Abstract
Background
Levodopa-carbidopa intestinal gel (LCIG) is a continuously delivered Parkinson's disease therapy intended to stabilize plasma levodopa levels. Patients receiving LCIG require education and follow-up. Some LCIG support programs use video-assisted telenursing.
Objective
To examine how videoconferencing impacts satisfaction with LCIG support programs.
Methods
FACILITATE CARE (
Results
Patients’ mean (standard deviation) ages were 67.9 (7.4, n = 26) and 71.1 (6.2, n = 15) years in the video and audio arms, respectively. Patients, caregivers, and physicians in both groups reported satisfaction scores of 8–10 with LCIG support personnel, communication access, and assistance with becoming independent. At week 12, the Modified Caregiver Strain Index least square means change from baseline was lower in the video vs. audio arm (−2.3 [1.0] vs. 1.6 [1.2]). LCIG support personnel travel time was lower in the video vs. audio arm (125.7 [70.2] vs. 203.0 [70.0] minutes).
Conclusions
LCIG support programs are associated with high patient, caregiver, and physician satisfaction; video and audioconferencing satisfaction are similarly high. Video-assisted telenursing may be a convenient communication avenue and may reduce caregiver burden.
Registration
ClinicalTrials.gov; NCT04500106.
Keywords
Introduction
Parkinson's disease (PD) is a common neurodegenerative disease identified by resting tremor, rigidity, akinesia, and postural instability that can lead to loss of mobility and a reduced quality of life.1,2 Although highly effective, oral administration of levodopa is often complicated by response fluctuations, resulting in dyskinesias and motor impairment. 3
Levodopa-carbidopa intestinal gel (LCIG) is continuously delivered via a portable pump placed through a percutaneous endoscopic gastrostomy with a duodenal/jejunal extension (PEG-J) tube with the aim of providing stable plasma levodopa concentrations. 4 Patients treated with LCIG therapy undergo a titration period, typically during a hospital stay, and often require education on the device system, monitoring, and follow-up once patients return home. 5
The LCIG support program, funded by AbbVie, is staffed by specialist nurses and consists primarily of 2 components (only in countries where direct contact of LCIG support personnel with the patient is allowed):
Providing preplanned contacts with patients and caregivers during the first few months after LCIG initiation, involving proactive education of the patient and caregiver on the device system (pump, tubes, connectors) and its use; monitoring the patient's understanding/handling of the device system; and following up on the patient's stoma condition. Performing reactive troubleshooting of device- and system-related issues, of which some can be resolved remotely, some require travelling to patients’ homes, and some require further medical intervention at a hospital. For reactive troubleshooting, patients or caregivers usually contact the LCIG support personnel for resolution of the unforeseen acute device system issues (e.g., pump, tubes, or connectors).
Currently, the primary contact is typically done via telephone (audio-connection only), but a reactive home visit may be required to reeducate the patient and caregiver on the device system, to check the technicalities of the system, or to examine the patient's stoma to determine the next steps.
Personnel in the LCIG support program can spend considerable time traveling to patients’ homes, particularly in remote or rural areas. Excessive travel times can potentially lead to a delay in resolving issues. Additionally, circumstances may arise that make direct patient contact challenging (e.g., the recent COVID-19 pandemic). Therefore, in some countries, LCIG support programs have introduced videoconferencing, which can be implemented using support personnel and patients’ existing communication devices (smartphones, laptops, tablets).
Videoconferencing success has been examined in previous studies. In a pilot study, 6 LCIG titration via stationary videoconferencing equipment proved to be resource efficient; technically feasible; well accepted; and deemed satisfactory by patients, neurologists, and nurses. A separate randomized controlled trial 7 enrolled 210 patients with PD and supplemented usual care with virtual house calls by remote specialists. While quality of life, quality of care, and caregiver burden were not significantly different for patients receiving virtual house calls compared with those having face-to-face interactions, the technology proved to be feasible, and each virtual house call saved patients a median of 88 min and 38 miles per visit. Results from another pilot trial 8 in Germany where 50 patients with PD used telemedicine showed an improvement in scores on the 39-Item Parkinson's Disease Questionnaire. Our observational study prospectively examines patient satisfaction scores with LCIG support programs associated with videoconferencing.
Methods
Observational study design
FACILITATE CARE (
In this observational study, patients and their caregivers assigned themselves to either the video arm or audio-only arm, based on their preference and familiarity with handling digital devices. Baseline visits took place after the patient had a PEG-J tube placed and before hospital discharge. Study initiation began at the time of patient discharge from the hospital. Patients were followed up for three months post-discharge; three months was chosen as the follow-up period because it is during this period when patients typically require the most support in resolving technical issues (Figure S1). Follow-up visits were conducted at weeks 4 and 12. Serious adverse events and product quality complaints were assessed in the 12-week follow-up period and for an additional 30 days after the end of the follow-up period using video- and audio-assisted devices. Patients used their own devices for videoconferencing. No additional devices were provided to patients or LCIG support personnel. In the video arm of this observational study, all preplanned and unplanned phone calls were addressed using the video function of the devices. The preplanned, face-to-face visits at patients’ homes remained face-to-face as scheduled by the local LCIG support program.
Clinical data were collected and reported electronically on electronic case report forms via a web address and secure passwords. Patient/caregiver self-administered questionnaires were completed via paper forms and responses were then entered into the electronic case report form by site staff.
Inclusion and exclusion criteria
Adult patients between the ages of 18–85 years were included in this observational study if they met the eligibility criteria for LCIG therapy in accordance with the approved local LCIG label. Patients had to be LCIG-naive, must have completed in-hospital titration, had a PEG-J tube placed, and were subsequently discharged from the hospital. The treating physician needed to have made the decision to treat patients with LCIG before approaching patients regarding study participation. Patients had to own a telecommunication device with videoconferencing capabilities and needed to be capable of managing their device.
Patients were excluded from the study if they had any contraindications as listed in the approved local LCIG label of their participating country, if patients lacked caregiver support, if they were participating in a concurrent interventional clinical trial, or if they weren’t motivated or had insufficient language skills to complete the study questionnaire (e.g., they were unable to understand the questionnaire language).
Assessments
Patient, caregiver, and investigator satisfaction was assessed at study visits. The primary endpoint of the study was patient acceptance as measured by patient satisfaction with the LCIG personnel support and communication access at week 12. Satisfaction was assessed using a visual analog scale (VAS) score from 1 to 10, with 10 being the highest level of satisfaction. Secondary endpoints included patient satisfaction with LCIG personnel support and communication access at week 4 (VAS from 1–10), percentage of patients with a VAS satisfaction score of 8–10 with LCIG personnel support and communication access at week 12, caregiver satisfaction/acceptance with LCIG personnel support and communication access at weeks 4 and 12 (VAS from 1–10), physician satisfaction with LCIG personnel support at week 12 (VAS from 1–10), patient satisfaction with the video function of the device specifically at weeks 4 and 12 (VAS from 1–10), and the change of caregiver burden (as measured by the Modified Caregiver Strain Index [MCSI] score) from baseline to week 12. Exploratory endpoints included duration of telephone calls by the LCIG support personnel from baseline to week 12 (including all preplanned and unplanned phone calls), LCIG support personnel travel time per patient spent for visiting patients at home from baseline to week 12 (unscheduled visits only), travel time required per patient to a hospital from baseline to week 12 (unscheduled visits only), percent of unplanned issues that were resolved by video-based troubleshooting (“Resolution” was defined as no home visit necessary, due to either the problem being resolved, or being advised by the LCIG personnel to directly visit a hospital), and drop out during study duration.
Statistical analysis
The sample size of approximately 50 patients, with 25 per arm, was calculated to control the precision of estimated mean within each treatment arm. Most studies on VAS score of patient satisfaction in the literature had standard deviation (SD) between 1.7 and 2.1 when converted to a 10-point scale. With a known SD of 1.7, a sample size per arm of 25 provides a two-sided 95% confidence interval (CI) with a precision of 0.666 for VAS scores on a 10-point scale. Results from previous studies indicated that the minimal clinically significant difference on patient satisfaction scores using VAS were 0.7–1.1, when converted to a 10-point scale. 9 Outcomes were summarized using descriptive statistics and based on observed data.
For continuous outcomes, the mean (SD) was provided, grouped by treatment arm. Frequency and percentage were provided for categorical outcomes. The least squared means change from baseline applied an exploratory mixed model repeated measures analysis using SAS® MIXED procedure with REPEATED sub command (SAS Institute, Cary, NC). The change from baseline was analyzed as the response-dependent variable. The model included terms for study arm, categorical post-baseline visit, study arm by categorical post-baseline visit interaction, baseline variable measure, and baseline variable measure by visit interaction. The model used the unstructured covariance matrix, the restricted maximum-likelihood estimation method, and the Kenward–Roger adjustment method for the degrees of freedom.
Results
Enrollment began on 2 September 2020, with the last data collection on 15 November 2021. Forty-one patients were enrolled in the intent-to-treat (ITT) population (all patients in this observational study); 39 patients were included in the full analysis set (all ITT patients with baseline and ≥1 post-baseline measurement), with 24 patients in the video arm and 15 patients in the audio arm (Figure 1). A total of 37 patients completed the study. The mean age (SD) of the patients in the ITT population was 67.9 (7.4) years in the video arm and 71.1 (6.2) years in the audio arm; 100% were White in both the video and audio groups. In the video arm, 53.8% of patients had a university education; in the audio arm, 20.0% of patients had a university education. Further baseline characteristics are described in Table 1.

Patient disposition. AE: adverse event; FAS: full analysis set; ITT: intent to treat.
Baseline and clinical characteristics (ITT population).

ITT: intent to treat; LCIG: levodopa-carbidopa intestinal gel; MMSE: mini-mental state examination; SD: standard deviation.
Primary endpoint
In the observed cases of the full analysis set, the mean (SD) patient VAS scores for satisfaction with LCIG personnel support and communication access were 9.1 (1.9; n = 22) in the video arm and 9.3 (1.0; n = 14) in the audio arm at week 12 (Figure 2a). The combined VAS scores for both arms for satisfaction with LCIG personnel support and communication access at week 12 were 9.2 (±1.6, n = 36). The median VAS scores were 10.0 in both the video and audio arms. In the video and audio arms, 95.5% and 92.9% of patients were completely satisfied (VAS score 8–10) (Figure 2b).

Primary endpoint of (a) patient satisfaction with LCIG personnel support and communication access at week 12, and (b) the distribution of patient VAS satisfaction scores. LCIG: levodopa-carbidopa intestinal gel; SD: standard deviation; VAS: Visual Analog Scale.
Secondary endpoints
The mean patient VAS scores with LCIG personnel support and communication access at week 4 were 9.5 (±1.0, n = 21) in the video arm and 9.7 (±0.6, n = 15) in the audio arm (Figure 3). At week 4, the mean caregiver VAS scores for satisfaction with LCIG personnel support and communication access were 9.2 (±2.1, n = 21) and 9.4 (±0.8, n = 14) in the video and audio arms, respectively (Figure 4); the mean caregiver VAS scores for satisfaction with LCIG personnel support and communication access were 9.2 (±2.0, n = 24) in the video arm and 9.0 (±1.1, n = 15) in the audio arm at week 12.

Patient satisfaction with (a) LCIG personnel support and communication access, (b) video conversations, (c) LCIG personnel support specifically, and (d) assistance in becoming independent. LCIG: levodopa-carbidopa intestinal gel; SD: standard deviation; VAS: Visual Analog Scale.

Caregiver satisfaction with (a) LCIG personnel support and communication access, (b) video conversations, (c) LCIG personnel support specifically, and (d) assistance in becoming independent. LCIG: levodopa-carbidopa intestinal gel; SD: standard deviation; VAS: Visual Analog Scale.
The mean patient VAS scores for satisfaction with the video conversations with the LCIG personnel at weeks 4 and 12 were 9.3 (±2.0, n = 21) and 8.8 (±2.2, n = 20), respectively (Figure 3). At weeks 4 and 12, the mean caregiver VAS scores were 9.4 (±2.0, n = 20) and 9.0 (±2.1, n = 21), respectively (Figure 4). The mean patient, caregiver, and physician satisfaction VAS scores with the LCIG personnel support specifically at week 12 were 9.1 (±2.0, n = 22), 9.1 (±2.0, n = 24), and 9.2 (±1.9, n = 22) for the video arms and 9.4 (±0.8, n = 14), 9.4 (±0.8, n = 15), and 9.3 (±0.6, n = 15) for the audio arms, respectively (Figures 3, 4, and 5).

Physician satisfaction at week 12 with (a) LCIG personnel support specifically, and (b) assistance in becoming independent. LCIG: levodopa-carbidopa intestinal gel; SD: standard deviation; VAS: Visual Analog Scale.
The mean patient satisfaction VAS scores at week 12 in assistance with becoming independent were 9.1 (±2.1, n = 20) in the video arm and 8.7 (±2.1, n = 14) in the audio arm (Figure 3). The corresponding caregiver satisfaction VAS scores with becoming independent were 8.8 (±2.2, n = 21) and 9.2 (±1.2, n = 15) for the video and audio arms, respectively (Figure 4). The mean physician satisfaction VAS scores for the video and audio arms were 9.1 (±1.9, n = 22) and 9.1 (±0.9, n = 15), respectively (Figure 5).
Change from baseline in MCSI scores to week 12 indicated that caregiver strain decreased in the video arm with a least squares mean change from baseline (standard error) of −0.7 (0.8) and −2.3 (1.0) at weeks 4 and 12, respectively (Figure 6a). Caregiver strain increased over time in the audio arm with a least squares mean change from baseline ± standard error of 0.4 ± 1.1 and 1.6 ± 1.2 at weeks 4 and 12, respectively. At week 12, there was a numerical difference of 3.9 ± 1.6 between the least squares mean of the video and audio arms (95% CI 0.6, 7.1). The MCSI values at baseline, week 4, and week 12 are provided in Figure 6b.

Modified Caregiver Strain Index score (a) LS means change from baseline and (b) absolute score at baseline, week 4, and week 12. LS: least squared; SD: standard deviation; SE: standard error.
Exploratory endpoints
The mean call duration was 71.6 (±54.9) minutes in the video arm and 59.1 (±61.4) minutes in the audio arm. The mean total travel times required of LCIG personnel for unscheduled home visits from baseline to week 12 were 125.7 (±70.2, n = 7) minutes for patients in the video arm and 203.0 (±70.0, n = 5) minutes for patients in the audio arm (Figure S2). The mean patient total travel times for unscheduled hospital visits from baseline to week 12 were 114.0 (±44.5, n = 5) minutes for patients in the video arm and 60.4 (±42.4, n = 5) minutes for patients in the audio arm. The most common issues were retraining requests in the video arm, and logistic/supply/accessories issues in the audio arm (Table S1). Most unplanned issues in both study arms were resolved by remote-based troubleshooting, with 261/285 issues (91.6%) in the video arm, and 124/142 issues (87.3%) in the audio arm being resolved.
Safety
There were four treatment-emergent serious adverse events in the video arm and none in the audio arm; one treatment-emergent adverse event led to study discontinuation (Table 2). There was one death in the video arm unrelated to LCIG and due to septic shock as a complication of post spinal surgery bacterial infection that the patient had experienced before initiation of LCIG. Of the four patients with treatment-emergent adverse events, one patient had mild muscular weakness, one patient had moderate general deterioration, one patient had moderate peritonitis and a severe abdominal abscess, and one patient had severe septic shock that led to death (described above). Of the treatment-emergent adverse events in the video arm, the moderate general deterioration in one patient was the only event considered possibly related to LCIG. There were two complaints related to the LCIG system in the video arm, one associated with the intestinal tube, and one associated with a PEG-J tube; there were no LCIG system-related complaints in the audio arm.
Summary of adverse events (ITT population).
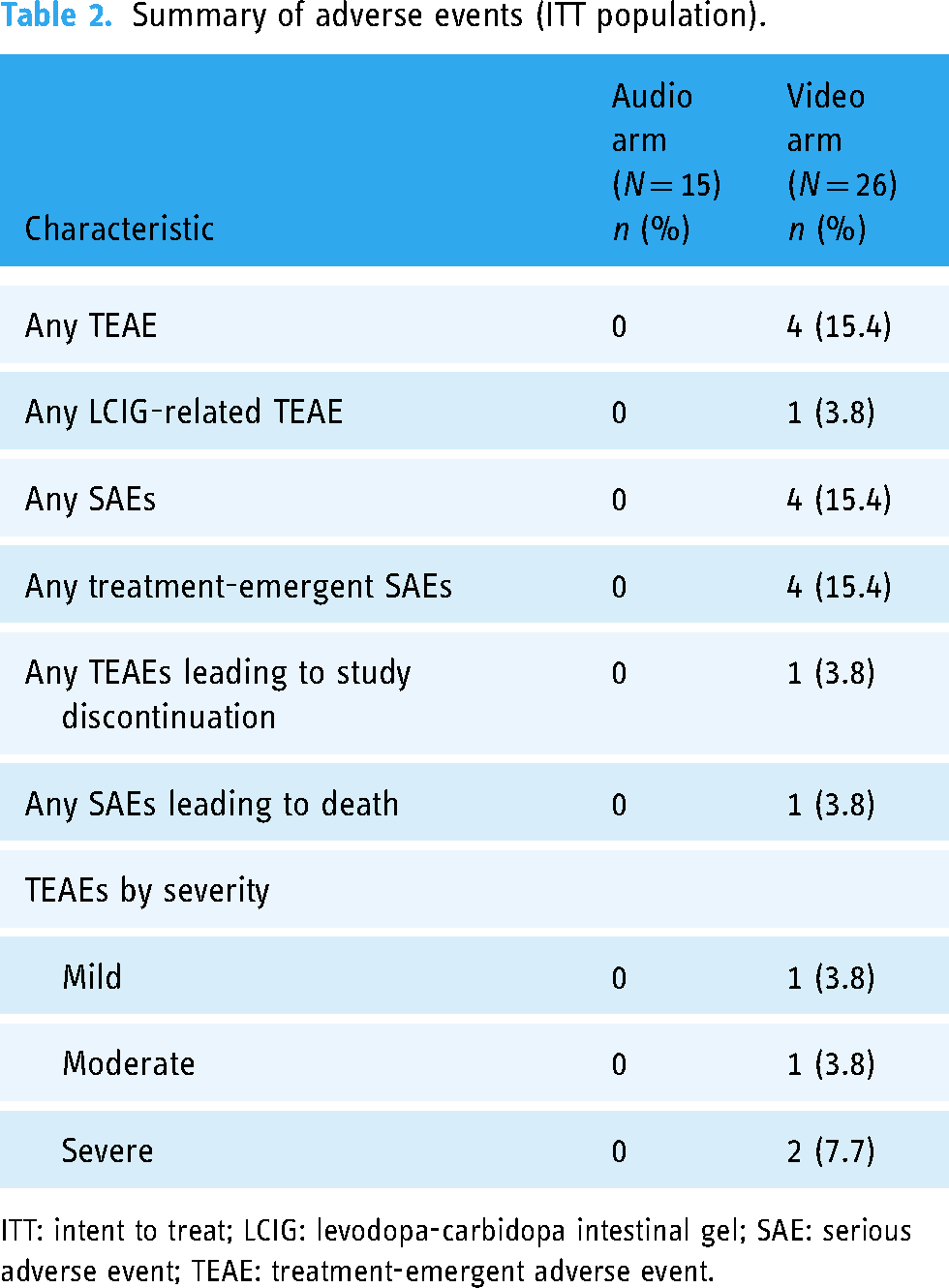
ITT: intent to treat; LCIG: levodopa-carbidopa intestinal gel; SAE: serious adverse event; TEAE: treatment-emergent adverse event.
Discussion
In this observational study, both video-assisted and audio-only telenursing demonstrated good satisfaction scores among LCIG patients, caregivers, and physicians in this study. Patients and caregivers were highly satisfied with LCIG support and communication access, with mean VAS scores at or above 9 in the video and audio arms at weeks 4 and 12. Over 90% of patients were completely satisfied with LCIG personnel support and communication access at week 12 in both video and audio arms. Patients and caregivers were also highly satisfied with the video conversations, reporting mean VAS scores above 8.5. Satisfaction with LCIG personnel support specifically and with assistance in becoming independent was high, with patients, caregivers, and physicians consistently reporting VAS scores above 8.5 for both the video and audio arms. Importantly, to our knowledge, this is the first prospective observational study demonstrating that satisfaction with the LCIG personnel support in a real-world setting was very high, regardless of the communication channel.
Caregiver burden as measured by the MCSI increased in the audio arm and decreased in the video arm, with numeric differences at week 12. This may suggest that video-assisted telenursing could have a particular impact on relieving the perceived caregiver burden, possibly due to more agile issue resolution.
Video-assisted LCIG telenursing had a higher call duration relative to the audio arm but saved time overall with reduced LCIG support personnel travel times for unscheduled home visits. On the other hand, patient travel times were higher in the video arm than the audio arm, suggesting that patients may have been advised more promptly to visit the treating physician to resolve an issue.
Limitations of this observational study include a nonrandomized allocation of patients to one of the two study arms, the open-label format, the small sample size, and the disparity in the numbers of patients included in each arm. This study was not designed to compare the head-to-head effectiveness of video vs. audio-only LCIG support; the sample size was instead calculated to control the precision of estimated mean within each treatment arm. Compared with patients in the audio-only arm, patients in the video arm were younger, had less advanced PD, had a higher level of education, and were predominantly male. As the study was nonrandomized and patients self-selected their study arm based on preference, the different demographics and characteristics of patients in the video vs. audio-only arm may reflect patients’ digital technology capabilities or affinity toward videoconferencing. Thus, results from this study may be prone to bias regarding the acceptance and capability of patients to handle videoconferencing technology. Additionally, patients were not provided with any additional equipment, rather the implementation of video-assisted telenursing was an option available to those who already had access to video-capable devices (i.e., tablet, smartphone, etc.). Adherence to LCIG treatment and the LCIG support program were not assessed. Adherence to LCIG treatment, device handling, and skin hygiene procedures are important to optimize the safety and tolerability of LCIG.
The median age for patients in the video arm of the study was 68 years; while internet usage has increased over time, its use is still disproportionately larger in younger populations (those aged <65 years) and in patients with higher levels of education. 10 Thus, this study may have limitations regarding the generalizability to more elderly patients or those in lower socioeconomic groups. However, video-assisted telenursing could prove useful to patients who are familiar with the internet and digital communication devices, who are more communicative, or who are living in rural areas to avoid unnecessary travel. Exploration of remote videoconferencing options is particularly relevant considering the unique challenges posed by the COVID-19 pandemic, as patients may wish to avoid in-person interaction. However, as the first patient in this study was recruited on 2 September 2020, the COVID-19 pandemic impacted this study as the recruitment was lower than expected due to decreased access of patients to study sites for LCIG initiation. Travel times for patients and LCIG personnel may be influenced by the country and specific location of the patient. Most patients were recruited in Israel, a country with relatively short travel distances. Patients from countries with further travel times or more rural populations might feel a greater degree of benefit from video-assisted telenursing, which should be confirmed in future trials.
In addition to having high satisfaction rates in resolving the special issues, video-assisted LCIG telenursing support offers unique benefits to patients and healthcare systems. Providing patients with 24-h convenient and quick access to LCIG support via video-assisted telenursing may increase their sense of security, which may improve the success of the therapy. Additionally, with an aging global population, increased adoption of technology that provides convenient communication channels, such as video-assisted telenursing, may reduce the burden on healthcare systems and expedite patient care. Findings from our observational study suggest that video-assisted LCIG support may be both feasible and satisfactory, and, thus, may encourage implementation of video-assisted telenursing support programs.
Conclusions
Video-assisted and audio-only LCIG support programs were associated with similarly high patient, caregiver, and physician satisfaction. Video-assisted telenursing is feasible to implement within LCIG support programs and may help provide patients with a needed avenue for health communication that is both convenient and comfortable.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241271847 - Supplemental material for Satisfaction with videoconferencing support for levodopa-carbidopa intestinal gel: An observational study
Supplemental material, sj-docx-1-dhj-10.1177_20552076241271847 for Satisfaction with videoconferencing support for levodopa-carbidopa intestinal gel: An observational study by Tanya Gurevich, Andrew Evans, Sharon Hassin-Baer, Georg Kägi, Dariusz Koziorowski, Anna Roszmann, Lars Bergmann, Juan Carlos Parra Riaza, Olga Sánchez-Soliño and Jarosław Sławek in DIGITAL HEALTH
Footnotes
Acknowledgements
AbbVie and the authors express their gratitude to the patients who participated in this study; their families and caregivers; the collaborating LCIG support programs; and the nurses, study investigators, and coordinators of the study. Medical writing support was provided by Sneh Mody, PharmD, MBA, BCCCP, of AbbVie, and Morgan Gingerich, PhD, of JB Ashtin, and funded by AbbVie. All authors have authorized the submission of this manuscript and approved any statements or declarations (e.g., conflicting interests, funding, etc.). Authors would like to acknowledge Ariela Hilel RN, NP, for her contribution to data acquisition.
Contributorship
TG, AE, SH-B, GK, DK, and JS contributed to execution of the research project and writing, review, and critique of the manuscript. AR contributed to execution of the research project and review and critique of the manuscript. LB contributed to the study design, execution of the research project, and writing, review, and critique of the manuscript. JCPR contributed to the study design and writing, review, and critique of the manuscript. OS-S contributed to the study design, review and critique of the statistical analysis, and writing, review, and critique of the manuscript. All authors approved this manuscript for publication.
Data sharing statement
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized individual and trial-level data (analysis data sets), as well as other information (e.g., protocols, clinical study reports, or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications. These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, statistical analysis plan, and execution of a data sharing agreement. Data requests can be submitted at any time after approval in the USA and Europe and after acceptance of this manuscript for publication. Data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit the following link: ![]() , then select “Home”.
, then select “Home”.
Declaration of conflicting interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TG is a study investigator and an advisor for AbbVie, Teva, Allergan, Neuroderm, Cytora, Truemed, and Medison. She has received research support from the Parkinson's Foundation, International Movement Disorders Society, and Israel Innovation Authorities, and travel support for her team and herself from AbbVie, Medison, Medtronic, and Allergan. AE holds shares in CSL limited and Global Kinetics Corporation and has received honoraria from AbbVie, Ipsen, UCB, Merck Sharp & Dohme (Australia) Pty Limited, STADA, and Pfizer. SH-B is a study investigator and an advisor for AbbVie. She has received funding/grant support from AbbVie; speaker honoraria from AbbVie, Medison, and Allergan; consultancy fees from Neuroderm, Takeda, and Teva; and advisory fees from Teva and AbbVie. GK is an advisor for Zambon SpA and has received an honorarium for consultancy from Bayer. DK is an advisor for AbbVie and has served as a consultant and speaker for AbbVie, Abbott, Ever Pharma, and Medtronic. AR is an advisor/speaker for AbbVie and EVER Pharma. LB, JCPR, and OS-S are employees of AbbVie and may hold stock or share options. JS is an advisor/speaker for AbbVie and EVER Pharma.
Ethical approval
The independent ethics committee or institutional review board at each study site approved the study protocol, informed consent forms, and recruitment materials before patient enrollment (Supplementary Appendix). The studies were conducted in accordance with the International Conference for Harmonisation guidelines, applicable regulations, and the Declaration of Helsinki. All patients provided written informed consent before screening. Caregivers were also required to provide written informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The LCIG support program was funded by AbbVie. AbbVie funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship.
Guarantor
TG
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
