Abstract
Background
Women dealing with cancer often face a myriad of physical and psychological challenges. Regular physical activity and mobile applications offering healthcare consultation, information, and health tracking have shown promise in aiding this demographic. Therefore, this study aims to examine the effectiveness of the i-CanManage program in improving physical activity, self-efficacy in symptom management, and the quality of life (QoL) among Vietnamese women after completion of cancer treatment.
Methods
A two-arm pilot randomized controlled trial will be conducted. Eligible participants will be randomly allocated to intervention and control groups. Members of both groups will be provided with a Cancer Management Handbook containing information on Life After Cancer and Symptom Management. Intervention group participants will be offered a 12-week personalized exercise program called i-CanManage via a Cancer Wellness Program smart application—an artificial intelligence-based mobile app developed for monitoring patients’ symptoms with a Fitbit Charge 5 wearable device. Data will then be collected at weeks 0, 6, and 12 concerning physical activity, symptom severity, self-efficacy, self-care, and QoL. App usability and user experience will also be explored using both quantitative and qualitative methods.
Discussion
This pilot study will assess the effectiveness of the internet-based cancer management program on physical activity, self-efficacy in symptom management, and QoL of women following cancer treatment in Vietnam, as well as evaluate users’ experience with the smart app. The findings will contribute to the growing evidence on the role of mhealth in oncology care, providing insights into how technology-driven interventions can empower patients in their cancer recovery journey. Datasets collected from this study will be openly available to the research community to foster new advances in the field.
Introduction
Globally, the incidence of diagnosed cancer has dramatically increased in recent years to approximately 18.1 million people by 2020, among whom 8.8 million confirmed cases were in women. 1 In the same year, according to the World Health Organization, in Southeast Asia alone, the number of new cases in the female population was 1,156,747, with breast, cervical, uterine, ovarian, colorectal, and lung cancers the six most frequently reported. 2 Industrialization and widespread adoption of unhealthy lifestyles, including tobacco usage, alcohol consumption, physical inactivity, and improper diets, have contributed to increased cancer incidence among women in both high-income countries and low- and lower middle-income countries in the region.3,4 In regard to Vietnam, according to the International Agency for Research on Cancer, breast, lung, and colorectal cancers were the top three most frequent cancers in females, accounting for 25.8%, 9.1%, and 9.0% of 83,647 newly diagnosed cases in 2020, respectively.5–9
Despite the increase in the number of people with cancer worldwide, the five-year survival rate is improving, thanks to earlier screening and improved care and treatment. 10 In recent years, the five-year survival rate for cancers in Vietnam has been estimated to be as high as 80–90%.11–13 While breast, cervical, and ovarian cancers, for example, are linked to distinct survivorship challenges, including pain, menopausal-like symptoms, and reproductive health issues,14–16 even among cancers that occur in both sexes, such as thyroid and colorectal cancers, women often report poorer posttreatment quality of life (QoL) due to gender-specific factors such as hormonal changes and societal roles. 17 Studies consistently show that female cancer survivors face higher psychological morbidity compared to male survivors.15,18 One cross-sectional study even found that being female is a significant factor associated with higher depression and anxiety in 1002 survivors five and 10 years after diagnosis. 19 This underscores the need for gender-sensitive support mechanisms, assisting women to adapt to their “new normal,” improve their health status and thereby enhance their overall QoL.11,20 This is especially relevant to women in Vietnam, where life following cancer treatment is not a healthcare priority.
Engaging in regular physical activity has been shown to have profound effects on both physical and mental wellness, particularly those persons who have undergone cancer treatment.21–24 People who are physically active are recognized to experience better sleep, improved mood, and reduced risk of anxiety and stress, leading to an improved overall QoL. 25 Also, psychosocial oncology, the specialist medical discipline that focuses on the psychological, social, and behavioral aspects of cancer, is also very important for supporting patients to manage their symptoms and therefore to enhance the QoL. 26 However, information from oncology professionals concerning exercises is not provided routinely, resulting in only relatively few women being able to meet the recommended physical routine.27–29 In Vietnam, there are also unmet needs identified in the mental health support and wellness system of screening, assessment, and management of symptoms for women after cancer.11,30 Physical activities and assistance in managing symptoms must therefore be given priority among the many elements impacting women living with, or following treatment for, cancer. 31 Different programs have been developed to assist such women through peer support groups, medical clearance, clinical consultations, and exercise recommendations.32,33 However, these programs have not placed a strong emphasis on smart process applications, commonly known as smart apps. These may be utilized on dedicated handheld devices such as mobile phones and tablets to manage and provide interventions.34,35
A wealth of recent research indicates that mobile apps could potentially act as healthcare professional consultation-related services, valuable information sources, and proactive tracking tools concerning exercise, diet, weight management, stress relief, and sleep monitoring.36–41 For this reason, oncology studies have started to make use of mobile health technology (so-called mHealth, e.g. wearable smart devices and mobile apps for health and well-being) to promote sustainable behavioral changes in sedentary patients following cancer treatment as a way to improve disease outcomes.42–44 The U.S. National Cancer Institute has designated the initiation and evaluation of mobile health for physical activity promotion in individuals with cancer a priority area for study. 45 In Vietnam, despite the fact that mobile health is acknowledged to reduce accessibility issues related to cost, time, geography, and other specific constraints, there is a dearth of data on interventions that emphasize exercise and symptom management.25,46 As Vietnam is ranked in the top 10 Southeast Asian nations for smart phone ownership, with around 70 million units currently in use among a population of 98 million, 47 research on the effectiveness and deliverability of mHealth has great promise for the nation's health. Our study's internet-based intervention tool, which helps female patients to control their symptoms and to practice physical activity on an individual basis, has the potential to improve QoL in this population and may serve as a stepping stone for future digital health innovations in Vietnam.
Our intervention development follows the U.K. Medical Research Council (MRC) framework, ensuring a systematic and evidence-based approach.
48
Drawing from Albert Bandura's Social Cognitive Theory, this program aims to enhance self-efficacy through strategies such as observational learning, role modeling, and goal setting.49,50 Our initiative commenced with a preparatory phase, adhering to the MRC framework. Prior to this trial, in the recruitment phase of the project, the research team conducted a survey to investigate current symptoms, self-efficacy in symptom management, physical activity, and QoL of Vietnamese women following cancer treatment. A systematic review was also undertaken to determine the optimal mode of delivery and the appropriate exercise dose for participants.
51
Based on that preparatory investigation, a smart health program called i-CanManage (an
Methods
Study design
This is a prospective two-arm study conducted over 12 weeks, evaluating the impact of an individual-based exercise program for women following cancer treatment. Figure 1 shows the flowchart of the study.

Flowchart of the study design.
Study setting
The study is a community-based exercise program conducted in Vietnam. The program is delivered virtually via the i-CanManage smart app, supplemented by in-person or video/audio phone meetings via Zalo—the most popular Vietnamese messaging app, with a member of the research team during their daycare or at their home.
Participants
Inclusion criteria:
- Be a Vietnamese woman and residing in Vietnam; - Be able to speak and write proficiently in Vietnamese; - Be 18 years of age or older; - Have been diagnosed with any form of cancer; - Have completed intensive cancer treatment in the last six months; - Have a smart phone; - Have the ability to perform exercises, as evidenced by a Karnofsky Performance Scale Index score of 80 or above, which translates to “Normal activity with effort; some signs or symptoms of disease.”
Exclusion criteria: women who are diagnosed with chronic mental illness.
Recruitment
In Phase 1 of this project, a survey will be conducted to identify the current symptoms, level of self-efficacy in symptom management, self-care practices, and QoL among Vietnamese women after completion of cancer treatment. General information on the background and purpose of the study is provided to all potential participants who participated in Phase 1, and those expressing an initial interest in the study will be contacted via their mobile phone number and/or email. A written invitation, study overview and an informed consent form will be sent to each participant who confirms her interest in participating.
Randomization and allocation
Following the screening for eligibility, each accepted participant will be assigned a number in numerical order. An online randomization program will be used to allocate participants to either the intervention or control group.
Intervention
The intervention to be used in this program is adapted from the Women's Wellness After Cancer Program (WWACP) developed by Anderson et al. for women in Australia. 52 This 12-week multimodal intervention designed for delivery via an e-health platform has been shown globally to be effective in improving physical activity, symptom management, and overall wellness in women after cancer treatment.53–55 In a pilot RCT conducted in Hong Kong among Chinese women treated for gynecological cancer, the culturally adapted WWACPHK demonstrated promising results, whereby the intervention group showed significantly greater improvement in self-efficacy for adhering to an exercise routine compared to the control group. 53 In order to meet our study's aim and objectives only the physical activity aspect of the WWACP has been adapted.
Figure 2 shows an overview of the study. Participants will be divided into two groups, an intervention group and a control group, after considering the importance of differentiating the impacts of various intervention components discussed as follows.
56
At the start of week 0, all participants will receive initial training on using the Cancer Management Handbook For Women After Cancer Treatment to enable them to self-monitor their symptoms; this will also enable a baseline measurement to be recorded by the investigators. Notably, this Handbook encompasses two key elements: knowledge about common symptoms and their appropriate management, and an emphasis on WELLNESS lifestyle goal setting (
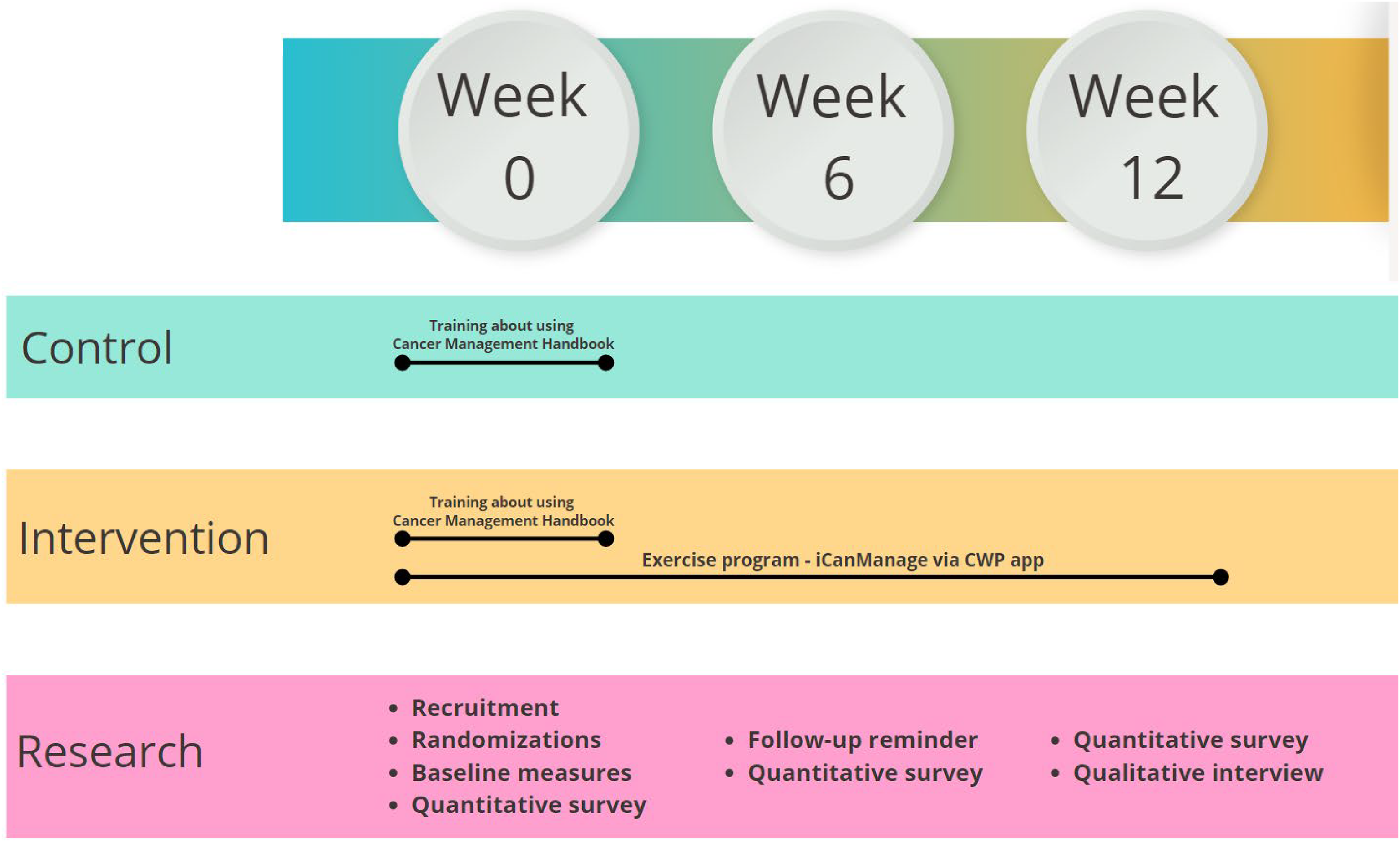
Study overview.
Thereafter, the control arm will follow up on their standard care procedures by monitoring each participant's self-detection of symptoms according to the Handbook's guidelines. At the same time, only those women in the intervention group will be equipped with a Fitbit Charge 5 wearable device and invited to participate in i-CanManage to receive the interventional program. While the Cancer Management Handbook For Women After Cancer Treatment chiefly emphasizes wellness, the i-CanManage program concentrates on physical activities via tutorial exercising pictures/videos to enhance symptom alleviation and overall QoL. This approach supports symptom monitoring, cultivates positive habits for wellness enhancement, boosts self-efficacy in symptom management, and thereby eventually ameliorates the symptoms themselves. Both control and intervention groups will be assessed and evaluated for their adherence to the physical activity, self-care practices, self-efficacy in symptom management, and QoL at three milestones (baseline, week 6, and week 12 of intervention). The app usability, user satisfaction, and user experience will be assessed only with feedback from participants in the intervention group.
The intervention involves exercises and monitoring symptoms for women following cancer treatment, which will be designed by the i-CanManage program. This offers exercises for flexibility, aerobic fitness, balance, endurance, etc., delivered in an individual-tailored format. For instance, participants may follow guided stretching routines for increased flexibility, perform brisk walking or cycling to improve aerobic fitness, practice yoga poses to enhance balance, and engage in low-impact cardio exercises for endurance. Over the course of 12 weeks, exercises gradually progress in level, intensity, frequency, and time, carefully adapting to each participant's needs while also aiming to meet goals to promote their overall wellness. All participants will receive guidance to monitor their symptoms using the program Handbook and will be encouraged to use the interactive report box via the smart app. Recommendations will be provided throughout the 12-week program based on the health status of participants. Table 1 highlights key topics of the 12-week program.
iCanManage program intervention content and delivery strategies.
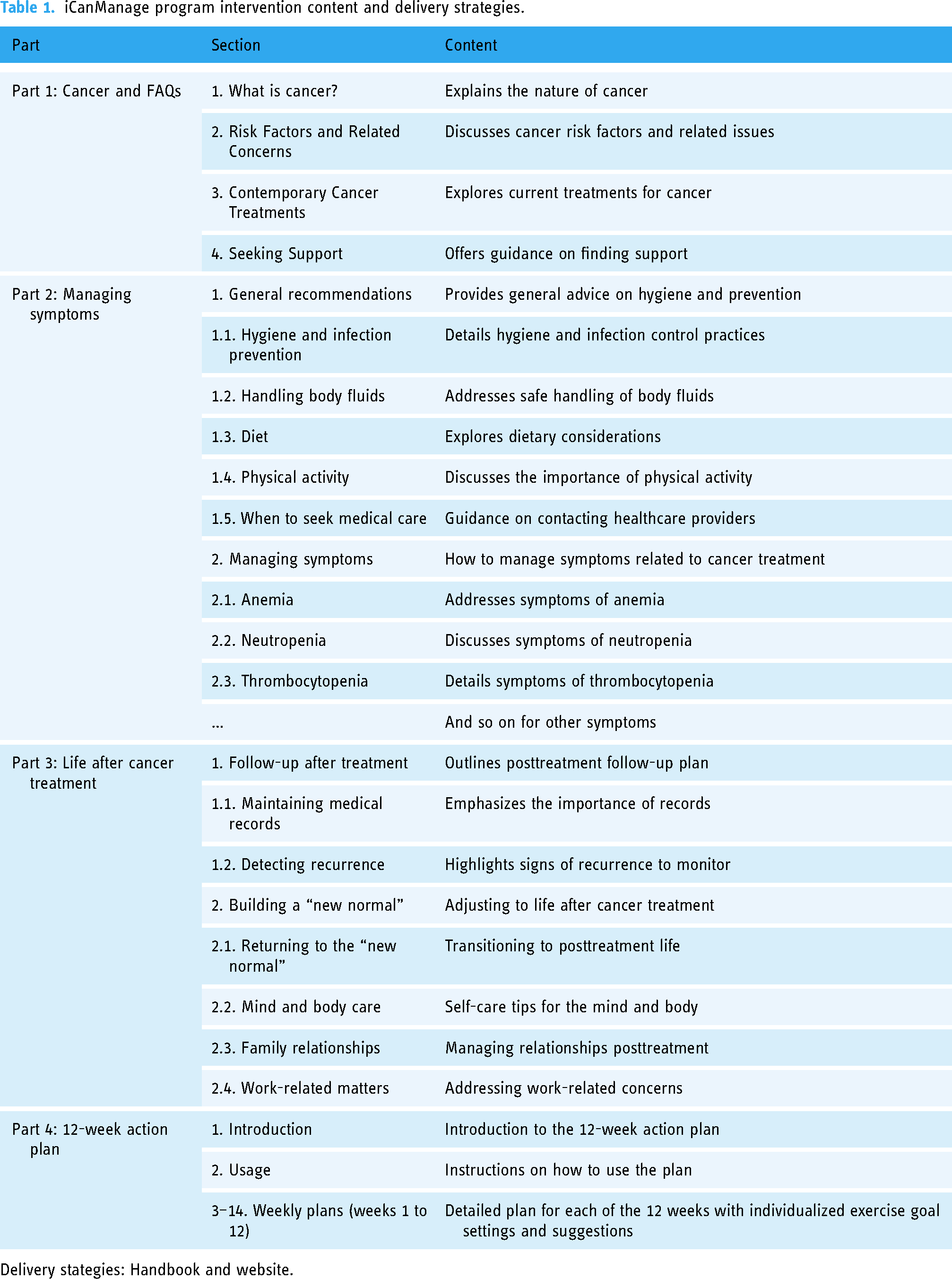
Delivery stategies: Handbook and website.
The CWP smart app
The acronym “CWP” stands for Cancer Wellness Program, a mobile app designed to help women to manage their common symptoms relating to recovery from cancer and to enhance physical exercise participation/performance/effectiveness following cancer treatment. This app is connected with a Fitbit Charge 5—an affordable, wireless-enabled activity tracker worn on the wrist like a watch, offering advanced insights into users’ exercise routines, sleep patterns, and overall wellness trends. Released in September 2021, the Fitbit Charge 5 has many features to support a patient's fitness and health tracking and to enable symptom management, allowing wearers to track runs, walks, and bike rides without carrying their mobile phones. In addition, it also has an electrodermal activity stress monitor plus an electrocardiogram sensor for checking patients’ heart health.55,56 All data will be collected in real-time and sent to the CWP app for further analysis. Fitbit Charge 5's reliability for fitness and health tracking, including measuring heart rate, step count, sleep tracking, and potential atrial fibrillation detection, is supported by previous studies on earlier Fitbit Charge models (Fitbit Charge HR, Charge 2, 3, and 4), showing accurate results with minimal error percentages.57–62 The Fitbit Charge 5 is provided to participants free of charge as part of the study intervention. This approach is intended to minimize financial barriers and thereby ensure that all participants in the intervention group have equal access to the wearable device.
This application runs on the two most commonly used mobile operating systems globally, Android and iOS, supported respectively by the leading mobile brands Samsung and Apple, and will be used on a participant's own smart phone. Intended users first need to download the app from the appropriate app store platform by clicking on a link sent to their smart phone by a member of the research team. After set-up and account registration, the app is ready to use. There are three main components of the CWP smart app. First, users will fill in details of their basic health information, such as age, height and weight, and self-report common symptoms and health conditions by answering short questions embedded in the app. Second, based on the general health assessment results collected in the first component, as well as data tracked via the Fitbit Charge 5 wearable device connected to the app, users will be recommended an individualized and tailored exercise plan with appropriate intensity level and goal. Third, users will be asked to follow that exercise plan as closely as they can. Toward the end of each day, information about performance and related health data for the preceding 24 h will be reported to help users keep track of their health and well-being.
Prior to starting the exercise program, each participant undergoes a thorough preintervention screening, assessing their health status and medical history. The AI-generated exercise plans are tailored to individual fitness levels and needs, progressing gradually to prevent injury. The app provides clear exercise instructions via video tutorials. Rest intervals and recovery periods are incorporated, for which a participant's Karnofsky Performance Scale Index score is considered. Participants are encouraged to report any discomfort, pain, or adverse effects, with continuous monitoring by the research team and designated healthcare professionals. These safety precautions, combined with personalized guidance, aim to enable participants to engage in exercises safely and with confidence.
User guides and ongoing technical support
Intended users will be invited to watch a webinar to guide them on how to set up and use the app. We also provide a detailed user guide that helps participants to set up and use both the Fitbit device and the app. The app development team will deliver this webinar in real-time that is suitable for most participants, and which will be recorded and made available for viewing by those unable to attend. During the smart app utility period, any technical issues or concerns will be supported by information technology technicians to ensure that participants can use the app proficiently. In particular, we have developed a mechanism to track collected data automatically in real-time. This approach allows us to determine when and why the data collection process will be disrupted by users or for any other reason. Hence, this enables support to be provided efficiently and at a time when it will have the most impact.
Measurement
This study will determine the effectiveness of the i-CanManage program in improving physical activities, managing common symptoms, and QoL among its users through several questionnaires and tools. Sociodemographic characteristics such as age, relationship status, educational level and occupation, and medical history will be collected during the baseline survey. At three assessment times during each participant's 12-week intervention program, the following tools will be utilized:
Physical activity measurement
Physical activity will be evaluated by the International Physical Activity Questionnaire Short Form (IPAQ-SF). 63 This questionnaire is freely available in the public domain, so no permission to use is required. The Vietnamese version of IPAQ, validated by previous studies,64,65 will be used to collect self-reported physical activity. The amount of time spent performing each of three levels of intensity—vigorous (e.g. running), moderate (e.g. jogging), and low (e.g. standard-paced walking)—will be recorded. Total daily physical activity corresponding to the specific type of activity will be aggregated (MET-min per week). The three levels of physical activity are categorized as low (3.3 METs), moderate (4 METs), and vigorous (8 METs). 63
Symptom measurement
The app's comprehensive approach extends to assessing a spectrum of symptoms, encompassing pain (assessed via the Visual Analog Scale), depression (evaluated using the Patient Health Questionnaire), anxiety (measured through the Generalized Anxiety Disorder), stress (evaluated with the Perceived Stress Scale-10), along with other symptoms like fatigue, appetite loss, sleep disturbances, hair loss, nausea and/or vomiting, and sexual health issues. The frequency, timing, and combinations of these symptoms will be evaluated. Participants will rate the frequency of each symptom on a scale of 0 to 6: 0 (never); 1 (rarely); 2 (occasionally); 3 (sometimes); 4 (often); 5 (usually); and 6 (always). These symptom assessments will be recorded directly within the app.
Self-efficacy measurement
The Self-Efficacy to Manage Chronic Disease (SEMCD) scale will be employed to assess participants’ perceived self-efficacy in managing their chronic health conditions. This scale was developed based on Bandura's Social Cognitive Theory, which emphasizes the role of self-belief in motivating and guiding behavior. 66 In the context of chronic disease management, self-efficacy reflects the confidence a person has in their ability to manage their condition effectively and to adhere to recommended self-care behaviors. Thus, enhancing self-efficacy in patients is a cornerstone of symptom self-management enhancement, with the SEMCD scale providing a reliable and valid measure of self-efficacy across a broad range of health conditions, including for cancer.67–70
Participants will be presented with a series of statements that pertain to their ability to manage different aspects of their chronic condition. These statements cover such areas as symptom management, adherence to treatment plans, coping with challenges related to the condition, and maintaining a healthy lifestyle. Participants will rate their level of confidence in each statement on a numerical scale, usually ranging from “not at all confident” to “very confident.”
Self-care practice measurement
Participants’ self-care practices will be assessed using the Self-Care of Chronic Illness Inventory (SC-CII) developed by Riegel.
71
The assessment is based on three primary dimensions: self-care maintenance, self-care monitoring, and self-care management.
- Self-care maintenance refers to behaviors aimed at maintaining health and well-being, such as taking prescribed medications. - Self-care monitoring involves the continuous surveillance of one's condition, monitoring bodily signs and symptoms. - Self-care management involves recognizing and responding to symptoms, for example by adjusting medications to alleviate symptoms.
Originally developed in English, the SC-CII has demonstrated strong psychometric properties across diverse populations, with confirmatory factor analysis showing robust fit indices (CFI ranging between 0.93 and 1.00) and acceptable reliability (coefficients ranging from 0.67 to 0.86).72,73
QoL measure
Overall perceived QoL will be assessed by the 12-item Short Form Survey (SF-12). 74 In particular, reference will be made to the Vietnamese-translated version. 75 Participants will be asked about the impact of physical and mental health on their everyday life during the last four weeks through 12 general health questions. This tool will be evaluated using a standard scoring procedure published previously. 74 Physical and mental component summaries will be attained by the summation of scores for all corresponding questions. As an outcome measure, the higher a person's total score the better their overall health.
App usability and user experience
A 14-item mHealth satisfaction questionnaire completed on weeks 6 and 12 of the program will be used to measure participants’ satisfaction and user experience of the i-CanManage app. 76 Responses are rated on a five-point Likert scale from 1 (strongly disagree) to 5 (strongly agree). The negatively stated questions will be reversed for data analysis. Items will be considered separately. On weeks 6 and 12 users will be invited to share their personal experience of using the i-CanManage smart app through open-ended questions.
Data analysis
Sample size
The effect of sample size on studies of mobile health apps for cancer management tends to range from small to medium.77,78 Based on the estimated stepped rules of thumb for the required pilot trial sample size, 15 participants in an arm is recommended for the planned trial. This involves a two-side type I error rate of 5% and a power of 80% to detect a medium standardized difference effect size. 79 In order to fulfill these criteria, we will initially recruit 30 participants (15 participants in each arm) for this pilot RCT. The sample size of 15 per group allows us to gather initial insights into the intervention's potential impact and to identify any unforeseen issues that could be addressed in a larger-scale study.
Considering the observed dropout rate of 11.5% in a related study conducted in Hong Kong featuring the culturally adapted WWACPHK, 53 we will prudently accommodate this in our recruitment strategy. Thus, an additional four participants will be recruited, allocating two participants to each study arm. This precautionary measure aims to ensure that the target sample size for the trial is effectively maintained.
Primary analysis
SPSS version 28 will be used for statistical analyses. The paired t-test will be used for before and after comparison within the intervention group. Depending on whether the variable of interest is normally distributed or skewed, an independent t-test, Mann–Whitney U test, one-way ANOVA, or Kruskal–Wallis test will be used to compare values between the intervention and control group. These comparative analyses will facilitate a comprehensive measurement of feasibility, allowing for robust insights into the program's efficacy and potential benefits. The effect size, mean differences and their confidence intervals will be reported. A significance level of 5% will be set with a 95% confidence interval.
Ethical considerations
Privacy protection measures
In order to address privacy concerns, stringent measures will be implemented to safeguard participants’ personal information. Prior to data analysis, all individual identifiers will be removed, ensuring that the data are reported in an aggregated manner that prevents any possibility of identifying individual participants. The information displayed on the i-CanManage app strictly avoids collecting any data related to a participant's geographical location. These steps are taken to uphold participants’ privacy rights and to maintain the confidentiality of their sensitive information throughout the study.
Ensuring fairness for the control group
Ethical considerations regarding fairness are paramount. In order to ensure equity for the control group, a balanced approach will be adopted. Control group participants still receive the Handbook in a manner that is consistent with routine distribution. Subsequently, after the program concludes, the opportunity to access the program through the app will be extended to the control group participants. This approach aims to ensure that all participants, regardless of their group assignment, have the chance to benefit from the program and to uphold the principle of fairness.
Ethical approval
Ethical approval was granted by the Institutional Ethical Review Board of Vinmec International General Hospital JSC—VinUniversity (No. 75/2022/QD-VMEC, dated 26 July 2022).
Quality assurance
Hospital approvals and research assistant training
Prior to commencing the initial data collection, approval will be sought from selected hospitals where we intend to conduct the study. Research assistants (Ras) involved in data collection will undergo comprehensive training in the research process. This training will equip them with the necessary skills to perform their roles effectively.
Compliance with good clinical practice
This study adheres rigorously to the standards set forth in the International Conference of Harmonisation Guideline E6—Good Clinical Practice (ICH-GCP), a comprehensive framework providing guidelines for conducting clinical trial. 80 This ensures that the study will be executed in accordance with established ethical and methodological principles.
Standardized data collection procedures
In order to maintain accuracy in anthropometric measurements and survey data, we have established standardized operating procedures. These protocols will guarantee consistent and reliable data collection across all participants. They will be implemented by our RAs only after each has satisfactorily completed an exhaustive one-day training workshop. The continuous monitoring of RAs’ performance by the research manager will further uphold the highest standards of data collection.
Minimizing bias and enhancing integrity
The study employs a nonblinded randomization process to allocate participants to the control and intervention groups. Given the limited opportunities for interactions between these groups during the 12-week project duration—primarily occurring during daycare and outpatient visits—the risk of inadvertent influences is minimized. Furthermore, at present there are no existing online group or peer-to-peer support platforms for cancer patients in Vietnam. Each patient will be provided with a physical copy of the intervention handbook. Additionally, for the trial's app involvement, only healthcare professionals and the research team will possess the authority to add patients from the intervention group to the app. This measure guarantees controlled access and safeguards the study against unintended influences.
Discussion
Following treatment, women diagnosed with cancers have particular sets of health needs. Globally, healthcare systems struggle to support the rising numbers of cancer survivors with these health issues and to address subsequent treatment-related chronic diseases. 81 The use of mobile apps offers a potentially timely, efficient, and cost-effective strategy for monitoring physical activity behavior changes and promoting wellness compared to traditional in-person consultations.45,82 However, few studies to date have examined the effectiveness of mHealth in supporting cancer survivors’ wellness and physical activity maintenance.
This study will be the first to explore the postcancer treatment experiences of Vietnamese women and to evaluate how well a technology-based intervention, the CWP smart app, can assist them in readjusting to their normal lives. This app, which was developed based on the principles of the MRC framework and drawing from Bandura's Social Cognitive Theory, focuses on enhancing self-efficacy, promoting physical activity, and offering support for symptom management. An expected outcome is evidence of long-term positive effects on patients’ physical and psychosocial wellness and overall QoL. The results may also be applicable to people receiving treatment for, or recovering from, other chronic conditions such as rheumatic disorders, diabetes, or cardiovascular diseases.
Most current interventions are clinic-based and use direct face-to-face delivery measures, whereas our research is a community-based approach that utilizes a smart app.83,84 The findings of this study should improve understanding of how to effectively deliver community-based interventions for cancer patients. Also, they may add to the growing knowledge regarding the utility of mHealth, a recently emerging healthcare discipline, by providing evidence-based information on the impact of new smart health technologies in oncology and primary care practice. This may help to pave the way for future digital health innovations within the public health system in Vietnam and other low- and lower middle-income countries that are resource-constrained and where mHealth solutions offer viable alternatives.
Footnotes
Availability of data and materials
The authors affirm the availability of the data and materials supporting the findings of this study.
Contributorship
H.T.H.N., and L.K.B., T.N.T., N.T.T.N., A.H.P., H.H.P., A.W.T.-R., T.Q.D., and H.T.T.N. collectively contributed to the conceptualization and design of the study. H.T.H.N., L.K.B., and T.N.T. were responsible for the acquisition of funding. H.T.H.N. and L.K.B. played a pivotal role in the development and implementation of the i-CanManage program. T.N.T., N.T.T.N., A.H.P., and H.H.P. were involved in the recruitment of participants and data collection. A.W.T.-R. and T.Q.D. provided oversight and guidance throughout the research process. T.N.T. led the drafting of the manuscript. All authors critically reviewed and revised the manuscript for important intellectual content. H.T.H.N. supervised the entire study. All authors approved the final version of the manuscript for submission.
Conflicting interests
The authors assert that they do not have any competing interests associated with the publication of this work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This research was supported by Grant No. VUNI.2223.SG06, a VinUniversity Seed Grant for the year 2023.
Guarantor
Huyen Thi Hoa Nguyen serves as the guarantor of this article. As the guarantor, she accepts full responsibility for the work, had access to the data, and controlled the decision to publish.
