Abstract
Objective
Frailty is a significant health problem that results in adverse outcomes, reduced quality of life, and increased medical and nursing care costs. This study aimed to review the effect of digital health interventions on improving physical activity, physical function, and social function in non-older individuals at risk of frailty.
Methods
Randomized controlled trials of digital health interventions reporting frailty-related outcomes on physical activity, physical function, and social function in non-older individuals at risk of frailty were collected. The literature was reviewed, and the risk of bias was assessed. Meta-analyses were conducted for each outcome, and the certainty of evidence was assessed using Grading of Recommendations Assessment, Development, and Evaluation.
Results
The review included a total of 63 records. The meta-analysis showed that digital health interventions positively affected the volume of physical activity, daily steps, walking test, walking speed, VO2peak, dynamic gait index, timed up and go test, and MOS 36-Item Short-Form Health Survey Social Functioning. However, no significant effect on the time of physical activity was found. Despite the absence of fatal bias, the overall certainty of the evidence was not high because of the small number of studies and participants included in the analysis.
Conclusions
Digital health interventions can improve physical activity, physical function, and social function in non-older individuals at risk of frailty. However, the evidence had limited certainty, and the evidence supporting the effect of digital health intervention is insufficient. Thus, more studies are needed to clarify the effects of these interventions.
Introduction
Frailty is a clinical syndrome characterized by decreased reserve and resistance to stressors caused by the deterioration of multiple physiological systems, leading to increased vulnerability. 1 Frailty affects approximately 17% of community-dwelling older adults and 47% of hospitalized patients, and its prevalence increases with age.2,3 Frailty is associated with adverse outcomes such as death, hospitalization, and falls.4,5
Social frailty is another significant issue. It is defined as a lack of social resources, activities, and self-management skills 6 and can adversely affect physical and cognitive function through limitations in social activities and a range of actions. 7 The adverse outcomes of frailty negatively affect an individual's health and quality of life and lead to increased medical and nursing care costs. Therefore, frailty prevention is crucial to mitigate these issues.
In recent years, digital health interventions have gained attention in the medical field. Digital health refers to the use of information and communications technologies, such as wearable devices, mHealth, and artificial intelligence (AI), to manage disease, mitigate health risks, and promote overall well-being.8,9 Furthermore, digital health is expected to enhance patient care by improving access to healthcare and playing a greater role in disease monitoring and management. 10 In addition, digital health is being used to improve the quality, efficiency, and productivity of medical and long-term care services, where labor shortages are a concern. 11 However, ensuring the security and privacy of patient data remains a challenge in the implementation of digital health. 10 Moreover, its potential effect on health disparities has raised concerns. 12 Although digital health presents both benefits and challenges, further exploration is needed to promote its effective utilization.
The effects of digital health interventions on disease prevention, management, and treatment have been examined across various conditions. A systematic review published in 2023 examined the effect of eHealth interventions for older adults with frailty. 13 However, their effects on physical activity, physical function, and social function in non-older individuals at risk for frailty remain unclear. Non-older individuals may demonstrate higher digital literacy and adaptability than older adults, 14 making digital health interventions potentially more effective. Because ageing, multimorbidity, and functional disability are risk factors for frailty, 15 prevention from midlife is essential. A review of the effects of digital health interventions on preventing frailty in non-older individuals at risk of frailty could be useful for future research and development of intervention programs. Thus, this review aimed to determine the effects of digital health interventions on physical activity, physical function, and social function in non-older individuals at risk of frailty.
Methods
This review complied with Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 Statement (Supplemental Material 1). 16 The review protocol was registered with PROSPERO (Registration no. CRD42023452784).
Criteria for considering studies for this review
This review evaluated the enrolled studies according to the inclusion/exclusion criteria provided in Table 1.
Inclusion and exclusion criteria.

Studies
The study included randomized controlled trials (RCTs) or cluster RCTs. No restrictions were set based on the number of participants, duration of intervention, or country.
Participants
This review included studies that focused on non-older individuals at risk for frailty. Individuals at risk of frailty were defined as those with comorbidities such as lifestyle-related diseases, obesity, cardiopulmonary diseases, cerebrovascular diseases, autoimmune disorders, and chronic pain. In addition, studies were considered to include non-older individuals if the mean age of the participants was <60 years. In this review, this group was defined as “non-older individuals.” Non-older people generally have higher adaptability to and experience with digital health, which leads to greater acceptability and feasibility of the intervention. Studies, where the mean age of the participants was ≥60 years, were excluded because older adults often have lower digital health literacy and difficulties with digital health interventions. 9 Studies with participants aged <18 were also excluded.
Interventions
Studies of digital health interventions aimed at improving frailty-related outcomes such as physical activity, physical function, and social function were also included. No restrictions on the duration of the intervention were set. However, studies on disease-specific digital health interventions, such as those aimed at improving motor or sensory paralysis, involuntary movements, or ataxia, were excluded.
This review included digital health interventions utilizing videoconferencing, sensors, games, virtual reality (VR), applications or websites, AI, and messaging for monitoring, health promotion, exercise therapy, counseling, communication, etc. The control group did not receive intervention or digital health interventions.
Outcome measures
Evaluations of physical activity, physical function, and social function are crucial for frailty-related outcomes. Therefore, the following measures were selected as primary outcomes, using values at the end of the intervention or the closest available time point: volume of physical activity, time of physical activity, daily steps, walking test, walking speed, VO2peak, dynamic gait index (DGI), timed up and go test (TUG), and MOS 36-Item Short-Form Health Survey (SF-36) Social Functioning. VO2peak reflects exercise tolerance, 17 DGI assesses dynamic stability during walking, 18 and TUG evaluates mobility and balance. 19 SF-36 is a tool that assesses health-related quality of life. 20 In this review, the social functioning score obtained using SF-36 was used to evaluate social activity and satisfaction, which are related to social frailty.
To examine the feasibility of digital health interventions, dropouts, and cost-effectiveness were set as secondary outcomes. Dropouts were defined as the number of participants who discontinued the intervention from the time of assignment to the end of the intervention.
Search strategy
A search strategy was developed with a librarian, and published studies were systematically searched. The search was limited to records published in English. The following databases were searched, and all relevant published studies were identified:
MEDLINE (Ovid): coverage date, 1 January 2000–31 March 2023 Web of Science Core Collection (Clarivate Analytics): coverage date, 1 January 2000–31 March 2023 Cochrane Central Register of Controlled Trials (CENTRAL): coverage date, 1 January 2000–31 March 2023
Searches in MEDLINE and the Web of Science Core Collection were last performed on 15 August 2023, and that in the CENTRAL on 5 September 2023. Please refer to PROSPERO (Registration no. CRD42023452784) or Supplemental Material 2 for search strategies.
Data collection and data extraction
After removing duplicates, the remaining studies were screened in two steps based on the inclusion criteria using Rayyan. 21 Seven review authors (YS, KU, YK, MS, IK, YS, and AH) screened the titles and abstracts in the first screening, and RM and MT checked the compatibility of the screening. Full texts of compatible records were obtained, and 17 review authors (MT, YS, KU, YK, MS, IK, YS, AH, MO, TT, HS, YN, KE, SH, NY, DM, and KF) screened the full texts and confirmed the compatibility with RM and MT. RM oversaw these processes and performed the final checks.
Seventeen review authors used Google Sheets to extract data on eligible records, including study design, setting, participants, interventions, and outcomes. Any questions or differences in opinions during the data extraction process were fully discussed and resolved among the review authors.
Data analysis
All data analysis in the meta-analysis was performed using Review Manager 5.4 (The Cochrane Collaboration, Copenhagen, Denmark). Continuous outcomes were presented with 95% confidence intervals (CIs) and mean differences (MDs) to indicate effect sizes, whereas standardized mean differences (SMDs) were used when data varied widely among studies. Dropouts were analyzed as a risk ratio. Only studies that provided postintervention means and standard deviations were included in the meta-analysis. If outcome data were not presented as means and standard deviations, other indicators (median, interquartile range, number of quartiles, standard error, and 95% CI) were converted to means and standard deviations for the analysis.
For the subgroup analysis, the studies included in the meta-analysis were divided into synchronous and asynchronous intervention groups. Synchronous interventions were defined as those in which interventions are implemented in real time, such as direct exercise interventions using video conferences, games, and VR. Asynchronous ones were those that were not implemented in real time, such as self-monitoring and feedback using applications and wearable devices. Subgroup analysis was conducted only for outcomes categorized as synchronous or asynchronous.
Quality assessment
Review authors assessed the risk of bias in each study based on the Cochrane Handbook criteria: 22 random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcomes data, selective reporting, and other biases.
The risk of bias in each domain was classified as “high risk,” “low risk,” or “unclear.” Given the nature of the study, the blinding of participants and assessors was challenged. Therefore, the effect of blinding on outcome measures was considered in the risk-of-bias assessment.
When five or more studies were included in the analysis, funnel plots were created to assess the risk of reporting bias.
I2 statistics were used to measure heterogeneity among the studies included in each analysis.
Summary of findings and assessment of the certainty of the evidence
A summary table of results was created using the following outcomes (Supplemental Table 1): volume of physical activity, time of physical activity, daily steps, walking test, walking speed, VO2peak, DGI, TUG, SF-36 Social Functioning, and dropouts. Cost-effectiveness was not summarized because of insufficient data.
Five considerations (risk of bias, inconsistency, indirectness, imprecision, and publication bias) were used to assess the certainty of the body of evidence for each outcome. 22 GRADEpro GDT was used to evaluate the certainty of evidence for each outcome at the level of “low,” “moderate,” or “high.” 23
Results
Search results
A total of 11,693 records were obtained through database searches performed on 15 August 2023 and 5 September 2023. After removing duplicates, the remaining 9434 records were screened, and 8962 records were excluded based on titles and abstracts. Full texts of the remaining 472 records were obtained and reviewed, resulting in the exclusion of 409 records for the following main reasons: the mean age of the study population was ≥60 years, or the control group also had a digital health intervention. Finally, 63 records were included in the review (Figure 1).24–86 Specifically, 62 studies were included because a single study had two records published.

PRISMA flow diagram.
Characteristics of studies
The included studies were published between 2012 and 2023 and conducted in the following 24 countries: Turkey, United States, Spain, Korea, Netherlands, China, Canada, India, Australia, Sweden, Czech Republic, Norway, Ireland, United Kingdom, Italy, Egypt, Thailand, Denmark, Germany, New Zealand, Pakistan, Belgium, Poland, and Jordan. In total, 7719 participants were included in this review.
Among the 62 included studies, 61 were parallel-group RCTs, and one was a cluster RCT.
The most common characteristics of the study populations were as follows: patients with cardiovascular diseases and multiple sclerosis (10 studies each), followed by chronic pain and cancer survivors (9 studies each), cerebrovascular diseases and overweight/obesity (7 studies each), diabetes or at-risk patients and osteoarthritis (4 studies each), and one study each on patients who recovered from COVID-19, HIV, epilepsy, ankylosing spondylitis, depression, and chronic fatigue syndrome.
The studies included interventions using videoconferencing (
Moreover, 34 studies conducted synchronous interventions with direct exercise interventions, and 29 conducted asynchronous interventions with monitoring and feedback to promote physical activity. The most common duration of intervention was 8 weeks, with a mean of 12.2 weeks.
Among the 63 records included, 32, 36, and 8 reported physical activity outcomes, physical function outcomes, and social function outcomes, respectively. Fifty-nine studies were included in the analysis of the dropouts, and two reported on cost-effectiveness. Please see Table 2 for detailed characteristics of the included studies.
Characteristics of included studies.
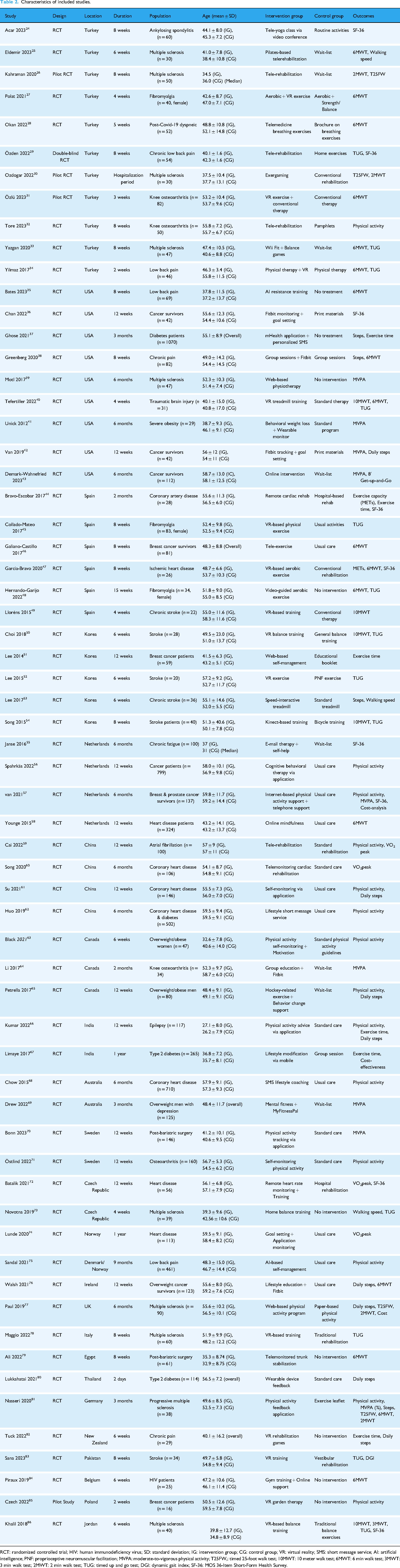
RCT: randomized controlled trial; HIV: human immunodeficiency virus; SD: standard deviation; IG: intervention group; CG: control group; VR: virtual reality; SMS: short message service; AI: artificial intelligence; PNF: proprioceptive neuromuscular facilitation; MVPA: moderate-to-vigorous physical activity; T25FW,: timed 25-foot walk test; 10MWT: 10 meter walk test; 6MWT: 6 min walk test; 3MWT: 3 min walk test; 2MWT: 2 min walk test; TUG: timed up and go test; DGI: dynamic gait index; SF-36: MOS 36-Item Short-Form Health Survey.
Risk of bias in included studies
The risk of bias was assessed based on the Cochrane Handbook criteria. 22 Figure 2 graphically represents the risk of bias for each domain. The risk of “blinding participants and personnel” was high because of the difficulty of blinding in these studies. The risk in other domains was generally not high.

Risk of bias graph.
Effects of interventions
The studies included in the meta-analysis consisted of 8 studies on the volume of physical activity, 7 on the time of physical activity, 7 on daily steps, 20 on walking tests, 12 on walking speed, 4 on VO2peak, 2 on DGI, 16 on TUG, 8 on SF-36 Social Functioning, and 59 on dropouts. Meta-analysis results showed positive effects of interventions on the volume of physical activity (SMD 0.23, 95% CI 0.13–0.32,

Results of meta-analysis on physical activity.
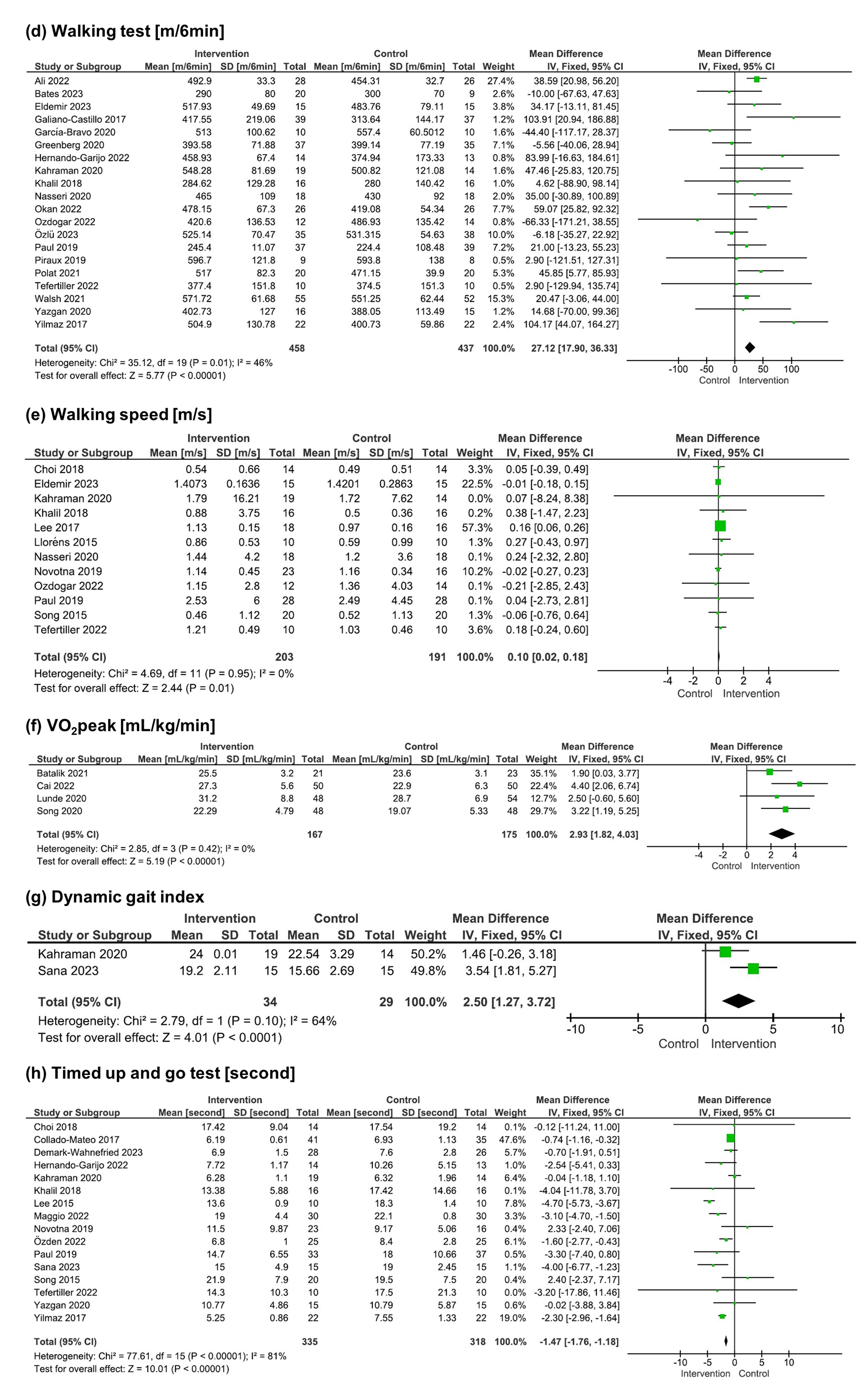
Results of meta-analysis on physical function.

Results of meta-analysis on social function and dropouts.
Studies included in the analysis were divided into synchronous and asynchronous interventions for the subgroup analysis. Synchronous interventions mainly involved aerobic, resistance, and balance trainings using video conferencing, motion sensors, VR, and games. In contrast, asynchronous interventions use wearable devices, applications, and websites for goal setting, monitoring, feedback, and providing information on physical activity. Subgroup analysis results revealed positive effects on the volume of physical activity [SMD 0.61 (95% CI 0.11–1.12) vs SMD 0.21 (95% CI 0.11–0.31)] and SF-36 Social Functioning [SMD 10.05 (95% CI 4.83–15.27) vs SMD 6.51 (95% CI 0.96–12.06)] in both synchronous and asynchronous interventions. Walking tests [MD 31.56 (95% CI 20.92–42.20) vs MD 13.79 (95% CI −4.65 to 32.23)], walking speed [MD 0.10 (95% CI 0.02–0.18) vs MD 0.24 (95% CI −2.32 to 2.80)], VO2peak [MD 2.99 (95% CI 1.81–4.17) vs MD 2.50 (95% CI −0.60 to 5.60)], and TUG [MD −1.52 (95% CI −1.82 to −1.22) vs MD −0.70 (95% CI −1.91 to 0.51)] showed positive effects only on synchronous interventions. Daily steps demonstrated positive effects only on asynchronous interventions [SMD −0.11 (95% CI −0.58–0.37) vs SMD 0.27 (95% CI 0.10–0.43)]. Dropouts were lower in the intervention group for synchronous interventions but higher for asynchronous interventions [0.86 (95% CI 0.63–1.17) vs 1.28 (95% CI 1.14–1.45)]. Because all studies on the time of physical activity and DGI were either synchronous or asynchronous, subgroup analysis could not be conducted. Please see Supplemental Figure 1 for details of subgroup analysis results.
Two studies reported cost-effectiveness. The RCT by Limaye et al. targeted individuals at risk of diabetes. 67 The intervention group received lifestyle advice through messages and websites in addition to face-to-face education, whereas the control group received only face-to-face education. Direct healthcare costs were 3401 Indian Rupees (35.8 GBP) in the intervention group and 2216 Indian Rupees (23.3 GBP) in the control group. Additional costs per individual who were overweight/obese for treatment and prevention (cost-effectiveness) were 10,665 Indian Rupees (112.3 GBP) annually. Paul et al. compared the effects of a web-based exercise program with standard treatment in patients with multiple sclerosis. 77 Costs were 954 and 1076 GBP in the intervention and standard treatment groups, respectively. The intervention group had higher quality-adjusted life-years (QALYs) than the standard treatment group, suggesting the potential for increasing QALYs at a lower cost with the web-based exercise program. However, estimated values from bootstrap analysis showed uncertainty.
Discussion
In this study, the literature was reviewed to determine whether digital health interventions improve physical activity, physical function, and social function in non-older individuals at risk for frailty. A total of 63 records involving 7719 participants were included.
Interpretation of results
Meta-analysis results indicated significant effects of digital health interventions on the volume of physical activity, daily steps, walking tests, walking speed, VO2peak, DGI, TUG, and SF-36 Social Functioning. Particularly, differences exceeding the minimal clinically important difference (MCID) were observed in walking tests (MCID 14–30.5 m), walking speed (MCID 0.10–0.17 m/s), VO2peak (1.82–4.03 mL/kg/min), and SF-36 Social Functioning (MCID 3 points), indicating clinically meaningful effects.87–90 Digital health interventions may enhance health outcomes by increasing user engagement through personalized content and behavior change techniques (BCTs). 91 BCTs are widely employed in interventions aimed at improving health behaviors, including techniques such as goals and planning, feedback and monitoring, knowledge building, and social support demonstrating efficacy in promoting health behaviors. 92 Many of the studies included in this review implemented interventions using these techniques. This meta-analysis included various digital health interventions, including applications, wearable devices, VR, and video counseling. These technologies may have facilitated behavioral change among participants, contributing to improvements in physical activity, physical function, and social function.
However, no significant effect was observed for physical activity time. Studies reporting this outcome implemented interventions involving goal setting, self-monitoring of physical activity, and providing feedback and information via text messages through an application or website. Conversely, several studies provided the control group with paper-based educational materials on physical activity and exercise diaries. A previous study demonstrated that providing exercise guidelines and exercise diaries can effectively promote physical activity, 93 suggesting that a certain level of behavior change may have occurred in the control group. This event may have reduced the differences between the intervention and control groups, potentially resulting in the absence of significant differences. In addition, the methods used to assess physical activity time varied, with some studies employing accelerometers, whereas others relying on self-reported estimates via questionnaires. Measurement discrepancies may have influenced the results, highlighting the need for future studies to adopt standardized, objective measurement methods.
Subgroup analysis by synchronous and asynchronous interventions indicated that the effect on outcomes may vary depending on the content of digital health interventions. Synchronous interventions were potentially effective in improving physical function, and these included aerobic, resistance, and balance training using video conferencing, motion sensors, VR, and games. Balance capacity is associated with walking capacity, and resistance training and aerobic exercise, as well as their combinations, have been reported to improve functional exercise capacity.94–96 These interventions likely enhanced the functional capacity and balance of participants, leading to improved walking capacity and endurance.
On the contrary, asynchronous interventions showed potential effects on increasing physical activity. Interventions involving self-monitoring and feedback promoted behavior change and improved adherence to physical activity over the long term. 97 Thus, asynchronous interventions may have effectively changed habitual physical activity.
In addition, subgroup analysis revealed that both synchronous and asynchronous interventions were effective for social function. Physical frailty and physical function decline can restrict social activities, potentially leading to social frailty.98,99 Synchronous and asynchronous interventions may have increased the physical function and physical activity of participants, thereby encouraging their social activity.
This review also analyzed dropouts to assess adherence to digital health interventions. The results revealed that dropouts were higher in the intervention groups than in the control groups overall. However, in the subgroup analysis, dropouts were lower in the synchronous intervention groups compared to the control groups. Synchronous interventions often included gamification elements such as training with games and VR, potentially enhancing enjoyment and sustaining participation. In addition, the difficulty of blinding participants given the nature of the studies might have led to higher dropouts in the control groups because participants who could not receive interventions may have lost motivation to continue in the study.
Cost-effectiveness could not be analyzed in this review given the limited studies reporting the cost-effectiveness of digital health interventions, leaving the degree of cost-effectiveness unclear.
Quality of the evidence
The overall certainty of the evidence for the outcomes evaluated ranged from very low to moderate. The outcomes with moderate certainty were the volume of physical activity and walking speed. The low certainty was mainly caused by the small number of studies and participants included for each outcome. For details on the overall certainty of the evidence, please refer to Supplemental Table 1.
Only one outcome was downgraded because of the risk of bias. The “blinding of participants and personnel” was deemed high risk in many studies because of the difficulty of blinding participants given the nature of the interventions. On the contrary, the “blinding of outcome assessment” had fewer studies judged as high risk than “blinding of participants and personnel.” Methods such as using assessors not involved in the allocation or intervention were employed to blind outcome assessors. Many studies were judged as having an unclear bias for “allocation concealment” and “selective reporting” owing to the lack of clear descriptions of the allocation methods or the inability to reference protocols. The bias risks for “random sequence generation” and “incomplete outcome data” were generally low.
Four outcomes were downgraded because of the risk of inconsistency, attributed to the high heterogeneity of the interventions. In addition, this review included studies not providing interventions in the control group and studies where the control group received interventions via face-to-face or paper-based methods. The high heterogeneity of interventions in the control group was also a contributing factor. Thus, limiting the content and duration of interventions could improve the risk of inconsistency.
No outcomes were downgraded because of the risk of indirectness. The studies included in this review limited the mean age of the study population to <60 years, resulting in a low risk of indirectness.
All outcomes were downgraded because of the risk of imprecision. Generally, few studies and participants were included in the analysis. This issue could be improved by increasing the number of studies.
Three outcomes were downgraded because of publication bias. VO2peak and DGI could not be assessed for publication bias owing to the small number of studies included in the analysis.
Potential biases in the review process
Review authors screened, extracted data, and assessed risks for this review. Because the included studies were limited to English records, there may have been studies in other languages that were not identified. Bias in study design is unlikely to have occurred because the studies were limited to RCTs. We were able to focus on the effects of digital health intervention as the inclusion criterion was studies that compared an intervention group using digital health with a control group not using digital health.
Overall completeness and applicability of evidence
The meta-analysis showed significant effects for the volume of physical activity, daily steps, walking test, walking speed, VO2peak, DGI, TUG, and SF-36 Social Functioning, whereas no significant effect was found for the time of physical activity. After assessing the certainty of evidence, the volume of physical activity and walking speed were rated as moderate, whereas the walking test was rated as low. This indicates a certain degree of confidence in the study results. In contrast, the certainty levels of evidence for the time of physical activity, daily steps, VO2peak, DGI, TUG, and SF-36 were evaluated as very low. When the certainty of the evidence is very low, intervention effects cannot be determined, even if significant effects are observed.
The small number of studies and participants included in the meta-analysis for each outcome inhibited generalization of the results of this review to a broader population. In addition, intervention durations among studies; thus, whether each study had a sufficient intervention duration to increase physical function is unknown.
Of the studies included in this review, 41, 17, and 4 were conducted in high-, upper-middle, and lower-middle-income countries. None of the studies were conducted in low-income countries. Low economic status was reported to hinder access to the Internet and digital health services and reduce digital health literacy. 14 Therefore, whether the results of this review can be applied to low-income countries is unclear.
Comparison with other reviews
To our knowledge, this is the first review that examined the effect of digital health interventions on improving physical activity, physical function, and social function in non-older individuals at risk for frailty. Two previous reviews have examined the effects of digital health interventions on older adults with pre-frailty or frailty.
Esfandiari et al. presented the effects of telemedicine interventions on physical function, quality of life, and frailty in older adults with pre-frailty or frailty. 100 They showed that telemedicine had a small effect on improving physical function and behavioral performance in older adults with pre-frailty or frailty (SMD 0. 31, 95% CI 0.15–0.47). However, the certainty of evidence was low. The results for physical function were consistent with those of the present review; however, the characteristics of participants and outcome measures differ. Therefore, the results cannot be directly compared.
Han et al. reviewed the effects of eHealth interventions on physical activity and physical performance, health-related quality of life, and activities of daily living in older adults with frailty. 13 Moderate-to-vigorous physical activity times increased significantly only in the intervention group. Walking speed increased in both groups; however, the rate of increase was higher in the intervention group than in the control group. Few studies were available for inclusion in the review for either outcome, and no meta-analysis was conducted. The effects on walking speed were consistent with the results of this review but not on physical activity time. However, Han et al. could not compare their results to our findings because they studied older adults with frailty and did not conduct a meta-analysis of these outcomes.
Limitations of the review
Despite the difference of approximately 20 days in the search periods across databases, it was unlikely to have significantly affected the included literature. This review targeted studies published up to March 31, 2023, which means that studies published thereafter may not have been included. To assess the potential effect of newly published studies, a preliminary search was conducted for articles published from 31 March 2023, to the present. Because we were unable to find any studies that fully met the inclusion criteria for this review, updating the search period is unlikely to significantly affect the overall findings. Therefore, we plan to address this issue in a future update of this review. The studies included in this review were conducted in high- or middle-income countries; therefore, whether the results would be generalizable in low-income countries is unknown. This review defined non-older individuals as those aged <60 years, considering factors such as digital literacy and adaptability to digital technologies. However, as the age group of who regularly use digital technologies has been expanding, it might be better to include older age groups in future reviews. In addition, this review included both purely digital health interventions (e.g. VR-based interventions) and digitally mediated interventions with human interaction (e.g. physical activity programs via videoconferencing). The effects of these different types of interventions may vary. Although subgroup analyses based on whether the interventions were synchronous or asynchronous were conducted, further categorization was difficult given the limited number of studies. The low number of studies and high heterogeneity levels of interventions limited the certainty of the evidence. These issues must be addressed with an expansion of the range of countries where studies are conducted and an increase in the number of studies.
Conclusions and implications
The results of this review indicate that digital health interventions have the potential to improve physical activity, physical function, and social function in non-older individuals at risk for frailty. However, the certainty of the evidence is low because of the small number of studies and participants included in the analysis. Higher-quality RCTs are needed to increase the certainty of the evidence, and more studies must be included in the analysis. In addition, studies must be conducted in a wider range of countries to broaden the applicability of evidence and examine the effects of interventions in countries with different economic backgrounds. This review lacked information on the cost-effectiveness of digital health interventions, which needs to be investigated to confirm the practical feasibility of these interventions.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251328566 - Supplemental material for Digital health interventions for non-older individuals at risk of frailty: A systematic review and meta-analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076251328566 for Digital health interventions for non-older individuals at risk of frailty: A systematic review and meta-analysis by Momoko Tohyama, Ryo Momosaki, Yuka Shirai, Kenta Ushida, Yuki Kato, Miho Shimizu, Issei Kameda, Yuya Sakurai, Asuka Hori, Masatsugu Okamura, Takahiro Tsuge, Hiroki Sato, Yuki Nakashima, Kaori Endo, Shota Hayashi, Norio Yamamoto, Daisuke Matsumoto, Kenichi Fudeyasu and Hidenori Arai in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251328566 - Supplemental material for Digital health interventions for non-older individuals at risk of frailty: A systematic review and meta-analysis
Supplemental material, sj-docx-2-dhj-10.1177_20552076251328566 for Digital health interventions for non-older individuals at risk of frailty: A systematic review and meta-analysis by Momoko Tohyama, Ryo Momosaki, Yuka Shirai, Kenta Ushida, Yuki Kato, Miho Shimizu, Issei Kameda, Yuya Sakurai, Asuka Hori, Masatsugu Okamura, Takahiro Tsuge, Hiroki Sato, Yuki Nakashima, Kaori Endo, Shota Hayashi, Norio Yamamoto, Daisuke Matsumoto, Kenichi Fudeyasu and Hidenori Arai in DIGITAL HEALTH
Supplemental Material
sj-pdf-3-dhj-10.1177_20552076251328566 - Supplemental material for Digital health interventions for non-older individuals at risk of frailty: A systematic review and meta-analysis
Supplemental material, sj-pdf-3-dhj-10.1177_20552076251328566 for Digital health interventions for non-older individuals at risk of frailty: A systematic review and meta-analysis by Momoko Tohyama, Ryo Momosaki, Yuka Shirai, Kenta Ushida, Yuki Kato, Miho Shimizu, Issei Kameda, Yuya Sakurai, Asuka Hori, Masatsugu Okamura, Takahiro Tsuge, Hiroki Sato, Yuki Nakashima, Kaori Endo, Shota Hayashi, Norio Yamamoto, Daisuke Matsumoto, Kenichi Fudeyasu and Hidenori Arai in DIGITAL HEALTH
Supplemental Material
sj-docx-4-dhj-10.1177_20552076251328566 - Supplemental material for Digital health interventions for non-older individuals at risk of frailty: A systematic review and meta-analysis
Supplemental material, sj-docx-4-dhj-10.1177_20552076251328566 for Digital health interventions for non-older individuals at risk of frailty: A systematic review and meta-analysis by Momoko Tohyama, Ryo Momosaki, Yuka Shirai, Kenta Ushida, Yuki Kato, Miho Shimizu, Issei Kameda, Yuya Sakurai, Asuka Hori, Masatsugu Okamura, Takahiro Tsuge, Hiroki Sato, Yuki Nakashima, Kaori Endo, Shota Hayashi, Norio Yamamoto, Daisuke Matsumoto, Kenichi Fudeyasu and Hidenori Arai in DIGITAL HEALTH
Footnotes
Acknowledgements
We would like to thank Hiromi Rakugi, Masahiro Akishita, Shosuke Satake, Yasumoto Matsui, Hiroyuki Shimada, Keisuke Maeda, Sumito Ogawa, Katsuya Iijima, Koichi Yamamoto, Hiroshi Akasaka, Minoru Yamada, Koichi Kozaki, Ken Sugimoto, Hidetaka Wakabayashi, Hyuma Makizako, Hidehiko Komine, Keiko Yamada, Tatsuro Inoue, and the representatives of the citizens, Misao Kawakami and Katsuko Bando, for their support in planning our systematic review.
Contributorship
MT, RM, YS, and HA: conceptualization. MT, RM, YS, and HA: methodology. MT, RM, YS, KU, YK, MS, IK, YS, AH, MO, TT, HS, YN, KE, SH, NY, DM, and KF: formal ANALYSIS. MT, RM, YS, KU, YK, MS, IK, YS, AH, MO, TT, HS, YN, KE, SH, NY, DM, and KF: investigation. RM: resources. MT and RM: writing—original draft. YS, KU, YK, MS, IK, YS, AH, MO, TT, HS, YN, KE, SH, NY, DM, KF, and HA: writing—review & editing. RM: funding acquisition.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by AMED (Grant Number JP22rea522005).
Guarantor
RM.
Other identifying information
The review protocol was registered with PROSPERO (registration number CRD42023452784).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
