Abstract
Objective
Hematopoietic stem cell transplant is a life-saving procedure that treats patients with various conditions by transplanting hematopoietic stem cells from bone marrow. Mobile health apps could be useful in closing the digital divide and improving health equity among Spanish-speaking caregivers of children who undergo pediatric hematopoietic stem cell transplant. This study aims to transcreate the BMT4me adherence app originally designed for English-speaking caregivers for Spanish-speaking caregivers and evaluate the feasibility and usability.
Methods
This study consists of two phases. Phase 1 transcreates the existing BMT4me app for Spanish-speaking populations. App feedback is collected from three community advisory board focus groups (n = 10; each meeting will include the same 10 community advisory board members). Groups consist of members connected to the local Spanish-speaking population and participating in the Community Engagement Program at The Ohio State University Center for Clinical and Translation Science. Phase 2 tests the feasibility and usability of the Spanish BMT4me app with child–caregiver dyads (n = 30; 15 at site 1, n = 15 at site 2) whose primary language is Spanish. This phase is mixed methods and incorporates both a qualitative approach (caregiver interviews) and quantitative measures (system usability scale). It is expected that app users in phase 2 will report above average system usability scale scores (>68%). It is also expected that >75% of families approached in phase 2 will enroll and complete the surveys in our study.
Conclusion
This protocol paper details the transcreation process of the BMT4me app into a Spanish version. The findings of the study will demonstrate the level of acceptability of the Spanish BMT4me app with participants whose primary language is Spanish. As a digital health intervention for an underrepresented population that is increasingly online yet historically underserved, this app can overcome health barriers and disparities and improve overall health equity.
Keywords
Introduction
Hematopoietic stem cell transplant (HCT) is the transplantation of hematopoietic stem cells from bone marrow and is a treatment for life-threatening malignant and nonmalignant disorders. Patients who undergo HCT must adhere to complex medication schedules and multiple restrictions and guidelines, such as mask wearing and avoiding crowds. Adherence to these medication regimens and guidelines is extremely important, as adherence to immunosuppressant medication in the acute phase (first 100 days) after transplant is necessary to prevent graft-versus-host disease (GVHD). Low immunosuppressant medication concentration in the blood, an inevitable consequence of non-adherence, is associated with increased risk of GVHD exacerbations.1,2 GVHD exacerbations are associated with increased rates of HCT-related mortality and decreased overall survival. 3 While other factors may result in low medication levels (e.g. poor luminal absorption) 4 and poor health outcomes (e.g. GVHD exacerbation and death) in children post-HCT,5–7 the preventable nature of non-adherence makes understanding and mitigating its risk factors of critical importance.
Almost 5000 children receive HCT annually worldwide7–9; yet, few studies examine adherence. Current documented rates of adherence range from 52 to 73%.10–12 McGrady and colleagues found that children missed at least one dose of medication post-HCT approximately three days a week, and adherence rates declined over time. 12 Nonadherence to oral medications has been associated with a greater incidence of infections in children during the post-HCT phase. 11 However, there has been a lack of primary focus on nonadherence outcomes in non-English-speaking groups.
Adherence is poorly understood among diverse, Spanish-speaking populations. In general, little is known about adherence and the experiences of Spanish-speaking families during HCT, particularly processes underlying known disparities in outcomes. 13 However, phone-based interventions, among ethnically diverse populations of adults, have been shown to improve overall adherence across multiple chronic diseases. 14 Additionally, Foronda et al. 15 found that low adherence to medication is widely reported among families of Latino children with chronic conditions such as diabetes, cancer, and asthma.
Designing and testing effective mobile health (mHealth) interventions to improve adherence are also highly relevant to promoting health equity in the large and growing U.S. Latino population. 16 Spanish-speaking populations were the focus of this study for three important reasons. First, Latinos are the largest and fastest-growing minority group in the country and are projected to account for one-third of the U.S. population by 2060. 16 Second, the U.S. Latino community has been disproportionately affected by a number of health disparities and structural inequalities. 17 Increasing population numbers and a high prevalence of chronic illness require focused efforts in prevention and improvement of health outcomes for this underserved population. 16 Lastly, Latino mobile adoption in the United States has increased significantly in recent years, resulting in the highest rates of ethnic groups online, closing the digital divide.17,18 With the rise of digital connectivity and a trend toward individualized medicine, mHealth has been heralded as a path toward more equitable delivery of care and improved health outcomes.
BMT4me app
BMT4me is a phone application designed to improve medication adherence in pediatric HCT (Figure 1). Using a novel interface, the app reminds caregivers to give medication, tracks adherence, and captures real-time barriers if medication is missed. The app has additional features, including the ability to record symptoms and take notes both through typing and speech to text. Co-design of the app occurred in a multiphase approach with interviews and focus groups eliciting stakeholder (e.g. child, caregiver, and healthcare provider) input to create an English prototype for pilot testing. Twenty-one healthcare workers, consisting of nurses (n = 15), advanced practice nurses (n = 3) and physicians (n = 3), were interviewed to provide feedback on the BMT4me app prototype. Healthcare workers supported the usability of the app, stating “it seems really simple and user friendly” and “I think they’ll really enjoy filling it out.” Further, healthcare workers’ system usability scores (SUS) were substantially over the 68% cutoff (mean = 84.2), indicating positive perception of the app. Fourteen families enrolled in the pilot study, and similarly, user testing also revealed SUS substantially above the 68% cutoff (mean = 87.5). Parents have stated “it's easy to use” and “it keeps reminding, which is very important (acceptability).” During the pilot study, the necessity for a Spanish version of BMT4me became apparent as one-third of the families spoke Spanish, making them ineligible to participate.
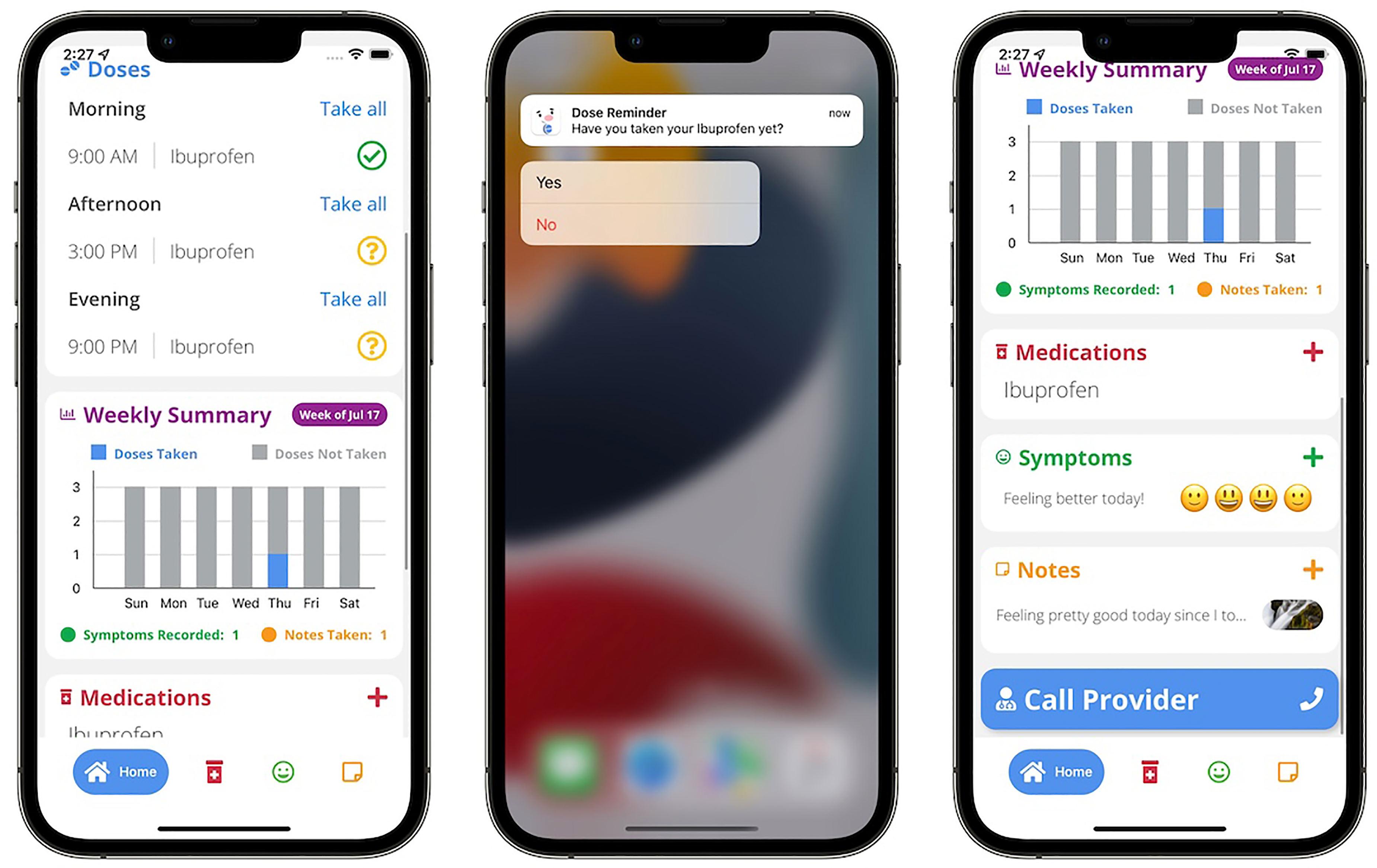
BMT4me application prototype.
We plan to transcreate the BMT4me app into a version for Spanish speakers. Transcreation is a process that involves creating a research team with relevant expertise, analyzing language and visual elements of the app, and implementing an iterative process of testing and reviewing the app. 16 Transcreation is also distinct from translation. In translation, the text is converted from one language to another. Transcreation in this study goes further by developing the app for and with its cultural and linguistic audience. By actively incorporating feedback from members of the Spanish-speaking community, we aim to elevate the app's cultural relevance and ease of understanding beyond the capabilities of the standard translation process.
Aims
To address pediatric health inequities, create an intervention to reach underserved, Spanish-speaking populations, and transcreate the BMT4me app into Spanish, this two-phase pilot study has the following aims:
Aim 1
The primary aim of the study is to conduct two to three community advisory board (CAB) focus groups to assist in the translation of the app and study measures into Spanish, as well as adapt cultural features and aesthetics of the app, to ensure it is appropriate for diverse families.
Aim 2
The secondary aim of the study is to conduct a multisite cross-sectional study to examine usability and acceptability of the transcreated Spanish BMT4me© app among 30 Spanish-speaking caregivers of children post-HCT.
Methods
Study design and setting
This is a multisite cross-sectional mixed methods study consists of two phases (Figure 2): (1) transcreate and adapt the current app into a Spanish language mHealth app and (2) usability and acceptability testing. Phase 1 is being conducted at a large, midwestern pediatric hospital, and phase 2 will be conducted in collaboration with a large academic medical center in New York City.

Study workflow across both phases.
Participants and recruitment strategy
The CAB will consist of members who are closely connected to the local Spanish-speaking/Hispanic community. The CAB will be convened with assistance from the Community Engagement Program at the Ohio State University Center for Clinical and Translational Science, which offers a community scientist academy to introduce stakeholders to the research process. Previous volunteers have come from organizations, such as the Ohio Hispanic Coalition, Ohio Latino Affairs Commission, and the League of United Latin American Citizens Ohio. Purposive sampling will be used to ensure a mix of demographic characteristics (e.g. sex, age, and country of origin) among invited CAB members.
For phase 2 of the study, which aims to assess usability and acceptability of the translated Spanish BMT4me app with caregivers, all children postallogeneic HCT at both study sites will be screened for eligibility. Primary caregivers of children (ages 2–17), whose primary language is Spanish, and are >18 years of age, will be invited to participate. Although most primary caregivers are expected to identify as female, a concerted effort will be made to include a mix of genders. A bilingual research coordinator at both sites will recruit 30 caregivers of children in the outpatient phase post-HCT from both sites to complete user testing, study measures and provide feedback on the app. Participants will be recruited using a script, and if interest is expressed, caregivers who would like to participate can either complete a paper copy or REDCap (Research Electronic Data Capture) consent. Access to technology is not expected to be a barrier to recruitment, as research phones will be used during clinic visits to obtain data from families during user testing. For both phases, the team will discuss recruitment and protocol activities at weekly lab meetings.
Phase 1 study design
The previously developed BMT4me app will be transcreated with help from the CAB and a translation company. Transcreation will minimize challenges common to standard translation-back-translation methods, such as idioms and colloquialisms being lost in translation, variation in reading comprehension levels after translation, and a lack of accounting for differences, especially in vocabulary, among cultural subgroups.19,20 First, CAB members will be invited to participate in a 60–90-min focus group via Zoom to provide important insight into nuanced cultural knowledge related to English versions and make recommendations. Virtual focus groups will be facilitated by the study Principal Investigators (PIs) and a bilingual research coordinator using a standard script to solicit input.
Language in BMT4me and study measures will be translated using Linguava, an external translation company for industries across many areas, including healthcare, education, business, and government agencies (linguava.com). Linguava uses accepted methodology for the translation and back-translation and cultural acceptability of translated documents via the following steps: (1) a professional linguist will translate the text from English to Spanish, while a second linguist edits the translation; (2) a team will reconcile the two Spanish versions to ensure consensus; (3) back-translation of the Spanish translated version into English will be performed by another native English-speaking translator; and (4) team review of the back-translation will occur again to ensure consensus. 19
Once consensus is assured and the content of the app is finalized, the app will be adapted for Spanish-speaking caregivers. The Spanish prototype and study measures will be presented to the CAB for evaluation and to ensure intent, tone, content strategy, and linguistic fidelity of the Spanish versions are as high quality as the original versions. Suggestions for changes in the visual representation and formatting of the app will be solicited and implemented, and BMT4me will be reappraised by the CAB in an iterative process until complete. Each member of the CAB will be compensated for time.
Phase 2 study design
There are usability testing procedures that have been used previously.
21
After the CAB approval, the Spanish prototype will be tested in a multisite study with 30 self-identified Spanish-speaking primary caregivers of children post-HCT to obtain feedback on the app interface, usability, and the flow and content of the software. Usability testing is the principle means of determining if the system meets its intended purpose. Purposive sampling of the entire eligible sample will be used to identify caregivers of children across age, sex, and diagnoses. A bilingual study coordinator (CRC) will recruit eligible caregivers and complete user testing. They will conduct one-on-one sessions, which will include facilitation, note taking, and audio recording. Caregivers will be evaluated for engagement and understanding of the app in the following steps:
Step 1 – Unobtrusive observation to measure the intuitiveness of the interface (10–15 min): Initially, the caregivers will be briefly introduced to the program and asked to start interacting with the adherence mHealth app. Research staff will not intervene or provide instruction and will observe and record participants’ progress. Step 2 – Interactive observation (10–15 min): The CRC will begin to interrupt individual participants during the interaction by posing questions regarding observed cues that implied success or delay in program use. Caregivers are encouraged to verbalize their criticisms of the mHealth app. Since discussion is based on evolving patterns of interaction, preprescribed questions are not possible. Step 3 – Debriefing (15–20 min): Caregivers will complete a 15-min semistructured interview with the bilingual coordinator related to participation in the intervention, as well as the Spanish version of the SUS. Caregivers will be asked to reflect on their experience with the app, its interface and contents, and indicate their opinions on the strengths and weakness of the app. These evaluations will be used to establish criteria for refining the content and interaction for future development and efficacy testing.
Ethical considerations
The study has central institutional review board approval from the primary site (STUDY00002478). Electronic informed consent will be signed by CAB member participants in phase 1, and written informed consent will be obtained by caregivers in clinic during phase 2. Data will only be available to the research team.
Outcome measures
SUS-Spanish version: The SUS is a 10-item questionnaire routinely used to evaluate the functionality and acceptability of mHealth apps.22,23 Items are rated on a 5-point scale and scores range from 0 to 100. Reliability (0.91) and validity (0.81 correlation with 7-point scale of “user friendliness”) have been well established. 24 A score of > 68% is considered above average. 23 The Spanish version of SUS has been previously developed and validated with the content validity index rating of 0.92 and the face validity index of 0.94. The Cronbach α was 0.812 (95% confidence interval 0.748–0.866; p < 0.001). 25
Caregiver Satisfaction Interview-Spanish version: Satisfaction will be assessed via semistructured interviews with caregivers. Caregivers will be asked using a semistructured interview guide for feedback regarding the mHealth app, benefit, burden, barriers, suggested modifications, and overall satisfaction. Suggested modifications to the app and advice to the healthcare team will also be solicited.
Data collection
A system to monitor recruitment, data collection, and general conduct of the study will include detailed training, weekly lab meetings, and secure collection and storage of data. All data will be checked in real time to ensure completeness, and if items are incomplete, the reason will be documented. Any paper copies will be marked with ID numbers and stored in a secure location. All data will be entered by the research coordinator and maintained in REDCap. The qualitative data will be collected through an audio-recorded semistructured interview via audio recorder with a bilingual research coordinator after caregivers complete app interaction. The quantitative data will be collected through REDCap at one time point at the end of the qualitative interview. A trained bilingual research coordinator will collect the data in clinic with caregivers enrolled in the study. Participants will be provided an option of answering the SUS verbally.
Data processing and analysis
Qualitative analyses
CAB focus groups and caregiver interviews will be audio-taped and transcribed verbatim by a bilingual research coordinator using NVivo and then translated to English. Then, two independent, trained, doctoral-level coders (MS and ES) will conduct content analysis using the constant comparison method. 26 In brief, the method will begin with immersion (i.e. repeatedly reading a subset of transcripts), cluster similar ideas to inform preliminary categories, review and revise coding schemes, apply the coding scheme to a second subset of transcripts (phase 2), revise themes, and repeat this process until reaching saturation and consensus.26–28 Member checking will be completed with a subsample to obtain family input on thematic codes as a final validity check, and frequency counts of final themes will be obtained using NVivo. Transcripts will be identified with ID numbers and stored electronically on a secure server.
Quantitative analyses
Descriptive statistics (e.g. mean, standard deviation, frequency, and percent) will be used to summarize the background characteristics of caregivers and children. Acceptability will be assessed by averaging total scores from the SUS. Per the literature, scores >68% on the SUS will be considered acceptable. 23 Quantitative data will be analyzed with SPSS version 28.
Discussion
Among Hispanic pediatric patients receiving HCT, patients with Spanish-speaking caregivers may be more likely to have longer hospitalization and increased pretransplant complications than patients with English-speaking caregivers. 13 Similarly, pediatric HCT patients of Hispanic and non-White background may be more likely to develop Cytomeglavirous infection posttransplant sooner and more frequently than their White, non-Hispanic counterparts. 29 Therefore, existing literature points to a link between limited English, Spanish-speaking caregiver status and increased symptom burden and worse outcomes in pediatric HCT. However, there is an absence of interventions that serve Spanish-speaking caregivers in pediatric HCT.
Digital interventions may hold promise for U.S. Latino, Spanish-speaking populations, as their digital connectivity has significantly increased in recent years.17,18 There are few existing pediatric apps in Spanish, which include hospital-owned mobile apps 30 and those to help paramedics overcome language barriers. 31 However, there is a lack of apps for pediatric chronic conditions tailored for the U.S. Latino, Spanish-speaking population. For example, there are existing apps that were designed and developed in Spain, such as those for pediatric asthma 32 and growth and nutritional disorders, 33 which may lack cultural nuance for other Spanish-speaking patients or caregivers. There also appears to be an effort to translate pediatric apps from English into Spanish, such as apps to support family management of congenital heart defects, 34 communicate via patient portal, 35 and provide pertinent information for pediatric cancer 36 ; however, these translated versions have not yet officially been transcreated and tested with Spanish speakers. Lastly, the development and usability of pediatric apps in Spanish may not be rigorously tested. For example, within pediatric cerebral palsy, there are several Spanish apps that are designed for or may be useful for the condition. However, few are scientifically validated. 37 Further, there appears to be no apps in Spanish for pediatric medication adherence or for HCT populations.
To our knowledge, this is the first study to use the transcreation model with community and family participants to adapt an app aimed to improve medication adherence outcomes for pediatric HCT Spanish-speaking populations. Through this study, we intend to improve equitable healthcare for the growing U.S. Latino population. The long-term goal of this multiphase study is to test the efficacy of the translated app with Spanish-speaking families through a randomized controlled trial. Efficacy in the future randomized controlled trial will be calculated by comparing percentage rates of adherence between those without the app versus those with the app. Adherence will be measured by self-report and electronic measurement devices (e.g. MEMS Cap and Wisepill Boxes), which record individualized adherence data. To further the accessibility and dissemination of the project, we also aim to make the translated app available in Google Play and the App Store and circulate the app to other medical centers to reach Spanish-speaking families on a national level and plan to eventually transcreate the app with literacy considerations (e.g. option to play written elements as audio). Lastly, we plan to transcreate the BMT4me app into other commonly spoken patient languages.
There are numerous strengths to the proposed study, including purposive sampling approaches in both phases 1 and 2 to identify CAB members from a variety of Spanish-speaking countries of origin and to identify caregivers of children across different ages, diagnoses, and gender. A strength of phase 1 specifically is the use of a CAB, which will ensure effective cultural tailoring of the app due to the involvement of community members in decision making. The multisite nature of phase 2 allows the study to increase generalizability and to reach more diverse Spanish-speaking patient populations. Another strength of phase 2 is its mixed-methods approach that leverages rigorous qualitative methods, and a validated quantitative measure of usability is it enables researchers to gain a more comprehensive understanding of app user experience and feedback. Despite the strengths, this study should be viewed considering potential limitations. The use of purposive sampling could produce the risk of sampling bias, due to nonrandom selection. Lastly, phase 2 includes a small sample size, which may prevent our study reaching a large enough sample to generalize findings to the wider, U.S. Spanish-speaking population.
Conclusion
This study extends the functionality of our existing medication adherence app, BMT4me, by transcreating it for Spanish-speaking populations. The transcreation process actively seeks and incorporates the feedback of community Spanish speakers. Demand for this app stems from the Latino population being historically underserved and one of the fastest growing minority groups in the United States. Additionally, as Latino mobile adoption has increased in recent years, mHealth apps provide an opportunity to access this previously underserved population. As a free digital health intervention, the BMT4me app can overcome health barriers and disparities, thus improving health equity. The findings from this study create and refine a mobile tool that improves pediatric medication adherence for Spanish-speaking users.
Footnotes
Acknowledgements
The authors are grateful to Cynthia Gerhardt and Erin Rodriguez for their mentorship during protocol development.
Author contributions
AB: contributed to writing the original draft; and developing tables; MW: contributed to writing the original draft, edited multiple versions, assisted with figure development, writing editing; MB: writing- review & editing, investigation, validation; ES: writing – review & editing, investigation, validation; MS: conceptualization, funding acquisition, investigation, methodology, supervision, writing original draft.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study is approved by the Nationwide Children's Hospital IRB (central IRB)
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by the National Institute of Minority and Health Disparities Grant Number 5R21MD019075.
Guarantor
MS
