Abstract
Objective
The significance of big data is increasingly acknowledged across all sectors, including medicine. Moreover, the trend of data trading is on the rise, particularly in exchanging other data for medical data to rejuvenate the medical industry. This study aimed to discern the facilitating factors of healthcare data trade.
Methods
We assessed five medical data market platforms on October, 2022, based on three criteria: (a) clarity in articulating the data for sale; (b) transparency in specifying the data costs; and (c) explicit indication that payment grants data access. This helped identify the traded medical data types. Additionally, we anonymously surveyed 43 representatives from medical device companies about their demand for medical data trading, achieving a response rate of 66%.
Results
Of the medical data traded on these platforms, 93.34% was structured, while 5.66% was unstructured, indicating an imbalance. Although there was a higher demand for structured medical data, there was also interest in purchasing unstructured medical data.
Conclusion
Unstructured big data are crucial for medical device development, fueling the demand for trading such data. Many stakeholders view the data market as essential and are willing to procure medical data. Consequently, medical device companies will need methods to acquire unstructured medical data for developing innovative and enhanced medical devices.
Keywords
Introduction
The importance of big data is increasingly recognized in all fields, including in medicine. In addition, trading data has tended to increase, with the trading of other data for medical data being increasingly performed to revitalize the medical industry. The demand for medical big data is also increasing significantly in medicine due to the development of machine learning analysis techniques. Medical big data have become essential in diagnosis, prediction, treatment, and precision medicine based on machine learning analysis techniques. The data is analyzed and utilized in real time to reduce health risks and harmful environmental exposure, which can create values that are beneficial for health. 1 The size of the global medical artificial intelligence market, which was about $2 billion in 2018, is expected to increase to over $7.2 billion by 2023, with an annual average growth rate of 28.44%. 2 Demands for medical big data will increase further, resulting in a corresponding increase in data supply. The worldwide size of big data in the healthcare market size was valued at USD 32.9 billion in 2021 and is predicted to reach USD 105.73 billion by 2030. 3 Data markets are currently emerging to trade medical data. These markets, however, are still in their infancy and have generally not been launched in Korea. When 131 companies were asked about their difficulties in data trading, 73 (55.7%) responded that there was a lack of usable, high-quality data, 68 (51.9%) reported that the cost of purchase was unreasonable, and 45 (34.4%) responded that there was a lack of data distribution channels. 4
Successful medical data trading requires understanding of the needs of healthcare data consumers and determining the scenarios and systems that can facilitate the trading of medical data in a positive direction. The purpose of this study was to identify the factors needed to enable trade in healthcare data in South Korea.
Materials and methods
Survey of medical data market platforms
Platforms that distribute existing medical data were evaluated on October 15, 2022 using three criteria. (a) The data to be sold must be clearly stated. (b) The cost of the data should be clearly stated. (c) It should be clearly stated that payment will allow access to the data. These five sites of current data trade markets were visited to determine the types of medical data being traded.
Survey of medical data trade demand by medical device companies
Medical device companies were surveyed about medical data trade demand. This study used a survey descriptive research method consisting of a questionnaire. The survey was sent to appropriate individuals at 65 affiliated medical device companies on or about October 26, 2022. The total research period from the creation of the questionnaire to the analysis of responses, writing of results, and drawing a conclusion was from June 2022 to December 2022, and 43 (66%) of participants responded by November 4, 2022. The purpose of the survey was explained to each of the respondents, and all provided written informed consent to participate in the survey. All data were anonymized to remove sensitive personal information prior to analysis.
Purpose of the survey
The survey was designed to determine whether the medical data desired by medical device companies were comparable to the medical data currently being circulated on the data trade market and to determine methods of enhancing trade in medical data.
Analysis
Frequency tables and cross tables were generated from survey data, with cross table results reported on a 100% stacked bar plot. All statistical analyses were performed using R 4.2.1 software. 10
Results
Types of structured and unstructured medical data for sale on data trade market platforms
Of the medical data sold on data trading market platforms, 93.34% was structured and 5.66% was unstructured, creating an imbalance. Evaluation of the formal medical data sold on these platforms showed that 32.2% of the data consisted of information on health behavior and eating habits; 15.9% consisted of health examination data; and 11.4% consisted of information on hospitals, medical staff, and insurance. The proportion of actual clinical data required by medical device development companies was about 40.45%. Among the atypical clinical data, clinical images accounted for the largest share at 65.12%, equivalent to 3.68% of the total data. Currently, the diversity of traded data is regarded as insufficient (Table 1).
Types of structured and unstructured medical data for sale on data trade market platforms

Characteristics of the respondents to the survey
Evaluation of the characteristics of the 43 survey respondents showed that 37 (86.4%) were affiliated with a medical device company, while 6 (14.6%) were affiliated with a healthcare company (Table 2). Five respondents (11.6%) had a data expert at their company, while 38 (88.4%) had not. Additionally, 26 respondents (60.5%) reported needing a data market platform, whereas 17 (39.5%) did not. Finally, when asked about the type of medical data they wanted, 18 (41.9%) respondents wanted structured data, 17 (39.5%) wanted unstructured data, and 8 (18.6%) wanted health insurance data.
Characteristics of respondents to the survey
Degree of awareness of data market platforms
Concerning the awareness of data market platforms among respondents, 8 (18.6%) of the respondents were aware of all data market platforms in South Korea, and 35 (81.4%) were not. Five people (11.6%) knew about the domestic medical data market platform, and 38 people (88.4%) did not. In addition, 2 respondents (4.7%) were aware of the overseas data market platform, 41 (95.3%) were not. One respondent (2.3%) was aware of overseas medical data market platforms, while 42 were not (97.7%) (Table 3).
Degree of awareness of data market platforms.

Benefits of data market platforms
Evaluation of the perceived benefits of data market platforms showed that 17 (39.5%) respondents reported that the convenience of data purchase was the most important, 14 (32.6%) stated that they would be able to save time by purchasing data quickly, and 12 (27.9%) reported that these data platforms were reliable transaction systems.
Examination of the perceived advantages to sellers of data through these data market platforms showed that 19 (44.2%) respondents believed that financial gains from data sales were the most important, 13 (30.2%) said they would be able to save time by selling data quickly, and 11 (25.6%) reported that these platforms were reliable transaction systems. Determination of the factors to be considered in data exchange found that 22 (51.2%) respondents emphasized security, 17 (39.5%) emphasized legal issues regarding medical data, and 4 (9.3%) emphasized data ownership. Evaluation of the type of transaction desired showed that 22 (51.2%) respondents wanted data transaction by subscription, 14 (32.6%) wanted direct data transaction, and 7 (16.3%) wanted API type. Moreover, 29 (67.4%) wanted to pay by credit card, 9 (20.9%) by account transfer, and 5 (11.6%) by other methods. Evaluation of the amount they regarded as reasonable for purchasing data showed that 33 (76.7%) respondents were willing to pay less than 3700 USD (5,000,000 KRW), whereas 10 (23.3%) were willing to pay more than this amount. When asked about matters to be considered in order to vitalize data exchanges, 16 (37.2%) each emphasized appropriate data prices and security, 6 (14.0%) responded that legal support was needed when using medical data, and 5 (11.6%) emphasized the brand value of the trading platform (Table 4).
Contents of data market platforms.

Respondent characteristics and intention to buy data
Of the respondents from medical device companies and healthcare companies, 56.8% and 50.0%, respectively, were willing to purchase data through the data market platforms. All respondents from both types of companies were willing to purchase data if a data expert worked in the company, whereas only 50% were willing to purchase data if a data expert was not present. Of the respondents who did and did not regard a data market as necessary, 76.9% and 23.5%, respectively, were willing to purchase data. Of the respondents who desired structured and unstructured data, 50.0% and 64.7%, respectively, intended to purchase these data (Figure 1).

Relationships between respondent characteristics and intention to buy data.
Awareness of data market platforms and intention to buy data
Of the respondents who were and were not aware of domestic data exchanges, 50.0% and 57.1%, respectively, were willing to purchase data. Moreover, of the respondents who were and were not aware of domestic medical data exchanges, 60.0% and 55.3%, respectively, were willing to purchase data. All respondents who were aware of overseas data exchanges and overseas medical data exchanges were willing to purchase data. Of the respondents who were not aware of overseas data exchanges and overseas medical data exchanges, 53.7% and 54.8%, respectively, were willing to purchase data (Figure 2).

Relationship between awareness of data market platforms and intention to buy data.
Characteristics of data market platforms and intention to buy data
Evaluation of the relationship between intention to purchase data and the perceived benefits of purchasing data through data markets showed that 70.6% of respondents reported that the convenience of purchasing data was important, 64.3% said that time saving through rapid transactions was important, and 25.0% said that the reliability of the transaction system was important. When asked about the relationship between willingness to purchase data and the perceived benefits of selling data through data markets, 47.4% of the respondents reported that they were willing to purchase data through data sales, 76.9% said that they saved time by selling data quickly, and 45.5% said that it was a reliable transaction system. Evaluation of factors to be considered in the data market showed that 75.0% of the respondents were willing to purchase data when data ownership was acknowledged, 52.9% reported that it was an issue related to medical law restriction, and 54.5% emphasized security. Determination of the relationship between intention to purchase data and the data purchase method showed that 50.0% of respondents were willing to purchase data through direct transaction, 59.1% by subscription, and 57.1% by API. In addition, 58.6% of respondents were willing to purchase data using credit cards, 55.6% through account transfers, and 40.0% by other methods. The survey found that 80.0% of respondents were willing to purchase data when the fair price was over 3700 USD, but only 48.5% were willing to purchase data for less than 3700 USD. When asked about matters to be considered for activating the data market, 75.0% of respondents emphasized the appropriateness of data prices, 66.7% emphasized the need for legal support related to medical data, 20.0% stated that the brand value of the trading platform was important, and 43.8% emphasized the security of the trading platform (Figure 3).
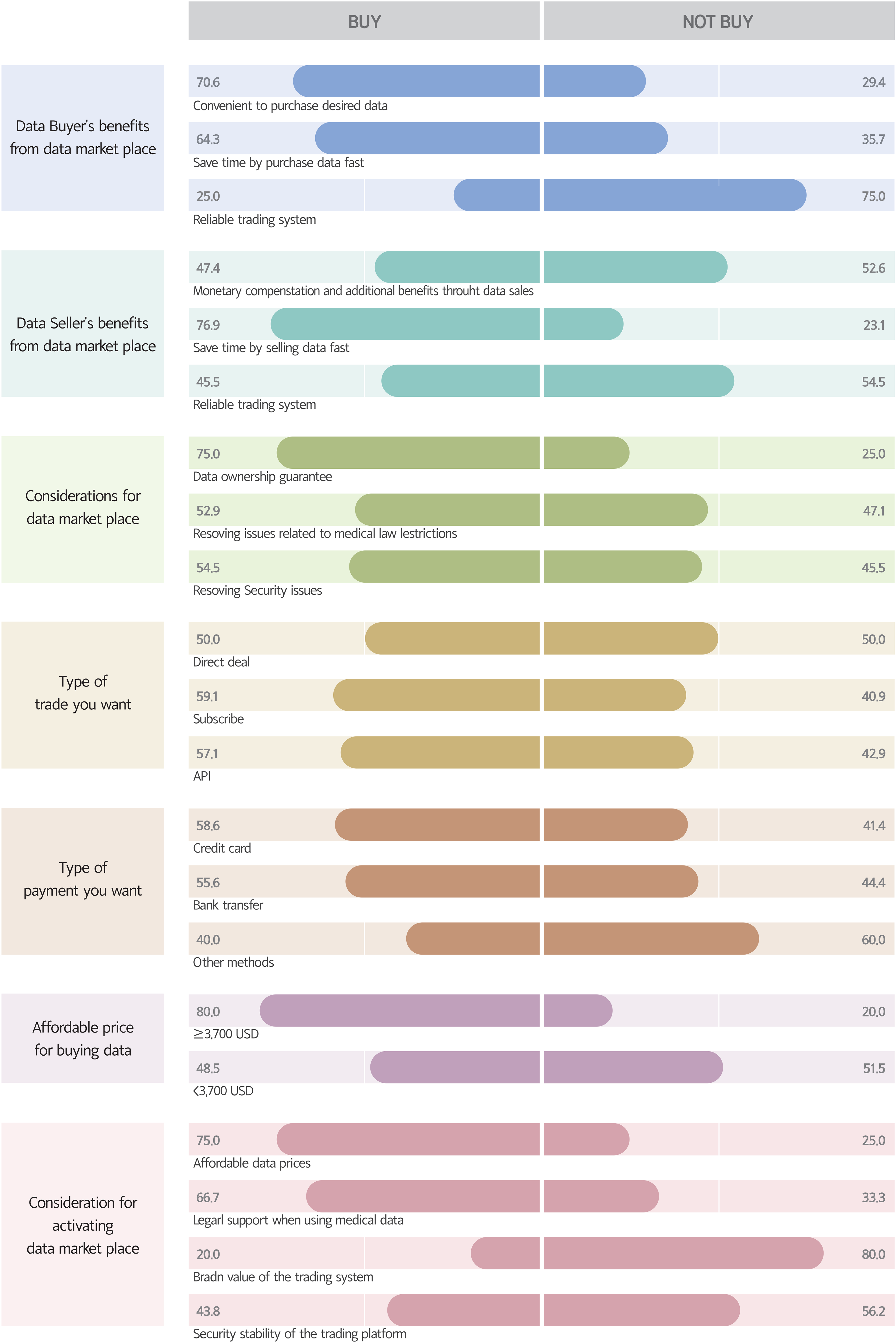
Relationships between the content of data market platforms and the intention to buy data.
Discussion
Overall, the study results clearly showed a high demand for the purchase of unstructured healthcare data. Unstructured medical data include the results of clinical imaging, consisting of both static and dynamic images. Static images include the results of x-rays, CT scanning, and ultrasound imaging, whereas dynamic images include the results of MRI and endoscopic imaging. Other types of unstructured medical data include voice results, methods of detecting symptoms of depression or anxiety, and auscultation of the lungs for diagnosis of asthma. AI-based industries are developing via the convergence of ICT with medical devices. 11 According to data in the Korean medical device development research list of 2020 to 2021, most medical device developers have developed AI software and systems mainly using unstructured data rather than structured data. 12 AI analyses require big data to fit mathematical and/or statistical models, 13 with unstructured big data directly needed for medical device development. Although many nonmedical companies are analyzing unstructured data for business uses, 14 it is difficult for companies seeking to develop medical devices to secure unstructured medical data. Although the amount of data currently available is considerable, companies still complain of a lack of data.15,16 Even if these companies are able to obtain unstructured medical data through their links with hospitals, they are limited in their use of these data due to issues surrounding the protection of personal information and regulatory issues under the Medical Act. 17 Nevertheless, the concept of data trading has emerged.15,18,19 If high-quality medical data is circulated at an appropriate price via medical data trade, many medical device development companies with innovative ideas will be able to challenge the medical device development industry and the entry barrier to development of medical devices will be lowered.
Limitation
This study is limited because it did not cover all types of currently existing data markets and failed to obtain the opinions of more medical device companies in international markets.
Conclusion
There is a clear demand for trading unstructured medical data, with many individuals who regard the data market as essential being willing to purchase medical data. Medical device companies will require methods of obtaining unstructured medical data to develop new and improved medical devices. Through medical data trading, not only can new medical devices be developed, but additional advancements can be made through cross-validation or transfer learning by utilizing these data after medical device development. This need may be met by activating medical data trading through the establishment of unstructured medical data trade platforms.
Footnotes
Acknowledgments
This paper was supported by a Korea Evaluation Institute of Industrial Technology (KEIT) grant funded by the Korea Government (MOTIE) (grant number: 20018689).
Additional information
Correspondence and requests for materials should be addressed to Hyeong Won Yu.
Contributions
Conceptualization was done by MG Kim, H Han, and HWY.
Methodology was done by MG Kim and I Lee.
Analysis was done by MG Kim and HWY.
Data visualization was done by I Lee.
Writing-original draft preparation was done by MG Kim and HWY.
Writing-review and editing were done by MG Kim and H Han.
Data availability
The data that supports the findings are available upon request from the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical committee approval
Considering that this study was a homepage and survey analysis study, ethical committee approval was not sought.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Korea Evaluation Institute of Industrial Technology (grant number 20018689).
