Abstract
Objective
Asthma is often inadequately controlled, which can result in exacerbations that lead to unplanned healthcare visits. Mobile application (app) use could help manage asthma exacerbations. We implemented the Amaze™ asthma disease management platform in clinical practice and assessed user satisfaction, usage, and usability.
Methods
Adults with asthma and healthcare professionals (HCPs) were enrolled from a community allergy practice (ClinicalTrials.gov Identifier: NCT04901260) and a large academic hospital (ClinicalTrials.gov Identifier: NCT04868500). Primary and exploratory outcomes included assessment of platform design, patient app usage, patient-reported daily asthma status, emergency room/urgent care visits, and ease of implementation by HCPs. The system usability scale and a post-clinic visit survey were also administered. HCPs/staff monitored the Amaze dashboard to assess patient needs and completed a post-study survey.
Results
Overall, 159 patients and five HCPs participated in the study. Patients’ mean (SD) age was 38.7 (16.4) years; most were female (78%) and White (78%). Mean patient app usage began at 3.6 days/week but declined to 1.0 day/week by the end of the study. Throughout the study, most daily entries (>69%) reported patient asthma status as “good.” Most patients were satisfied/very satisfied with the app (66%) and reported it helped them during discussions with their HCP (44%). Most patients rated the usability of Amaze as “excellent” (49%) or “good” (30%). Most HCPs (71%) reported that Amaze was “very easy” to implement.
Conclusions
Most patients and HCPs were satisfied with Amaze. The Amaze platform may help patients and HCPs monitor asthma status, which could improve asthma control.
Introduction
Inadequate disease control affects more than 50% of children and adults with asthma 1 and often goes unrecognized by patients, resulting in exacerbations that lead to unplanned emergency room (ER)/urgent care visits, hospitalizations, and systemic corticosteroid use.2,3 The rate of office visits for patients with asthma as the primary diagnosis was 308 per 10,000 people in 2016 in the United States. 4 Unfortunately, asthma is inadequately controlled in most patients, likely due to suboptimal symptom management, which typically results in a need for healthcare services. 5 Consequently, healthcare costs for patients with uncontrolled asthma are significantly higher than in those without asthma. 6 Asthma significantly impacts patients’ quality of life, affecting their social interactions, educational achievements, and mental health. Poorly controlled asthma can lead to missed school or work days, limitations in physical activities, and a general decline in overall well-being. 7
Patients are often unaware that they have uncontrolled asthma because they are not well informed about disease symptom variability and signs of poor asthma control. 8 Uncontrolled asthma can result from poor medication adherence, difficulty treating the disease due to inadequate response to treatment, or poor healthcare professional (HCP) adherence to treatment guidelines. 9 Asthma control can typically be improved with maintenance therapy, such as inhaled corticosteroids, if the patient maintains adherence to their medical regimen. 9 A system that assists patients in obtaining further care from HCPs could help address early signs of exacerbation and poor asthma control.
Because asthma is a chronic disease that requires frequent symptom assessment and medication adjustment, patients may benefit from mobile technologies to help monitor and control their asthma symptoms. 10 Mobile technologies can be used to provide treatment reminders to patients and aid in disease prevention and management, symptom monitoring, and communication between HCPs and patients. 11 Mobile technologies currently being used for health assessment and disease management include personal digital assistants, tablets, wearable biometric devices, short messaging service, cell phone applications (apps), and Bluetooth-compatible devices.11,12 Mobile technologies not only improve patient engagement by providing educational resources and self-management tools but also enable personalized treatment plans based on real-time data. This individualized approach can lead to more effective asthma control and a better patient experience. 13 A 2015 systematic literature review found that 56% of studies reported a significant improvement in medication adherence and 39% reported a significant improvement in clinical outcomes when mobile technologies were used to manage different chronic diseases. 12 An estimated 350,000 health-related mobile apps are available. 14 However, very few of them relay information to HCPs via connection to electronic health records (EHRs). 15
The Amaze™ digital platform was developed to enhance communication between patients with asthma and their HCPs by utilizing a patient mobile app and an HCP EHR-integrated dashboard that generates actionable insights to improve patient care. In the Amaze app, patients can enter daily symptoms and communicate with their HCP. In the EHR-integrated dashboard, HCPs can review patients’ asthma control and provide guidance based on patient-reported data. The goal of this study was to evaluate usage, usability, and satisfaction with the platform; patient-reported asthma experiences; and ease of implementation of the app.
Methods
Study design
This was a prospective, observational study conducted over several months at a community allergy practice (Austin, TX; November 2020 to June 2021) and a pulmonology practice at a large academic hospital (Boston, MA; April to November 2021).
Adult patients (aged ≥18 years) with a clinically confirmed asthma diagnosis and access to a smartphone with internet access (operating system iOS 13 or newer/iPhone 8 or newer or Android operating system 8 or newer) were enrolled on a rolling basis. Patients were excluded if they had a concurrent diagnosis of chronic obstructive pulmonary disease or other non-asthma pulmonary disease. Best efforts were made to recruit patients who had moderate-to-severe asthma, as defined by treatment with inhaled corticosteroids/long-acting beta agonist combination therapy with or without add-on therapy, or as determined by their treating HCP. After providing informed consent, patients were asked to download the Amaze app (Figure 1).

Amaze app screenshots showing key features. (A) Patient app interface and (B) clinician dashboard.
HCPs and clinical support staff monitored the Amaze dashboard to assess changes in patient asthma symptoms over the study period and implemented changes in care based on clinical judgment. Patients and clinic staff from each site were asked to complete several surveys throughout the study and provide feedback on Amaze. The overall study duration was 7 months, and each patient's participation lasted 3 to 6 months depending on when they enrolled in the study. A subset of patients was randomly selected by block randomization of 1:4 to participate in longitudinal qualitative telephone interviews at months 1 and 3 and at the end of the study to gain a deeper understanding of the usability of the Amaze app and to elicit recommendations for improvement. Written informed consent was obtained prior to survey completion, and written and verbal informed consent was obtained before the first interview and reconfirmed before each additional interview.
Patient-completed measures
Patients completed questionnaires via a web survey and the Amaze platform. Permission to use copyrighted questionnaires was obtained by the study sponsor. Patients first completed a baseline sociodemographic questionnaire. At months 1 and 3 and at the end of the study, they completed a study-specific user experience survey. The survey consisted of 12 questions about their app experience with a 5-star response scale in which 1 = poor and 5 = excellent, the proposed frequency of use for app features (daily, weekly, monthly, or other), and four questions about satisfaction and concerns with the app. At the end of the study, patients completed the system usability scale (SUS), 16 which is a 10-item questionnaire that evaluates the usability of a system based on user feedback. The SUS uses a 5-point Likert scale by which users rate their experiences from 0 = strongly disagree to 4 = strongly agree. The total score is then multiplied by 2.5 to convert the scores to 0–100. 17 Finally, patients completed a visit experience survey any time they had an in-person or telehealth visit. The survey evaluated satisfaction with their visit and any benefits the app provided.
The Amaze app prompted users to submit a daily asthma log, including for overall asthma condition (“bad,” “okay,” or “good”), as well as asthma symptoms, triggers, medication use (including maintenance, oral steroids, and rescue), and ER/urgent care visits. The app allowed patients to send messages to clinical site staff using pre-defined subject lines. The app also provided air quality reports, trends of daily logs, patient asthma plans, appointment viewing and scheduling, and asthma education. Additionally, patients were asked to complete the Asthma Control Test (ACT™) monthly. The ACT is a standardized questionnaire with five items used to monitor asthma control. Based on ACT scores, asthma was classified as well-controlled (≥20), not well-controlled (16–19), or very poorly controlled (≤15). 18
Clinical site staff-completed measures
HCPs completed a form at baseline and at the end of the study for each enrolled patient to collect information on current medication use, changes to controlled medicines, HCP-rated asthma severity and control, exacerbation history, and number of clinic visits. Medication use (by drug class) was used to approximately classify asthma severity according to Global Initiative for Asthma (GINA) classes 1/2 (mild) or classes 3/4/5 (moderate to severe). Physicians were asked to report whether patients had a step-up/step-down in treatment, whether their medication dose was increased/decreased, whether a new medication was added/removed, or whether their GINA class increased/decreased. 19 Frequency of asthma exacerbations, defined as a patient requiring steroids and/or a hospitalization/ER visit, was calculated over the study period. Clinical site staff also completed a study-specific post-study survey of the app to provide feedback on their experience implementing it and to suggest other useful information that the app could provide in the future.
Statistical analysis
Data were analyzed using SAS version 9.4 (SAS Institute, Cary, NC, USA) and descriptive statistics were calculated. Because this was an implementation study and no inferential statistics were planned, a formal sample size calculation was not performed.
Primary outcomes
Primary outcomes included patient app usage, clinician dashboard usage, patient-perceived usability and satisfaction with the app, patient-perceived advantages and disadvantages of the app, ease of implementation by HCPs, and qualitative feedback on the daily log. App usage by patients was formally tracked using user IDs and was defined by the average number of days per week they completed the daily log (a maximum of once per day). Levels of app usage in 4-week intervals were defined as follows: high (5–7 days/week), medium (3–4 days/week), low (1–2 days/week), and very low (<1 day/week). Clinician dashboard usage was formally tracked via user IDs and assessed as the mean number of days the dashboard was utilized per month. Individual item responses were descriptively reported for the user experience survey and HCP post-study survey. Patient SUS scores were categorized as excellent (>80.3), good (>68 to ≤80.3), okay (68), poor (≥51 to <68), or awful (<51).
Exploratory outcomes
Exploratory outcomes included exacerbations, healthcare resource utilization (e.g. ER/urgent care visits), and change in asthma therapy during the study, as reported by the clinical sites based on a medical chart review at month 6, and patient-reported effects of the app on clinic visit experiences. Symptoms, triggers, and rescue medication use reported on the app were analyzed according to daily log entries for overall asthma condition.
Results
Sociodemographic and clinical characteristics
A total of 159 patients were enrolled across the two sites (community allergy practice: n = 117; large academic hospital: n = 42) (Figure A.1). Two patients from the community allergy practice were deemed ineligible due to other pulmonary diagnoses. From the academic center, three patients were withdrawn due to their HCP withdrawing from the study, and one patient was eligible but did not open the app. Therefore, 153 patients (community allergy practice: n = 115; large academic hospital: n = 38) participated. One patient at the community allergy practice discontinued the study early, leaving 152 patients at the end of the study (community allergy practice: n = 114; large academic hospital: n = 38).
Five HCPs (community allergy practice: n = 1; large academic hospital: n = 4) and four clinic staff members (community allergy practice: n = 1; large academic hospital: n = 3) participated in the study. One HCP discontinued the study early, leaving a total of eight HCPs/clinic staff members in the study.
Baseline sociodemographics were reported by 120 patients (78%) (Table 1). The mean (SD) age of patients was 38.7 (16.4) years. Most were female (n = 93, 78%), White (n = 94, 78%), not Hispanic (n = 98, 82%), and had at least some college education (n = 81, 61%). At baseline, HCPs reported that most patients (n = 118, 77%) had well-controlled asthma (Table 2). However, baseline ACT scores showed that only half of patients (n = 77, 50%) had well-controlled asthma.
Patient sociodemographic characteristics.

SD: standard deviation.
Of the 115 eligible patients at baseline, only n = 82 completed the baseline survey.
Other includes: Hispanic; Southern European/English/Irish; Spanish, Jewish.
Not mutually exclusive.
Artist (n = 1), project manager (n = 1), self-employed full-time (n = 1).
General Educational Development (GED).
HCP-reported clinical characteristics of patients.

ER: emergency room; GINA: Global Initiative for Asthma; HCP: healthcare professional.
HCPs reported that more patients had moderate persistent asthma (n = 96, 63%) than severe persistent (n = 31, 20%) or mild persistent asthma (n = 20, 13%). A larger percentage of patients at the large academic hospital had moderate persistent asthma (n = 28, 74%) at baseline compared to patients at the community allergy practice (n = 68, 59%). From the baseline assessment, a larger percentage of patients from the community allergy practice (n = 47, 41%) were on at least one biologic therapy compared to patients at the large academic hospital (n = 7, 18%).
Primary outcomes
Amaze app and dashboard usage
Overall, app usage declined over time (Figure 2). The mean (SD) number of times the app was used was 3.6 (1.9) days/week during weeks 1–4 and 3.1 (2.3) days/week during weeks 5–8. Thereafter, app usage declined to a mean (SD) of 1.0 (2.0) day/week by weeks 21–24. Overall, most patients had high (5–7 days/week; 26.1%–28.1%) or medium (3–4 days/week; 28.8%–35.3%) usage of the daily log during the first 8 weeks of use.

Amaze patient usage per 4-week period (N = 153). The white boxes indicate mean number of entries per week.
Across both sites, clinical site staff review of the dashboard increased from weeks 1–4 to weeks 21–24 (mean [SD] days per month: 7.6 [5.1] vs 9.0 [5.7]) (Figure A.2).
Usability of Amaze
At the end-of-study visit, 70 patients completed the SUS. Most patients rated the Amaze app as “excellent” (49%) or “good” (30%). The remaining patients rated the app as “poor” (11%) or “awful” (10%).
Perceived advantages and disadvantages of Amaze
Seventy-seven patients (51% response rate) completed the end-of-study user experience survey. Patients reported wanting to track their symptoms, triggers, and rescue and maintenance medication use on a daily or weekly basis (Table A.1). However, they preferred to track their ER/urgent care visits and peak flow monthly or less frequently. Most patients reported “forgetting to use the app” (51%) and “things missing in the app that they wanted” (22%) as their top concerns.
For the categories of daily logs, air quality, and trends of daily logs, most patients provided 4- or 5-star ratings (Figure 3). Features that were not frequently used included the patient asthma plan (not used by 47% of patients), appointment viewing and scheduling (52%), asthma education (57%), and messaging the clinic (65%). Most patients were satisfied or very satisfied with the app (n = 51, 66%) and would likely or very likely recommend the app to other patients with asthma (n = 51, 66%).
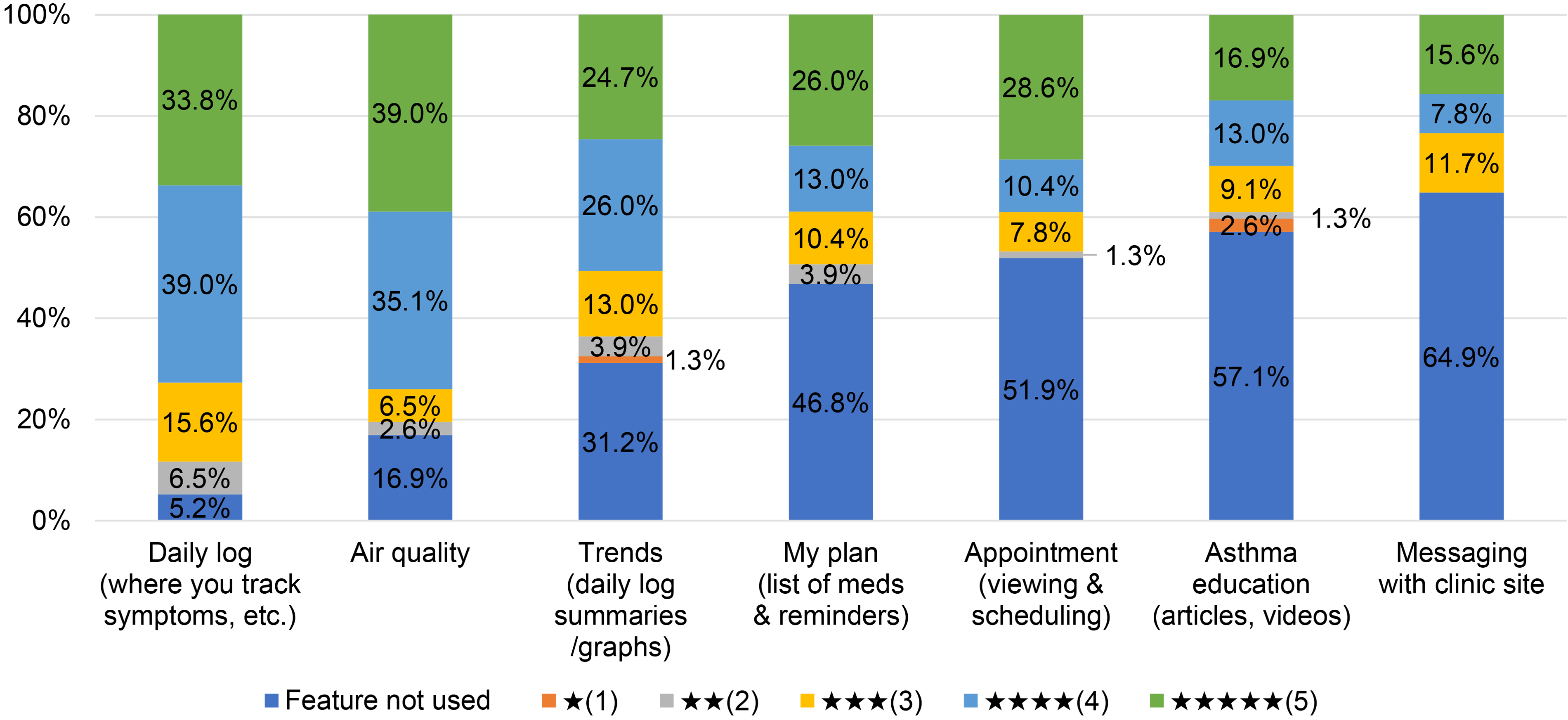
End-of-study patient ratings for Amaze features (N = 77). Patients were asked to rate each feature of the app on a 5-star rating. Ratings ranged from 1 = poor to 5 = excellent.
Ease of implementation
Five of the seven HCPs (71%) who completed the post-study survey reported that Amaze was “very easy” to implement in their practice (Table A.2). They reported that the most useful aspects were the ability to track patient asthma symptoms (n = 3, 43%) and the ability to integrate the platform into their EHR system (n = 3, 43%). Most HCPs reported wanting to track their patients’ asthma symptoms and rescue and steroid medication use daily or weekly. However, they wanted to track patient asthma triggers, ER/urgent care visits, and peak flow weekly or less often. Most HCPs reported that Amaze “moderately” (n = 2, 29%) or “slightly” (n = 2, 29%) helped them to manage their patients.
Qualitative feedback on the daily log
Longitudinal patient qualitative interviews were conducted at month 1 (n = 22), month 3 (n = 15), and at the end of the study (n = 14). Almost all patients at month 1 (n = 21, 96%) thought that the app was easy to use, straightforward, and intuitive. At the end of the study, nine patients (64%) provided feedback on areas for improvement on the daily log, including providing more options for symptoms and triggers (n = 2/14, 14%) and revising the response scale (n = 1/14, 7%). Other desired changes suggested by one patient each (n = 1/14, 7.1%) included being able to retroactively complete the log, being able to visually review trends, and reducing the frequency of completion for those with well-controlled asthma.
Exploratory outcomes
A minority of patients (n = 21, 14%) experienced an asthma exacerbation during the study. By the end of the study, 23 patients (15%) received stepped-up therapy, 27 patients (18%) received stepped-down therapy, and 102 patients (67%) had no change in inhaled corticosteroid dose, GINA level, or class of medication.
Clinic visit experience
Of the 43 survey responses, 19 patients (44%) agreed or strongly agreed that “The app helped me discuss my asthma with my HCPs during my most recent visit” (Table A.3). Sixteen patients (37%) agreed or strongly agreed that “the app helped the appointment with my HCP go more smoothly.”
Daily logs, symptoms, triggers, and medication usage
Daily asthma status assessed via the daily log entries for patients was consistent throughout the study, with most entries (>69%) reported as “good.” When evaluating the relationship between daily log entries and symptoms and triggers, no clear patterns were identified for triggers (Table A.4). Chest congestion (28%) (Table A.5) and use of quick relief/rescue medication (76%) (Table A.6) were reported more frequently for patients rating their daily asthma status as “bad” than for other log entries.
Discussion
The Amaze digital platform was implemented at two distinct clinical practices and provided useful information for patients and HCPs regarding asthma symptoms, experiences, and exacerbations. Most patients felt that the app was easy to use and intuitive. Features that were rated highly included the daily log, air quality reports, and daily log trends. However, features such as the patient asthma plan, appointment viewing/scheduling, asthma education, and messaging the clinic site were not widely used, suggesting a need for improvements. HCPs reported that Amaze was easy to implement in their clinical practice and valued patient tracking of their asthma symptoms, which could help improve shared decision-making with patients. Limited use of the messaging feature may in part have been because the clinics were already using messaging platforms. Interestingly, HCP and patient perceptions of asthma control status differed: 77% of HCPs believed that their patients’ asthma was well-controlled, whereas ACT results indicated that only 50% of patients had well-controlled asthma.
Qualitative patient feedback indicated that the Amaze app was highly usable. This is significant, as an earlier systematic review identified usability issues as a key barrier to successful delivery of digital health solutions. 20 Usability was similarly high in pilot testing of an app developed to monitor inhaler use and gather surveys from children and adolescents with asthma. 21 Although app usage was initially high, it declined over time. This may partly have been due to lack of training on the app and because not all patients enabled app reminder notifications. Additionally, some patients had technical issues logging into the app when their passwords expired. Establishing and maintaining app usage is a frequent problem with asthma digital health solutions. A retrospective real-world analysis of a web-based asthma treatment platform for adults with asthma found that over a quarter of patients who were registered on the platform never used it. 22 Interestingly, patients who did use the platform tended to have more severe asthma than non-users. In a recent randomized controlled study of a commercially available app for asthma self-management, two-thirds of participants stopped using the app within the first eight weeks of the study. 23 Adding more options for symptoms and triggers, using a revised daily log scale, providing a visualization of trends (as proposed by one patient), and making the app more game-like may help to maintain long-term Amaze usage. Symptom and trigger monitoring and visualization of trends were similarly highlighted as potentially useful digital health features by adults with asthma who participated in a qualitative interview study in Ireland. 24
Based on patient and HCP feedback, daily use of the Amaze app may not be necessary. Notably, one interview participant expressed that the frequency of log completion could be reduced for patients with adequately controlled asthma. Because most patients had adequately controlled asthma at baseline, some of these patients may have considered the requirement to complete the log daily excessive. Adapting the frequency of log completion to asthma control status may increase user engagement with the Amaze app.
A strength of the present study was random selection of telephone interview participants to avoid selection bias. Also, obtaining information from both a community allergy practice and large academic hospital likely increased the generalizability of the findings. However, the socioeconomic status of patients was generally high, suggesting the need to consider the ability of patients of lower socioeconomic status to access and use the app. The location of the clinics and the mobile device requirements for participation, together with the high education and limited racial heterogeneity of patients, means that the sample of patients in our study may not be fully representative of patients with asthma in the United States. Moreover, reduced app usage at the end of the study resulted in small sample sizes, making comparisons across subgroups challenging. Future studies should evaluate a larger sample of patients with greater geographic and socioeconomic diversity. This study was conducted during the COVID-19 pandemic, which limited the ability for in-person clinical visits. This may have reduced app usage and communication between patients and HCPs. Finally, the patient user experience survey and HCP post-study survey are unvalidated instruments. However, the lack of validation of these questionnaires should be considered a minor limitation because these were only used to descriptively characterize the study sample.
Conclusions
The present study demonstrated that both HCPs and patients were able to successfully monitor asthma severity, medication use, and asthma management through an integrated digital platform. Most patients were satisfied with the Amaze app and found it easy to use. However, this study highlights the need for additional training in future implementation of Amaze to ensure patients are aware of the full range of app features available to them. Although both HCPs and patients demonstrated high levels of engagement, future studies may be needed to evaluate how often patients should use a mobile disease management platform as well as strategies to enhance continued patient engagement. Future research should also focus on optimizing features based on user input and determining whether use of a digital platform is associated with improved asthma control.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241282380 - Supplemental material for Observational study of the Amaze™ asthma disease management platform
Supplemental material, sj-docx-1-dhj-10.1177_20552076241282380 for Observational study of the Amaze™ asthma disease management platform by Jehan Alladina, Peter P Moschovis, Hitesh N Gandhi, Donna Carstens, Elizabeth D Bacci, Katelyn Cutts, Karin S Coyne, Karen Goldsborough, Dawei Jiang, Conor O’Brien and T Bernard Kinane in DIGITAL HEALTH
Footnotes
Abbreviations
Acknowledgements
The authors thank Dr Eric Schultz, Investigator at Greater Austin Allergy in Austin, TX, for his support and feedback during the study. The authors also thank Remona Kanyat, Dalton Lancaster, Jennifer Rooney, and Nora Salo at Massachusetts General Hospital for their support in participant recruitment, data management, and preliminary analyses. Medical writing support, under the direction of the authors, was provided by Holly Richendrfer, PhD, and Stephen Gilliver, PhD, of Evidera, which was in accordance with Good Publication Practice (GPP 2022) and funded by AstraZeneca. Editorial and manuscript submission support was provided by Kiley Margolis, PharmD, of Lumanity Communications Inc. (Yardley, PA, United States), which was in accordance with Good Publication Practice (GPP 2022), funded by AstraZeneca, and authorized by all listed authors. The Amaze app was developed in collaboration with BrightInsights.
Contributorship
JA conceptualized the study; conducted the formal analysis, investigation, visualization, supervision, and project administration; and participated in the writing, review, and editing of the manuscript. PPM conducted the investigation, supervision, formal analysis, and project administration; and participated in the writing, review, and editing of the manuscript. HNG and DC conceptualized the study; developed the methodology; conducted the formal analysis, investigation, and supervision; and participated in the funding acquisition and the writing, review, and editing of the manuscript. EDB, KC, and KSC conceptualized the study; developed the methodology; conducted the formal analysis, investigation, supervision, data curation, visualization, and project administration; provided resources; and participated in the writing, review, and editing of the manuscript. KG, DJ, and CO participated in project administration and in the writing, review, and editing of the manuscript. TBK conceptualized the study; conducted formal analysis, investigation, visualization, and project administration; provided resources; and participated in the writing, review, and editing of the manuscript. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JA has received research funding from AstraZeneca and a grant from the National Institutes of Health (grant #5KL2TR002542-04). PPM has received a grant from the National Institutes of Health (grant #K23ES030399) and has served on an adjudication committee for Pfizer. HNG was an employee of AstraZeneca at the time this work was completed and is a current employee of Alexion (AstraZeneca Rare Disease). DC is an employee of AstraZeneca. KG was an employee of AstraZeneca at the time this work was completed. EDB, KC, and KSC are employees of Evidera, which was contracted by AstraZeneca to conduct this study. DJ, CO, and TBK have no relevant conflicts of interest to disclose. All listed authors approve the funding and conflict of interest disclosure statements.
Ethics approval
The study was approved by two independent Institutional Review Boards (IRBs): Ethical and Independent IRB approved the study for the community allergy practice (protocol #20181-01) and Mass General Brigham IRB approved the study for the large academic hospital (protocol #2021P000260).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by AstraZeneca. Three of the named authors are employees of AstraZeneca and therefore contributed to the design of the study, the analysis and interpretation of the results, and the decision to publish.
Trial registration
The study was conducted at a community allergy practice (Austin, TX, USA; ClinicalTrials.gov identifier: NCT04901260) and a pulmonology practice at a large academic hospital (Boston, MA, USA; ClinicalTrials.gov identifier: NCT04868500).
Guarantor
JA
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
