Abstract
Objective
With advancements in digital health technologies, telepractice has become crucial for providing accessible medical interventions. Cerebral palsy (CP) frequently results in comorbidities including dysarthria and dysphagia, leading to restrictions in activities. This study evaluates the efficacy of both in-person and telepractice-based intensive voice therapy in improving speech, swallowing functions, and related quality of life measures in adults with CP.
Methods
The Lee Silverman Voice Treatment (LSVT) LOUD®, a speech-behavior therapy, was administered to 16 CP subjects (9 men and 7 women; mean age = 43.4 ± 10.43 years) via in-person extended version of LSVT LOUD® (LSVT-X), and telepractice program of LSVT-X (LSVT-X e-LOUD®). Outcomes, including maximum phonation time (MPT), voice intensity, diadochokinetic rate (DDK), Voice Handicap Index (VHI), swallowing quality of life (SWAL-QOL), and Videofluoroscopic Dysphagia Scale (VDS), were assessed pre- and post-treatment.
Results
Significant improvements were noted in speech functions: MPT (p = 0.006), voice intensity (p = 0.004), DDK /puh/ (p = 0.043), and DDK/puh-tuh-kuh/ (p = 0.031). Swallowing function improved significantly in the pharyngeal phase on the VDS. Quality of life measures showed improvement in physical (p = 0.003), emotional (p = 0.000), total score of VHI (p = 0.001), fear (p = 0.031), sleep (p = 0.013), fatigue (p = 0.003), and total score of SWAL-QOL (p = 0.019). These improvements were consistent in both LSVT-X and LSVT-X e-LOUD® groups.
Conclusions
Both in-person and telepractice-based LSVT-X enhance speech, swallowing functions, and quality of life in adults with CP. LSVT-X e-LOUD® can be utilized as a digital therapeutic intervention for dysphagia and dysarthria, offering accessible and flexible treatment options aligned with digital health advancements.
Keywords
Introduction
Cerebral palsy (CP) is a group of permanent neurodevelopmental disorders that affect movement and posture. 1 Characterized by lifelong impairments, CP symptoms such as dysphagia and dysarthria persist throughout one's lifespan.2,3 Previous studies show that 90% of CP patients exhibit dysarthria, with symptoms including phonation breaks, fluctuations in pitch and loudness, slow rate of speech, strained voice quality, hypo- or hyper-nasality, and imprecise articulation.4–6 These communicative impairments resulting from dysarthria cause social restrictions and limitations on participation, negatively impacting the quality of life of individuals with CP. 5 For example, speakers with CP are often perceived as less intelligent, less likable, and less sociable, and they experience more negative emotions and behavioral intentions compared to non-dysarthric speakers. 7
Additionally, approximately 50% of CP patients develop swallowing disorders.8,9 Difficulty in swallowing and poor oromotor skills are among the first noticeable symptoms of CP in neonates. 10 Children with CP tend to swallow less frequently and less efficiently than their typically developing peers. 11 Those with dysphagia have difficulty controlling the muscles of mastication, exhibit abnormal respiratory-swallow coordination and may have insufficient intrathoracic and subglottic pressure. 12 These symptoms increase the risk of aspiration during swallowing, potentially leading to aspiration pneumonia, a leading cause of death in children with CP. 13 Besides adverse health outcomes, dysphagia significantly reduces quality of life due to restrictions in social participation.14,15
The Lee Silverman Voice Treatment (LSVT) LOUD® has been demonstrated in multiple studies to improve symptoms of dysarthria and dysphagia in Parkinson's disease (PD) and other neurological disorders through motor learning and intensive intervention.16,17 Studies have shown that LSVT LOUD® and its modifications have shown significant improvements in speech intelligibility, vocal loudness, and communication abilities not only in stroke patients but also in other populations including multiple sclerosis, and traumatic brain injury.18–20 Recent studies have also shown its benefits in addressing dysarthria in pediatric patients with CP.3,21 LSVT LOUD®, a speech-behavior therapy, aims to increase vocal cord adduction and voice intensity. The standardized program, conducted by a qualified speech-language pathologist, includes 16 one-hour treatment sessions over four consecutive days per week for one month. Exercises involve repeated sustained vowel phonation, pitch glides, functional phrase repetition, and reading or conversational tasks, with an emphasis on using a loud voice with good voice quality and generalizing or calibrating the louder voice.
Two alternative modes of LSVT LOUD® have been studied: LSVT-X and LSVT e-LOUD®. LSVT-X is an extended in-person program following the same protocol as LSVT LOUD® but with sessions reduced to twice per week over eight weeks. LSVT e-LOUD® is an adaptation delivered via telepractice. Studies have shown that LSVT LOUD®,22–24 LSVT-X,25–27 and LSVT e-LOUD®28,29 result in increased vocal loudness, maximum phonation time (MPT), voice quality, and pitch range due to repetitive and intensive exercise of the larynx and vocal folds. Additionally, LSVT-X e-LOUD® combines aspects of both LSVT-X and LSVT e-LOUD®, providing treatment twice a week through telepractice. Recent studies have verified speech function improvements, such as vowel speech and paragraph reading, following LSVT-X e-LOUD® application. 27 Evidence also indicates that LSVT LOUD® alleviates dysphagia symptoms by improving tongue and tongue-base function during the oral and pharyngeal stages of swallowing, due to the shared anatomy and physiology between speech and swallowing mechanisms.16,23,30
The rapid advancements in digital health and telepractice technologies have opened new possibilities for the treatment of various neurological disorders.31–33 Digital health solutions can overcome physical accessibility barriers, providing essential treatment opportunities, especially for patients in remote or underserved areas. 34 Telehealth and telemedicine have significantly grown in recent years, and their importance has been particularly highlighted during the COVID-19 pandemic.35,36 Digital health technologies improve cost efficiency and time management, benefiting both patients and healthcare providers. 34 For example, telepractice-based physical or speech therapies facilitate easy access from home, enhancing consistency and participation in treatment.29,37 This is especially beneficial for patients with mobility limitations or those who face barriers to accessing traditional in-person therapy. Multiple studies have demonstrated that telepractice-based rehabilitation therapies are as effective, if not more, than in-person therapies.37–39 The remote speech therapy program LSVT e-LOUD®, delivered through digital platforms, removes physical barriers and ensures smooth delivery of therapy. 29 Digital platforms help maintain continuity of care, extending the total duration of therapy and maximizing overall treatment outcomes.
This study aimed to evaluate the effectiveness of both in-person and telepractice-based LSVT-X as appropriate interventions for reducing symptoms of dysarthria and dysphagia in adults with CP. The primary objective was to evaluate the improvements in speech and swallowing functions, as well as related quality-of-life measures, following these interventions. Additionally, the research sought to explore the potential utilization of LSVT-X e-LOUD® as a digital therapeutic intervention for CP patients, considering the growing importance and accessibility of telepractice within the digital health framework. By comparing the outcomes of in-person and telepractice delivery modes, this study aimed to provide comprehensive evidence on the viability and efficacy of LSVT-X e-LOUD® as a sustainable and flexible treatment option for adults with CP, potentially shaping future therapeutic approaches in digital health.
Materials and methods
Study design and participants
This study took place from March 2021 to November 2023 at Severance Hospital, Seoul, Republic of Korea. Patients with CP, who complained of facing reduced speech intelligibility and difficulty in swallowing, were recruited for the trial through a referral process in which eligible participants were identified by the attending physicians based on the inclusion criteria. Based on the G*Power analysis for a Wilcoxon signed-rank test with matched pairs, the required sample size was calculated to ensure sufficient statistical power for detecting a large effect size (dz = 0.9) from a similar study on LSVT by Mahler et al. With an alpha level (α) set at 0.05 and a desired power of 0.95, the analysis determined that a total sample size of 16 participants is needed. A total 26 were first recruited. 9 patients were allocated to the in-person group and 7 patients were allocated to telepractice group. A total of 26 patients were assessed for eligibility, with 10 excluded. Inclusion criteria were: 1) diagnosis of CP, 2) signs and symptoms of dysarthria, 3) signs and symptoms of dysphagia, and 4) ability to fully understand and follow the treatment process. Exclusion criteria included: 1) COVID-19 infection, 2) inability to adhere to the treatment schedule, and 3) physical incompetence preventing completion of the treatment (Figure 1). Participants completed the Mini-Mental State Examination (MMSE), a general assessment tool for cognitive function that scores from 0 to 30. A score under 24 could suggest potential cognitive impairment. 40 This study was approved by the Institutional Review Board (No. 4–2021–1009).
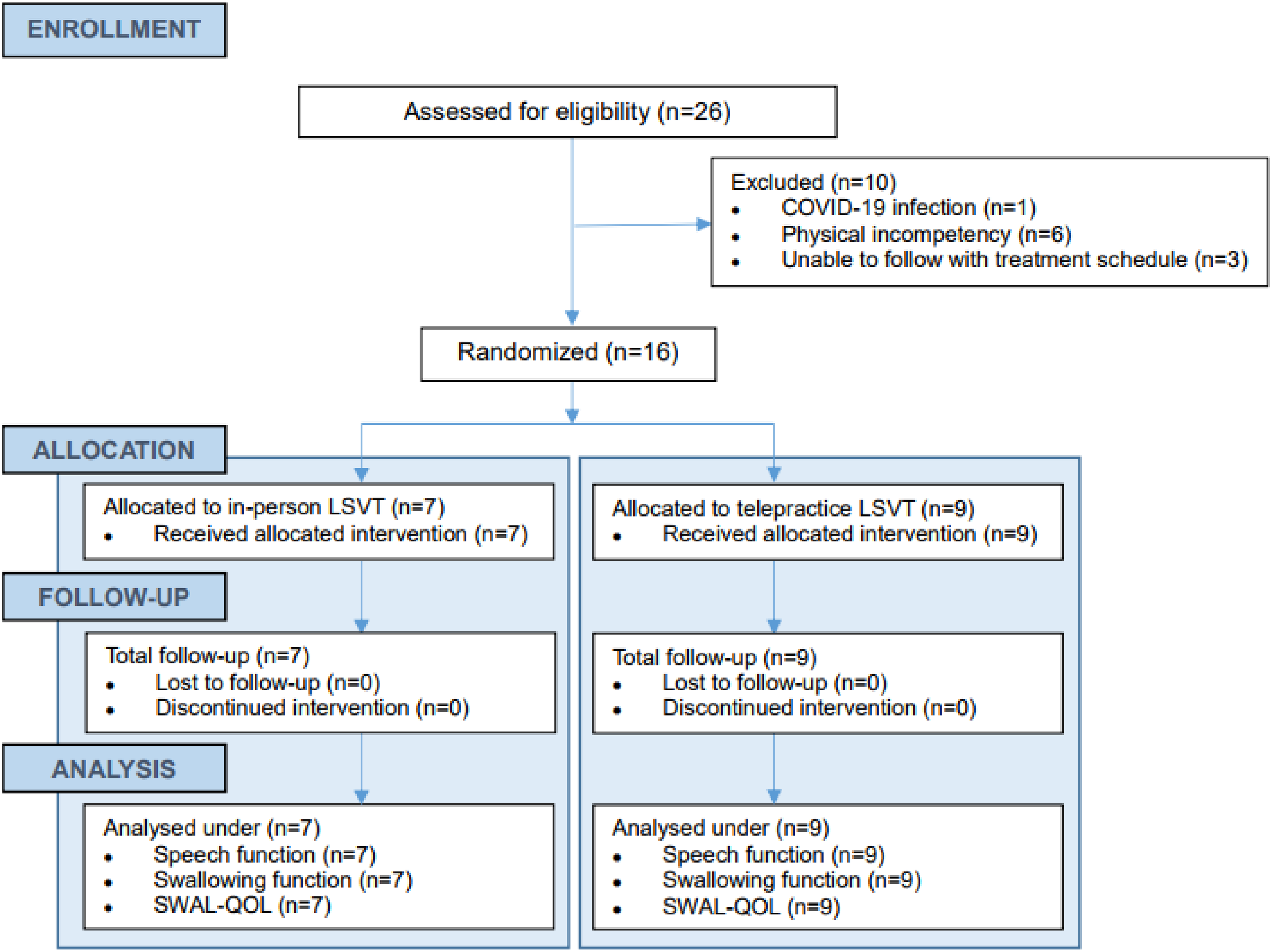
CONSORT flow diagram for patient participation.
Treatment
LSVT LOUD®, a speech-behavior therapy, aims to increase vocal cord adduction and voice intensity and was conducted by a qualified speech-language pathologist. Seven participants received in-person LSVT-X treatment with sessions twice per week for eight weeks (total of 16 sessions). Nine participants received LSVT-X e-LOUD® telepractice treatment with the same frequency and duration. Participants were randomly allocated to LSVT-X or LSVT-X e-LOUD® groups by an independent research coordinator using simple randomization. LSVT LOUD® consists of 16 sessions, each lasting 60 min and held four times a week over four weeks. LSVT-X is a modified version delivering the same number of sessions but spread over two sessions per week over eight weeks. LSVT e-LOUD® provides treatment through telepractice, and LSVT-X e-LOUD® is the modified version with the same session count but conducted twice a week for eight weeks via telepractice. These modifications were implemented during the study to accommodate facility limitations and patient schedules. Each LSVT-X and LSVT-X e- LOUD® session follows the same structure of an LSVT LOUD® session, consisting of 30 min dedicated to functional exercises (such as prolonged /a/ vowel phonation, low-to-high and high-to-low pitch glide phonation, and functional phrase recitation), followed by 30 min of hierarchy exercises, progressing from word/phrase level (sessions 1–4) to sentence level (sessions 5–8), paragraph level (sessions 9–12), and conversational level (sessions 13–16). 41 Participants were assigned homework after each session, requiring 5–10 min of practice once on treatment days and twice on non-treatment days.
Data collection and analysis
Participants completed quality-of-life questionnaires related to speech, voice, and swallowing handicaps. Subsequently, speech and swallowing evaluations were conducted before (pre-evaluation) and 8 weeks after the first voice treatment program (post-evaluation), when the intervention had ended.
Speech function data collection and analysis
All speech assessment tasks were recorded in a quiet room with environmental noise lower than 50 dB. Participants were seated comfortably upright. The SONY ECM-MS907 (SONY Corp., Tokyo, Japan) condenser microphone was positioned 10 cm away at a 90-degree angle from the participant's mouth, connected to an ICD-UX560F (SONY Corp., Tokyo, Japan) voice recorder. The voice recorder was set to a sample rate of 44.1 kHz and a bit depth of 16 bits with recording level fixed at −12 dB. Voice data were collected by the same speech-language pathologist for both pre- and post-treatment assessments, conducted within one to seven days before and after treatment, respectively. Speech data included prolonged /a/ vowel phonation, MPT, DDK, functional phrases, phonetic feature phrases, and connected speech samples.
To determine the dysarthria type and severity, auditory-perceptual rating was conducted by analyzing recorded prolonged vowel phonation, diadochokinetic rates (DDK), reading (functional phrases, phonetic feature phrases, standardized Autumn/Travel passages), and connected speech samples of each participant. Features were scored on a five-point scale: 0 = normal, 1 = mild, 2 = moderate, 3 = severe, 4 = profound. 42 The speech data files were analyzed using the Praat software (Ver. 6.0.39).
To minimize bias, all pre- and post-intervention assessments of speech and swallowing outcomes were conducted by a licensed speech pathologist. Furthermore, independent physicians analyzed the results, providing an additional layer of objectivity and enhancing the reliability of the findings for both in-person and telepractice interventions.
Swallowing function data collection and analysis
During the Videofluoroscopic Swallow Study (VFSS), participants were given 5 mL and then 15 mL of a liquid solution containing barium sulfate to swallow. The liquid as administered with a tablespoon and patients were given the instruction to swallow it, with an assessment made to observe the concurrent hyoid bone position change. The equipment used in this study was SONIALVISION G4 Digital Radiography/Fluoroscopy system (Shimadzu, Kyoto, Japan, 2020). This test was assessed using the Penetration-Aspiration Scale (PAS) and Videofluoroscopic Dysphagia Scale (VDS).43,44 VDS quantifies the swallowing function of the oropharyngeal stage, classifying 14 items into the oral phase (items 1 to 7) and pharyngeal phase (items 8 to 14). The total score ranges from 0 to 100 (oral phase = 40; pharyngeal phase = 60), with higher scores indicating greater severity of dysphagia.
Speech and swallowing-related quality of life data collection and analysis
The Speech Handicap Index-15 (SHI-15), with two subcategories and 15 questions, was used as a self-report tool to determine the effect of speech on daily life. 45 Higher scores indicate lower quality of life associated with speech. The Voice Handicap Index (VHI), consisting of 30 items rated on a five-point scale, assessed the effects of voice on daily routine, with higher scores indicating greater severity of handicap. 46 Swallowing-related quality of life (SWAL-QOL), with 11 subcategories and 44 questions, was measured to assess quality of life in patients with oropharyngeal swallowing disorders. 15 Higher scores indicate better quality of life-related to swallowing impairment. PAS is used in grading the dysphagia severity observed in the VFSS, with the total score ranging from 1 to 8 and higher scores indicating higher severity (1 = normal, 2 = mild, 3 = moderate, 4 = moderate, 5–8 = severe). 47
Statistical analysis
IBM Statistical Package for the Social Sciences (version 26.0) was used for statistical analysis in Windows. Two-way analysis of variance (ANOVA) was performed to analyze the differences in data over time and presence of group, time, and group-by-time interaction effects for the speech function, swallowing function, and quality of life measures. Wilcoxon signed rank tests were conducted to evaluate significant differences in speech function, swallowing function, and quality of life measures between pre- and post-treatment for LSVT-X versus LSVT-X e-LOUD®. Statistical significance was determined at p < 0.05 for all analyses.
Results
Participants
A total of 16 patients with CP (9 men and 7 women) were recruited with informed written consent (Table 1). The mean age of the participants was 43.4 ± 10.4 years. The mean MMSE score was 27.8 ± 1.76 out of 30. Dysarthria types among participants were mixed (62.5%), spastic (18.8%), and flaccid (18.8%). The mean dysarthria severity was 2.75 ± 0.93, with 12.5% classified as mild, 18.8% as moderate, 50.0% as severe, and 18.8% as profound. All participants were on oral diets with no modifications and had not been previously treated with gastrostomy tube feeding.
Participant information.

P: Participant; K-MMSE: Korean Mini-Mental State Examination.
Changes in speech function after treatment of LSVT-X and LSVT-X e-LOUD®
Significant post-treatment improvements were observed in both the LSVT-X and LSVT-X e-LOUD® groups for MPT (p = 0.006), voice intensity (p = 0.004), DDK /puh/ (p = 0.043), and DDK /puh-tuh-kuh/ (p = 0.031) with two-way ANOVA analysis (Table 2). LSVT-X group showed a significant difference before and after treatment in MPT (p = 0.018) and LSVT-X e-LOUD group showed a significant difference in voice intensity with Wilcoxon signed rank test (p = 0.028). When comparing changes in speech function between the LSVT-X and LSVT-X e-LOUD® groups, the LSVT-X group showed significant improvement in MPT, while the LSVT-X e-LOUD® group showed significant improvement in voice intensity. Therefore, both in-person and telepractice delivery of LSVT-X were confirmed to positively impact speech function in adults CP patients.
Comparison of speech function in LSVT-X and LSVT-X e-LOUD® groups.

Two-way ANOVA was performed to analyze differences in data over time and to assess the effects of group, time, and the interaction between group and time on speech function. Additionally, Wilcoxon signed-rank tests were conducted to evaluate significant differences in these measures between pre- and post-treatment for LSVT-X versus LSVT e-LOUD®. Statistical significance was set at *p < 0.05, **p < 0.01. The values were expressed as mean ± standard deviation (SD).
LSVT-X: extended version of LSVT LOUD®; LSVT-X e-LOUD®: telepractice of LSVT-X; MPT: maximum phonation time; DDK: diadochokinetic rate.
Changes in swallowing function after treatment of LSVT-X and LSVT-X e-LOUD®
Significant post-treatment differences were observed in both the LSVT-X and LSVT-X e-LOUD® groups for the liquid 5 mL pharyngeal phase (p = 0.015) and liquid 15 mL pharyngeal phase (p = 0.046) on the VDS scale with two-way ANOVA analysis (Table 3). No significant differences between the LSVT-X and LSVT-X e-LOUD® groups were observed on the VDS and PAS scales with Wilcoxon signed rank test, but the LSVT-X group showed a numerical decrease in all items and the LSVT-X e-LOUD® group showed a numerical decrease in all items except Liquid 5 ml Oral phase. There were some findings of no change which was possibly due to low sensitivity of the assessment. Moreover, our intervention was mainly focused on pharyngeal region instead of oral region and therefore laryngeal elevation showed the most sensitive and significant improvement. Therefore, both in-person and telepractice delivery of LSVT-X positively impacted swallowing function improvements in adult CP patients.
Comparison of swallowing function in LSVT-X and LSVT-X e-LOUD® groups.

Two-way ANOVA was performed to analyze differences in data over time and to assess the effects of group, time, and the interaction between group and time on swallowing function. Additionally, Wilcoxon signed-rank tests were conducted to evaluate significant differences in these measures between pre- and post-treatment for LSVT-X versus LSVT e-LOUD®. Statistical significance was set at *p < 0.05, **p < 0.01. The values were expressed as mean ± standard deviation (SD).
LSVT-X: extended version of LSVT LOUD®; LSVT-X e-LOUD®: telepractice of LSVT-X; PAS: Penetration-Aspiration Scale.
Changes in quality of life after treatment of LSVT-X and LSVT e-LOUD-X®
Significant improvements were observed in physical (p = 0.003), emotional (p = 0.000), and total scores (p = 0.001) of the VHI, as well as in fear (p = 0.031), sleep (p = 0.013), fatigue (p = 0.003), and total scores (p = 0.019) of the SWAL-QOL when analyzed with two-way ANOVA (Table 4). When comparing changes in quality of life between the LSVT-X and LSVT-X e-LOUD® groups, the LSVT-X group showed significant improvements in the emotional and total scores of the VHI and the sleep and fatigue scores of the SWAL-QOL. The LSVT-X e-LOUD® group showed significant improvements in the physical, emotional, and total scores of the VHI and the total score of the SWAL-QOL (Table 4). LSVT-X group showed significant differences in VHI emotional (p = 0.028), VHI total score (p = 0.028), sleep (p = 0.042), and fatigue (p = 0.027) with Wilcoxon signed rank test (Table 4). LSVT e-LOUD-X group showed significant differences in VHI Physical (p = 0.019), emotional (p = 0.018), VHI total score (p = 0.021), SWAL-QOL total score (p = 0.028) before and after treatment with Wilcoxon signed rank test (Table 4). Therefore, both in-person and telepractice delivery of LSVT-X had a positive effect on voice and swallowing-related quality of life in adults with CP.
Comparison of quality of life in LSVT-X and LSVT-X e-LOUD® groups.

Two-way ANOVA was performed to analyze differences in data over time and to assess the effects of group, time, and the interaction between group and time on quality-of-life measures. Additionally, Wilcoxon signed-rank tests were conducted to evaluate significant differences in these measures between pre- and post-treatment for LSVT-X versus LSVT e-LOUD®. Statistical significance was set at *p < 0.05, **p < 0.01,*** p < 0.001. The values were expressed as mean ± standard deviation (SD).
LSVT-X: extended version of LSVT LOUD®; LSVT-X e-LOUD®: telepractice of LSVT-X; SHI-15: Speech Handicap Index-15; VHI: Voice Handicap Index; SWAL-QOL: swallowing quality of life.
Discussion
The LSVT LOUD® voice treatment has been demonstrated to be effective in improving speech, voice, swallowing, and quality of life.16,17,48 However, most studies focus on PD patients, with limited research targeting CP patients.49,50 This study confirms the efficacy of both in-person and telepractice delivery of LSVT-X on speech, swallowing function, and quality of life in adults with CP.
Post-treatment, significant improvements were observed in MPT, voice intensity, and DDK (Table 2). The results observed in this study are consistent with previous research demonstrating significant improvements in MPT with in-person LSVT-X treatment28,50 and in voice intensity with LSVT-X e-LOUD® treatment. 46 Moreover, these findings align with existing studies16,22,50 indicating that LSVT LOUD® enhances breathing, vocalization, and articulation agility in adults with CP. CP often leads to abnormal muscle contraction and postural control due to abnormal muscle tone, which can adversely affect trunk muscle development and respiratory function.51,52 Additionally, these abnormal movements can lead to thoracic spine malformations, contributing to respiratory difficulties. 52 CP-related speech abnormalities include phonation breaks, fluctuations in pitch and loudness, slow speech rate, strained voice quality, hypo- or hyper-nasality, and imprecise articulation.4–6 Setting goals related to managing loudness may become more desirable as a speech intervention for individuals with CP exhibiting these characteristics. Muscle weakness or coordination problems in the respiratory and vocal subsystems are also common in CP, leading to inaccurate production of speech sounds, which involves comprehensive issues extending beyond just oral articulation to include breathing, laryngeal, pharyngeal, and oral articulation problems.3,6 Therefore, both in-person and telepractice delivery of LSVT-X, focusing on breathing and vocalization exercises, successfully improve speech function in CP. These treatments reduce speech impairment and improve communication efficiency and engagement, thereby improving overall communication and reducing the risk of social isolation. 53
Functional improvement observed in the 5 mL and 15 mL pharyngeal phases on the VDS scale (Table 3) implies a positive effect of LSVT LOUD® as mentioned in previous literature.16,30 CP causes significant challenges in moving food into the esophagus and initiating the involuntary swallowing response due to decreased oral muscle function. Adults with CP often exhibit inadequate chewing and reduced tongue movement in the oral phase, which can lead to aspiration and result in a reflexive cough after swallowing even small amounts of liquid. 2 Additionally, they present with inadequate laryngeal elevation, vallecular residue, penetration, and aspiration during the pharyngeal phase, suggesting a compromised swallowing reflex. 2 LSVT LOUD® has been shown to improve tongue movement and endurance, which contributes to better oral phase swallowing by enhancing bolus formation and manipulation.54,55 An improved tongue movement likely contributes to reduced oral transit time. Efficient contact time between the tongue base and the pharyngeal wall is required during the pharyngeal phase to generate adequate bolus driving pressure. 42 When tongue-base retraction is inadequate, vallecular residues appear. LSVT LOUD® enhances tongue-base retraction, reducing residue during swallowing. 30 The treatment's primary aim is to improve respiratory and laryngeal muscle functions through voice strength training. 56 Breathing cessation during swallowing is essential to protect the airway and prevent aspiration, facilitated by various movements of the cartilages and muscles within the larynx to close the airway entrance. 46 The hyoid bone, functioning as a cornerstone for the tongue, is closely interrelated with the oral cavity, tongue, and larynx in the swallowing process. In-person and telepractice delivery of LSVT-X may have strengthened respiration and laryngeal muscles in CP, thereby enhancing laryngeal elevation and overall swallowing function.
Significant improvements were observed in the total scores of SHI-15, functional, physical, emotional, and total scores of the VHI, and symptom frequency, fear, fatigue, and the total score of SWAL-QOL (Table 4). These results are consistent with previous studies53,57 indicating that LSVT LOUD® improves speech, voice, and swallowing-related quality of life. The improvements in sound quality, voice intensity, and intelligibility likely result from enhanced laryngeal function. Consequently, patients’ perceptions of their voice problems improved post-LSVT LOUD® intervention. The reduction in dysphagia symptoms and swallowing-related fears suggests that patients experienced fewer instances of severe dyspnea when eating, thereby positively influencing social activity and participation.
This study provides evidence that both in-person LSVT-X and telepractice LSVT-X e-LOUD® effectively improve speech, swallowing functions, and quality of life in adults with CP. While both treatments share the core principles of LSVT, emphasizing intensive vocal exercises and motor learning, some differences in outcomes emerged. LSVT-X led to greater improvements in certain speech functions, possibly due to the direct tactile feedback and physical guidance provided by the therapist. On the other hand, LSVT-X e-LOUD® demonstrated more pronounced improvements in specific quality-of-life measures, likely because of its enhanced accessibility and convenience for patients. LSVT-X e-LOUD® offers greater flexibility and convenience for patients, particularly those with mobility limitations or those who have difficulty accessing traditional therapy. This increased accessibility has a significant positive impact on social participation and quality of life for individuals with CP. In addition, telepractice can enhance patient engagement and contribute to sustained treatment effects. Future research is warranted to explore the nuanced aspects of each service model more thoroughly, focusing on factors such as patient adherence, satisfaction, and the specific roles played by different aspects of each treatment in contributing to unique outcomes. Further investigation could lead to a better understanding of the optimal treatment strategies for individuals with CP, considering their unique needs and preferences. Research is needed to develop new strategies and technologies that leverage the strengths of LSVT-X e-LOUD® and ultimately improve the quality of life and social participation of individuals with CP.
The advancement of digital healthcare is transforming the treatment landscape for various conditions. 58 LSVT-X e-LOUD®, a digital adaptation of the LSVT-X program, allows CP patients to interact with therapists remotely. This model eliminates geographical barriers, making high-quality therapy accessible to residents of remote or underserved areas who might otherwise lack access to specialized neurological therapy. Telepractice addresses mobility issues, enabling patients with severe restrictions to receive therapy at home, thereby increasing participation rates and adherence to treatment plans. The flexibility in scheduling inherent in telepractice supports a more patient-centered approach, ensuring therapy sessions do not conflict with other daily activities. Additionally, the familiar and comfortable home environment can enhance patient engagement during therapy sessions. Cost reduction is another significant benefit, as travel expenses are eliminated and time efficiency for both patients and therapists is enhanced. 59 These cost savings make therapy more affordable and accessible to a broader population. Continuous support is facilitated by digital communication channels, enabling patients to receive guidance and answers to queries outside regular therapy sessions. Therapists can remotely update and adjust treatment plans based on real-time patient progress, ensuring optimal therapeutic interventions. Telepractice also offers scalability advantages, allowing a single therapist to manage a larger patient roster without the limitations of physical space, thereby enhancing overall patient reach. Integration with digital health tools, such as virtual reality and AI-based applications, can further augment therapy effectiveness and personalization. The digital nature of telepractice enables a data-driven approach to therapy, with continuous data collection facilitating quantitative evaluation of patient progress. This data can be analyzed to assess the efficacy of therapeutic interventions and contribute to the development of improved treatment methodologies. In conclusion, LSVT-X e-LOUD® leverages digital health advancements to deliver accessible, convenient, and effective therapeutic interventions for CP patients. The numerous advantages, including improved accessibility, real-time feedback, cost-efficiency, continuous support, scalability, and data-driven insights, make telepractice a valuable modality for managing dysarthria and dysphagia in adult CP patients.
This study has several limitations. First, with a sample size of only 16 participants, the generalizability of the results may be restricted, potentially leading to an overrepresentation of specific group characteristics. Future research should involve a larger, more diverse sample to enhance statistical significance. Second, the study focused on the immediate effects of treatment without assessing the long-term sustainability of the intervention. Long-term follow-up studies are necessary to observe the persistence and changes in treatment effects over time. Third, the analysis did not encompass key advantages of telepractice, specifically cost-effectiveness and accessibility. Evaluating cost-effectiveness is essential for determining the economic efficiency of remote therapy compared to traditional in-person treatment, while accessibility should be assessed with respect to the ability to overcome geographic, physical, or social barriers for a broader patient population. Consequently, future research should include data analysis on these factors to establish the effectiveness of digital therapy and enhance its practical applicability. Addressing these limitations will increase the reliability of the findings and validate treatment effects across a wider population.
In the context of LSVT LOUD® for individuals with CP, several limitations of telepractice are noteworthy. First, the lack of a therapist's physical presence may hinder the delivery of essential tactile feedback and real-time adjustments critical for interventions requiring precise physical engagement, potentially diminishing therapy efficacy, especially for improving speech and swallowing functions. Second, maintaining patient motivation and participation in remote platforms can be challenging. If telepractice lacks the immersion and interactivity of in-person therapy, patients may lose interest and struggle to remain engaged. Finally, not all CP patients may be suitable for telepractice, particularly those with severe motor or cognitive impairments who may require intensive, hands-on interventions not adequately provided in a remote setting. Addressing these limitations in the design and implementation of telepractice will be essential to optimize treatment efficacy for this population.
Conclusion
This study is notable as it is among the first to investigate the potential efficacy of both in-person and telepractice-based LSVT-X in adults with CP. The findings suggest that both treatment modalities may positively enhance speech and swallowing functions, as well as related quality of life aspects. Moreover, LSVT-X e-LOUD® represents an innovative solution that, in alignment with advancements in digital health, may offer a more effective and accessible treatment option for patients with reduced mobility. This digital therapeutic intervention for dysphagia and dysarthria holds promise for broader application in clinical settings, contributing to improved care and outcomes for CP patients.
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Contributorship
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Sang Ah Park. The first draft of the manuscript was written by Kyung Min Kim, Sang Ah Park and Jihye Hwang and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Critical revision of the manuscript for important intellectual content: Kyung Min Kim, Jihye Hwang, Alyssia Park and Hyang Hee Kim. Supervision: Jihye Hwang and Sung-Rae Cho.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
All participants in this study were provided with sufficient explanation prior to receiving any interventions mentioned in this study. Their written consent was obtained. The study protocol was approved by the Institutional Review Board of Yonsei University Health System (No. 4-2021-1009).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Korean Fund for Regenerative Medicine (KFRM) grants (21C0715L1 and 21A0202L1) funded by the Ministry of Science and ICT and the Ministry of Health & Welfare, Republic of Korea; and the Korean Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (HI22C1588).
