Abstract
Telemedicine is an innovative medical model that utilizes information technology, including communication, computing, and networking technologies. It can transcend geographical barriers, enhance medical efficiency, facilitate the sharing of medical resources, and foster the diversification of healthcare services. However, the advancement of telemedicine in China encounters specific legal risks, including those associated with professional qualifications and licensing of healthcare institutions and personnel, determination of medical liability, infringement of patient rights, and cross-border telemedicine practices. To promote the innovative development of telemedicine in China, targeted measures must be implemented in areas such as improving the legal framework for healthcare, strengthening industry regulation, enhancing patient rights protection, and bolstering technical safeguards and standardization.
Introduction
The rapid advancement of information technology has facilitated the emergence of telemedicine as an innovative healthcare model that is transforming conventional medical service delivery. Telemedicine employs communication, computer, and network technologies to deliver healthcare services remotely, enabling patients to receive professional medical diagnoses and recommendations regardless of geographical location. This approach not only overcomes geographical barriers posed by the inequitable distribution of medical resources, thereby enhancing access to quality healthcare for patients in remote areas, but also improves healthcare delivery efficiency and reduces treatment costs. 1
The COVID-19 pandemic since 2020 has driven a surge in demand for contactless healthcare, positioning telemedicine as a critical supplementary service. Data from China's National Health Commission indicates that by 2018, telemedicine services were available in 52.9% of public hospitals, with adoption exceeding 90% among tertiary hospitals. By 2022, China had established over 1700 internet hospitals, while more than 7700 Level II + hospitals offered telemedicine services, collectively delivering over 10 million consultations annually. By late 2023, internet hospitals nationwide surpassed 3000 facilities, with annual telemedicine consultations exceeding 120 million instances. While alleviating healthcare access challenges and optimizing resource allocation, this rapid expansion has exposed critical legal gaps and risks, including: diagnostic and treatment legitimacy, patient privacy protection, cross-border data flows, and multi-party liability frameworks. 2 These issues impede telemedicine's standardized development while jeopardizing patient rights and interests. Consequently, investigating China's telemedicine legal risks and corresponding preventive measures holds significant theoretical and practical value. Addressing these issues will enhance telemedicine's regulatory framework, establish robust legal safeguards for its sustainable development, optimize medical resource distribution, and ultimately improve healthcare delivery for patient populations. 3
This study systematically analyzes primary legal risks in China's telemedicine sector and proposes targeted preventive countermeasures. The study's significance manifests in three dimensions: theoretically, it establishes a telemedicine legal framework tailored to China's national context; practically, it delivers risk prevention guidelines for healthcare institutions, professionals, and regulatory bodies; and policy-wise, it offers reference points for legislative enhancements. Through examining legal risks across all operational stages of telemedicine, this research aims to advance its sustainable development and support the “Healthy China” strategy.
Literature review and theoretical analytical framework
Literature review
An American scholar defines telemedicine as “long-distance monitoring and medical knowledge sharing through remote communication,” emphasizing its technical foundations. The World Health Organization (WHO) focuses on service scope, defining telemedicine as encompassing all medical activities—including diagnosis, treatment, and consultation. In 2014, China's National Health Commission (formerly the National Health and Family Planning Commission) issued Opinions on Promoting Telemedicine Services in Medical Institutions, defining telemedicine as “medical activities where one institution utilizes communication, computer, and network technologies to solicit technical support from another institution for patient diagnosis and treatment”.4,5
Recent years have seen increased academic focus on legal issues surrounding China's rapidly developing telemedicine sector. Domestic scholarship exhibits a “three more, three less” pattern: greater emphasis on experience introduction than risk analysis; more policy interpretation than normative analysis; and abundant single-subject perspectives but limited interdisciplinary studies. Research on practice thresholds highlights regulatory constraints: Chinese law restricts physicians from practicing solely within registered institutions, creating significant barriers for telemedicine implementation. Permitted telemedicine models remain limited to two institutional forms, with private practice prohibited. 6 Medical liability attribution faces regulatory conflicts between Ministry of Health provisions and the Civil Code, compounded by multi-stakeholder involvement—including requesting institutions, consulting institutions, and third-party platforms—that obscures responsibility determination. 7 Patient rights protections raise concerns about telemedicine's vulnerability to informed consent violations and privacy breaches. 8 During remote consultations, incomplete illness disclosure by requesting institutions can compromise informed consent, while network transmission of electronic health records creates privacy disclosure risks. 9 Absence of uniform standards exacerbates qualification-related risks, while legislative disparities across jurisdictions constrain cross-border telemedicine development. 10
Theoretical analytical framework
This study develops an analytical framework integrating medical jurisprudence, tort liability law, and network security law. Through a medical jurisprudence lens, this study examines practitioner qualifications and clinical practice standards for telemedicine providers, ensuring compliance with healthcare access regulations and diagnostic protocols. 10 Applying tort liability principles, the research delineates stakeholder accountability in telemedicine processes to safeguard patient rights and interests. 11 Leveraging network security law, the study addresses protection of patient data confidentiality, integrity, and availability—mitigating risks of unauthorized disclosure, alteration, or destruction to establish a secure telemedicine infrastructure. 12 By synthesizing these theoretical frameworks, the study identifies systemic legal risks in telemedicine and proposes targeted mitigation strategies.
Materials and methods
Material source
This study draws upon three primary sources.
Legal and policy frameworks
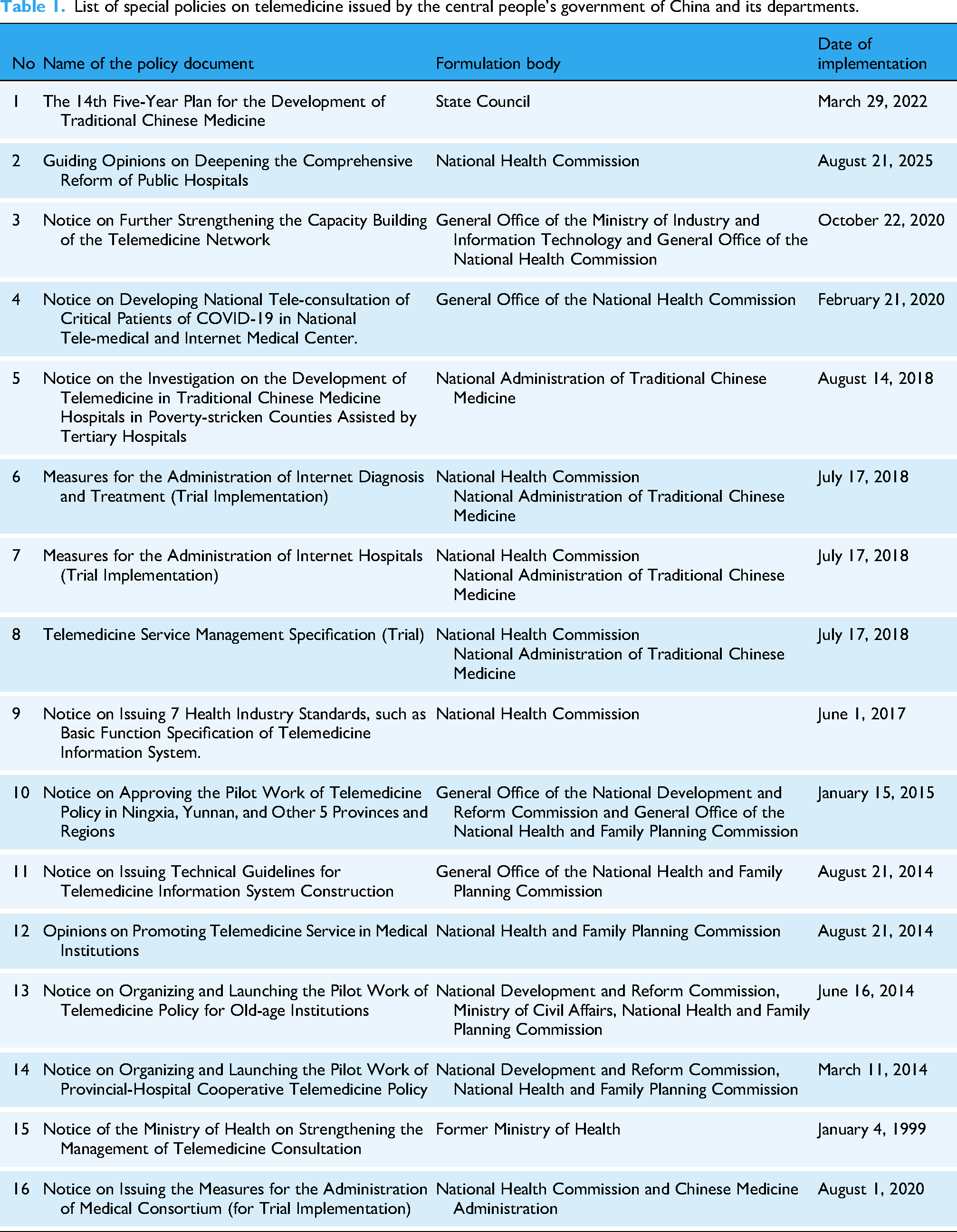
Key instruments include the Law on Doctors of the PRC, Law on the Promotion of Basic Medical and Health Services of the PRC, Regulations on the Administration of Medical Institutions, Measures for the Administration of Internet Diagnosis and Treatment (Trial), and Management Specifications for Telemedical Services (Trial) (see Table 1, Figure 1). These instruments establish the normative foundation for analyzing telemedicine regulation.

Statistics on the number of laws, regulations, and policy documents related to telemedicine issued by the central and local governments in China.
List of special policies on telemedicine issued by the central people's government of China and its departments.
Scholarly literature
Systematic searches across international (Web of Science, PubMed) and Chinese (CNKI, Wanfang Data) databases yielded extensive academic articles and research reports addressing telemedicine's legal dimensions. This corpus provides multifaceted theoretical perspectives on risk identification and mitigation strategies.
Empirical legal analysis
Examination of Chinese telemedicine-related judicial cases—including medical disputes and information disclosure proceedings (see Figure 2)—and online referee documents provides grounded insights into practical legal challenges.
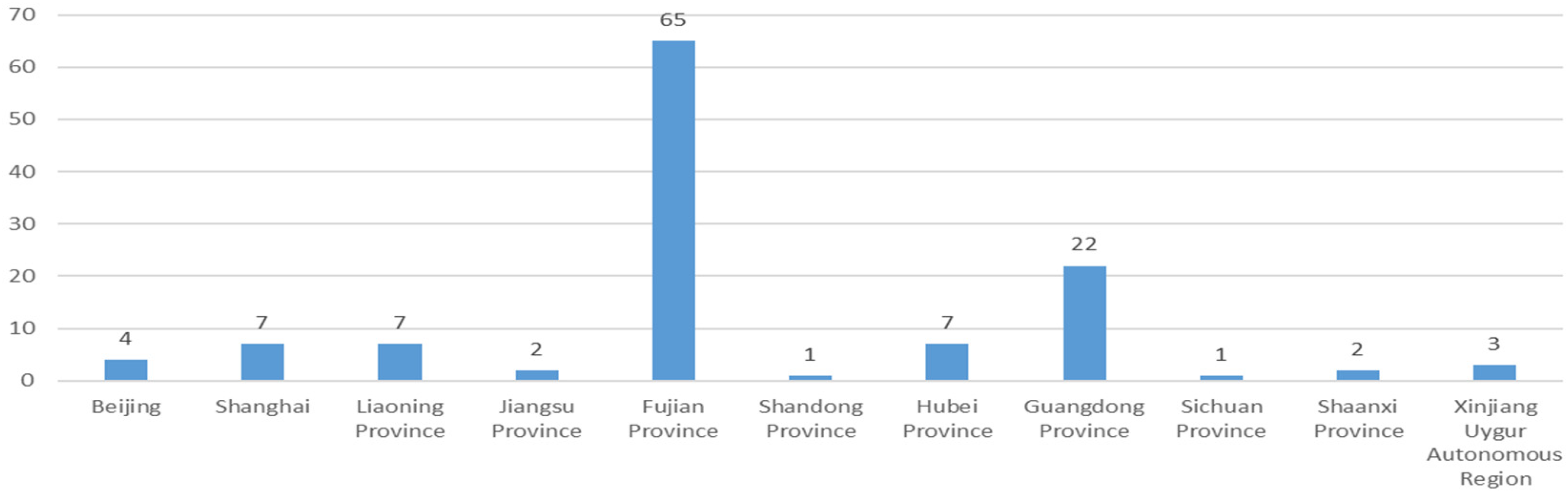
Statistics on telemedicine cases accepted by local courts in China.
Research methods
In this paper, various research methods are adopted. First, the research method of the literature, which systematically combs the legal and regulatory documents and the academic literature and comprehensively understands the current situation and research trends in the legal regulation of telemedicine in China and abroad, lays the foundation for follow-up research. Second, the case analysis summarized the specific forms of legal risk in telemedicine and the causes to present preventive measures to provide a practical basis. Finally, the comparative study method is used to compare the experiences and practices of different countries and regions in the legal regulation of telemedicine and to provide a reference to improve the legal system of telemedicine in China. We ensure comprehensive, in-depth, and scientific research through the comprehensive use of various research methods.
The rise of telemedicine and its innovation of the traditional medical model
Rise of the telemedicine background
Amidst socioeconomic advancement, the escalating demand for healthcare services has intensified. Concurrently, geographical disparities in medical resource allocation have grown more pronounced. Urban centers and developed regions concentrate high-quality medical infrastructure, whereas remote and primary care facilities face critical shortages of advanced equipment and specialized personnel, resulting in restricted healthcare access and treatment delays. 13 The rapid evolution of information technology presents a viable solution to these systemic challenges. Advances in internet infrastructure, communication networks, and computing systems have enabled telemedicine implementation. Recent integration of 5G technology has enhanced telemedicine's operational efficiency, service quality, and adoption rates across healthcare systems. 14 During public health emergencies like the COVID-19 pandemic, telemedicine experienced accelerated adoption due to its capacity to limit population aggregation and minimize cross-infection risks, thereby ensuring healthcare service continuity.15,16
China's telemedicine expansion has been significantly shaped by proactive policy leadership and strategic governmental initiatives (see Table 2). The term “telemedicine” first appeared in national policy through the 2015 State Council guidance on “Internet Plus” healthcare initiatives. Subsequently, the 2018 Administrative Measures for Internet Diagnosis and Treatment—jointly issued by three ministries—established regulatory frameworks for internet hospital accreditation. The 2022 14th Five-Year Plan further prioritized “expanding telemedicine coverage” as a national health objective. These cumulative policies have catalyzed telemedicine's rapid proliferation throughout China's healthcare landscape. 17
Key nodes of telemedicine development in China.

Telemedicine as an innovation of the traditional medical model
Telemedicine is a new medical model that is changing and innovating beyond the traditional medical model.
Breakthrough of the geographical restrictions
Conventional healthcare models require patients to physically access medical facilities, creating significant barriers for individuals in remote regions or with mobility impairments. Telemedicine leverages information technology to overcome geographical constraints. This enables virtual consultations between patients and specialists across locations, allowing individuals to receive expert diagnoses and treatment recommendations from home or local facilities without long-distance travel, thereby substantially enhancing healthcare accessibility. 18 For instance, patients in remote mountainous regions utilize telemedicine platforms to transmit clinical data to urban specialists, who then develop diagnostic and therapeutic protocols, ensuring timely and effective care delivery. 15
Improving the medical efficiency
Telemedicine enables efficient transmission and sharing of medical data while reducing patient treatment delays. Conventional healthcare models often involve significant time expenditure for registration, queuing, and examinations, whereas telemedicine streamlines processes through online scheduling and remote diagnostics. Concurrently, the telemedicine system provides clinicians with immediate access to patient records and diagnostic reports, enhancing diagnostic accuracy and efficiency. In emergency scenarios, telemedicine facilitates real-time multidisciplinary consultations, enabling specialists across locations to collaboratively diagnose patients, develop optimized treatment protocols, and significantly reduce critical time-to-treatment. 19
Promoting medical resource sharing
Telemedicine facilitates widespread sharing of specialized medical resources. Through telemedicine platforms, specialists from tertiary hospitals deliver care to patients in primary care facilities while concurrently training and mentoring primary care physicians to enhance service quality. For instance, tertiary hospitals have established telemedicine centers and collaborative networks with numerous primary care facilities. These centers routinely facilitate telemedicine consultations, case reviews, and academic seminars, enabling knowledge transfer of medical expertise and reducing regional disparities in healthcare standards. 18
Promoting medical services diversification
E-medicine has facilitated diverse medical service modalities, including internet hospitals, online consultations, and remote monitoring. Patients select appropriate service modalities based on their clinical requirements. Internet hospitals enhance patient accessibility through services like online follow-ups and e-prescribing. Remote monitoring devices enable real-time tracking of patients’ vital signs—such as blood pressure and blood glucose—optimizing chronic disease management. These diversified service modalities expand healthcare service availability and enhance patient experience.18,20
Legal risks associated with telemedicine
Telemedicine has emerged as an innovative healthcare modality with the proliferation of internet technologies. Across jurisdictions, including China and other nations, legal frameworks inherently exhibit lag and rigidity. Systematic analysis of telemedicine-associated legal risks is imperative to align with contemporary regulatory standards.
Qualification practice and access risk
Issues in medical institution practice qualification
Telemedicine service providers often exhibit inconsistent credentialing standards. Certain internet hospitals operate without requisite institutional licensing or exceed their authorized scope of practice. 21 This includes purported internet hospitals established by third-party platforms that deliver remote care without regulatory approval. Such operations contravene medical institution regulations while introducing significant patient safety risks. Furthermore, current regulations lack explicit provisions regarding the institutional qualification requirements for emerging telemedicine service models, creating regulatory lacunae. 22
Practice qualification of medical personnel and multi-point practice problem
Chinese laws and regulations explicitly restrict physicians to practice within registered medical institutions and geographic scopes following licensure. Telemedicine, however, requires physicians to serve patients across disparate locations, necessitating flexibility in practice location regulations. Despite existing provisions for multipoint practice, the filing and management mechanisms remain inadequate for telemedicine contexts. 23 Some physicians fail to file for multipoint practice when delivering telemedicine services, raising questions about the legality of their practice. Furthermore, credential verification for foreign or Hong Kong, Macao, and Taiwan (HMT) physicians in telemedicine is complicated by regulatory disparities across jurisdictions, creating loopholes in qualification screening that compromise service quality and patient safety. 24
Identification of medical liability risk
Ownership of multisubject medical liability is unclear
Telemedicine involves multiple stakeholders, including patients, inviting medical institutions, invited medical experts, and third-party platforms. Liability attribution becomes complex when medical disputes arise. The 2009 Notice on Strengthening Telemedical Consultation Management, issued by the former Ministry of Health, stipulates that institutions initiating consultations bear liability for resulting disputes. This provision contradicts the Civil Code's liability attribution framework for diagnostic and treatment activities. Under the Civil Code, invited experts bear liability for patient harm resulting from erroneous diagnostic or treatment opinions. Conversely, the Notice may exempt invited experts from liability, creating regulatory ambiguity in responsibility assignment. Furthermore, regulatory gaps persist regarding third-party platforms’ liability for technical failures during data transmission—such as distortion or loss of information—that compromise diagnostic integrity and treatment outcomes. 22
The fuzzy standard for determining liability for medical damage
Telemedicine diverges from traditional medical treatment in its diagnostic processes and operational environment, rendering existing medical malpractice liability standards inapplicable without modification. Technical variables—including device performance limitations and network transmission deficiencies—may compromise diagnostic accuracy in remote consultations. Current frameworks lack standardized criteria to distinguish between inaccuracies stemming from technological constraints versus medical negligence. Remote surgery necessitates distinct liability standards to evaluate: (1) surgical robot compliance with operational specifications, and (2) timely management of intraoperative emergencies. China's regulatory framework remains underdeveloped in this domain, consequently precipitating doctor-patient disputes over liability attribution following adverse medical events. 25
Risk of infringement of patient rights
Informed consent rights of patients violated
Informed consent in telemedicine encounters significant challenges. The technical complexity and specialized nature of telemedicine may impede patients’ comprehensive understanding of diagnostic procedures, treatment protocols, and associated risks. For instance, during diagnostic procedures utilizing advanced telemedicine devices, patients may lack comprehension of operational principles and precision metrics, while physicians may encounter limitations in fully explaining device functionality due to communication constraints or time restrictions. Moreover, telemedicine's multi-stakeholder framework and multi-stage processes create susceptibility to deviations or omissions during information transmission. Inadequacies in the referring institution's presentation of consulting experts’ credentials, consultation protocols, and procedural details may preclude patients from achieving fully informed consent. Furthermore, communication barriers inherent in telemedicine—compared to traditional in-person consultations—may delay patient notification and exclude them from real-time decision-making during treatment plan modifications, thereby violating informed consent rights.17,26
Violation of the patient's right to privacy
Telemedicine necessitates network transmission and storage of patients’ personal information and medical records, elevating risks to privacy rights. Inadequate cybersecurity measures expose patient data to threats including unauthorized access, malware infiltration, and resultant breaches. 27 Security vulnerabilities in telemedicine systems may permit unauthorized acquisition of sensitive data, including medical records and diagnostic reports. Concurrently, insufficient privacy protection awareness among healthcare personnel persists. Operational errors or protocol violations during telemedicine may precipitate unauthorized information disclosure. Moreover, the multi-stakeholder nature of telemedicine—with complex information flows between entities—lacks a comprehensive regulatory framework ensuring secure data storage and appropriate utilization by all parties. 28
Cross-border telemedicine risks
Obstacles caused by differences in legal and regulatory
Globalization has accelerated cross-border telemedicine adoption, yet significant jurisdictional fragmentation persists in medical laws and regulations. Practice qualification requirements vary substantially across jurisdictions, creating credential recognition barriers. Chinese physicians may encounter foreign non-recognition of their licensure when delivering telemedicine services internationally. Similarly, credential verification for foreign practitioners providing telemedicine services in China presents considerable administrative complexity. Medical liability determination faces further challenges due to divergent legal frameworks and judicial practices across jurisdictions. Jurisdictional complexity complicates applicable law selection in medical malpractice disputes. Disparities in liability attribution principles between China and other jurisdictions may produce inconsistent legal outcomes for patients. Moreover, inconsistencies in patient rights protections and cross-border data transfer regulations create significant regulatory impediments to global telemedicine expansion. 29
Lack of supervision and difficult quality control problems
The absence of unified regulatory standards and oversight mechanisms characterizes cross-border telemedicine. Jurisdictional variations in access protocols, operational requirements, and supervisory frameworks create regulatory fragmentation, marked by both gaps and redundancies. Consequently, regulatory inconsistencies enable participation by unaccredited providers and personnel, complicating quality assurance. The transnational nature of service delivery—spanning multiple jurisdictions and stakeholders—further exacerbates quality control challenges. Service quality depends on technological infrastructure (equipment reliability, network stability) and provider competence. Without standardized quality benchmarks or monitoring protocols, patients face significant challenges in evaluating service safety and efficacy.30,31
Countermeasures to legal risk prevention in telemedicine
Improving the legal system
Explicit legal status and responsibilities of telemedicine topics
Specialized telemedicine regulations have been proposed, with scholars urging China to establish comprehensive telemedicine legislation that explicitly defines the legal status, rights, and obligations of all telemedicine stakeholders, including medical institutions, personnel, and third-party platforms. Such legislation must stipulate qualification standards and approval procedures for medical institutions, alongside telemedicine service protocols, diagnostic norms, and liability parameters. For medical personnel, regulations should refine multipoint practice provisions, specifying practice jurisdictions, scope, filing requirements, and telemedicine-specific liability standards. Third-party platforms must have clearly defined service provider responsibilities, encompassing network security obligations, data transmission integrity guarantees, and liability allocation protocols for system failures. Clarifying the legal status and responsibilities of all stakeholders would mitigate legal disputes in telemedicine. 32
To develop a unified liability standard for medical damage
Specialized standards for identifying medical damage liability in telemedicine must be established, accounting for its unique operational characteristics. Determinations of medical failure must comprehensively evaluate the impact of equipment performance, network transmission quality, and other technical factors on diagnostic and treatment outcomes. Liability for medical damage resulting from equipment failure or network interruption should be assessed based on the contractual obligations of medical institutions and third-party platforms regarding equipment maintenance and network reliability. Compensation liability determinations must holistically consider patient harm severity, the degree of fault among all parties, and contributory factors. Concurrently, a formal telemedicine liability identification mechanism should be established, comprising specialized assessment institutions and qualified personnel to conduct impartial, scientifically rigorous evaluations of medical damage. This framework would provide an objective basis for determining responsibility and resolving disputes. 24
Strengthening industry supervision
Establishing strict access and exit mechanisms
Health administrative departments must implement stringent access controls for telemedicine institutions and service personnel. Medical institutions must rigorously evaluate their operational capacity, including scope of medical services, qualified technical personnel, and IT security infrastructure, before providing telemedicine services. 33 Personnel qualifications and practice credentials must be formally verified and documented. Third-party platforms require assessment of technical capabilities and security assurance protocols. A credit rating system for telemedicine providers and personnel must be established. Entities demonstrating regulatory non-compliance or substandard service quality should face escalating sanctions: formal warnings, financial penalties, practice suspension, and ultimately license revocation. Non-compliant entities must be blacklisted and permanently barred from re-entering the telemedicine market. Strict entry and exit mechanisms are essential to ensure telemedicine service quality and safety.25,32
Strengthen the supervision of the entire telemedicine process
Implementing information technology requires establishing a telemedicine supervision platform capable of real-time monitoring throughout the diagnostic and treatment process. This platform must incorporate monitoring capabilities for patient data security, medical conduct compliance, service quality metrics, and other critical aspects. For instance, real-time monitoring of patient data security during transmission and storage prevents information breaches; it supervises healthcare professionals’ adherence to clinical protocols and detects regulatory violations; and evaluates service quality through diagnostic accuracy and treatment efficacy metrics. 34 Concurrently, enhanced surveillance of cross-border telemedicine is necessary, requiring information-sharing frameworks and collaborative protocols with international regulatory counterparts to ensure compliance and standardization of transnational telemedicine services.22,30
Strengthening the protection of patients’ rights
Protecting the informed consent of patients
A standardized telemedicine informed consent template should be developed to comprehensively inform patients about service contents, procedural protocols, potential risks, and the qualifications of participating medical institutions and healthcare providers. Healthcare providers must deliver detailed, comprehensible explanations of consent documentation, verifying patient comprehension prior to service initiation. Supplementary educational resources, including specialized videos and materials, should be provided to elucidate complex telemedicine technologies and equipment. Information technology should facilitate bidirectional communication channels between patients and healthcare providers. Patients must have real-time access to healthcare providers for clarification during service delivery to ensure voluntary and informed participation.26,35
Ways to strengthen the protection of patient privacy
Patient information access is strictly limited to authorized personnel with legitimate work-related purposes, with comprehensive audit trails maintained for traceability. Mandatory privacy protection training programs are implemented for all medical personnel to enhance data security awareness, standardize operational protocols, and prevent inadvertent privacy breaches resulting from negligence. 27
A structured patient information security management system must be established, defining clear responsibilities for departments and personnel regarding data protection, implementing periodic security self-assessments, and ensuring prompt identification and remediation of vulnerabilities. All medical data containing protected health information must comply with regulatory requirements, with enhanced safeguards mandated for highly sensitive data, including HIV/AIDS status and mental health records.27,36
Implement rigorous oversight of third-party collaborators through enforceable confidentiality agreements that explicitly define their obligations and responsibilities regarding patient information utilization and protection. Periodic audits and evaluations of third-party information security protocols must be conducted to verify effective patient privacy safeguards. Any breach of agreement necessitates immediate termination of partnership and initiation of legal liability proceedings. 37
Furthermore, a comprehensive breach response protocol must be established to enable rapid mitigation of damages following patient privacy incidents. This protocol must encompass incident reporting procedures, immediate containment measures, patient notification frameworks, remediation strategies, and compensation mechanisms. Concurrently, patients must receive timely notification of incidents, transparent explanations of remedial actions and outcomes, and guaranteed rights to information access and decisional involvement. 37
Integrating these multifaceted approaches establishes a robust, multilayered privacy protection framework that comprehensively safeguards patients’ rights and interests within telemedicine ecosystems. 38
Strengthening the technical support and standard construction
Formulation and promotion of telemedicine technical standards
Establish unified telemedicine technical standards encompassing performance parameters, data transmission formats, interface specifications, and other critical equipment requirements. For instance, standards specifying image resolution and data acquisition precision for remote diagnostic equipment must be defined to ensure interoperability and seamless data exchange across systems. Concurrently, telemedicine service providers and equipment manufacturers must rigorously adhere to these standards to enhance technological standardization across the telemedicine sector. Unified technical standards mitigate medical risks stemming from technological incompatibility or equipment non-compliance, thereby providing a foundational guarantee for telemedicine quality and safety. 29
Data security and technical support system construction
A comprehensive telemedicine data security management system must be established to govern the entire lifecycle of patient data, including acquisition, transmission, storage, and utilization. A dedicated data security management department should be created to develop security protocols, contingency plans, and conduct regular evaluations, testing, and risk mitigation. Increased investment in R&D is essential to develop proprietary telemedicine security technologies—including data desensitization and access control—to enhance data protection. Additionally, a technical support team must be established to deliver prompt maintenance and operational support to telemedicine service providers, ensuring system stability. 17
Strengthening personnel training and industry self-discipline
Legal and operational training for medical personnel
Telemedicine medical personnel must receive comprehensive legal training to ensure familiarity with relevant laws, regulations, and policies governing telemedicine, thereby enhancing their legal literacy and accountability. Regular operational training should be provided to enhance personnel's proficiency with telemedicine technologies and equipment, strengthen clinical communication skills, and elevate diagnostic and treatment capabilities. Training curricula should cover telemedicine diagnostic and treatment protocols, patient rights protection, and medical dispute resolution mechanisms. This training ensures compliance with legal and regulatory requirements during telemedicine service delivery while mitigating risks associated with operational errors or insufficient legal knowledge.26,31
Self-discipline and regulatory role of industry associations
Industry associations should spearhead telemedicine self-regulation by establishing industry-wide self-discipline conventions, clearly defined codes of conduct, and ethical standards to guide compliance among telemedicine providers and practitioners. These associations should conduct systematic quality assessments of telemedicine services, recognize exemplary institutions and individuals through public commendation, and impose informed criticism and disciplinary measures on entities violating self-regulation standards. Concurrently, industry associations must enhance collaboration with governmental bodies, promptly report sectoral challenges and developmental needs, and provide expert recommendations to foster sustainable telemedicine industry growth.30,39
Improvement of the dispute resolution mechanism
Establishment of diversified dispute resolution channels
Diversified telemedicine dispute resolution channels—including consultation, mediation, arbitration, and litigation—should be established. Parties should be encouraged to resolve disputes through consultation; if unsuccessful, they may seek mediation from a medical dispute mediation committee. A specialized telemedicine arbitration institution should be established, with arbitration rules tailored to telemedicine's unique characteristics, providing efficient and accessible arbitration services for all parties. 40 For litigation, specialized medical dispute tribunals should be created to enhance the efficiency and quality of remote medical case adjudication. These diversified resolution mechanisms expand options for stakeholders while enabling efficient and timely resolution of telemedicine disputes. 7
Settlement mechanism for cross-border telemedicine disputes
A cross-border telemedicine dispute resolution mechanism must be established to address the unique characteristics of such disputes. Judicial cooperation should be strengthened through mutual legal assistance agreements with other jurisdictions, clearly defining jurisdictional boundaries and applicable laws for cross-border telemedicine disputes. 41 A specialized cross-border mediation institution, comprising international legal and medical experts, should be established to deliver professional mediation services for these disputes. Furthermore, the international community should develop uniform standards for managing cross-border telemedicine disputes to promote fair and efficient resolution. 28
Conclusions
As an emerging domain within healthcare, telemedicine demonstrates significant utility in overcoming geographical barriers, optimizing medical resource allocation, and enhancing service efficiency, thereby revitalizing China's healthcare sector. However, amid rapid expansion, telemedicine confronts multifaceted legal risks, including qualification and access challenges, medical liability attribution complexities, patient rights protection vulnerabilities, and cross-border regulatory uncertainties. Such risks impede telemedicine's standardization while simultaneously jeopardizing patients’ legal entitlements and interests.
This study proposes targeted preventive measures to address legal risks in China's telemedicine sector. Strengthening the legal framework constitutes the foundational step, involving: clarifying stakeholders’ legal status and responsibilities, and establishing unified standards for medical liability attribution. Enhanced industry supervision is critical for implementing stringent access and exit protocols, while monitoring the full telemedicine service continuum to guarantee quality and safety. Fortifying patient rights protection is central to building trust in telemedicine, specifically through safeguarding informed consent procedures and privacy rights. Concurrently, technical standardization, personnel training, industry self-regulation, and enhanced dispute resolution mechanisms contribute substantively to legal risk mitigation in telemedicine. 38
Despite current deficiencies in China's telemedicine legal framework, effective risk control will be achieved through progressive enhancement of legislation, regulatory mechanisms, and industry self-discipline. Ongoing attention must be directed toward emerging challenges in telemedicine development, necessitating adaptive refinement of preventive strategies to foster sustainable and compliant sector growth while effectively addressing escalating healthcare demands. 40
Footnotes
Acknowledgments
We would like to thank all the participants for their contribution to this study in all ways.
Ethical considerations
This paper is a public policy analysis paper that does not use personal information or personal data and, therefore, does not require consent or permission from the individual. Therefore, we hereby declare that this paper does not require IRB approval or permission.
Author contributions
QW contributed to the conception and design of study. HH contributed to the acquisition of data. QW and HH contributed to the analysis and/or interpretation of data and drafting the manuscript. LH and QW contributed to revising the manuscript critically for important intellectual content. QW, HH, and LH provided approval of the version of the manuscript to be published.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The Youth Fund for Humanities and Social Sciences Research of the Ministry of Education, (grant number 23YJC820013).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Guarantor
Lansong Huang
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
