Abstract
Objective
Patients with advanced chronic kidney disease (CKD) face challenging decisions about kidney replacement therapy (KRT) options. This study aimed to evaluate the effectiveness of an Interactive Patient-Decision-Aid App for Kidney Replacement Therapy (iPtDA-KRT App) in improving decision self-efficacy, knowledge, and readiness while reducing decisional conflict and regret among patients with advanced CKD.
Methods
A prospective, two-arm clinical trial was conducted with 70 patients who used either the mobile app-based iPtDA-KRT or a paper-based PtDA for 4 weeks. Key measures included decision self-efficacy (DSES), decisional conflict (DCS), KRT knowledge (KRTKS), preparation for decision-making (PrepDM), and decision regret (DRS). These measures were assessed immediately post-intervention and at 4 weeks post-intervention. Additionally, outcomes such as DSES, DCS, and DRS were tracked at 8 weeks post-intervention. Data were analyzed using generalized estimating equations.
Results
The experimental group included 19 male and 16 female patients with a mean age of 66.4 ± 14.0 years, while the control group included 22 male and 13 female patients with a mean age of 59.7 ± 16.1 years. The experimental group demonstrated significantly greater and sustained improvements in self-efficacy, knowledge, and preparation for decision-making compared to the control group. Reductions in decisional conflict and regret were also maintained over time.
Conclusions
The iPtDA-KRT App demonstrated substantial and lasting benefits over paper-based PtDAs in supporting patients’ complex decision-making. These findings suggest that digital, interactive PtDAs may enhance informed decision-making, reduce long-term decisional conflict, and improve patient confidence in clinical settings.
Trial Registration
This study was registered at ClinicalTrials.gov (Identifier: NCT05666544).
Keywords
Introduction
Chronic kidney disease (CKD) is a major global health issue, projected to be the fifth most common chronic condition by 2040. 1 Advances in medical technology have extended survival rates for CKD patients; however, most progress to kidney failure, requiring decisions about treatment options like kidney replacement therapy (KRT). Taiwan, which has the highest prevalence rate of kidney failure worldwide, has become known as a “dialysis kingdom,” 2 where the number of individuals undergoing dialysis continues to rise. 3 Dialysis, often a lifelong treatment, not only directly affects the physical health of patients but also indirectly affects their families, employment, and the ability to participate in social activities. 4 Therefore, patients’ preferences should be considered and patients should be involved in the decision-making process to ensure patients effectively adapt to treatments.
KRT options include hemodialysis (HD), peritoneal dialysis (PD), kidney transplantation, and conservative management. Each modality has specific pros and cons, which can be overwhelming for patients, leading to anxiety and decision-making challenges. 5 Traditional, one-way information delivery often fails to address patients’ unique preferences and concerns. Shared decision-making (SDM), a collaborative process between healthcare providers and patients, considers patients’ perspectives crucial in forming a mutually agreed treatment plan. 6 SDM has been shown to effectively increase medical knowledge, enhance decision-making self-efficacy, and reduce decisional conflict in patients with CKD.7,8 In Taiwan, SDM is encouraged in CKD treatment planning. However, patients often struggle to bridge the gap between information received and real-life dialysis experiences, especially with home-based self-management requirements of PD. 9 This discrepancy between expectation and reality can lead to confusion, regret, and feelings of powerlessness.
Patient decision aids (PtDAs) are essential components of the shared decision-making (SDM) process. 10 As part of our ongoing research, we developed a web-based PtDA called the Interactive Patient-Decision-Aid App for Kidney Replacement Therapy (iPtDA-KRT App). This application integrates simulated scenarios, compares various KRT options, and translates medical evidence into patient-friendly language. Computer simulations that use sound, text, and graphics provide a practical method for presenting realistic scenarios.11,12 Simulated scenarios engage patients in a real-life-like environment, enabling them to better integrate skills and knowledge while assisting them in overcoming the gap between expectations and reality. 13
The Internet, as a globally accessible medium, offers significant advantages over traditional paper-based PtDAs in delivering these tools.14–16 In recent years, information technology-based PtDAs have been widely applied in SDM across diverse healthcare fields, receiving positive evaluations for their usability and impact.7,16,17 Historically, research on the use of web-based PDAs for KRT options has been limited. In a study conducted in the United States, Subramanian et al. (2019) provided participants with information on chronic kidney disease, peritoneal dialysis, and hemodialysis, along with a value clarification exercise, through a study website. Participants accessed this information via their personal electronic devices to support the complex decision-making process between center-based hemodialysis and peritoneal dialysis. 7 To our knowledge, no web-based PtDA specifically designed for CKD patients choosing among KRT options has been developed in Taiwan. This could be due to the fact that traditional strategies for health education primarily involve face-to-face sessions with medical personnel. Therefore, this study aims to evaluate the impact of the developed iPtDA-KRT App on decision-making outcomes among patients with advanced CKD.
Methods
Study design
This was an exploratory pilot study using a two-arm, open-label design. The study was conducted at the nephrology outpatient clinics of two university-affiliated hospitals in central Taiwan. Both hospitals implemented a CKD care program led by the Ministry of Health and Welfare, with nephrology nurses at both sites trained in CKD care. To minimize potential institutional differences, we selected two hospitals with an established collaborative relationship, ensuring that both adhered to the same CKD care protocol and that their nephrology nurses received standardized training. One hospital was randomly assigned to the experimental group and the other to the control group through a lot-drawing method. Each group consisted of 35 participants. The experimental group used the iPtDA-KRT App, whereas the control group used the paper-based PtDA. The study was conducted from October 2022 to July 2023. Both groups completed a pretest (T0) prior to the intervention, followed by three post-tests conducted immediately after (T1), 4 weeks after the intervention (T2), and 8 weeks after the intervention ended (T3; Figure 1). Prior to data collection, researchers conducted meetings with nephrology nurses from both hospitals to ensure accuracy and procedural consistency. This study adhered to the STROBE cohort guidelines.

Study flow.
The Research Ethics Committee of a medical university hospital in central Taiwan approved this study (CMUH109-REC3–085).
Decision aid development
The iPtDA-KRT App was developed in two phases, following the International Patient Decision Aid Standards. 18 Phase 1 involved creating the app's prototype content through semi-structured interviews with patients and feedback from experts. A total of 15 patients, who had started dialysis within the past month and had experience with SDM and paper-based PtDAs, were recruited from a medical center. These semi-structured interviews were conducted to identify their PtDA needs. Based on these interviews, a questionnaire was developed by reviewing the literature on KRT and using an existing PtDA survey. The questionnaire included five sections covering CKD symptoms, basic information about KRT, comparisons of KRT modalities, patient preferences, and CKD/KRT information. Feedback was gathered from an expert panel using the modified Delphi method. The expert panel consisted of two nephrologists, four dialysis nurses, and a health education expert. Two rounds of the modified Delphi process were conducted to finalize the iPtDA-KRT App prototype.
The second phase focused on evaluating the iPtDA-KRT App prototype and making necessary revisions. The iPtDA-KRT App, a web-based application, was developed using JavaScript and HTML5. Testing of the prototype was carried out by the expert panel and five patients with CKD, all of whom had an educational level equivalent to junior high school. The expert panel and the patients assessed the suitability, comprehensibility, and readability of the prototype of the iPtDA-KRT App by using a structured questionnaire with items rated on a 5-point scale. The iPtDA-KRT App was refined according to the feedback from these questionnaires.
The iPtDA-KRT App (Figure 2) improves patient understanding of KRT options through three main features: (1) Interactive Guidance (Figure 2A), which provides a step-by-step walkthrough of clinical procedures; (2) Interactive Simulation (Figure 2B), which allows patients to virtually experience clinical operations via interactive videos; and (3) Interactive Decision-Making (Figure 2C), which facilitates decision-making by enabling interactive comparisons of methods, preference assessments, knowledge evaluations, and final reviews. These features empower patients to make well-informed decisions about their KRT options.

The iPtDA-KRT App.
Participants
The sample size was calculated using G*Power 3.1. Based on the recommendations of the Ottawa Hospital and Health Research Institute (OHHRI), sample size estimation using the decisional conflict scale as the outcome variable should be based on an effect size of 0.3–0.4. 19 The minimum required sample size was 58 participants, which was increased to 70 (approximately 35 per group) to account for potential attrition.
Participants were recruited from the outpatient clinics of two nephrologists. All potential subjects were screened to determine eligibility based on the subsequent inclusion/exclusion criteria. The inclusion criteria were as follows: (1) adults aged 20 years or older who were conscious and able to communicate in Mandarin or Taiwanese; (2) patients had an estimated glomerular filtration rate (eGFR) of less than 30 mL/min/1.73 m² (according to the recommendations of the National Kidney Foundation in the United States, education on different KRTs should begin when eGFR falls below this threshold 20 ); and (3) patients who voluntarily decided to participate in this trial and follow the trial protocol. Patients were excluded if they had absolute contraindications preventing free choice between hemodialysis and peritoneal dialysis, or if they suffered from severe illnesses or mental conditions that hindered questionnaire completion. Patients who met the inclusion criteria were referred for inclusion by nephrologists. Once patients agreed to participate, written informed consent was obtained from them prior to study initiation.
Measures
Kidney Replacement Therapy Knowledge Scale
The Kidney Replacement Therapy Knowledge Scale (KRTKS) was developed based on the Taiwan Chronic Kidney Disease Clinical Guidelines. 21 This 10-item scale assesses knowledge of CKD and KRT, with each correct answer scoring 1 point, totaling a maximum score of 10 points. Incorrect answers receive no points.
Preparation for Decision-Making Scale
The Preparation for Decision-Making (PrepDM) Scale, developed by OHHRI, assesses patient readiness for decision-making. This 10-item scale uses a 5-point rating (1 = not at all to 5 = a great deal). Scores are calculated by subtracting the average score from 1, and then multiplying by 25, yielding a range from 0 to 100. Higher scores indicate greater readiness for decision-making. 22 The scale has been confirmed to have high reliability and validity. 23
Decisional Conflict Scale
The Decisional Conflict Scale (DCS), developed by OHHRI, measures patients’ decisional conflict in medical decision-making. It consists of five subscales: informed, clarity of values, supported, uncertainty, and effective decision. Each item is rated on a 5-point scale (0 = strongly agree to 4 = strongly disagree). Scores are calculated by multiplying the average score by 25, resulting in a range from 0 to 100, with higher scores indicating more conflict. 19 The DCS has demonstrated strong reliability and validity.24–26
Decision Self-Efficacy Scale
The Decision Self-Efficacy Scale (DSES) by OHHRI measures a patient's confidence in their decision-making ability. This 11-item scale uses a 5-point rating (0 = not at all confident to 4 = very confident). The average score is multiplied by 25, yielding a score range of 0 to 100, with higher scores indicating greater decision self-efficacy. 27 The DSES has been validated in diverse populations.28,29
Decision Regret Scale
The Decision Regret Scale (DRS), developed by OHHRI, evaluates the level of regret associated with health-related decisions. It includes five items rated on a 5-point scale (1 = strongly agree to 5 = strongly disagree). The second item, “I regret the choice I made,” and the fourth item, “This choice caused me a lot of harm,” are reverse-coded. Scores are converted to a 0–100 scale by subtracting 1 from each item score and multiplying by 25. Final scores are averaged, with 0 indicating no regret and 100 indicating high regret. 30 The DRS has been demonstrated to have high reliability and validity.31,32
Intervention and data collection
Experimental group (iPtDA-KRT group)
Before the intervention, nephrologists and nurses at the hospital enrolling patients in the experimental group received training on the iPtDA-KRT App. At T0, demographic and disease data were collected, and participants completed the DSES, DCS, and KRTKS. The experimental group used the iPtDA-KRT App within the SDM process under the guidance of nephrology nurses, who also addressed patient questions. Participants progressed through five decision aid steps: (1) CKD symptoms and overview, (2) introduction to KRT options, (3) comparative KRT information (advantages, disadvantages, risks), with patients rating risks and benefits, (4) assessment of patient preferences and values, and (5) knowledge check on CKD and KRTs. The patient's final treatment choice was documented in the app to facilitate treatment discussions with the nephrologist.
After completing these steps, patients used the iPtDA-KRT App to discuss their choice with nephrologists and confirm their treatment plan. Following this, the first posttest (T1) was administered immediately, during which patients completed the DSES, DCS, KRTKS, and PrepDM. Patients could access the iPtDA-KRT App at home, using their accounts to review information and engage in interactive dialysis simulations for 4 weeks. Patients could determine the frequency of using the iPtDA-KRT App based on their individual preferences and needs throughout the study period. They could choose to perform any of the five decision aid steps individually or complete all steps sequentially. At the end of the 4-week period, each patient completed the second posttest (T2) at the outpatient clinics. This posttest required patients to complete the following scales: DSES, DCS, KRTKS, PrepDM, and DRS. Eight weeks after the intervention ended, all patients had a follow-up appointment, where they completed the third posttest (T3) at the outpatient clinics. This posttest required patients to complete the DSES, DCS, and DRS scales.
Control group (paper-ptDA group)
The control group received a paper-based PtDA during a face-to-face session with a nephrology nurse prior to their outpatient appointment. After this session, patients discussed their treatment options with a nephrology specialist to finalize their treatment plan. Patients were also given the paper-based PtDA to review at home. The data collection time points for the control group were identical to those of the experimental group.
Data analysis
Data were analyzed using SPSS version 24.0 (IBM, Armonk, NY, USA). Continuous variables are presented as means and standard deviations, with normality assessed using the Kolmogorov–Smirnov test, while categorical variables are shown as frequencies and percentages. Chi-square and independent t-tests were conducted to compare demographic and disease characteristics between the groups. An independent t-test analyzed pretest differences in DSES, DCS, and KRTKS scores between groups. To evaluate intervention effectiveness, paired and independent t-tests assessed changes in PrepDM and DRS scores over time within each group and between groups at each time point. A generalized estimating equation (GEE) analyzed changes in DSES, DCS, and KRTKS scores between groups post-intervention.
Results
Participant characteristics and baseline comparison
A total of 70 patients with advanced CKD were recruited, evenly divided into two groups of 35 each. No participants were lost during the study. All participants completed post-tests 4 weeks after the intervention concluded and underwent follow-up assessments at 8 weeks. The mean age of participants was 63 years (SD = 15.4), and the majority in both groups were male (58.6%), had less than a primary school education (44.3%), were married (74.2%), were unemployed (65.7%), and lived with their spouse and children (37.1%). Regarding disease status, participants had an average of 1.8 chronic conditions (SD = 1.1), a mean eGFR of 12.2 mL/min/1.73 m² (SD = 7.9), and the majority were at CKD stage 5 (67.1%). There were no significant differences in baseline demographic characteristics between groups (Table 1). At T0, no significant differences were observed between groups in DSES and DCS scores (Table 2), although the experimental group had significantly lower KRTKS scores than the control group. Further analysis of DCS subscales revealed that the experimental group scored significantly higher on the informed, values clarity, and uncertainty subscales compared to the control group.
Patient demographic and disease characteristics.

Abbreviations: eGFR, estimated glomerular filtration rate.
Baseline DSES, DCS, and KRTKS scores in each group.

Abbreviations: DSES, Decision Self-Efficacy Scale; DCS, Decision Conflict Scale; KRTKS, Kidney Replacement Therapy Knowledge Scale.
Analysis of DSES, DCS, and KRTKS scores
Table 3 and Figure 3A show the DSES scores. In the control group, DSES increased by 20.45 points at T1 compared to T0 (

(A) DSES, (B) DCS, and (C) KRTKS score at different times.
Intergroup differences in intervention effectiveness of DSES, DCS, and KRTKS.

Abbreviations: DSES, Decision Self-Efficacy Scale; DCS, Decision Conflict Scale; KRTKS, Kidney Replacement Therapy Knowledge Scale; T0, baseline; T1, first posttest (immediately after intervention); T2, second posttest (4 weeks after intervention); T3, third posttest (8 weeks after the intervention ended).
The DCS scores are presented in Table 3 and Figure 3B. In the control group, DCS decreased significantly at T1 (−15.89,
The KRTKS scores are shown in Table 3 and Figure 3C. At T0, the experimental group scored 1.54 points lower than the control group (
Analysis of prepDM scale and DRS scores
The PrepDM scale scores at T1 and T2 are presented in Table 4 and Figure 4A. Significant differences between groups were observed at both T1 and T2 (
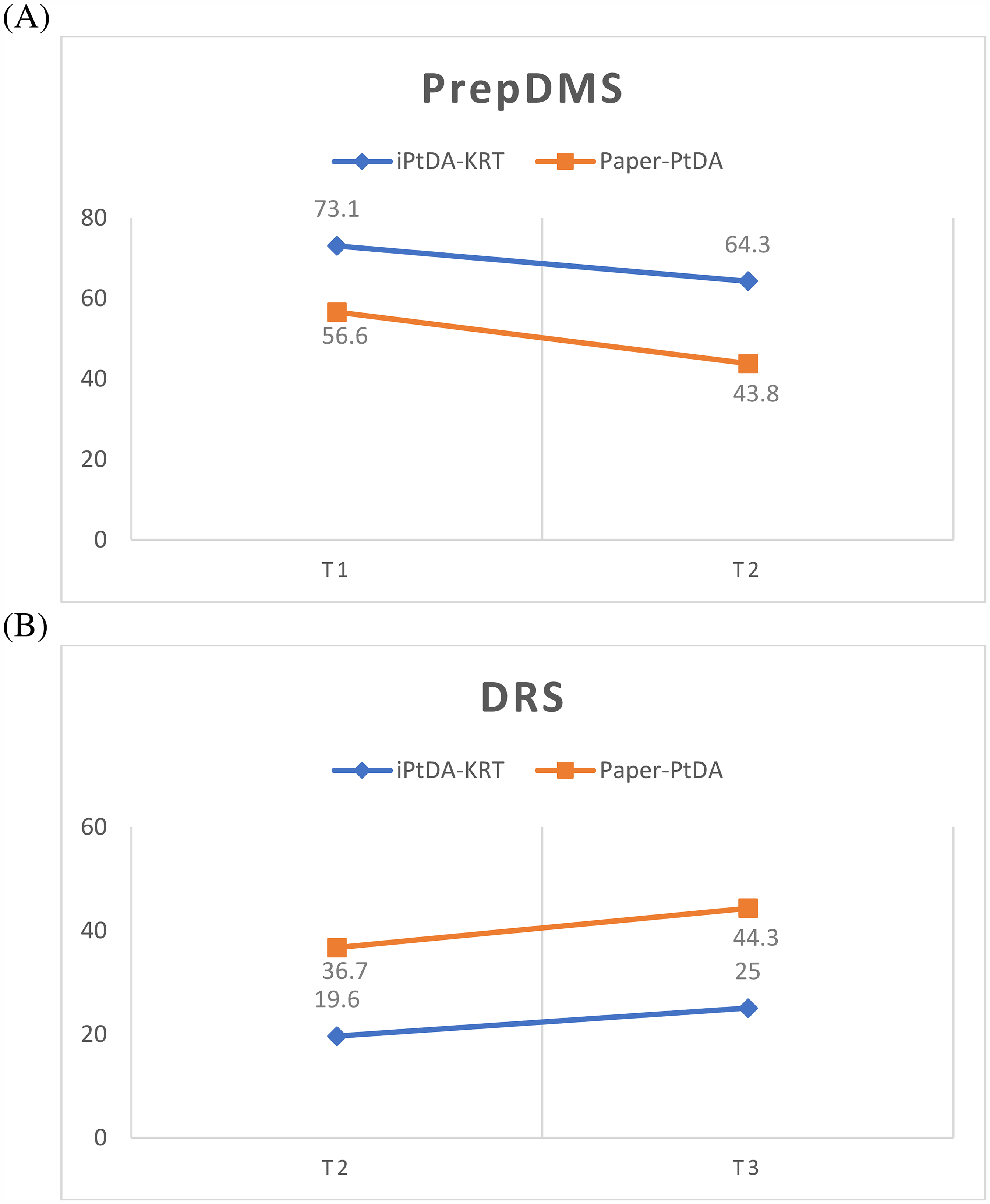
(A) PrepDMS and (B) DRS score at different times.
Intergroup and intragroup differences in intervention effectiveness of PrepDMS and DRS.
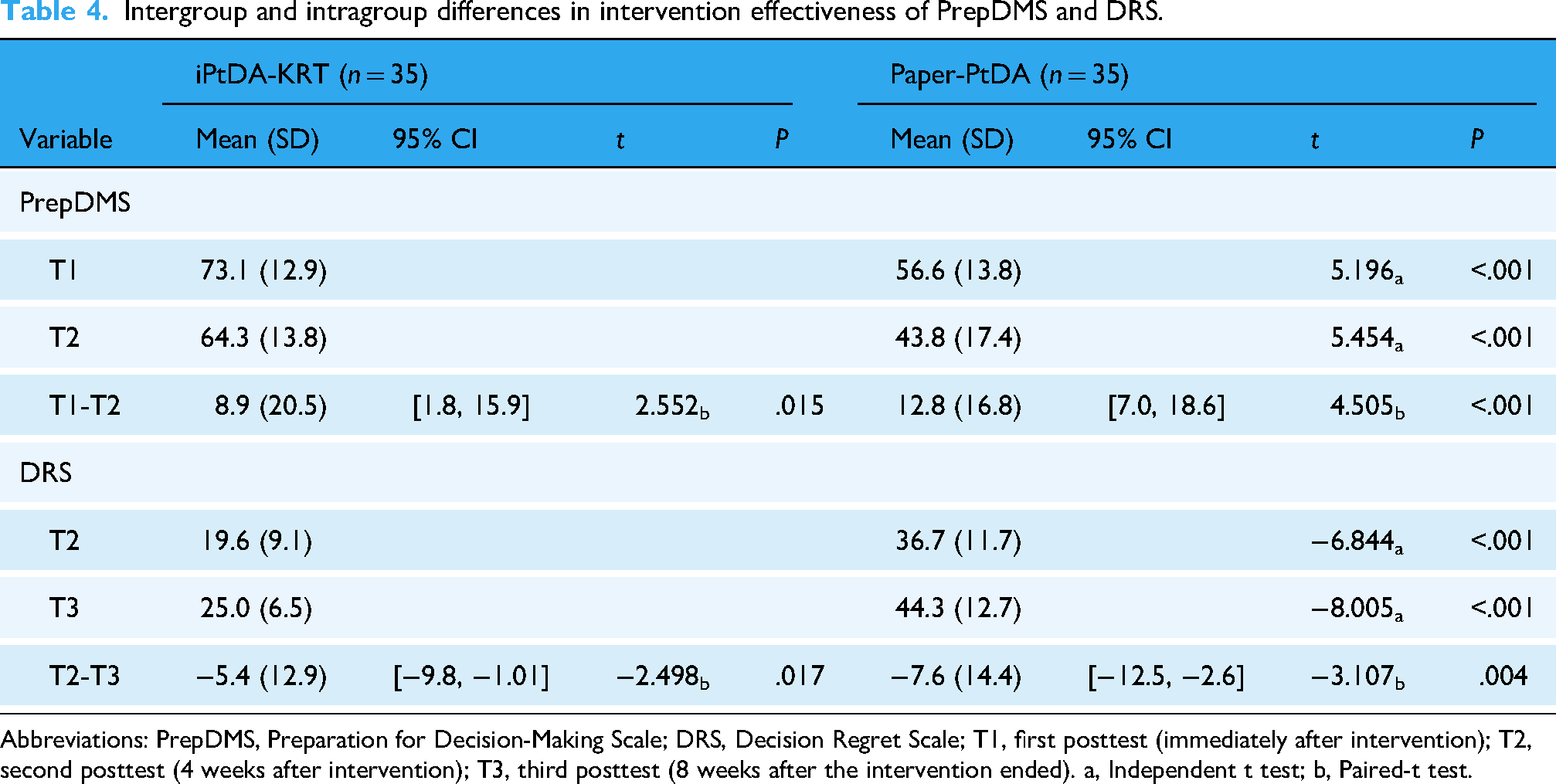
Abbreviations: PrepDMS, Preparation for Decision-Making Scale; DRS, Decision Regret Scale; T1, first posttest (immediately after intervention); T2, second posttest (4 weeks after intervention); T3, third posttest (8 weeks after the intervention ended). a, Independent t test; b, Paired-t test.
The DRS scores at T2 and T3 are displayed in Table 4 and Figure 4B. Significant differences were observed between groups at both time points (
Discussion
This study developed a digital and interactive PtDA for patients with advanced CKD to assist in deciding on KRT options. The iPtDA-KRT App was effective in enhancing patients’ decision self-efficacy and knowledge, improving readiness for decision-making, and reducing decisional conflict and regret, with these benefits sustained over time. These findings support the clinical value of digital PtDAs with simulation features to improve decision quality, achieve sustainable outcomes, and reduce decisional difficulties.
Before the intervention, the experimental group had significantly less knowledge about KRT than did the control group. Additionally, the experimental group had significantly higher baseline decisional conflict scores in the subscales of being informed, clarity of values, and uncertainty compared to the control group. This finding aligns with those of other studies indicating that patients with lower knowledge scores tend to experience greater conflict related to information and uncertainty. 33 Insufficient knowledge may prevent patients from fully understanding the benefits, risks, and side effects of treatment options, thereby increasing value conflicts as they struggle to evaluate the importance of these factors.
Our findings indicate that although the control group experienced significant short-term improvements in decision self-efficacy, knowledge, and decisional conflict immediately after the intervention, these effects diminished over time, suggesting that paper-based PtDAs do not provide lasting benefits. In contrast, the experimental group demonstrated significant and sustained improvements. Other studies have confirmed that PtDAs enhance decision self-efficacy and knowledge while reducing decisional conflict in the short term,7,8 consistent with our findings. We demonstrated that both paper-based PtDAs and digital, interactive PtDAs elicit immediate improvements in decision-making ability. However, the immediate improvements observed in the experimental group were significantly greater than those in the control group. Although both groups experienced a decline in decision self-efficacy 4–8 weeks post-intervention, the decrease in the experimental group was significantly smaller than that in the control group. In the control group, decision self-efficacy was lower, and decisional conflict was higher at the 8 weeks after the intervention ended compared with pretest levels. Regarding knowledge, the experimental group showed a significantly greater increase in knowledge scores compared to the control group post-intervention, with these scores remaining stable over time. These findings suggest that the iPtDA-KRT App not only provides more immediate improvements in knowledge, self-efficacy, and decisional conflict compared to the paper-based PtDA but also delivers longer-lasting effects. This aligns with findings from a meta-analysis by Wu et al. (2024), which demonstrated that web-based PtDAs outperform paper-based PtDAs in reducing decisional conflict and enhancing knowledge. 15 Over time, their efficacy in reducing decisional conflict remains significant. Digital tools can leverage interactive and flexible features to provide more effective support and sustain improvements in patients’ decision-making abilities.
From the perspective of long-term effects, the limitations of paper-based PtDAs are apparent. Paper-based PtDAs elicit only short-term improvements in decision self-efficacy and knowledge, likely because they lack continuous interaction and real-time feedback. Over time, patients may face recurring issues with information asymmetry and reduced self-efficacy, which can lead to increased decisional conflict. By contrast, the interactive features of the iPtDA-KRT App facilitate long-term improvements in knowledge and accuracy in risk perception. Additionally, the iPtDA-KRT App provides simulated scenarios in realistic contexts, helping patients enhance their understanding and familiarity with treatment options while reducing uncertainty about KRT choices. The convenience and flexibility of the iPtDA-KRT App enable patients to maintain higher levels of knowledge and self-efficacy even during follow-up appointments, thereby reducing decisional conflict. This highlights the dynamic adaptability of digital tools in supporting medical decision-making and addressing the personalized needs of patients more effectively.
The experimental group also demonstrated greater improvement in preparation for decision-making compared to the control group. This finding aligns with research emphasizing the benefits of interactive web-based PtDAs in improving patients’ decision readiness.34,35 The experimental group maintained significantly higher preparation for decision-making scores up to 4 weeks post-intervention compared to pre-intervention levels, indicating that the iPtDA-KRT App effectively enhances patients’ readiness to make medical decisions. Furthermore, although preparation for decision-making began to decline 4 weeks post-intervention, the experimental group still had significantly higher scores than the control group at the 8-week follow-up. Notably, while the experimental group continued to outperform the control group in preparation for decision-making at 4 weeks, both groups exhibited a downward trend. This may reflect that patients are prone to experiencing uncertainty or emotional fluctuations during the decision-making process over time. However, the experimental group exhibited a smaller decline, indicating that the iPtDA-KRT App offers sustained decision-making support by helping patients maintain high levels of preparedness, especially in scenarios requiring long-term management or complex treatment decisions.
The experimental group demonstrated significantly greater improvement in decision regret compared to the control group. At both 4 weeks postintervention and 8 weeks after the intervention ended, the experimental group consistently had lower decision regret scores compared to the control group. Although decision regret scores in both groups increased between the 4- and 8-week follow-up visits, the experimental group maintained significantly lower scores, indicating that the iPtDA-KRT App effectively reduces patients’ negative emotions and feelings of regret, providing long-term decision support. This is particularly important because decision regret following complex and life-impacting decisions, such as those related to KRT, can have lasting effects on patients’ mental and physical well-being. Becerra et al. (2016) identified factors associated with decision regret, including higher decisional conflict, lower decision satisfaction, poorer physical health outcomes, and higher levels of anxiety. 36 This aligns with the present study's findings, as the experimental group demonstrated lower decisional conflict scores than the control group post-intervention, which may explain their lower decision regret scores. Thus, the continued support provided by the iPtDA-KRT App not only facilitates informed decision-making but also alleviates patients’ future psychological burdens.
Strengths and limitations
To our knowledge, this study is the first to utilize the iPtDA-KRT App to assist patients with advanced CKD in selecting a KRT modality in Taiwan, with decision quality indicators collected at multiple time points to enable a comprehensive assessment of its impact. However, several limitations should be noted. The small sample size of this pilot study may have introduced bias, and findings from two hospitals in central Taiwan may not be generalizable to other regions. Furthermore, the quasi-experimental design limited our ability to control for potential confounders, and the reliance on self-reported data may have introduced information bias. Future studies should address these limitations by increasing the sample size, expanding the geographic scope, and employing randomized controlled trials to provide more robust evidence.
Conclusions
This study highlights the benefits of digital, interactive PtDAs in enhancing patients’ self-efficacy, knowledge, and decision readiness while reducing decisional conflict and regret. The interactivity and simulated scenarios of the iPtDA-KRT App effectively support informed decision-making. As healthcare becomes increasingly digitalized, integrating PtDAs into clinical practice can enhance decision quality, reduce regret, and promote better health outcomes, highlighting their scalability and potential for widespread implementation.
Supplemental Material
sj-pdf-1-dhj-10.1177_20552076251332832 - Supplemental material for Interactive decision aid on therapy decision making for patients with chronic kidney disease: A prospective exploratory pilot study
Supplemental material, sj-pdf-1-dhj-10.1177_20552076251332832 for Interactive decision aid on therapy decision making for patients with chronic kidney disease: A prospective exploratory pilot study by Jun-Ming Su, Huey-Liang Kuo, Kai-Ling Yang, Chih-Jung Wu and Ya-Fang Ho in DIGITAL HEALTH
Supplemental Material
sj-pdf-2-dhj-10.1177_20552076251332832 - Supplemental material for Interactive decision aid on therapy decision making for patients with chronic kidney disease: A prospective exploratory pilot study
Supplemental material, sj-pdf-2-dhj-10.1177_20552076251332832 for Interactive decision aid on therapy decision making for patients with chronic kidney disease: A prospective exploratory pilot study by Jun-Ming Su, Huey-Liang Kuo, Kai-Ling Yang, Chih-Jung Wu and Ya-Fang Ho in DIGITAL HEALTH
Footnotes
Acknowledgments
We highly appreciate the patients who participated in the study, which contributed greatly to this work. We also thank Cheng-Yu Hsieh for his assistance with system programming.
Ethical considerations
The Research Ethics Committee of China Medical University Hospital in Taiwan approved this study (CMUH109-REC3–085). Once patients agreed to participate, written informed consent was obtained from them prior to study initiation.
Author contributions/CRediT
YFH was responsible for this study's conceptualization, methodology, data analysis, writing, editing, and supervision. JMS was responsible for methodology, data collection and analysis, and writing. YFH and JMS drafted the manuscript. HLK and KLY were responsible for data collection and critically reviewed the manuscript. All authors have agreed on the final version.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science and Technology Council, Taiwan (NSTC 112–2314-B-039-028 and NSTC 113-2410-H-024-012-MY2).
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
