Future research should prioritize implementation science approaches that systematically document implementation processes, barriers, and facilitators alongside outcome evaluations. It would be beneficial for future research to explore opportunities to improve workflow for programs and providers, which could include geo-spatial analysis to improve planning and referral, or quality assurance tools for patient feedback. There is a critical need to move beyond outcome-focused research to understand the “how” and “why” of implementation success and failure.
Conclusion
This systematic scoping review provides a comprehensive analysis of implementation factors influencing digital health technologies that support the TB care cascade in high TB-burden countries. Moreover, this review synthesizes implementation considerations in integrating such digital technologies to avoid patient attrition and improve continuity of care. The identification of three key domains—Interoperability and Integration, Digital Infrastructure, and User Experience—with six underlying mechanisms offers a framework for understanding and improving implementation success. Key findings demonstrate that successful implementation requires comprehensive attention to systems-level integration, robust technological infrastructure, and careful consideration of user experiences and training needs. Backbone technologies show greater sustainability through deeper institutional integration but require substantial upfront investments, while add-in technologies offer flexibility and innovation opportunities but face integration and sustainability challenges.
The significant gap in comprehensive technologies supporting the entire TB care pathway represents a critical opportunity for future development. The documented implementation “black box” in current literature underscores the urgent need for implementation science approaches that systematically document and analyze implementation processes alongside outcome evaluations. For practitioners and policymakers, these findings emphasize the importance of holistic implementation planning that addresses technical, organizational, and user factors simultaneously. Investment in user training, digital infrastructure, and systematic integration processes appears essential for sustainable implementation.
Future research should prioritize mixed-methods implementation studies that document both barriers and facilitators, develop adaptive implementation strategies for evolving technologies, and create comprehensive platforms supporting complete care pathways. Only through such systematic attention to implementation science can the global TB community realize the full potential of digital health technologies to strengthen care cascades and improve patient outcomes in high-burden settings. The transformation of TB care through digital technologies will ultimately depend not only on technological innovation but on our collective ability to implement these tools effectively within complex health systems serving the world's most vulnerable populations.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251410991 - Supplemental material for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review
Supplemental material, sj-docx-1-dhj-10.1177_20552076251410991 for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review by Laura Jane Brubacher, Charity Oga-Omenka, Bridget Beggs, Monica Bustos, Petra Heitkamp, Plinio P Morita and Warren Dodd in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251410991 - Supplemental material for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review
Supplemental material, sj-docx-2-dhj-10.1177_20552076251410991 for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review by Laura Jane Brubacher, Charity Oga-Omenka, Bridget Beggs, Monica Bustos, Petra Heitkamp, Plinio P Morita and Warren Dodd in DIGITAL HEALTH
Supplemental Material
sj-docx-3-dhj-10.1177_20552076251410991 - Supplemental material for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review
Supplemental material, sj-docx-3-dhj-10.1177_20552076251410991 for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review by Laura Jane Brubacher, Charity Oga-Omenka, Bridget Beggs, Monica Bustos, Petra Heitkamp, Plinio P Morita and Warren Dodd in DIGITAL HEALTH
Supplemental Material
sj-docx-4-dhj-10.1177_20552076251410991 - Supplemental material for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review
Supplemental material, sj-docx-4-dhj-10.1177_20552076251410991 for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review by Laura Jane Brubacher, Charity Oga-Omenka, Bridget Beggs, Monica Bustos, Petra Heitkamp, Plinio P Morita and Warren Dodd in DIGITAL HEALTH
Supplemental Material
sj-docx-5-dhj-10.1177_20552076251410991 - Supplemental material for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review
Supplemental material, sj-docx-5-dhj-10.1177_20552076251410991 for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review by Laura Jane Brubacher, Charity Oga-Omenka, Bridget Beggs, Monica Bustos, Petra Heitkamp, Plinio P Morita and Warren Dodd in DIGITAL HEALTH
Supplemental Material
sj-docx-6-dhj-10.1177_20552076251410991 - Supplemental material for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review
Supplemental material, sj-docx-6-dhj-10.1177_20552076251410991 for Profiling digital technologies used to support the tuberculosis care cascade and their implementation across high burden countries: A systematic scoping review by Laura Jane Brubacher, Charity Oga-Omenka, Bridget Beggs, Monica Bustos, Petra Heitkamp, Plinio P Morita and Warren Dodd in DIGITAL HEALTH
Footnotes
Contributorship
LJB: Conceptualization, methodology, formal analysis, investigation, data curation, and writing—original draft. CO-O: Conceptualization, methodology, writing—original draft, supervision, and funding acquisition. BB: Investigation, data curation, and writing—original draft. MB: Methodology, investigation, and writing—review & editing. PH: Conceptualization, methodology, and writing—review & editing. PPM: Conceptualization and writing—review & editing. WD: Conceptualization, methodology, formal analysis, investigation, writing—original draft, supervision, and project administration.
Funding
This study was supported by the Graham Seed Fund (University of Waterloo) and the Centre for Bioengineering and Biotechnology Seed Fund (University of Waterloo).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
