Abstract
Introduction
eHealth-facilitated integrated care models (eICMs) offer promising enhancements to healthcare delivery in stem cell transplantation, but entail implementation and ethical challenges. While qualitative process evaluation methodologies can effectively address such issues through interdisciplinary collaboration, no previous studies have integrated approaches from implementation science and applied ethics into eICM evaluations.
Objective
Exploring an integrated approach to the qualitative process evaluation of a stem cell transplantation eICM (SMILe–ICM), our objectives were threefold: (1) assess the current SMILe–ICM's implementation through stakeholder perceptions; (2) examine relevant ethical issues; and (3) develop strategies to mitigate identified implementation and ethical challenges.
Methods
Semistructured individual interviews were conducted with 12 patients and 3 relatives. Additionally, 8 clinicians in total participated in two focus groups. Data analysis followed an inductive-deductive thematic approach built on interpretative frameworks from implementation science and medical ethics and supported by ethical consultations.
Results
We isolated three main themes, centered on the patient's treatment journey and recovery, to explain patients’, their relatives’ and clinicians’ perceptions regarding the SMILe–ICM. While the SMILe–ICM was generally viewed as valuable, perspectives varied regarding standardized procedures, including eHealth, and care coordination practices. These themes encompass implementation and ethical issues related to individual, intervention, inner setting, and outer setting factors, leading to the development of 17 implementation strategies.
Conclusion
This study provides nuanced insights into patient- and provider-level eICM implementation and ethical challenges. By identifying these issues early, the integrated research design and resulting strategies facilitated well-informed, timely solutions.
Keywords
Introduction
Advances in allogeneic stem cell transplantation (alloSCT) practices have significantly improved hematological disease survival rates. 1 However, the growing population of survivors often faces persistent physical and psychosocial challenges, such as graft-versus-host disease or anxiety. This necessitates ongoing health management, including sustained medication use and routine monitoring for complications.2,3 Therefore, although they have recovered from their primary illness, these patients are still considered chronically ill and have correspondingly complex healthcare needs. 2 Traditional care models often fall short in addressing these needs, as they are primarily designed for acute care. 4 Together with these care models’ typically fragmented nature, this can cause deficits in informational and psychosocial support for survivors5,6 and their families. 7
To address such gaps in healthcare delivery, eHealth-facilitated integrated care models (eICMs) are emerging as promising solutions.8–10 By combining innovative care coordination with personalized self-management support, eICMs can empower patients to effectively manage their health conditions.11–13 Using eHealth solutions, that is, the use of new information and communication technologies in healthcare, eICMs enable continuous connections between patients and clinicians throughout the follow-up phase. 14 This allows for early detection of health deterioration and timely interventions, potentially preventing severe complications.15,16
In addition to eICMs’ benefits, the literature highlights their implementation and ethical challenges,10,17–21 which we elaborate on in the following sections. Despite these challenges’ undisputed relevance, the evidence needed to better understand and address them remains insufficient,18,21–27 pointing to significant knowledge-based and methodological gaps.
Implementation and ethical issues in the eICM context: Distinct perspectives from implementation science and applied ethics
Within the eICM context, it is crucial that implementation and ethical issues be addressed early and continuously through ongoing evaluations,28–31 as challenges evolve and may vary from project to project.30,32 Implementation and ethical issues are relevant to two distinct fields—implementation science and applied ethics—which have traditionally approached them separately:
Implementation science, which focuses on improving the introduction of evidence-based interventions into real-world settings,
33
investigates implementation issues. Acting as either facilitators or barriers to successful adoption, these include individual aspects, for example, personal attitudes toward eICMs, as well as intervention-related aspects, such as the functionality of eHealth solutions. If relevant implementation issues are not adequately addressed, costly innovations such as eICMs are more likely to fail, leading to wasted resources.
30
To mitigate this risk and support adoption, strategies such as gathering ongoing stakeholder feedback and implementing educational initiatives are proposed.
34
Tailoring strategies to disparate contexts requires process evaluations, which can reveal how, why, and for whom an intervention succeeds or fails.
35
Qualitative research designs are crucial to this process, offering unique insights into key stakeholders’ perceptions and experiences.36,37 By examining factors critical to these perceptions, including outcome drivers such as intrinsic motivation, or contextual factors such as healthcare working patterns, process evaluations help clarify the specific reasons behind an implementation's success.
35
Implementation frameworks such as the Consolidated Framework for Implementation Research (CFIR)30,38 have proven valuable in eICM evaluations.
23
By offering structured approaches to identifying key implementation determinants, the CFIR and similar frameworks provide theoretical foundations upon which to develop effective and contextually appropriate implementation strategies.
39
Ethical issues relating to eICM contexts are matters of applied ethics. As a field of study, applied ethics aims to guide human actions in relation to ethical ideals—what is good, right, or desirable—in specific contexts.
40
In the area of eHealth, ethical issues range from minor psychological impacts
41
and privacy concerns
42
to broader social issues such as health disparities.
43
Neglecting those issues means overlooking potential harms or injustices in eICMs’ practices. For this reason, they are best addressed from the outset.28,29 To ensure a stable ethical infrastructure, it is recommended to accompany each implementation science project with a systematic ethical analysis. This should encompass a thorough description of the current situation (the “IS” stage), reflections on desirable outcomes (the “OUGHT” stage), and a formulation of corresponding recommendations to move toward an ethical ideal (the “ACTION” stage).44,45 To define the intended ethical ideal, an accepted ethical framework is necessary. In healthcare contexts, one particularly useful framework is principlism. This includes ethical principles such as beneficence, nonmaleficence, respect for autonomy, and justice. Its use can facilitate the identification of ethical issues, guide decision making, and support recommendations for ethical health care practices.
46
A plea for integrated approaches following the IS-OUGHT-ACTION stages
The combination of implementation science and applied ethics forms a stable foundation for transdisciplinary approaches to eICM evaluations. Transdisciplinary approaches integrate knowledge, methods, and perspectives from multiple disciplines to address challenges such as those encountered in eICM implementation—problems too complex for any single field to resolve. 47
Integrating methods and knowledge across disciplines aligns with recent health technology guidance, which emphasizes the limitations of using empirical or ethical approaches in isolation.48,49 Empirical research-based decisions, for example, the reporting of specific results, are inherently value-laden,29,50 while ethical considerations detached from evidence can be overly hypothetical.32,51 However, by balancing ethical and empirical considerations as complementary aspects of evaluation, researchers can combine both fields’ strengths to overcome their limitations. 52
Implementation science excels in describing and understanding the complex dynamics of implementation processes, while providing a solid foundation for ethical reflection.45,51 Upon that foundation, applied ethics can construct a moral framework to guide and inform our research actions. Moreover, it enables the systematic screening and analysis of ethical issues in the implementation process, serving as an early warning system for emerging concerns.31,53,54 Finally, actionable implementation strategies can be developed by synthesizing empirical and ethical insights, including observations and reflections from all previous stages. 52 By integrating these perspectives, strategies can be designed that not only improve the possibility of an eICM's successful implementation but also uphold and enhance the ethical value of its intervention components.
Although guidelines are available for empirical 55 and ethical 56 eHealth evaluations, as well as for integrated research practices, 48 none specifically address the combination of eICMs, implementation science, and applied ethics in evaluation research. To fill this methodological gap, that is, to integrate ethical considerations into the development and implementation of eICMs, the new DAta new REsponsibilities project (DARE, funded by the German Federal Ministry of Education and Research (BMBF)) has developed the innovative IS-OUGHT-ACTION approach. This is built around a pragmatic three-part framework. The “IS” component deals with the current situation, including any problems; the “OUGHT” component is the ideal/target situation or goal; and the ACTION component includes methods of achieving that goal. The IS-OUGHT-ACTION approach guides researchers to choose and systematically combine distinct implementation science methods with appropriate applied ethics principles within qualitative process evaluations (see Figure 1).
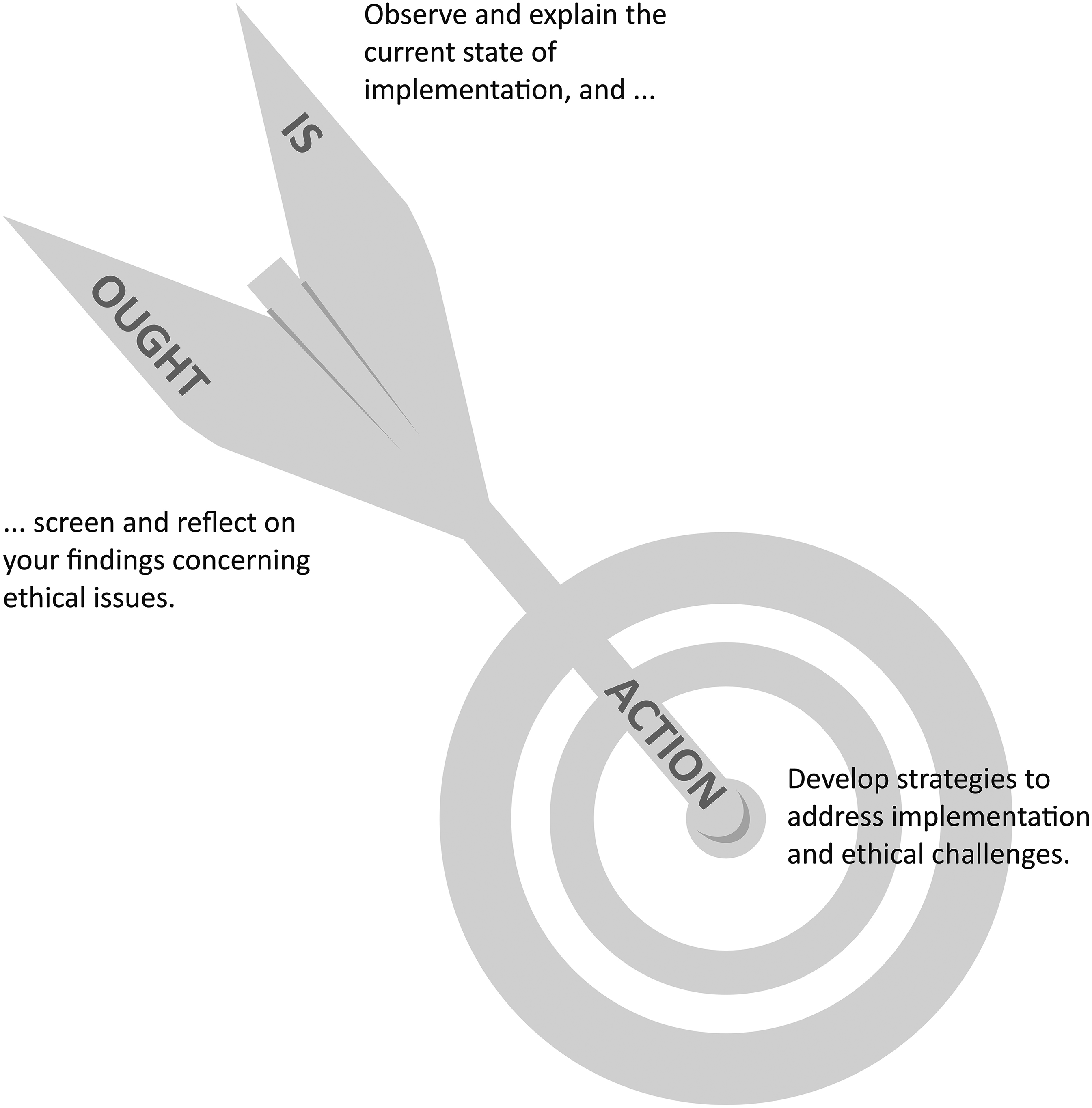
Transdisciplinary process evaluation.
The IS-OUGHT-ACTION structure is particularly effective when the eICM's eHealth component is created through agile software development. Following this methodology, each component is not formulated and delivered as a final product, but evolves incrementally via an iterative process that facilitates the continuous addition and refinement of software solutions throughout the implementation process.57,58 Therefore, in addition to the clinical leaders who decide on an intervention's implementation, and the clinicians who carry out that intervention, a transdisciplinary process evaluation of an eICM can also inform the software engineers’ decisions. In addition to coordinating the software engineers, clinicians and clinical leaders, this helps align the proposed eHealth application with ethical standards. 42
Objectives
To recap, eICMs are inherently linked to implementation and ethical challenges. Better understanding and addressing these challenges demands ongoing systematic evaluations of eICMs’ complex systems. Within this context, a transdisciplinary approach, combining implementation and ethical methods, has not yet been applied in eICM evaluation studies. Therefore, to fill the identified gaps both in knowledge and in methodology, this study explored a transdisciplinary research design within a qualitative process evaluation conducted during the implementation of a stem cell transplantation eICM—the SMILe–ICM.
59
Using the above-noted IS-OUGHT-ACTION stages as an overarching structure, we pursued three objectives:
To observe and explain the SMILe–ICM's current state of implementation by considering the perceptions of patients, their relatives, and clinicians (IS). To critically reflect on the current state of implementation in light of a healthcare-focused ethical ideal (OUGHT). To enhance the SMILe–ICM's implementation success and ethical value by developing strategies to address identified implementation and ethical issues (ACTION).
Methods
Study design
This process evaluation study employed a transdisciplinary research design that combined empirical and ethical approaches within the IS-OUGHT-ACTION structure. It was approached from a contextualist epistemological standpoint, straddling the realms of natural science and humanities. 60 For the empirical part, we used a qualitative evaluation design, collecting data via semistructured individual interviews and focus groups. The ethical perspective was fulfilled through a principlism 46 and moral case deliberation 61 approach suitable for health interventions. Throughout this manuscript, we adhered to the Standards for Reporting Qualitative Research (SRQR). 62
Study context and intervention
This substudy is the offspring of two broader parent-projects. The first is the interdisciplinary DARE project, which aims to provide recommendations for integrating ethical considerations into the development and implementation of eHealth interventions. As a practical example, the DARE research team referred to the SMILe–ICM implementation process, which is the focus of our second parent-project. At the time of data collection, the SMILe team had developed57,59,63,64 and begun implementing and testing the SMILe–ICM as part of an international multicenter implementation science project.59,65 The reported substudy focuses on the German SMILe–ICM, which was conducted at the University of Freiburg Medical Center's alloSCT clinic. Ethical approval for the study was obtained from the local ethics committee (EK 309/19).
The SMILe–ICM supplemented standard care practices by involving Advanced Practice Nurses (APNs) and eHealth (SMILeTechnology). The study APNs (SMILeAPNs) with advanced expertise in oncology coordinated care and provided self-management support through 12 face-to-face visits (SMILeVisits), between pretransplantation and 1-year post-alloSCT. SMILeVisits encompassed aspects such as monitoring and follow up of medical, behavioral, and symptom-related parameters, infection prevention, physical activity promotion, and immunosuppressant medication adherence. Additionally, SMILeAPNs screened patients’ health data remotely via SMILeTechnology, which included the SMILeApp for patients and the SMILeCare monitoring system for use by transplant center clinicians. Using the SMILeApp, patients recorded and transferred data on daily health parameters (e.g. temperature), the occurrence and severity of symptoms (e.g. diarrhea), and their overall wellbeing to their clinical team (described in detail in Leppla et al. 59 ).
Participants
Patient and relative sample: patients were recruited as a subsample of the SMILe–ICM intervention group at the Medical Center Freiburg. Therefore, the sample pool was predetermined by the inclusion and exclusion criteria set by the SMILe study (German Registry for Clinical Trials: DRKS00020347). To be included in the SMILe–ICM, patients had to be at least 18 years old, proficient in both written and spoken German, capable of performing self-management activities, have access to home internet, and be capable of providing informed consent. To be selected for the DARE study, they also had to have approximately 6 months of post-alloSCT experience with the SMILe–ICM.
Within this sample pool, we employed purposeful sampling to gather divergent and extreme cases. 60 This involved selecting patients from various social roles (e.g. younger mothers and retired males) and with varied post-alloSCT trajectories (e.g. with and without complications such as infection and graft-versus-host disease). Additionally, we selected patients flagged by the SMILeAPNs as either noncompliant or significantly engaged with their SMILe interventions. Patients’ relatives were not explicitly contacted, but were invited to participate in the interviews if they accompanied participating patients.
Clinician sample: We aimed to include clinicians from various disciplines who regularly interacted with the SMILe–ICM. No additional inclusion criteria were applied. Clinicians were approached based on a list of potential participants compiled by the SMILeAPNs. After inviting them to participate in our focus groups, we selected participants using convenience sampling, that is, we invited those who were willing and available to participate.
Data collection procedures
Open-ended interview questions were designed to capture participants’ perceptions of the SMILe–ICM. To mitigate the potential for social biases by enabling participants to articulate criticism from a third-person perspective,66,67 we incorporated a self-developed vignette presented as a concise fictional story. The interview guide underwent two rounds of feedback from the research team, followed by a pilot test with three patients. It was then iteratively refined based on interim findings throughout the data collection period. Before the interviews, both verbal and written informed consent were obtained from all participants. All interviews were audio-recorded. Example questions are presented in Supplemental material 1.
Individual interviews: In-person interviews were conducted with patients and their relatives (where appropriate) at the Medical Center Freiburg from November 2020 to June 2021. During the interviews, COVID-19 pandemic hygiene regulations were implemented, including mask-wearing and maintaining physical distance. The interviewer—the first author, who had expertise in nursing science and applied ethics—was unknown to the participants.
In addition to collecting qualitative interview data, each patient's relevant characteristics were recorded, including an assessment of their subjective socioeconomic status (SES). To define this variable, we used the validated German MacArthur Scale, which prompts patients to self-assess their SES on a scale from 1 to 10. 68 Patients’ demographics (age, gender, and living status) and disease-related details (days post-transplant) were retrieved from the SMILe Study records (see Table 1). Patient recruitment concluded when the initial themes were substantiated through two consecutive interviews, indicating data saturation. This required a total of 12 interviews. Of the 12 primary interviewees, three male patients were accompanied by their wives. In two of these cases, this was necessitated by the men's physical frailty. The interviews lasted 48 minutes on average.
Participants characteristics.

FG: focus group; IQR: interquartile range; SES: socioeconomic status.
Focus groups: Clinician focus groups convened online (via Zoom) in March 2022 and in person in February 2023. Overall, 27 clinicians were invited to participate via email. The invitation included study information and three potential focus group dates. Due to the small response rate, participants were additionally invited in person by the SMILeAPNs, resulting in one session with 5 clinicians and another with 3. The focus groups were moderated by the first and second authors (the latter of whom holds a PhD in philosophy and has particular expertise in applied ethics), neither of whom were known to the participants. In addition to gender, each participant's professional role and experience in the field of hematology-oncology (see Table 1) was assessed during focus group discussions. The sessions lasted an average of 47 minutes.
Data analysis
The PSPP software (Version 2.0.0) was used to conduct basic descriptive statistical analyses of participant demographics.
Interview data analysis was supported by MAXQDA software (version 2020). We conducted a codebook thematic analysis, 69 following Gale et al.'s Framework Method for interdisciplinary health research: 70 Interviews were transcribed verbatim and pseudonymized, followed by mixed inductive-deductive coding by the first and second authors. Based on the first three transcripts, the coders established a set of structural codes. These were then organized thematically to construct the categories of an analytical framework, which was then applied to the remaining transcripts and their data charted. The analytical framework was updated regularly based on interim results. The charted data were interpreted concurrently, as the IS and OUGHT stages were performed iteratively. In line with thematic analysis, we focused our data interpretation on the meaning of the data rather than on their quantitative aspects. 69
Within the IS step, after initial inductive coding was completed, the qualitative data were interpreted using the CFIR framework.30,38 This helped distinguish implementation outcome-linked determinants focusing on acceptability, appropriateness, and feasibility. 71 To address the OUGHT step in the output domain, we additionally applied beneficence, nonmaleficence, autonomy, and justice, these are, the four core principles of medical ethics (details in Supplemental material 2).46,72 The same principles were used as a screening matrix within the qualitative data to identify ethical issues. During weekly meetings between the first and second authors, they also guided further ethical reflections.
Additionally, moderated ethical consultations (n = 7) were conducted throughout the study process. The interdisciplinary research team—comprised of ethicists, software engineers, and nursing scientists—employed the Nijmegen method, 61 a moral case deliberation approach. This method involves identifying an ethical problem, collecting relevant facts, discussing and weighing underlying norms and values, and finally reaching a well-supported moral judgment.
During analysis, emerging ideas, concepts, and explanations were explored using visual tools on Miro, an online team-oriented workspace that aids in visualizing content and data. Structured written analytical memos, as proposed by Gale et al. 70 also proved useful. The first and second authors then synthesized the findings and developed actionable strategies. They employed the implementation strategy format proposed by Proctor et al., 39 which involves describing the “actor, action, action targets, temporality, dose, implementation outcomes addressed, and theoretical justification.” The theoretical basis was the CFIR, complemented by the four core ethical principles, 46 which provide moral justification and support the transdisciplinary approach. Additionally, to specify the eICM element targeted by the strategy, we incorporated “modality” into the format. All draft strategies were shared with the members of the interdisciplinary study team for refinement.
Results
The IS and OUGHT stages are presented in terms of three central themes: “patients’ arduous journey,” “patients’ diverse paths toward normalcy,” and “the value of companionship on the journey.” Along with their associated subthemes, these themes focus on the patient’s journey with the disease and steps toward recovery. They provide insights not only into each patient's perspective regarding the SMILe–ICM, but also those of their relatives and clinicians.
The “Strategies for ACTION” subsection first summarizes the study's findings and reflections on implementation and ethical issues, then proposes seventeen strategies (Strat.) to address them (Table 2).
Strategies for ACTION.
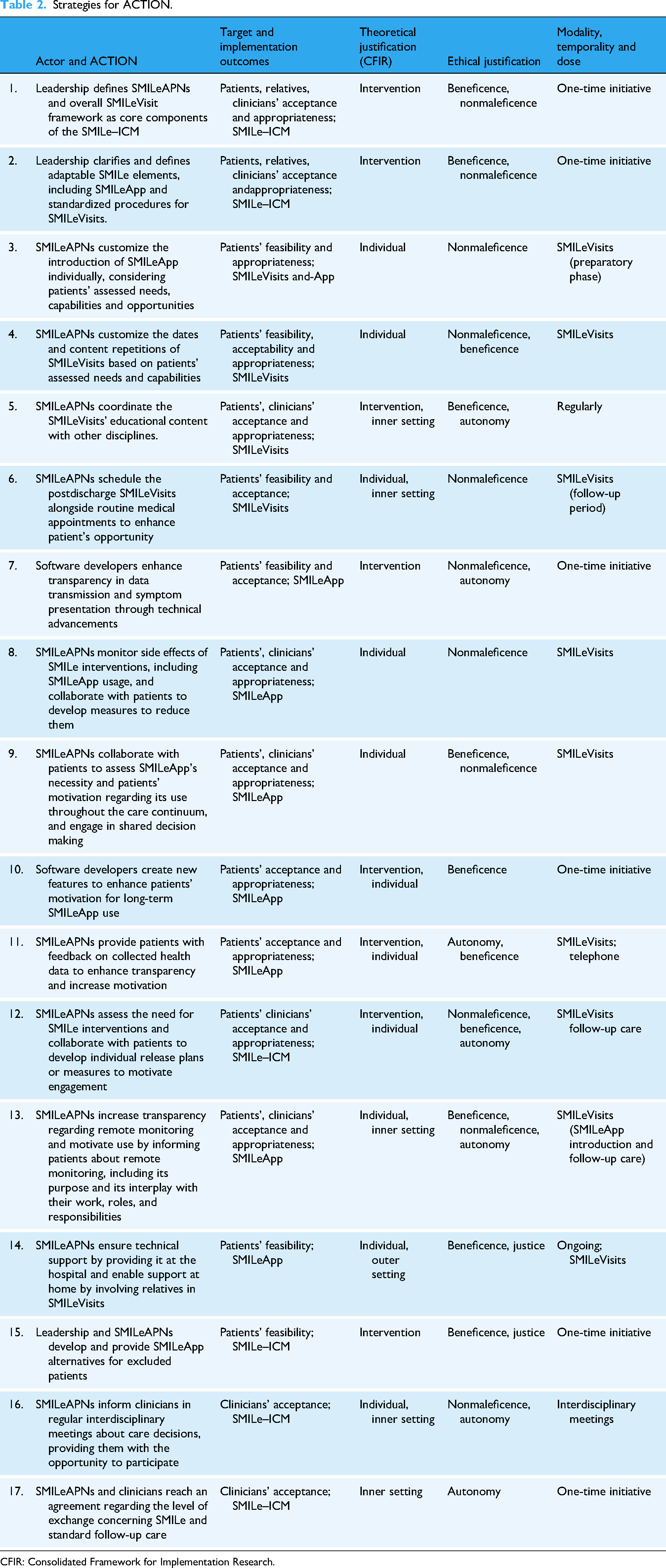
CFIR: Consolidated Framework for Implementation Research.
Theme 1: Patients’ arduous journey
“Patients’ arduous journey” encompasses the phases of preparation, hospitalization, and transitioning back to home life. Throughout these phases, we observed factors within the individual, inner setting, and innovation domains that we linked with patients’ and relatives’ perceptions of the SMILe–ICM. We dichotomized these perceptions as appropriate/inappropriate, acceptable/unacceptable, and feasible/unfeasible.
Preparing for an unknown journey and outcome: At the beginning of their journey, patients faced the task of preparing for the unknown. The burden of not knowing the outcome of their treatment was a psychological burden. This was especially noticeable among single persons responsible for caregiving duties. During this phase, SMILeAPNs provided patients insights about their upcoming hospitalization, which in some cases alleviated the patients’ emotional distress. Moreover, acceptance of the SMILe–ICM was achieved largely through the SMILeAPNs’ assurances of care continuity. Aligning well with the principle of beneficence (Table 2, Strat. 1), this was perceived as especially positive compared to the sporadic involvement of various physicians. It's a good feeling to have at least someone who continues to care for you from the beginning. (Patient 21, female, line 349) The thing with the app is kind of a side issue…really, concerning survival. (Patient 10, female, line 835–836)
Away from home: The next phase was hospitalization: Patients left their familiar surroundings, routines, and support networks for inpatient life. To minimize infection risks, treatment began in isolation, which brought forth an array of health problems. During the SMILeVisits, patients’ capacity to process information was once again compromised by the conditioning therapy and multidrug treatment, which led to concentration difficulties. To combat this effect, topics were repeated to enhance retention. Aligning with the principle of beneficence, this was perceived by patients as appropriate. However, for those with no cognitive impairment, it extended the SMILeVisits’ learning sessions with little to no added benefit. Especially for patients with poor health status, this posed challenges, conflicting, once again, with the principle of nonmaleficence (Table 2, Strat. 4). But the training, that was incredibly much at the beginning. Sometimes it went on for over two hours, and I couldn't sit anymore. (…) It often just repeated itself. (Patient 18, female, line 543–548) I didn’t just get a lot of info from the nurses, but also from the dietitian on the ward. And then again in rehab. (…) Sometimes it left me feeling a bit confused. (Patient 18, female, line 552–554) You really need someone who knows their stuff, not someone who has no clue what you’re talking about. (…) That was always nice, and we ended up building a real relationship of trust. (Wife, Patient 13, line 213–217)
The homecoming: Despite feeling well-prepared by the SMILeVisits for their homecoming, the need to adapt self-management demands to individual circumstances led to insecurities. Especially among patients without direct support at home, physical impairments added further challenges. Nevertheless, despite the need to travel, the ongoing SMILeVisits were generally perceived by patients as feasible, acceptable, and appropriate during this period. Aligning with the principles of nonmaleficence and beneficence (Table 2, Strat. 1 and 6), this effect was amplified partly by SMILeAPNs’ scheduling of visits alongside regular medical visits, and partly by the patients’ and relatives’ preference to address concerns and questions that had arisen at home to a familiar person. The training sessions and stuff were always super doable. I just combined them with my appointments at the ITZ [Interdisciplinary Tumor Center]. (Patient 11, male, lines 231–233) She called right away and asked what was going on—whether I was feeling bad. (…) It worked. (…) If something goes off track, they’re immediately there and they move things around or make changes. (Patient 8, male, line 274–281) The hip pain is always there, but if other things come along and I’m in pain, they don't know that those are different pains. (Patient 21, female, line 406–408)
Theme 2: Patients diverse paths toward normalcy
We identified diverse patient paths toward normalcy and corresponding levels of engagement with the SMILe–ICM. These ranged from early independence to sustained dependence, described in the subthemes “Shortcuts,” “Detours,” and “The Defined Path.” These subthemes summarize patients’ and clinicians’ perceptions, associating them with factors both in individual patients and in the intervention domains.
Shortcuts: Premature disengagement from active disease management, contrary to SMILe recommendations, was associated with taking “shortcuts” in the disease journey. We identified two distinct motivations for patients to take such paths, resulting in decreased acceptance and appropriateness toward the SMILe–ICM. One was linked to SMILe interventions triggering negative emotions. This was observed in a single mother who avoided disease-related information materials and frequently missed health data transmissions. Subsequent telephone calls by the SMILeAPNs triggered feelings of anxiety. Considering that calls from the hospital number were associated with the possibility of bad news, this was a fairly common response. Nevertheless, both this patient and all others interviewed appreciated the calls. They assured the SMILeAPNs that the SMILe–ICM worked and that they knew their nurses aimed to inquire about their wellbeing. The calls were generally perceived as caring, aligning with the principle of beneficence (Table 2, Strat. 1 and 11). When you see the number from the university hospital, you get a bit of a racing heart because you don't know whether it's good news or not. But otherwise, it's fine. (Patient 21, female, line 383–384) Some find it beneficial, while others don't. (…) I do notice if it's not working, if it causes them distress or fear. But those are individual cases. (FG1, clinician 3, line 581–583) By now, I find it a bit annoying. Maybe it's because things are going well for me, and I don't feel the need to constantly track everything anymore. (Patient 11, male, line 472–478) It would be nice if, as a patient, you could see a personal statistic on the app. Because right now, it feels kind of blind. You have to go back individually if you want to know something. (Patient 18, female, line 687–689)
Detours: A sustained focus on disease management tasks—even one partially relieved by SMILe recommendations—was linked to “detours” in some patients’ disease journeys. Two female patients, reporting anxiety, depressive feelings, and limited social support, made their medical conditions central to their journey to regain control over their lives. One observed that this focus was an effective diversion from depression. As a result, both engaged strongly with the SMILeAPNs and were clearly motivated to implement their recommended SMILe interventions. Activities such as self-surveillance, strict dietary adherence, and meticulous hygiene precautions replaced their lost daily routines. Their perceived acceptance and appropriateness of the SMILe–ICM were high, initially appearing to align with the principle of beneficence. This is for me, so that I don't fall into depression or anything, so that I don't somehow lose the ground beneath my feet. It's like a framework or like a skeleton for me, that I control everything I do. (…) The app also fits well into this control system. (Patient 10, female, line 923–926) [Getting] back to life: we need to support them in that (…), not always making them feel ill. (…) We pathologize them to some extent because of this need for security. (FG1, clinician 1, line 544–549)
The defined path: A third pathway became evident due to the concurrent SMILe Study: The “defined path” connected patients’ and their relatives’ intervention acceptance to personal feelings of trust, gratitude, and altruism. Therefore, trusting that the SMILeAPNs’ recommendations were beneficial, they sought to help future patients through health data collection. Independently of perceived personal health benefits, this motivation led patients to accept and perform interventions daily. The app is just to help, you know. (Wife, patient 13, line 818) Yes, exactly. For the study. (Patient 13, male, line 820) What's important to me is that Ms. X [SMILeAPN] and Ms. Y [SMILeAPN] check in on how I’m doing and give me some feedback, whether in person or by phone or whatever, like saying, ‘You’re doing well. We’re satisfied,’ or ‘You need to train more.’ (…) Both their opinions really matter to me. (Patient 13, male, line 1207–1213)
Theme 3: The value of companionship on the journey
Theme 3 explores patients’ “Companions from their private surroundings” and “Companions from the healthcare setting” throughout their journeys. These two subthemes cover influential factors related to individuals, external and internal settings, and aspects of the intervention itself that influenced patients’ and clinicians’ perceptions of the SMILe–ICM.
Companions from private surroundings: Patients’ narratives indicated that contacts with companions from their private surroundings had diverse characteristics. We considered the lack of support as a factor that increased patients’ motivation to engage with the SMILeAPNs. Patients sought contact not only to clarify medical questions but also to address psychosocial needs. This increased the perceived appropriateness and acceptance of the SMILe–ICM, aligning with the principle of beneficence (Table 2, Strat. 1). Maybe also just talking a little to someone, you know; also, about really private things; just so you can look forward to participating in a normal life again someday—also with the worries, somehow (…) if I’ll ever be able to go bowling again. (Patient 11, male, line 298–303) We have a target group in which not everyone is proficient in using digital media. (…) They would probably cope better if you just wrote them a letter or called them every two weeks. (FG2, clinician 3, line 243–246) If it [SMILeApp] stops working, I can’t do anything about it myself. I have to ask for help. Usually, it's my sons who fix it for me. (Patient 8, male, line 349–342)
Companions from the healthcare setting: Changes in clinicians, poor accessibility, and strict time limits regarding medical visits complicated the establishment of trustworthy relationships other than those with the SMILeAPNs. As a result, some patients restricted their clinicians’ responsibilities, that is, by allowing physicians to handle only medical issues. They preferred to discuss sensitive matters with their SMILeAPNs. I love my husband, and he loves me. What about physical contact? Is that allowed? (…) I wouldn’t have had the confidence to ask a doctor or Professor F that. (Patient 17, female, line 478–490) We also [experienced] this practically through Ms. X [SMILeAPN] and Ms. Y [SMILeAPN]. Just the option to say, ‘Well, you could have a port [catheter] at home, and the homecare service can handle it, and you can use it at night’ or something like that. So, getting shown different options that no one else mentioned along the way. (Wife, patient 19, line 620–626) The doctors didn’t really show much interest in it. (…) I kept getting asked, ‘How's your blood pressure?’ And I’d say, ‘Well, you can see it in the app.’ (Patient 11, male, line 583–587) If it's really advice, consultations, or more general things, or even just monitoring or the graphical analysis of the data input, we don't have the time to review that every day. (FG2, clinician 3, line 336–341) In follow-up, we need to be cautious that this care person doesn't automatically take over all disciplines just because they are the primary contact. (FG1, clinician 2, line 503–505)
Strategies for ACTION
We identified implementation issues relating to various factors, including the characteristics of the intervention itself (e.g. standardized SMILeVisit procedures, care coordination tasks, SMILeApp functionality, and process transparency), individual factors (e.g. patients’ lack of capabilities and motivation), inner setting factors (e.g. physician turnover, limited time resources, and information resources for patients), and outer setting factors (e.g. patients’ lack of social and technical support at home).
Ethical issues discussed included the importance of human contact (i.e. with SMILeAPNs) for patients. We also considered any risk of harm, as well as fairness concerns regarding standardized procedures. Additionally, we addressed transparency issues in remote monitoring processes and examined the ambiguous effects of eICM implementation on clinicians’ roles, responsibilities, and relationships.
This integrated empirical and ethical analysis resulted in the creation of 17 strategies aimed at clinical leaders, SMILeAPNs, and software engineers (Table 2). Two overarching strategies involve clarifying and defining core and adaptable elements within the SMILe–ICM (Strat. 1, 2). Additionally, we formulated 15 specific strategies for ACTION, including:
Customizing standardized SMILe interventions for patients based on assessments (Strat. 3, 4, 6, 8, and 9), Supporting and improving communication and collaboration with transplant team clinicians (Strat. 5 and 16), Enhancing SMILeApp functionalities (Strat. 7 and 10), Increasing patients’ motivation, capability, and opportunities to adapt SMILe (Strat. 11, 14, and 15), Clarifying SMILeAPNs’ roles vis à vis those of clinicians, for example, dieticians, physicians (Strat. 12 and 17), Enhancing the transparency of SMILe–ICM processes for patients (Strat. 13).
Discussion
In this study, we explored the perceptions of patients, their relatives, and clinicians regarding the SMILe–ICM. Based on our assessments of those perceptions in terms of both implementation-related and ethical issues, we derived actionable strategies aimed at enhancing both implementation success and ethical acceptability.
While the SMILe–ICM was generally perceived as a valuable addition to current alloSCT care, varied stakeholder perceptions about specific SMILe elements reflect the complexity of eICM implementation,17,18,73 which is influenced by multifaceted determinants. 23 Besides implementation issues, our results support concerns about eICM-related ethical issues. 74 To prevent such issues from becoming irreversible problems, our 17 action strategies provide proactive approaches to addressing them during the SMILe–ICM implementation. A number of these strategies have already been implemented in practice. These include creating an overview of symptom and vital value progression for the patients, expanding input fields (pain/rash locations), adding a free text message field, providing data transfer status notifications, and increasing the SMILeAPNs’ flexibility regarding their delivery of the intervention elements.
Beyond these results, we now highlight two central findings that are crucial, from both implementation science and ethical perspectives, to future eICM practices: the central role of interpersonal relationships in eICM implementation, and the challenge of balancing standardization with customization in eICM element delivery.
The central role of interpersonal relationships in implementing eICMs
Interpersonal relationships played a central role in our findings. This involved close cooperation both between the directly involved parties—SMILeAPNs, patients, and relatives—and across the broader interdisciplinary transplant team.
Strong, durable relationships between SMILeAPNs, patients and relatives enhanced positive perceptions of the SMILe–ICM. These relationships developed through regular face-to-face visits during inpatient care and continuous eHealth connections during follow up. Similar findings by Piras and Miele,
75
Darley et al.,
76
and McCann et al.
77
highlight how, by fostering familiarity and providing a sense of security, a well-designed eHealth care model can enhance beneficial relationships. Such psychosocial effects challenge the common ethical concern that the increasing use of eHealth will lead to depersonalization in healthcare.78,79 This concern may not be relevant to care-enhancing interventions such as the SMILe–ICM. Rather than replacing human contact, the SMILe–ICM uses technology to connect patients with familiar reference persons and round-the-clock support. Moreover, privacy concerns and lack of trust—two commonly cited barriers to eHealth implementation42,80—were not issues in our interviews. We attribute this to the patients’ strong interpersonal relationships with the SMILeAPNs. Based on these findings, we suggest that human components, including interpersonal relationships, should be considered core elements of eICMs. In addition to potentially promoting implementation success by enhancing eHealth-related perceptions, this strategy aligns with the WHO's proposal to “place people at the center of digital health.”
14
Further, by considering cancer patients’ and their relatives’ preference for personal contact,
5
as well as individual interventions’ impacts on their overall wellbeing, it also aligns with the core ethical principle of beneficence. Our findings also revealed challenges regarding relationships within the transplant team. These were particularly affected by the introduction of a new SMILeAPN role regarding existing care processes.
17
This new role unintentionally affected certain team members’ decision-making authority, as well as their relationships with patients. In this respect, our findings support Bamber and Marshall's
17
outline of how any encroachments on members’ professional identities can lead to implementation barriers. Consideration of such implementation issues is also relevant from the perspective of applied ethics, which upholds the integrity of healthcare relationships as a criterion for the ethical evaluation of eHealth.79,81 Thus, when implementing eICMs in any setting, it is essential to advocate for ongoing, continuous communication and a feedback culture among stakeholders.17,82 This pertains not only to physicians, who are often the focus of studies, but also to “lower-profile” members of transplant teams, including therapy staff.
Striking the right balance between standardization and customization
Another key finding relates to the challenge of balancing standardization and customization in delivering eICM interventions. As noted by Mannion and Exworthy,
83
there exists a tension in healthcare trends aiming to achieve both. Our findings underscore this dynamic, especially concerning the SMILeApp and standardized procedures in SMILeVisits.
The SMILeApp met most participating patients’ needs, likely due to their involvement in the design, development, and implementation phases.
64
In addition to enhancing patients’ safety, as noted by Darley et al.,
84
the SMILeApp also helped patients regain a sense of control lost during their arduous journey. However, considering that patient traits vary and needs change along the cancer care continuum,
5
even the best eHealth model has only a limited capacity to respond to patients’ individualities and dynamics. Results include ambiguous perceptions (e.g. acceptance vs nonacceptance) and effects (e.g. benefit vs harm). As this study suggests, the introduction of new features (e.g. a chat function) may allow the care team to meet more patients’ individual needs. However, even at this stage, eHealth customization will certainly face limitations, particularly among vulnerable patient groups. The literature highlights concerns about unequal access to eHealth, especially for those with low technological and language skills, or restricted internet access, all of which can make people more vulnerable to health inequities.
85
To address these and other limiting factors, implementation strategies must first maximize eICMs’ availability. This includes, for instance, providing technical support (as demonstrated in the SMILe–ICM) and assessing patients’ ability to access assistance from home. Still, even while healthcare systems commit to promoting equitable healthcare, not all barriers can be fully overcome by technical solutions or additional support. While we recognize that customization is vital, we do not advocate for unconditional adaptations or the rejection of standardization. On the contrary, with a sharp focus on ensuring the quality and safety of healthcare interventions, we agree with the WHO High 5 project's emphasis on standardization as a weapon against resource constraints.
86
Therefore, the SMILe team developed standardized protocols for SMILeVisits. However, as Chae et al.
87
note, information overload can result not only from the size of a standardized information package but also from the patients’ individual characteristics, for example, lower education or high anxiety, all of which should be considered when providing educational elements as important as the SMILeVisits.
As illustrated, achieving an effective balance between standardization and customization in eICMs is a significant challenge. Needham 88 highlights the need for healthcare systems to deliver “the best of both worlds.” For SMILe, this involves assessing highly standardized components, for example, the SMILeApp and SMILeVisits, to better meet individual needs without sacrificing overall quality. Regular patient assessments (e.g. of cognitive impairment and technical skills), monitoring for side effects (e.g. information overload), and offering alternatives as necessary (e.g. email or telephone instruction) are all recommended. This approach mirrors prescriptive medication practices and has been advocated across various eHealth fields,28,31 ICMs 12 and cancer education. 89 It also aligns with Kirst et al.'s 90 findings, which emphasize flexibility as a key factor for success in implementing ICMs.
Strengths and limitations
This study is the first to merge methods from implementation science and applied ethics in the qualitative eICM process evaluation. Structuring this transdisciplinary approach through the IS-OUGHT-ACTION stages provides valuable insights across all of the involved disciplines. While empirical findings offered robust descriptions of implementation, the ethical perspective broadened the focus to include alternative viewpoints. This inclusivity is critical, as research tends to emphasize acceptance over nonacceptance, thereby risking bias toward neutral or positive outcomes. Regarding this tendency, Stevens et al. 91 cautioned against the under-reporting of negative outcomes in eHealth research. The incorporation of applied ethics encouraged us to think critically, nudging us toward the exploration of alternative perspectives, which is essential in evidence-scarce areas such as eICM research.92,93 Throughout the processes of evaluation research, we believe that the benefits of critical ethical reflection far outweigh the challenges typically associated with transdisciplinary research, particularly its resource-intensiveness. 47
Besides its strengths, the study has limitations. These include the generally small number of study participants, which meant the perspectives of several key stakeholders were either underrepresented or missing. This is particularly true regarding our reporting of the viewpoints of relatives, whose role in implementation warrants more attention. Further, some disadvantaged patient groups (e.g. those with language barriers) were excluded from this substudy. We also encountered a pronounced gender imbalance within the focus groups: proportions of 80% to 100% female participants reflect the limitations of convenience sampling. 60 This limits the interpretation of our findings, as male perspectives and opinions may differ. Moreover, SMILeAPNs’ perspectives were only indirectly included through their involvement in the research process; and the organizational domain, known to be significant in eICMs’ implementation, was not considered. Such omissions limit insights into issues such as resource allocation and workload management. These factors are vital to the formulation of strategies not only for practice in the target context but also for the outscaling of ICMs to other institutions. 23
Another limitation pertains to the chosen principlist approach, which, as noted by general critique, does not provide sufficient guidance in conflicting situations. 94 In line with published recommendations,81,95 we used moral case deliberation as a supplementary method.
One additional point of criticism refers to the use of the four principles of medical ethics as the main framework for our ethical analysis, as they lack the specificity necessary for eHealth. While approaches such Marckmann's evaluation matrix 81 exist, they do not fit the SMILe–ICM, which involves both human and technological components. Therefore, we adopted an interim solution: utilizing the four (abstract) principles of medical ethics while employing Marckmann's criteria to add specificity (see Supplemental material 2).
Conclusion
This study offers nuanced insights into the implementation process and related ethical implications of an eICM. It explains how interpersonal relationships play a crucial role in eICM implementation, with the potential to enhance or hamper success. In this context, we discuss ethical issues related to changing healthcare roles, responsibilities, and relationships. Another key finding concerns the provision of highly standardized eICM elements, such as SMILeApp and SMILeVisits: While these elements proved valuable for most patients most of the time, they were not suitable for everyone at all times. Such gaps in suitability prompted reflections on the potential harm of overly standardized elements and the risks of exclusion due to unequal opportunities.
In response to the observations and reflections this research evoked, we developed 17 actionable strategies to address implementation barriers, facilitators, and ethical implications on the patient and provider levels. These include recognizing the SMILe–ICM's human elements and related interpersonal relationships as core eICM components, while understanding the need to better tailor adaptable elements (i.e. SMILeApp and SMILeVisits) to individual traits and needs.
Alongside implementation challenges, ethical issues often emerge as subtle impacts. In process evaluation studies, a transdisciplinary approach can detect and address these issues early on, thereby preventing serious ethical dilemmas. By facilitating interdisciplinary collaboration, the IS-OUGHT-ACTION stages offer a structured framework to put this early detection system into practice.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251336859 - Supplemental material for Implementation and ethical issues of an eHealth-facilitated integrated care model in stem cell transplantation: A qualitative process evaluation study
Supplemental material, sj-docx-1-dhj-10.1177_20552076251336859 for Implementation and ethical issues of an eHealth-facilitated integrated care model in stem cell transplantation: A qualitative process evaluation study by Linda Wienands, Julia Krumme, Sabina De Geest, László Kovács, Alexandra Teynor, Sabine Valenta and Lynn Leppla in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251336859 - Supplemental material for Implementation and ethical issues of an eHealth-facilitated integrated care model in stem cell transplantation: A qualitative process evaluation study
Supplemental material, sj-docx-2-dhj-10.1177_20552076251336859 for Implementation and ethical issues of an eHealth-facilitated integrated care model in stem cell transplantation: A qualitative process evaluation study by Linda Wienands, Julia Krumme, Sabina De Geest, László Kovács, Alexandra Teynor, Sabine Valenta and Lynn Leppla in DIGITAL HEALTH
Footnotes
Acknowledgements
We would like to thank all the participants in this study for sharing their thoughts and experiences with us. We also thank Chris Shultis for editing the manuscript.
Disclosure
Writing AI assistants ChatGPT-3.5 and Smodin were used for language improvement. No scientific content in this manuscript was created by these or any other AI system.
Guarantors
LW, SV, and LL
Ethical considerations
During the study process, we adhered to all ethical principles applicable to empirical health research, including taking every reasonable step to produce valuable research results, ensure scientific validity, and maintain fair subject selection and fully informed consent. 96 Ethical considerations influenced key methodological decisions, including sample composition, data collection procedures, and presentation of the results. All externally produced tools and questionnaires used in this study were used within the scope of respective copyright holders’ stipulations. This study was approved by the Ethics Committee of the University of Freiburg in Germany (EK 309/19).
Consent to participate and consent for publication
Informed consent was obtained from all study participants in both verbal and written form.
Author contributions/CRediT
LW, JK, LL, SV, and LK were involved in conceptualization; LW and JK, in formal analysis; LL, LK, and AT in funding acquisition; LW and JK in investigation; LW, JK, LL, and SV in methodology; LL in project administration; LL, SV, SDG, LK, and AT in supervision; LW in writing—original draft; and JK, LL, SV, SDG, LK, and AT in writing- review and editing.
Funding
The author(s) disclose receipt of the following financial support for the research and publication of this article: This work was supported by the Federal Ministry of Education and Research (BMBF) (grant number 01GP1909B) and the Deutsche José Carreras Leukämie-Stiftung (grant number DJCLS R20/2018).
Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The research team included members of the SMILe team (LL, SV, SDG, and AT), who developed and implemented the intervention. They contributed to the conceptualization of the study, the recruitment of clinicians for the focus group, and the review and editing of the manuscript. To mitigate their influence on the evaluation results, they were not involved in the data collection or analysis.
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to the fact that they consist of qualitative data in the form of transcripts, which could potentially identify individuals, but are available from the corresponding authors on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
