Abstract
Background
Children's mental health (MH) places significant strain on their caregivers, who often experience disrupted sleep and heightened stress. Given the close relationship between caregiver and child MH symptoms, understanding how digital MH interventions (DMHIs) influence both child and caregiver symptoms is essential for optimizing family-centered treatment approaches.
Objectives
The purpose of this study was to (a) examine associations between child MH and caregiver sleep problems and stress before beginning MH treatment, (b) explore whether a child's engagement level with a DMHI moderates their rate of MH improvement, and (c) determine whether a child's rate of MH improvement is associated with their caregiver's rate of improvement in sleep problems and stress.
Methods
Child–caregiver pairs (N = 3104) completed assessments approximately monthly over at least three months of care to assess caregiver sleep problems and stress, and child MH outcomes. Associations between caregiver and child MH symptoms at baseline were tested. Linear mixed-effects models tested whether engagement was associated with child symptom improvements, and to test whether child outcomes were associated with monthly improvements in their caregiver.
Results
Caregiver sleep problems were associated with child internalizing symptoms (anxiety, depression, and sleep problems; P < .001). Stress was associated with child externalizing symptoms (inattention, hyperactivity, and opposition; P < .001) and younger child age (P < .001). Improvements in symptoms were marginally larger for children with more frequent coaching and therapy sessions (P = .066). Larger improvements in child symptoms were significantly associated with larger improvements in caregiver sleep problems and stress (Ps < .001).
Conclusion
Caregivers of children with more severe symptoms experienced higher stress and sleep disturbances. Greater session engagement was linked to greater child symptom improvement, and greater child symptom improvement was associated with greater reductions in caregiver sleep problems and stress. This highlights how a pediatric DMHI can drive meaningful improvements in MH that extend beyond the child, creating positive ripple effects for caregivers as well.
Introduction
One in seven children in the United States under the age of 18 has a diagnosed mental health (MH) condition. 1 Anxiety, depression, and behavioral disorders are among the most prevalent, affecting millions of children and adolescents each year and disrupting their emotional, social, and academic development.2–4 These challenges not only affect children but also place significant strain on their caregivers, who often experience disrupted sleep and heightened stress.5–8 Child–caregiver sleep disturbances are commonly linked, as children with MH disorders frequently struggle with falling and staying asleep, leading to frequent nighttime awakenings that affect caregivers’ rest.5,6,9 Additionally, managing a child's internalizing or externalizing symptoms can increase emotional, financial, and time-related stress, particularly for caregivers of children with severe behavioral difficulties.7,10 These ongoing stressors can impair a caregiver's ability to regulate emotions, fulfill daily responsibilities, and engage effectively in their child's treatment.11,12 Conversely, when caregivers experience improved sleep and reduced stress, they are better equipped to support their child's progress, highlighting the need for interventions that consider both child and caregiver well-being.
Behavioral health (BH) care (e.g. coaching and therapy) delivered via digital MH interventions (DMHIs) has emerged as a scalable, accessible solution to address pediatric MH concerns, potentially overcoming common treatment barriers including cost, long wait times, and geographic limitations. 13 While research suggests that DMHIs can be effective in reducing symptoms of anxiety, depression, and behavioral disorders in pediatric populations,14–16 less is known about how these interventions impact caregivers. Some evidence suggests that structured caregiver involvement in treatment can enhance parental well-being and self-efficacy,17,18 yet caregiver distress can also serve as a barrier to treatment engagement. 19 Additional evidence shows that when caregivers ruminate (i.e. repeatedly focus on stressful thoughts), their children are more likely to do the same, which in turn is linked to greater anxiety, depression, and behavioral problems. 20 Given the close relationship between caregiver and child well-being, understanding how DMHIs influence caregiver sleep and stress—and what factors contribute to these changes—can help refine intervention strategies.
Our preliminary study of caregiver wellbeing during child participation in a DMHI found that 77% of caregivers reported improvements in sleep problems, and 73% experienced reductions in caregiver stress. 21 These improvements increased over the child's time in care, showing that longer durations in treatment were associated with greater caregiver symptom relief. While these findings provide an initial understanding of how caregivers may indirectly benefit from their child's participation in a DMHI, several limitations remain. Our sample size for longitudinal analyses was small, with only 88 caregivers reporting on stress and 119 on sleep problems. Additionally, children and adolescents were in care for an average of less than three months, limiting our ability to assess longer-term impacts on caregiver wellbeing. We also did not examine potential moderators of changes in caregiver sleep and stress, which is needed to understand how various factors influence these outcomes. Addressing these gaps is essential for ensuring that DMHIs are designed to effectively support both children and their caregivers to maximize their impact on family well-being.
The purpose of this study was to (a) examine associations between child MH and caregiver sleep problems and stress before beginning MH treatment, (b) explore whether a child's engagement level with a DMHI moderates their rate of MH improvement, and (c) determine whether a child's rate of MH improvement is associated with their caregiver's rate of improvement in sleep problems and stress. By addressing these specific aims, we will provide a clearer understanding of how caregiver improvements are linked to their child's treatment progress, particularly in the context of longer-term participation in a DMHI.
Methods
Study design and participants
This retrospective, longitudinal observational analysis examined data from caregivers and their children (ages 6–17) enrolled in MH care with Bend Health Inc. (Bend), a pediatric DMHI, between January 1, 2023, and March 1, 2025 (a 26-month period). Participants were not recruited for research purposes. Instead, eligible child–caregiver pairs were identified using Bend's internal database. Pairs were included if: (a) the child completed at least three coaching or therapy sessions in their first three months in care, and (b) the caregiver completed baseline MH assessments within one month of starting care (N = 3104).
At enrollment, children ages 13 to 17 years and caregivers (on behalf of themselves and their child) agree to Bend terms, including data collection during care and analysis of de-identified data. All study procedures were approved by the Biomedical Research Alliance of New York (Study 23-12-034-1374). The study was determined “exempt,” and requirements of informed written consent were waived. All data were de-identified prior to analysis.
Treatment
Bend is a measurement-based DMHI offering BH care, including virtual video-based BH coaching, therapy, and medication consultation, to children (ages < 18 years) and their caregivers, as well as young adult members (ages 18–25 years). Bend uses a whole family, collaborative approach to tackle child MH challenges, as well as caregiver symptoms. Members access care via primary care provider (PCP) referral, employer or insurance benefits, or direct-to-consumer offerings.
During enrollment in Bend's secure online portal, caregivers provide their child's demographic information (date of birth, sex at birth, gender, and race/ethnicity; see Supplement 1 for details) and clinical history. The caregiver and child also complete a series of MH screeners and assessments to identify key areas of concern (see “Mental health assessments” section). Following enrollment, the caregiver schedules an intake appointment with a behavioral care manager (BCM). The BCM reviews enrollment information, insurance or employer coverage, and discusses care options with the caregiver, ultimately determining the appropriate type of care for the family's circumstances. All children have a BH coach (coach), and they may also be assigned a licensed therapist (therapist) or psychiatric provider (e.g. nurse practitioner or psychiatrist) for medication consultation. The first virtual video-based coaching or therapy session is typically scheduled within two weeks of intake, and the frequency of follow-up sessions is tailored to each family, with most children attending two to three coaching sessions and one therapy session (when applicable) per month.
Coaches are trained in evidence-based behavioral techniques, including cognitive behavioral therapy (CBT), dialectical behavioral therapy (DBT), and mindfulness-based stress reduction, to help members develop coping strategies and behavior change skills. Therapists provide additional clinical expertise for more complex cases, offering diagnostic clarity and therapeutic intervention. Coaches and therapists are both trained in parenting management strategies, delivering interventions directly to caregivers to support the use of positive parenting techniques at home. Psychiatric providers are available when medication management or further psychiatric evaluation is needed. For members referred by an external provider (e.g. PCPs), Bend providers communicate and collaborate with external providers, implementing the collaborative care model to integrate primary and BH care. BCMs oversee care coordination, ensuring seamless communication between different Bend MH providers, as well as external providers. This multidisciplinary approach ensures that children and their families receive comprehensive, developmentally appropriate care tailored to their unique needs.
Coaching and therapy sessions follow structured, module-based care programs tailored to the member's age and primary symptom target—the main area of concern the care program is designed to address (e.g. anxiety)—with each module delivering evidence-based tools to both the child and caregiver, including parenting tools for the caregiver. During all live video-based sessions, caregivers are required to be in the same area (e.g. at home) as their child to ensure safety and to address any issues that may arise. For children ages 1 to 13, caregivers are required to actively participate in sessions to receive parent management training. To further support skill development between sessions, children and caregivers have access to an online learning resource center containing care program materials, and caregivers can securely communicate with providers via instant messaging.
Mental health assessments
At enrollment and approximately monthly as follow-up during care caregivers and children complete MH screeners. Symptoms flagged by the screeners are further measured using full validated assessments. Screeners and assessments measure child anxiety, depression, inattention, hyperactivity, opposition, and sleep problems, as well as caregiver sleep problems and stress. For children < 13 years old, caregivers report their child's symptoms with input from the child. Children ≥ 13 self-report their symptoms except inattention, hyperactivity, and opposition, which are reported by caregivers per standard procedures for these symptoms. 22 Caregivers self-report their own symptoms. Once all assessments are completed, the caregiver and child receive a summary of their results in the online portal. Caregivers are reminded to complete follow-up assessments via email and verbally in-session. All measures used are publicly available and do not require permission for use.
For children < 13 years old, anxiety, depression, inattention and hyperactivity are screened using questions derived from the DSM-5 cross-cutting measure. 23 Opposition is screened by caregiver-report of how often their child has had problematic behaviors in relation to others, and sleep problems are screened by caregiver-report of the frequency of sleep problems. Screeners assess symptom frequency over the past two weeks using a five-point. Likert-type scale from “Not at all” (score = 0) to “Nearly every day” (score = 4). If a child scores ≥ 2 (“Several days” or more) on any anxiety (3 questions), depression (2 questions), or sleep problems (1 question) screener question, caregivers complete the corresponding Patient-Reported Outcomes Measurement Information System (PROMIS) assessment.24,25 PROMIS assessments query about symptom frequency over the past 7 days. The PROMIS anxiety and depression assessments are 10 and 11 items, respectively, with responses on a five-point Likert-type scale from “Never” (score = 1) to “Almost always” (score = 5). The PROMIS sleep assessment is 8 items about sleep disturbance and quality, with responses on five-point Likert-type scales appropriate to the question wording. If a child scores ≥ 1 (“Rare, less than a day or two” or more) on the inattention or opposition screener questions, caregivers complete the 26-item Swanson, Nolan, and Pelham (SNAP-IV)26,27 to measures inattention (questions 1–9; e.g. distractibility), hyperactivity (questions 10–18; e.g. fidgeting), and opposition (questions 19–26; e.g. arguing) symptoms over the past month, with responses on a four-point Likert-type scale from “Not at all” (score = 0) to “Very much” (score = 3).
To screen for anxiety and depression, children aged ≥ 13 years complete the first two questions from the Generalized Anxiety Disorder-7 (GAD-7; called the GAD-2), 28 and the first two questions from the Patient Health Questionnaire-9 adolescent version (PHQ-9A; called the PHQ-2), 29 respectively. Responses are on a four-point Likert-type scale from “Not at all” (score = 0) to “Nearly every day” (score = 3). If the sum of their GAD-2 or PHQ-2 responses is 2 or higher, they complete the remaining questions from the GAD-7 (7-items) or PHQ-9A.30,31 To ensure safety during online self-response, the PHQ-9A is the 8-item version with the original item on suicide and self-harm removed. 32 For sleep problems, children aged ≥ 13 years respond to the same sleep screener as children aged < 13 years (framed as self-report). A response of ≥ 2 (“Several days” or more) triggers the PROMIS sleep assessment for children self-report.
To screen for caregiver sleep problems, caregivers report how often (in the past two weeks) they have experienced a sleep problem. Responses are on a five-point Likert scale from “Not at all” (score = 0) to “Nearly every day” (score = 4). If they score ≥ 2 (“Several days” or more), they complete the 7-item Insomnia Severity Index (ISI), 33 which measures sleep issues (e.g. trouble falling asleep), overall sleep satisfaction, and perception of sleep problems over the past 2 weeks. Responses are on five-point Likert-type scales corresponding with the question (e.g. 0 = “None” to 4 = “Very severe”). To screen for stress, caregivers respond to two questions from the Parental Stress Scale (PSS): “The major source of stress in my life is my child” and “Having a child leaves little time and flexibility in my life”.34,35 Responses are on a five-point Likert-type scale from “Strongly disagree” (score = 1) to “Strongly agree” (score = 5). If they select “Undecided” or more on either question, they complete the remaining 16 items of the PSS. The full 18-item PSS captures caregivers’ subjective experiences of parenting, including items about stressful aspects (e.g. “The major source of stress in my life is my child”) and positive aspects (e.g. “I am happy in my role as a parent”) to capture both the challenges and rewards of parenting.34,35 Scores for positively framed items are reverse-coded. Additional details on screeners and assessments, and copies of measures are in Supplement 2.
Measure calculation
The outcomes of interest are in Textbox 1. Primary outcomes were assessed at baseline (the assessment before intake with Bend) and during follow-up (assessments completed after the first session). Using standardized procedures, the sum of question scores was used to determine assessment score, with PHQ-9A scores adjusted to account for the one removed question, and then symptom severity was categorized using pre-defined ranges for low, mild, moderate, and severe symptoms for each assessment.24–26,30,33,36 Screened-out assessments were categorized separately. For sleep problems, caregivers with mild to severe symptom severity were flagged as having sleep problems. The PSS does not have an established cutoff to identify elevated caregiver stress.34,35 To maintain consistency with our prior work—with mild to severe sleep problems identified in 36% of caregivers
21
—the cutoff for caregiver stress was defined as PSS scores > 32, capturing elevated stress in 40% of caregivers. Elevated child symptoms were defined as moderate to severe symptom severity at baseline and coded as binary (1 = elevated, 0 = not elevated). Assessment score, symptom severity category, and elevated symptoms were used to assess symptom severity, as appropriate for the analysis (see Statistical Analysis).
Outcomes of interest. Primary outcomes:
Caregiver symptom severity Child symptom severity Child symptom improvement Additional outcomes:
Child symptom type Child symptom category Child symptom burden Care type Months in care Session count Average sessions per month Child age (in years) Child age group Child sex Child gender–sex conformity Child race and ethnicity
Child symptom improvement was calculated as the percent improvement (from baseline) in the child's primary symptom target for each follow-up assessment, with positive values indicating improvement. Primary symptom target was identified based on each child's first assigned care program (Table S1) or most severe symptom at baseline (when care program was not applicable; see Supplement 1). For children with both anxiety and depression as primary symptom targets, percent improvement was averaged. For children with multiple ADHD or opposition primary symptom targets, their scores on each domain were added before percentage improvement calculations. All screened-out follow-up assessments were considered as 100% improvement.
Child symptom types were categorized into internalizing (anxiety, depression, and sleep) and externalizing (inattention, hyperactivity, and opposition) categories, coded “1” if any relevant symptom type was elevated and “0” otherwise. Child symptom burden was the count of elevated child symptoms at baseline (range: 0–6). Care type included therapy (1+ therapy sessions) and no therapy (only coaching sessions). Months in care were defined as the number of 30-day intervals since intake at the time of each follow-up assessment. Session count was the sum of coaching and therapy sessions attended (at last follow-up assessment), and average sessions per month was session count divided by months in care (at last follow-up assessment). Age in years was determined at intake and follow-up assessments, and age at intake was grouped as <13 years and 13+ years. Child sex included female and not female. Gender–sex conformity was “conforming” if sex at birth matched gender, and “non-conforming” otherwise. Race and ethnicity responses were summarized as White, Asian, Hispanic/Latino, Black/African American, and Multi-racial or other.
Statistical analysis
A de-identified subject ID (assigned to each child–caregiver pair in Bend's database) was used in all analyses to ensure accuracy. All unique instances of completed assessments within the first six months of care were considered for analysis, and only baseline and follow-up assessments were retained. Data were analyzed in three sections to align with the purpose of the study: (a) baseline child–caregiver symptoms, (b) DMHI engagement and child symptom improvement, (c) child symptom improvement and change in caregiver symptoms.
Sample sizes and additional exclusionary criteria are reported in Figure 1. Throughout, model estimates (and accompanying statistical tests) are reported to identify significant associations, and odds ratios (ORs) and 95% confidence intervals (95% CI) are reported to quantify effect sizes. Models were evaluated for multicollinearity using variance inflation factors (VIFs) or generalized VIFs, as applicable, and all values were < 5, indicating that no concerning correlations were flagged. Correlations between elevated child symptoms were also performed to identify any notable comorbidities, all R values < .10 between internalizing and externalizing symptoms (Table S2). The alpha level was set at .05 for all analyses. P-Values < .10 are discussed as statistical trends given their potential relevance in the context of behavioral data and importance of considering the magnitude of P-values rather than a strict dichotomy, 37 though trends should be interpreted with caution.
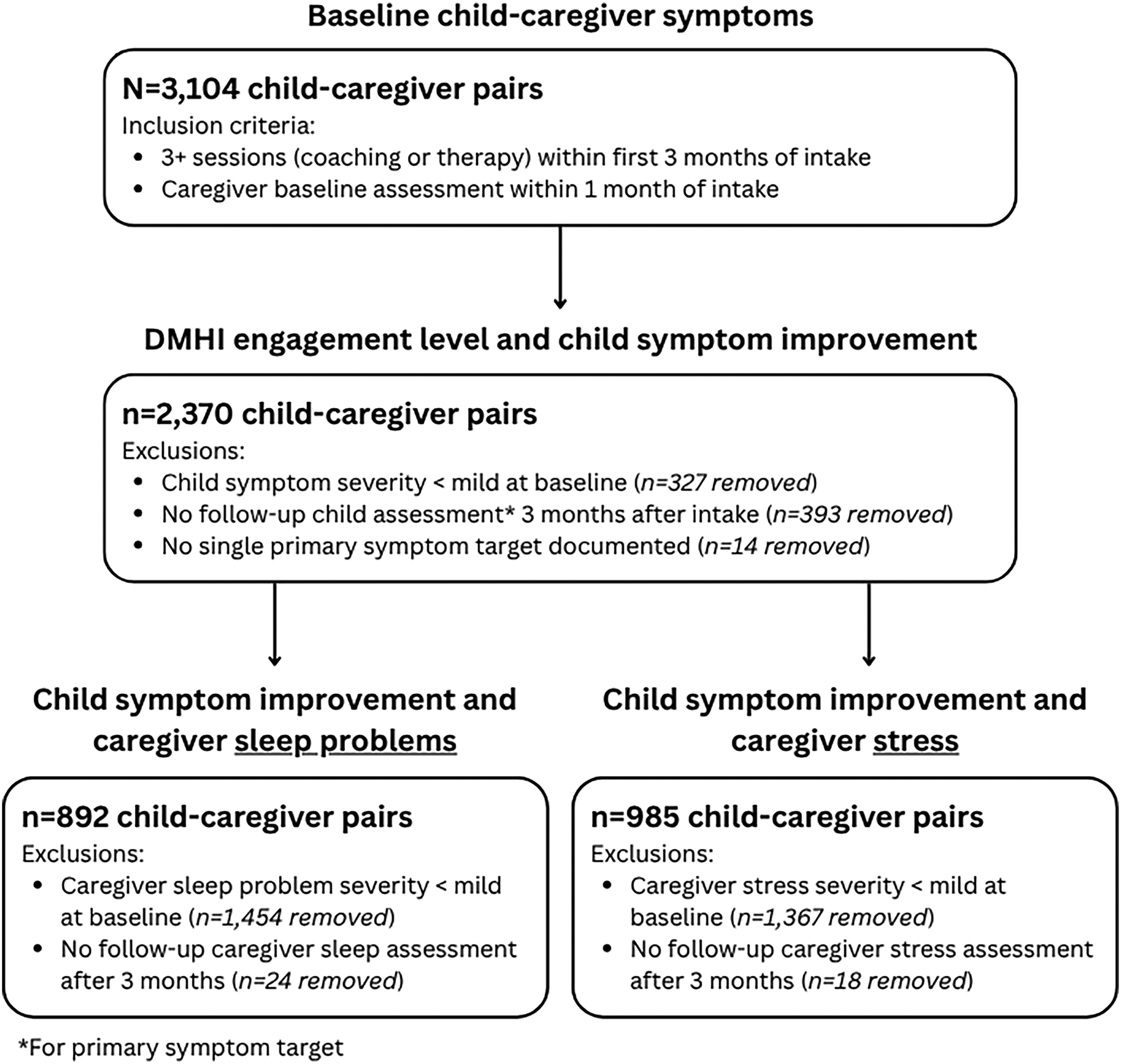
Participant flow chart with study inclusion criteria and exclusion criteria for each set of analyses.
Baseline child–caregiver symptoms
Full-sample (n = 3104) characteristics, including child demographics, and child and caregiver symptom severity, were described. Then, models tested associations between child and caregiver symptoms at baseline. Each caregiver symptom was model separately, with symptom severity category as the outcome. The multivariate models included fixed effects of child internalizing and externalizing symptoms (both binary) and their interaction. The first set of univariate models included a fixed effect of child symptom burden. Then, additional univariate models tested whether elevated child symptoms of each type (e.g. anxiety) predicted caregiver symptom severity category. Child age (in years) and child sex (female and not female) were included in all models as demographic covariates.
DMHI engagement level and child symptom improvement
Only children with mild to severe baseline symptom severity (of primary symptom target) and a follow-up assessment of the same type after at least 3 months in care were included in analyses of child symptom improvement. A linear mixed-effects model tested associations between child symptom improvement and DMHI engagement. Fixed effects were months in care (continuous), sessions per month (continuous), and their interaction. Baseline symptom severity, symptom type (anxiety or depression, and ADHD or opposition), care type, sex, and age group were included as covariates. A random effect of subject (internal ID) was placed on the slope of months in care to account for differences in responsiveness to care. Each child's rate of symptom improvement was calculated as the sum of their random slope plus the estimate of months in care.
Child symptom improvement and caregiver outcomes
For caregivers with baseline sleep problems or stress, we reported the percentage with any decrease in symptom severity and timing of first reported decrease. The percentage of caregivers with a clinically significant decrease in symptom severity (50% ≥ decrease from baseline) was also reported. Then, linear mixed-effects models were used to assess caregiver symptoms (score) over time. The fixed effects included rate of child symptom improvement and months in care, and their interaction. There was a random effect of subject ID on the slope of months in care.
Results
Baseline child–caregiver symptoms
Baseline characteristics of child–caregiver pairs (n = 3104) are in Table 1. Elevated child symptoms were reported by 77.42% (n = 2403), with 24.48% (n = 760) reporting one type, 21.81% (n = 677) reporting two, and 31.13% (n = 966) reporting three or more. Baseline assessments were completed by caregivers a median of 8 days (IQR: 13–5) before starting care with the DMHI. Caregivers who completed the full sleep assessment (n = 1510) had an average score of 11.34 ± 4.99, and 37.38% (n = 1152) of all caregivers had sleep problems. For stress, the average score was 37.25 ± 8.55, and 44.27% (n = 1374) of caregivers had parental stress.
Child demographics and prevalence of elevated child symptoms, and caregiver symptom severity reported for all child–caregiver pairs.

Multivariate model results
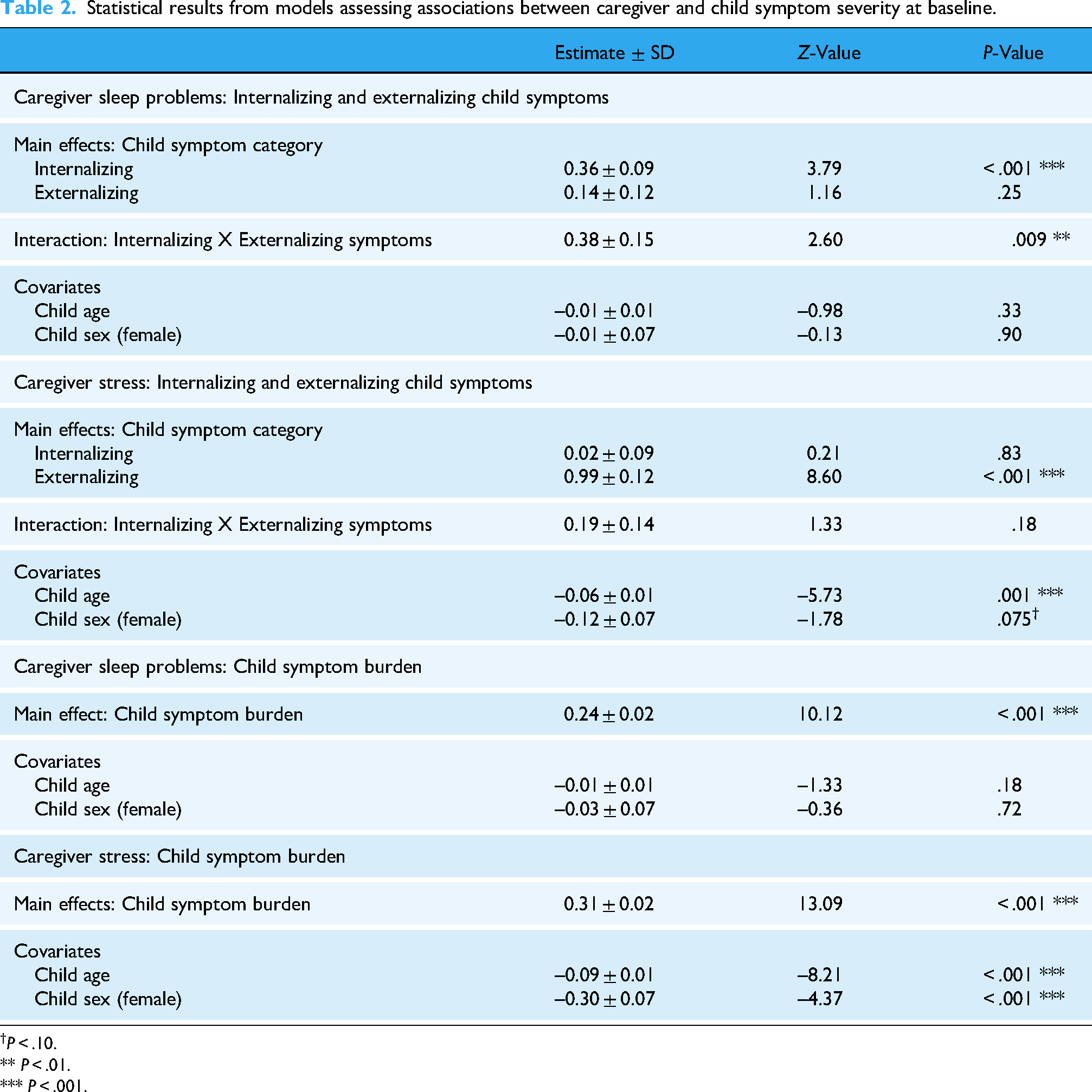
Caregiver stress and sleep were uniquely associated with child symptoms and characteristics in multivariate models (see Table 2). Elevated internalizing symptoms was associated with 1.43 times greater likelihood of more severe caregiver sleep problems (95% CI: 1.19–1.72; Z = 3.79, P < .001). Child externalizing symptoms were not significantly associated with caregiver sleep problems (Z = 1.16, P = .25). The interaction of child internalizing and externalizing symptoms was significant (Z = 2.60, P = .009), both elevated internalizing and externalizing symptoms were associated with 1.47 times greater odds of more severe caregiver sleep problems (95% CI: 1.10–1.95). Child age (Z = –0.98, P = .32) and child sex (Z = −0.13, P = .90) did not significantly associate with caregiver sleep problems. For caregiver stress, elevated child externalizing symptoms were associated with 2.70 times greater odds of more severe caregiver symptoms (95% CI: 2.15–3.38; Z = 8.60, P < .001), and child internalizing symptoms were not associated with caregiver stress (Z = 0.21, P = .83). The interaction of child internalizing and externalizing symptoms was not significant for caregiver stress (Z = 1.33, P = .18). Younger child age was associated with more severe caregiver stress (Z = –5.73, P < .001), and female sex marginally predicted less severe caregiver stress (statistical trend; Z = –1.78, P = .075).
Statistical results from models assessing associations between caregiver and child symptom severity at baseline.
P < .10.
** P < .01.
*** P < .001.
Univariate model results
Regarding child symptom burden, each additional symptom was associated with 1.27 times greater likelihood of more severe caregiver sleep problems (95% CI: 1.21–1.33), and 1.36 times greater likelihood of more severe caregiver stress (95% CI: 1.30–1.43; see Table 2). Child demographic factors were again non-significant for caregiver sleep problems (age: Z = –1.33, P = .18; sex: −0.36, P = .71), but remained significant for caregiver stress (age: Z = –8.21, P < .001; sex: Z = −4.37, P < .001).
Elevated symptoms across all child symptom types were associated with more severe caregiver sleep problems (all Ps < .01; see Table 3). Notably, elevated child sleep problems were associated with a 1.92 increase in the likelihood of more severe caregiver sleep problems (95% CI: 1.67–2.20). All child symptom types were associated with more severe caregiver stress (Ps < .001), except for anxiety (P = .29). Oppositional symptoms had the strongest association; elevated opposition predicted a 3.78 times greater likelihood of more severe caregiver stress (95% CI: 3.20–4.46). Caregivers were more than twice as likely to report more severe stress if their child had inattention (OR = 2.45, 95% CI: 2.11–2.84) or hyperactivity (OR = 2.08, 95% CI: 1.72–2.51). Child age and sex were not associated with caregiver sleep problems (all Ps > .05), whereas younger child age and non-female child sex were both associated with more severe caregiver stress (all Ps < .05).
Univariate models of caregiver sleep problems and stress at baseline, as predicted by elevated child symptoms and with child demographics included as covariates.

Odds ratio (OR) is reported for each main effect of elevated child symptoms.
P < .10.
** P < .01.
*** P < .001.
DMHI engagement level and child symptom improvement
Children (n = 2370) were engaged with the DMHI for an average of 4.76 ± 0.87 months (41.56% in therapy; n = 985 of 2370), and they participated in 9.00 ± 3.32 total coaching and therapy sessions (2.01 ± 0.57 sessions per month). Symptoms improved over months in care (t7207 = 2.67, P = .008; see Table S3), and sessions per month was not associated with symptom improvements (t2365 = 0.78, P = .44). The interaction of months in care with sessions per month approached statistical significance (t7207 = 1.84, P = .066), such that marginally larger improvements in symptoms were reported by children with more sessions per month. The model estimated 2.03% ± 3.04% average rate of improvement (range: −14.10% to 16.28%).
Child symptom improvement and caregiver outcomes
Overall, 87.89% (n = 784 of 892) caregivers with sleep problems reported improvements in the first six months, first reported after an average of 1.70 ± 1.11 months in care. Clinically significant improvements were reported by 47.42% (n = 423 of 892), first reported after an average of 2.41 ± 1.39 months in care. For stress, 82.94% (n = 817 of 985) reported improvements (first reported after 1.77 ± 1.20 months), and 31.88% (n = 313 of 986) reported clinically significant improvements (first reported after 2.69 ± 1.47 months). Table 4 reports comprehensive results of both models testing associations between improvements in child symptoms and caregiver symptoms during care. Caregiver sleep problems and stress decreased over months in care (Ps < .001), and these symptoms were not associated with child symptom improvement (Ps > .05). Notably, decreases in sleep problems and stress were larger for caregivers whose child had larger child symptom improvement (Ps < .001).
Statistical results from the model assessing whether child symptom improvement is associated with decreases in caregiver symptoms.

*** P < .001.
Discussion
Principal findings
The purpose of this study was to (a) examine associations between child MH and caregiver sleep problems and stress before beginning MH treatment, (b) explore whether a child's engagement level with a DMHI moderates their rate of MH improvement, and (c) determine whether a child's rate of MH improvement is associated with their caregiver's rate of improvement in sleep problems and stress. Caregivers tended to have more severe sleep problems and stress if their child had elevated symptoms at baseline, with sleep problems more strongly associated with child internalizing symptoms and parental stress linked to child externalizing symptoms, younger child age, and male child. Children who engaged in more sessions per month tended to have larger symptom improvement. Improvements in caregiver symptoms were associated with their child's response to MH treatment, such that larger reductions in caregiver sleep problems and stress were linked to greater improvements in child symptoms during treatment.
Caregiver sleep problems were more severe for those whose child presented with internalizing versus externalizing symptoms, though elevated symptoms of any type were associated with greater sleep disturbance. Children with depression, anxiety, or ADHD often have impaired sleep, which may negatively impact caregiver sleep due to the increased caregiving demands and nighttime disruptions.38,39 Other studies have also reported that caregivers of children with developmental disorders have significant sleep problems.40,41 Further, given the genetic contributions to many MH and sleep disorders,42,43 caregivers of children with MH issues are more likely to suffer from their own MH challenges, and associated impairments in sleep quality and quantity.44,45 Our findings highlight how interconnected child and caregiver well-being can be, suggesting that sleep problems in children with MH challenges may contribute to a broader cycle of disrupted sleep and emotional strain within families. More severe stress was reported by caregivers whose child had externalizing versus internalizing symptoms. Oppositional symptoms were associated with over three times the odds of more severe caregiver stress, while ADHD symptoms were associated with double the risk. Caregiver stress was also higher in those caring for younger and male children. Externalizing behaviors and developmental factors may be primary drivers of caregiver stress, as U.S. research shows higher stress in those caring for children with ADHD or oppositional defiant disorder. 41 Stress was also marginally associated with children's depressive symptoms and sleep problems, consistent with research linking severe youth depression and sleep disturbances to greater caregiver stress.8,41,46 Taken together, these findings underscore the strong connection between caregiver sleep and stress and their child's specific MH challenges.
Unlike traditional therapy models, which often operate on fixed or infrequent session schedules, and other DMHIs with asynchronous and automated tools, and limited clinician access, Bend's scalable and flexible approach allows children with diverse symptom presentations to access care at a frequency that maximizes their progress. We found preliminary evidence that children with higher DMHI session frequency had greater improvements in their MH symptoms. This aligns with studies demonstrating that higher treatment intensity leads to better outcomes, particularly for conditions such as anxiety and depression.47,48 More frequent sessions support steady progress by reinforcing skills, reducing setbacks, and improving symptom outcomes.49–51 Given that higher session frequency was linked to greater symptom improvement, DMHIs can reduce barriers and help families access the consistent, high-quality treatment needed for better MH outcomes.52,53
Bend's DMHI uniquely supports both child and caregiver wellbeing, as improvements in child treatment outcomes were associated with improvements in caregiver sleep problems and stress. These results build on previous research demonstrating that Bend's family-centered treatment model drives meaningful progress for both children and caregivers,21,54–56 by highlighting the potential that these improvements are not independent of one another. Although other commercial DMHIs have reported improvements in caregiver MH, 57 to our knowledge, we are the first to demonstrate that the magnitude of caregiver improvements are linked to their child's MH improvements. The bidirectional relationship between child and caregiver MH may explain our findings58–62; child symptom reductions may improve caregiver wellbeing by easing caregiver burden, 63 while improvements in caregiver sleep and stress may enhance caregivers’ ability to support their child's treatment and implement positive parenting.64,65 While our findings demonstrate the connection between child MH treatment progress and caregiver wellbeing, future research is needed to identify which aspects of caregiver and child engagement within a DMHI contribute most to these improvements, helping to optimize support for the entire family. At the same time, our results highlight the need for policies that recognize and fund family-centered DMHIs (like Bend) as an essential part of MH care that supports both individual outcomes and entire family well-being.
Strengths and limitations
This study has several strengths. First, the large sample size of children and their caregivers, all of whom meaningfully engaged with the DMHI for at least 3 months and participated in at least three sessions, enhances the reliability and generalizability of our findings. Building on our previous work with a smaller sample size and shorter treatment duration, 21 this study replicates findings of improvements in caregiver symptoms during pediatric DMHI engagement. While our previous study focused on clinically elevated levels of caregiver sleep problems and stress (moderate to severe symptoms), the present study included those with subclinical symptoms (mild severity). By expanding upon our initial study sample, we demonstrate that our initial findings are repeatable and robust, applying to a broader context of caregivers involved in a pediatric DMHI. The rigor of our study is strengthened by the use of validated MH measures, ensuring that both child and caregiver symptoms were evaluated repeatedly with established and psychometrically sound tools. Research done by others in this space has used limited or less-established tools, 57 and without frequent follow-ups during treatment.57,66 Finally, this study addresses previously uncovered associations by including treatment engagement metrics (e.g. session frequency) and rate of child symptom improvement in linear models. While previous research has focused primarily on child outcomes, our study considers how different levels of participation, as well as child treatment responsiveness, may influence caregiver outcomes.
The findings of our study should be considered in the context of a few limitations. Given all analyses were retrospective, causal inferences could not be made between predictors and outcomes of interest. Indeed, our findings may reflect causal relationships between caregiver and child outcomes, or co-occurring but unrelated improvements related to joint participation in care. We also cannot verify that MH improvements are a direct effect of DMHI treatment. A priori power analysis was not conducted and, though large (N = 3104), our sample may have been insufficient to detect small effects. While caregiver-reported assessments of child MH symptoms are commonly used, they may be less accurate in capturing internalizing symptoms compared to child self-reports. Future research should incorporate both caregiver and child self-reports to assess whether these findings remain consistent across different perspectives. The inclusion of self-reports for younger children (ages < 13 years) would minimize any biases in proxy-report that may be influenced by changes in caregivers’ own symptoms. For example, caregivers with improvements in their sleep and stress may be more likely to have a more positive perspective on their child's outcomes. Our findings are based on the outcomes of members participating in care with a specific DMHI, so they may not be entirely applicable to the therapeutic models or populations of other DMHIs or traditional therapy interventions, limiting the generalizability of our findings. Similarly, the study did not compare outcomes of the DMHI to other treatment modalities. To better understand which treatment modality is most effective, future research should compare DMHIs to traditional in-person therapy and other family-based interventions. Addressing these comparisons would determine whether the observed caregiver benefits are unique to DMHIs, or whether they occur across different approaches. This study did not account for caregiver-specific factors (e.g. age, sex, and MH history), which are likely to contribute to observed relationships between family MH outcomes.43,67–70 This is an important line of research to be addressed in future studies.
Concluding remarks
This study highlights the strong connection between child MH and caregiver well-being, demonstrating that Bend Health's family-centered DMHI supports both children and their caregivers. Caregivers of children with more severe MH symptoms reported higher levels of sleep disturbances and stress, emphasizing the challenges families face when managing child MH concerns. Children who attended more sessions per month experienced greater improvements in their MH symptoms. In turn, we found that greater improvements in children's MH symptoms contributed to greater reductions in caregiver sleep problems and stress. These results highlight how effective pediatric MH treatment can have meaningful, whole-family benefits.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251361589 - Supplemental material for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement
Supplemental material, sj-docx-1-dhj-10.1177_20552076251361589 for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement by Darian Lawrence-Sidebottom, Kelsey L McAlister, Donna McCutchen, Monika Roots and Jennifer Huberty in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251361589 - Supplemental material for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement
Supplemental material, sj-docx-2-dhj-10.1177_20552076251361589 for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement by Darian Lawrence-Sidebottom, Kelsey L McAlister, Donna McCutchen, Monika Roots and Jennifer Huberty in DIGITAL HEALTH
Supplemental Material
sj-docx-3-dhj-10.1177_20552076251361589 - Supplemental material for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement
Supplemental material, sj-docx-3-dhj-10.1177_20552076251361589 for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement by Darian Lawrence-Sidebottom, Kelsey L McAlister, Donna McCutchen, Monika Roots and Jennifer Huberty in DIGITAL HEALTH
Supplemental Material
sj-docx-4-dhj-10.1177_20552076251361589 - Supplemental material for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement
Supplemental material, sj-docx-4-dhj-10.1177_20552076251361589 for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement by Darian Lawrence-Sidebottom, Kelsey L McAlister, Donna McCutchen, Monika Roots and Jennifer Huberty in DIGITAL HEALTH
Supplemental Material
sj-docx-5-dhj-10.1177_20552076251361589 - Supplemental material for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement
Supplemental material, sj-docx-5-dhj-10.1177_20552076251361589 for Impact of a pediatric digital mental health intervention on caregiver sleep and stress: A retrospective exploration of moderators of improvement by Darian Lawrence-Sidebottom, Kelsey L McAlister, Donna McCutchen, Monika Roots and Jennifer Huberty in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors would like to acknowledge the exceptional care team at Bend Health Inc. for their dedication, expertise, and commitment to providing high-quality care to families.
Ethical considerations
All study procedures were approved by the Biomedical Research Alliance of New York (Study 23-12-034-1374), and the study was determined “exempt.”
Consent to participate
To participate in care with Bend Health Inc., caregivers (on behalf of themselves and their child aged < 13) and children ages 13 to 17 agree to Bend Health Inc. terms and conditions, which include a statement regarding data collection during care and analysis of de-identified data. Given that this study was determined exempt, there were not additional requirements of informed consent.
Contributorship
KM and DL-S contributed to conceptualization, formal analysis, and writing the original draft. DM, MH, and JH contributed to conceptualization and funding acquisition, and JH supervised the project. All authors reviewed, edited, and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in its entirety by Bend Health Inc.
Declaration of conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employed by or contracted with Bend Health Inc., the company that delivered the treatment in this study. However, the authors’ employment status and compensation are not contingent on the findings of their research.
Data availability
The data used in this study is not publicly available, given Bend Health Inc's privacy policy and HIPAA compliance. The authors will consider all reasonable requests (directed to the corresponding author) for aggregated and anonymized data.
Guarantor
JH.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
