Abstract
Objective
To evaluate the cost-effectiveness of a digital supportive healthcare pathway in patients with type 2 diabetes mellitus (T2DM) compared to usual care.
Methods
An age-dependent Markov model was applied from a healthcare payer perspective projecting results of a clinical trial study over a time horizon of 22 years assuming a continuous implementation of the intervention every year, 24/7. The setting was Flanders (Belgium). One-way and probabilistic sensitivity analyses were performed.
Results
The hybrid care pathway led to a quality-adjusted life year (QALY) gain of 5.97, while the costs increased with €663,036. This resulted in an incremental cost-effectiveness ratio of €110,989/QALY. With a cost-effectiveness threshold of 45,000€/QALY, the hybrid care pathway was found not cost-effective compared to the usual care trajectory. Sensitivity analyses showed that over 50% of iterations exceeded the threshold, with a cost-effectiveness probability of 13.12% at €45,000/QALY.
Conclusions
This cost-effectiveness analysis indicates that a hybrid care pathway is unlikely to be a cost-effective approach compared to the standard care trajectory in patients with T2DM. Nevertheless, the exploration of technology-driven healthcare pathways are vital for advancing patient well-being, emphasizing the need for further research to optimize resource utilization and enhance outcomes effectively.
Introduction
With changing lifestyles and the increasing number of obese people, the prevalence of type 2 diabetes mellitus (T2DM) is rapidly rising worldwide. 1 The latest International Diabetes Federation Atlas reported that the global prevalence of diabetes has reached 10.5%, with over 90% of people with diabetes having T2DM. 2 T2DM is a chronic metabolic disease requiring lifelong medical care to reduce the risk of life-threatening health complications. 3 Uncontrolled diabetes is associated with diminished life expectancy and health-related quality of life, and elevated treatment costs. 4 Diabetes self-management combined with appropriate lifestyle changes and ongoing guidance from healthcare providers is crucial in improving health outcomes in patients with T2DM and in reducing healthcare expenditures.
With the goal of improving both glycemic control and outcomes through self-management, various digital health interventions have been developed in recent years as an alternative or additional method for conventional diabetes monitoring. Telemedical devices, such as mobile apps, record and transmit self-monitored blood glucose levels to clinicians in remote locations in order to monitor patients’ health. The clinical effectiveness of telemedicine demonstrated positive effects on HbA1c levels (i.e. blood glucose level) in comparison with traditional diabetes care.5–8 In addition, it has been reported that telemedicine has the potential to decrease costs on the health system. 9 A recent systematic review, summarizing health economic evidence, concluded that digital health interventions for T2DM are cost-effective and, in some cases, even cost-saving. 10
With T2DM being more prevalent in older populations who are more often unfamiliar with technologies utilized in telemedicine and the higher risk of digital exclusion in individuals of lower socioeconomic status, the existing standard care for T2DM patients should not be entirely digitalised.11,12 Hence, a hybrid care pathway, in which T2DM patient care at home with face-to-face clinical guidance is alternated with digital support, was set up. The clinical and cost-effectiveness of such a T2DM program has not been previously investigated in Belgium. The present study aims to evaluate the cost-effectiveness of a hybrid care pathway in patients with T2DM and to compare it with the usual care model for T2DM, in Flanders (Belgium).
Methods
Data are reported following the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) 2022 statement.13,14
Markov model
A Markov decision-analytic model was used to evaluate the cost-effectiveness of a supportive digital care pathway for T2DM. The model structure was based on earlier published Markov models of T2DM 15 and has been further developed using Microsoft Excel® 2016. To ensure the model's internal validity, we conducted extensive checks, including sensitivity analyses and expert reviews, confirming its robustness and alignment with established trends in diabetes cost-effectiveness studies. Taking into account the potential irreversibility of the diabetes disease progression, the model includes five health states (Figure 1): (1) T2DM without complications, (2) T2DM with microvascular complications (i.e. complications affecting small blood vessels such as end-stage renal disease, retinopathy, neuropathy, and foot ulcers), (3) T2DM with macrovascular complications (i.e. primarily diseases of the coronary arteries, peripheral arteries, and cerebrovascular such as coronary heart disease and ischemic stroke), (4) T2DM with micro- and macrovascular complications, and (5) death. Health state transitions were restricted to maintenance of the current health state or deterioration. Transitions between health states were allowed once a cycle. The setting was in Flanders (Belgium). We used a healthcare payer perspective, including direct medical costs for the national healthcare insurance in Belgium (National Institute for Health and Disability Insurance (NIHDI)) and for patients (co-payments). 16 The cost-effectiveness analyses allowed to project effects over a longer time horizon than the duration of the trial. As care trajectories for chronic diseases mostly have consequences over a patient's lifetime, a 22-year's (≈lifetime) time horizon was adopted, based on the mean age (64.4 years) of study participants and their life expectancy (86.3 years) in Belgium. 17 The cycle length was 1 year. Discount rates were 3.0% for costs and 1.5% for health outcomes. 18

Structure of the Markov model.
The incremental cost-effectiveness ratio (ICER) is the main outcome of cost-effectiveness analysis and can be expressed as costs per quality-adjusted life year (QALY) gained. A QALY measures both quantity (i.e. number of years lived in a certain health state) and health-related quality of life (i.e. utility value, ranging between 0 “death” and 1 “perfect health”). The ICER is the ratio between the difference in costs and the difference in QALYs between the intervention cohort and the control cohort:
Intervention
A new hybrid care pathway for T2DM was developed (Figure 2). In addition to the standard care trajectory (explained in the section “comparator”), the hybrid program is expected to be more efficient and effective through patient self-monitoring, 24/7 monitoring of parameters, and just-in-time interventions. The clinical trial (n = 50) was carried out for 9 months. During this period, patients were medically monitored remotely via a digital app. Patients were requested to measure their blood glucose levels using a self-monitoring blood glucose device at predetermined moments during the day. Patients were asked to record the values in a validated m-health app. Parameters were 24/7 monitored, and in case of hypo- and hyperglycemia, a medical intervention was performed by trained nurse operators from an alarm dispatch center who were promptly connected with diabetes educators. These interventions include short phone calls or an educational session via consultation between diabetes educator and patient. In addition, patients received a blood pressure device and a pedometer (i.e. device that counts the number of steps taken) in order to monitor blood pressure and physical activity, respectively. Patients were trained with information about hypertension/physical activity, the procedure for self-monitoring, advice on equipment and its proper use, and interpretation of the data. Prior to the intervention, patients’ health were assessed by analysis of the following parameters: blood glucose, blood pressure, blood lipids, body mass index, and physical activity. These parameters were also examined at the end of the intervention.

Flowchart of participants in intervention and control groups.
Inclusion criteria for T2DM participants were being included in the usual care trajectory for T2DM (explained in the section “comparator”), being an adult (≥18 years), having sufficient understanding of Dutch, and having digital skills (i.e. having an own smartphone and/or tablet and internet connection, passed a digital proficiency test). Finally, patients were excluded if they had type 1 diabetes, were pregnant, had an active desire to have children in the near future, or if they had cognitive impairment or severe comorbidities (cardiovascular and/or renal disease). Written informed consent was obtained from all the subjects prior to study initiation. Eventually, the Markov model started with a cohort consisting of 70% of diabetic patients without complications, 24% with minor complications (e.g. retinopathy), and 6% with major complications (e.g. cardiovascular disease).
The sample size was determined based on HbA1c levels, a key indicator of glycemic control in diabetes management, with a target level of 7% (53 mmol/mol). Using literature-based parameters—single mean, coefficient of variation (0.5–2% 20 ), and significant difference (0.5% or 5 mmol/mol 21 )—47.18 participants were required to detect a 0.5 mmol/mol difference with 90% power and a 5% significance level. To account for potential dropout, 50 participants were included in both the intervention and control group, totaling 100 participants.
Comparator
The control group (n = 50) only received the usual care trajectory for T2DM. In 2009, the NIHDI in Belgium introduced “standard care trajectories” for T2DM with the aim to organize and coordinate patient monitoring; to stimulate interaction and collaboration between patients, general practitioners (GPs), specialists and other caregivers; and to optimize quality of care. 22 The benefits of a care trajectory for patients include receiving high quality care; having access to information, materials (i.e. blood glucose device every 3 years, 150 glucose strips every 6 months, 100 lancets every 6 months) and consultations; and receiving financial benefits (i.e. full reimbursement of consultations with the GP, endocrinologist, and diabetes educators; and reimbursement of two treatments per year with a podiatrist, and two sessions per year with a dietician). Patients with T2DM are eligible for a standard care trajectory if they receive one or two insulin injections a day or having inadequate regulation despite receiving the maximum dose of oral antidiabetics. The following patients are excluded: patients with type 1 diabetes and pregnant women or women with a pregnancy wish.22,23
Data input
An overview of the included input parameters is provided in Table 1.
Overview of input parameters used in the Markov model.

Age-dependent.
T2DM_0: type 2 diabetes mellitus without complications.
T2DM_MICRO: type 2 diabetes mellitus with microvascular complications.
T2DM_MACRO: type 2 diabetes mellitus with macrovascular complications.
T2DM_MICRO_MACRO: type 2 diabetes with micro- and macrovascular complications.
CI: confidence interval; NA: not applicable; RRR: relative risk reduction; SE: standard error.
Transition probabilities
Transition probabilities between disease states were based on a recent study using a multistate model for T2DM progression demonstrating the probability of transfer to microvascular or macrovascular complications. 24 To derive a single annual transition probability for the model, an average probability was calculated, weighted by the distribution of transitions over time, ensuring consistency with long-term trends in disease progression. To address concerns about generalizability, the applicability of the reported transition probabilities was carefully reviewed. Age-specific mortality probabilities were derived from nationally available mortality data 25 and were multiplied with the diabetes-specific mortality probabilities. 24
Health outcomes
Age-dependent utilities for patients with T2DM were retrieved from the Belgian Health Interview Survey 2018, which administered the EQ-5D-5L questionnaire.26,27 Utility decrements for micro- and macrovascular complications were sourced from the literature. 31 A utility value of zero was applied for the health state “death.”
Cost data
Disease cost data, expressed in euros (€), were sourced from the literature. As we used a public healthcare payer perspective, only direct medical costs were taken into account. The healthcare cost of diabetes without complications was calculated by Odnoletkova et al. 29 The healthcare cost associated with microvascular complications was calculated based on the fact that the presence of microvascular complications leads to a 70% increase in costs. The cost for macrovascular complications is found to be twice as high as the cost of diabetes without complications. Having both micro- and macrovascular complications appears to increase costs by 3.5-fold over diabetes without complications. 30 All costs were updated to 2023 Euros by using the Belgian healthcare inflation rates. 32 No costs are linked to the state of “dead.”
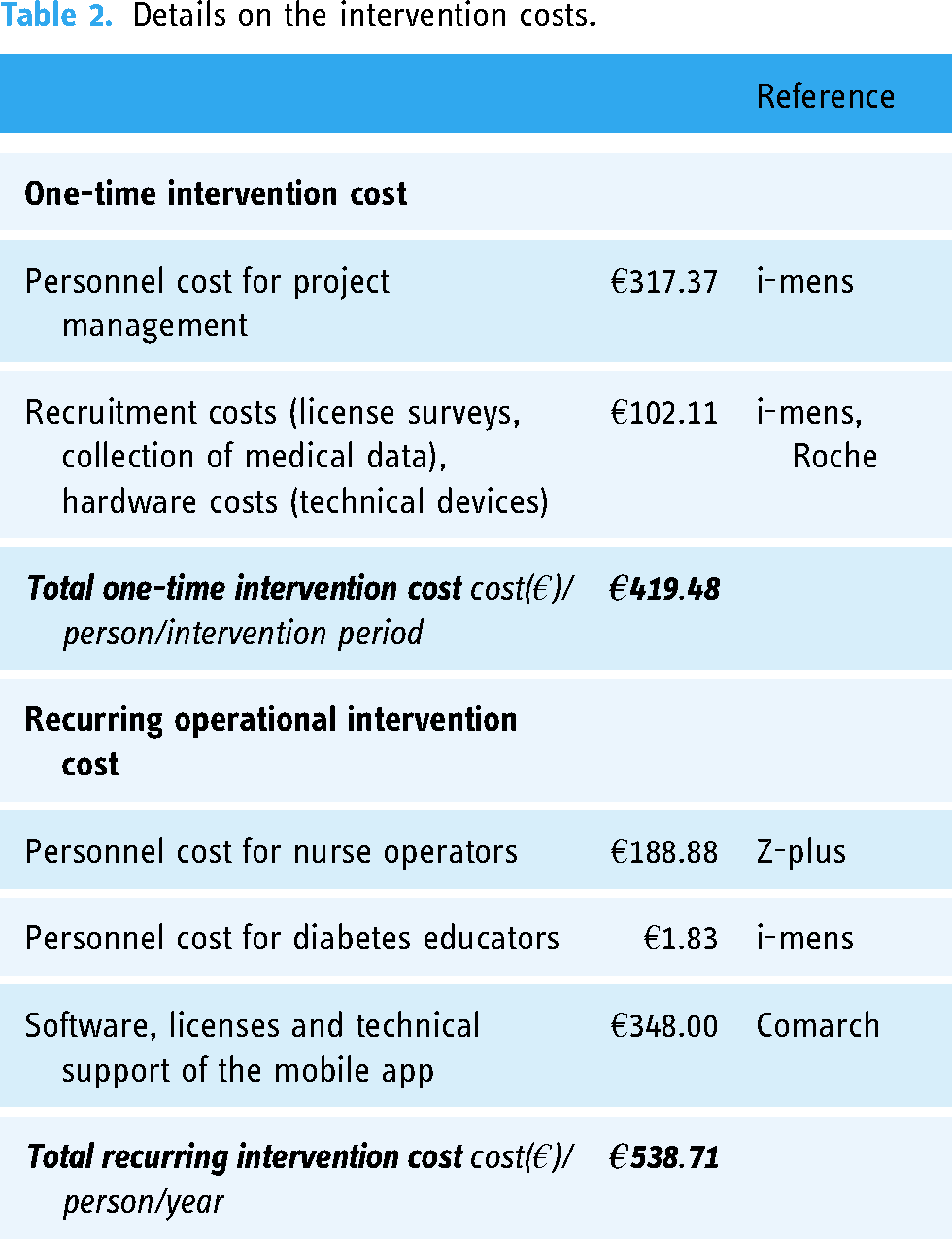
The mean one-time intervention cost was €419.48 per patient for the whole intervention period. These costs included recruitment costs, cost of technical devices, collection of medical data, and personnel costs of the project managers. The recurring operational intervention cost was €538.71 per patient per year. These costs included technical support of the app (software, licenses), personnel cost for nurse operators, and time of the diabetes educators. Costs were prospectively documented during the trial, based on individual time and contractual prices. Costs were calculated by dividing the total costs by the number of participants in the intervention group. Potential variability of the intervention costs was explored in the sensitivity analysis. Details on the intervention costs can be found in Table 2.
Details on the intervention costs.
Effect of the intervention
The primary outcome measure of the intervention was the change in HbA1c level from baseline to 9 months. The measurement of HbA1c is internationally recognized to be a valid predictor for future diabetes-related complications. 33 To simulate the effect of the intervention, transition probabilities were reduced by a relative risk reduction (RRR). Data on RRR were based on the United Kingdom Prospective Diabetes Study, which found that each 1% reduction in HbA1c (equivalent to a change of 10.9 mmol/mol) over 10 years is associated with relative reductions in risk of 37% for microvascular complications, 14% for myocardial infarctions, and 21% for deaths related to diabetes.28,34 The effect of the intervention was modeled only during the years in which the intervention was applied. Specifically, the HbA1c change and corresponding RRR were modeled annually for each year the intervention was administered.
After completion of the intervention, at 9 months, the mean (95% confidence interval) between-group difference in HbA1c change (%) was 0.242 (−0.048 to 0.532, P = 0.051).
Sensitivity analyses
Both one-way sensitivity analysis (Tornado diagram) and probabilistic sensitivity analysis (Monte Carlo simulation) were conducted to test the robustness of the model. 35 The former evaluates the effect of each input parameter on the ICER, by increasing and decreasing the value of each parameter separately by 30%. The probabilistic sensitivity analyses assess the uncertainty for all parameters by varying them simultaneously (10,000 iterations), according to the input variables’ probability distributions.
Cost data were modelled using a gamma distribution, utilities and transition probabilities using a beta distribution, and risk reductions using a log-normal distribution. 36
Results
The hybrid care pathway led to a QALY gain of 5.97, while the costs increased with €663,036 from a healthcare payer perspective. This resulted in an ICER of €110,989/QALY. With a cost-effectiveness threshold of 45,000€/QALY, the hybrid care pathway was found not cost-effective compared to the usual care trajectory (Table 3).
Base case results per 100 patients over 22 years.

QALY: quality-adjusted life years; ICER: incremental cost-effectiveness ratio.
Sensitivity analyses
The results of the one-way sensitivity analyses are presented in Tornado diagrams (Figure 3) and show the most important determinants that contribute to the lack of cost-effectiveness: (1) reduction in HbA1c, (2) annual cost of the hybrid care pathway, (3) RRR for T2DM with micro- and macrovascular complications, and (4) cost of standard care for T2DM with micro- and macrovascular complications.

One-way sensitivity analyses.
The results of the probabilistic sensitivity analyses, generated through 10,000 simulations, are presented in a cost-effectiveness plane (Figure 4). More than half of the dots fall above the predetermined willingness-to-pay threshold (€45,000/QALY), showing robustness of the model. More specifically, 9500 dots are in the north-east quadrant, 492 in the north-west quadrant, 0 in the south-west quadrant, and 8 in the south-east quadrant. In Figure 5, the cost-effectiveness acceptability curve shows that the probability of the hybrid care pathway to be cost-effective (compared with the usual care trajectory) was 13.12% at the threshold of €45,000/QALY.

Results of the probabilistic analyses.
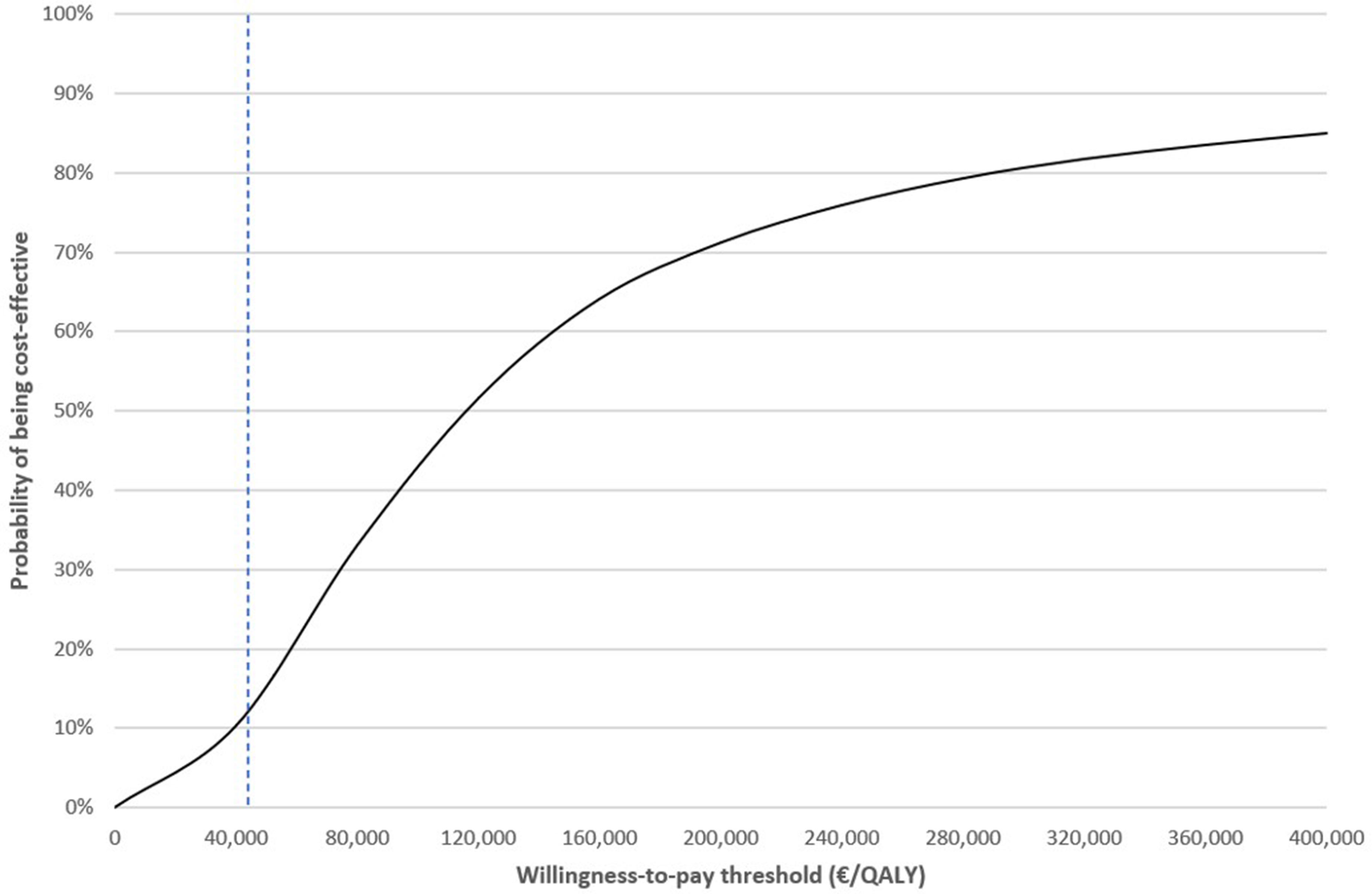
Cost-effectiveness acceptability curve.
Discussion
This cost-effectiveness analysis evaluated the economic implications of a hybrid care pathway in patients with T2DM compared with usual care over a 22-years’ time horizon. During these years, it was assumed that intervention group dyads received this hybrid support every year, day in and out. From a healthcare payer perspective in Flanders (Belgium), the base case analysis showed an ICER of €110,989/QALY. Given a threshold value of €45,000/QALY, it can be concluded that the hybrid care pathway is highly unlikely to be cost-effective compared with the standard care trajectory in people with T2DM. However, deterministic analyses, while informative, may not fully capture the uncertainty inherent in non-linear models.37,38 Hence, the sensitivity analyses showed that over 50% of iterations exceeded the threshold, with a cost-effectiveness probability of 13.12% at €45,000/QALY.
Although mobile health interventions have already shown their clinical effectiveness in the treatment of T2DM,39,40 there are only a limited number of cost-effectiveness studies showing that mobile healthcare is cost-effective. 41 More importantly, to the best of our knowledge, there is currently no published literature on the cost-effectiveness of hybrid care pathways, which combine diabetes education with telemonitoring and remote care. Although mobile healthcare has been proven to be cost-effective, our research does not demonstrate these results. The main drivers for the hybrid care pathway not being cost-effective are the high annual recurring costs for the software (i.e. the annual purchase price for patients of the smartphone application) and the continuous support (24/7) of nurse operators. Reducing these costs could potentially make the hybrid care process cost-effective, for example, by economies of scale though widespread adoption of the mobile app. In addition, technological advancements, such as artificial intelligence (AI) and machine learning, may offer promising opportunities to reduce these costs. For instance, automating telemonitoring processes could minimize the need for constant manual supervision, allowing healthcare providers to allocate resources more efficiently.42–44 Additionally, the study also revealed limited health benefits, which turned out to be another major driver for the lack of cost-effectiveness. Due to the short duration of the clinical trial, it was expected that no strong significant improvement in HbA1c would occur in the intervention group. However, future research will investigate the hybrid care pathway over an extended duration and with a larger sample size. Ideally, future studies should explore AI-driven data analysis to enable more personalized treatment recommendations. Additionally, gamification and adaptive interfaces in mobile health apps could boost patient adherence, ultimately leading to better health outcomes.
Although the intervention group experienced reductions in HbA1c levels that led to a reduced risk of diabetes-related complications and potentially improved life expectancy and QALYs, the additional costs of the intervention were not sufficiently offset by cost savings from preventing diabetes-related complications. Consequently, it is important to note that although the hybrid care pathway provided limited clinical benefits for individuals with T2DM, it failed to deliver value for money for healthcare payers in Belgium. This underlines the uncertainty surrounding the implementation of such innovative care pathways in optimizing both clinical outcomes and cost-effectiveness in the management of T2DM.
However, a threshold analysis (Supplemental file) was conducted to ascertain the conditions under which the hybrid care pathway could be cost-effective. Specifically, in order for the hybrid care pathway to be cost-effective compared to usual care, the reduction in HbA1c in the intervention group has to be greater than or equal to 0.421%. Furthermore, a reduction greater than 0.848% would result in the intervention being dominant compared to usual care. These thresholds provide valuable insights into the efficacy and cost-effectiveness of the hybrid care pathway in managing HbA1c levels. Additionally, it was determined that the annual costs associated with the hybrid care pathway would need to be reduced by 52.6% in order to achieve cost-effectiveness, given the current clinical effect. This highlights the significance of cost considerations in evaluating the overall feasibility and sustainability of implementing the hybrid care pathway.
However, it is important to recognize that while several studies have demonstrated the cost-effectiveness and potential cost savings associated with mobile healthcare interventions,10,45–49 a significant portion of these studies demonstrate inadequate reporting quality and important methodological shortcomings.10,41 Specifically, there is a lack of transparency regarding the inclusion of costs and the methods used for cost measurement, leading to results where interventions are proposed with questionably low intervention costs. 10 Moreover, many of these studies focus solely on digital interventions, which tend to have lower resource demands compared to hybrid care pathways like ours that combine digital and in-person support. These factors, along with differences in study duration, patient adherence, and clinical outcomes, make direct comparisons challenging. Without detailed insight into the cost methodology used in these studies, it becomes challenging to assess their generalizability and make comparisons with our study results. Therefore, while acknowledging the potential benefits of mobile health interventions in terms of cost-effectiveness and cost savings, it is essential to interpret these findings with caution. Further research utilizing robust methodologies and transparent reporting practices is needed to better understand the economic implications of mobile health interventions in the management of various health conditions, including diabetes.
Strengths and limitations
A strength is that the model includes largely relevant and robust evidence for many of the parameters. This evidence is not only derived from literature reports, but also from the intervention carried out, increasing generalizability to the T2DM population. By using empirical evidence from the intervention, such as patient outcomes and intervention costs, the model reflects the practical applicability of the proposed hybrid care pathway in a clinical setting. This approach not only increases the credibility of the model, but also strengthens its potential for informing healthcare decision making. As a result, stakeholders, including healthcare providers, policymakers, and payers, can have increased confidence in the applicability and generalizability of the model's findings when considering the implementation of similar interventions or healthcare strategies for managing T2DM.
Several limitations need to be taken into account. First, the evaluated intervention may also involve indirect costs (e.g. productivity loss) and non-medical costs (e.g. additional costs in accessing healthcare such as transportation or meals) that can impact both the individual patient and overall economy. However, as a logical first step, the healthcare payer perspective was chosen over the societal perspective. Second, it is important to acknowledge that the model—like any model—simplifies reality. Although the model strives to capture the complex dynamics of treating T2DM, it inherently involves simplifications and abstractions of the disease and its treatment. For example, Riddle and colleagues 50 emphasized the potential for diabetes remission as an important consideration. In our model, however, remission was not explicitly included as a separate state due to its relatively low probability of occurrence in the studied population. In addition, the model is based on several assumptions, introducing some degree of uncertainty in the findings. These assumptions may relate to various aspects of the model, including the progression of T2DM and the long-term effects of interventions. However, the sensitivity analyses explored the impact of different assumptions on the model outcomes and provided insights into the robustness of the findings under various scenarios. Next, this study's focus on Flanders may impede its transferability to other regions with differing healthcare systems, cost structures, and policies. There are at least two pathways to assess its transferability: research in diverse settings or transferability analysis. Future research in diverse settings can explore how hybrid care pathways should be adapted to align with various healthcare contexts, enhancing their broader applicability. Moreover, several transferability instruments have been developed to assess the applicability of cost-effectiveness results in other regions or healthcare systems, in order to faster bridge the gap between science and practice. Prioritized transferability instruments include the qualitative instrument of Welte and colleagues 51 or the more quantitative approach of Antonanzas and colleagues. 52 Lastly, patient perspectives might enrich the understanding of the adoption, adherence, and ultimately the cost-effectiveness of digital health interventions. While several studies have explored this topic,53–55 our study also included patient satisfaction data. However, these results were not directly analyzed in this cost-effectiveness evaluation.
Conclusion
In conclusion, the findings of this cost-effectiveness analysis indicate that a hybrid care pathway is unlikely to be a cost-effective approach compared to the standard care trajectory in patients with T2DM. Nevertheless, it is crucial to emphasize the ongoing need to support and investigate new healthcare pathways using technology. These efforts remain pivotal in advancing patient well-being. Yet, there is a call for additional research to better understand how healthcare resources can be utilized optimally to enhance outcomes effectively.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251319169 - Supplemental material for Cost-effectiveness of a digital supportive healthcare pathway for type 2 diabetes compared to usual care in Belgium
Supplemental material, sj-docx-1-dhj-10.1177_20552076251319169 for Cost-effectiveness of a digital supportive healthcare pathway for type 2 diabetes compared to usual care in Belgium by Lisa Van Wilder, Delphine De Smedt, Lieselotte Sandra, Dorien Vandormael, Joyce Kaes, Ruben Willems and Jeroen Trybou in DIGITAL HEALTH
Footnotes
Acknowledgements
We would like to thank all partners (in particular i-mens and Odisee) of the project for the fruitful collaboration.
Contributorship
LVW was involved in conceptualization, methodology, data analysis, and writing—original draft, review, and editing. DDS was involved in conceptualization, methodology and review. LS contributed to ethical approval and patient recruitment. DV contributed to ethical approval, patient recruitment, and writing—review and editing. JK was involved in ethical approval, patient recruitment, and writing—review and editing. RW was involved in conceptualization, methodology, data analysis, and writing—review and editing. JT was involved in conceptualization, and review and supervision.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee Research UZ/KU Leuven, Belgium (reference number: S66951 - B3222022000924).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Agentschap Innoveren en Ondernemen (grant number HBC.2021.0917).
Guarantor
JT.
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

