Abstract
Objective
This study aimed to develop an individual WeChat Mini Program to provide pharmaceutical care to better manage cancer pain patients and to evaluate its feasibility and the differences in analgesic efficacy, medication adherence and safety versus conventional pharmacy interventions.
Methods
In this parallel randomized clinical trial, 42 cancer pain patients were equally allocated into the experimental group and the control group. The experimental group received individualized pharmaceutical care based on the “Yao Nin You Wo” WeChat Mini Program, while the control group received conventional care during the 4-week period. Main outcomes contained pain scores, medication adherence, incidences and relief rates of breakthrough pain, and incidences of adverse events. Relief rates of pain were also calculated according to pain scores.
Results
At the beginning of intervention, none of the pain scores and medication adherence showed relevant differences between the two groups (all P > .05). After intervention, the experimental group had significantly lower pain scores compared to the control group (P = .003). Breakthrough pain of both groups was alleviate; not only the incidence of breakthrough pain considerably was lower at 4 weeks than at baseline, but the relief rate of breakthrough in the experimental group was higher than that in the control group. Compared with the control group, the medication adherence rate of the experimental group was significantly improved (P = .02). Types of adverse events that happened in experimental and groups were similar, but the total incidence of adverse events in the experimental group was lower than that in the control group.
Conclusions
WeChat Mini Program is a useful and facilitative tool with the potential to improve cancer pain self-management ability in discharged patients. In addition, pharmacists could play a key role through the Mini Program to connect with patients successfully by providing personalized pharmaceutical services.
Introduction
Pain is one of the most common syndromes of cancer patients; over 66% of advanced cancer patients experience severe pain. 1 Pain is miserable and debilitating, restricts the patient's daily activities, affects sleep and diet, even can profoundly reduce their quality of life. 2 Although standardized analgesic drug therapy is the main method to control cancer pain and guidelines to evaluate and treatment have been issued to facilitate management, a majority of patients still have inadequate pain control, even suffer from transient exacerbations of severe pain, also known as breakthrough pain, especially for discharged patients.
The reasons for the lack of analgesia are various, such as inadequate understanding of cancer pain and breakthrough pain, inappropriate evaluation methods, lack of effective management of adverse events, and poor treatment adherence.3–6 These multifaceted barriers highlight the need for multidisciplinary interventions during pain management. Pharmacists play a crucial role in the assessment and have a positive impact on the treatment of cancer pain patients.7–9 Regrettably, health care services still rely on face-to-face interventions to manage pain conditions, which means that patients must return to the hospital to receive treatments. In addition, patients were anxious about exposure to viruses, and the cancer treatment has gradually shifted toward domestic care during the COVID-19 pandemic. Investigation reveals that about 70% to 100% of patients preferred antitumor therapy at home. 10 For patients discharged from the hospital or living in remote areas, they cannot benefit from face-to-face pharmaceutical care if they are not able to follow up in time, particularly during the COVID-19 pandemic. During the strict epidemic prevention and control period, the extended treatment of cancer pain patients is limited by time and space. Moreover, due to the dynamic process of cancer pain, patients may be subject to memory bias, inaccurate descriptions and incomplete paper records, resulting in suboptimal analgesic treatment. Hence, as they are more likely to deviate from the doctor-recommended analgesic treatment regimens, personalized pharmaceutical care is especially important for discharged patients without continuous medical supervision.
Nowadays, electronic health (eHealth) and mobile health (mHealth) have been widely utilized in health care management and have entered the cancer pain field, including the use of the mobile applications (Apps) and WeChat Mini Programs. Significant evidence revealed that patient self-management interventions delivered via mHealth are more effective than that delivered via traditional methods or usual services in cancer pain.11,12 Apps were efficient for the management of pain in discharged patients with cancer pain. At present, WeChat, as a smart terminal instant messaging service and a multifunctional tool, is widely used by a large number of people in China, and WeChat Mini Programs are embedded in WeChat in the form of Apps. Compared with Apps, WeChat Mini Programs have the advantages of low hardware performance requirements, low development cost and short development cycle. WeChat Mini Programs provide a new platform with low technical difficulty to develop. WeChat Mini Programs can be tailored to the specific needs of cancer pain patients and are able to provide longitudinal support to present the dynamic characteristics of cancer pain experience. WeChat Mini Programs also makes it possible to achieve real-time reevaluation, reduce recall bias and optimize individual pharmaceutical services according to the patients’ response. Therefore, WeChat Mini Program interventions may be a scalable approach to improve cancer pain management. Our objective was to develop a “Yao Nin You Wo” WeChat Mini Program to harness the unique potential of technology solutions which can deliver personalized pharmaceutical care to cancer pain patients in a way that meets their needs. More importantly, we performed a randomized controlled trial to evaluate the differences in analgesic efficacy, medication adherence and safety versus conventional pharmacy interventions for cancer pain patients.
Methods
Study design
This study was conducted as a two-arm randomized controlled trial (RCT; NCT05049811). To analyze whether reasons for the lack of analgesia could be modified under a pharmacist-led intervention in a short term, assessments were repeated at baseline, weekly, and at the end of 4 weeks. Assuming an alpha value of 0.05 and two-sided power of 0.80, the sample size was calculated to be 26 (alternative m = 1.70, and SD = 2.17) according to previous published studies.13,14 Allowing for a 30% attrition or drop-out rate, the minimum number of patients enrolled is 34. The study protocol was approved by the Fujian Cancer Hospital ethical committees. Our protocol is shown as Figure 1.

Flow diagram of patient enrollment and analysis.
Participant inclusion/exclusion criteria
Adult cancer pain patients enrolled in this study were older than 18 years and had histologically or cytologically confirmed advanced or metastatic tumor with a life expectancy of at least 3 months. Eligible patients were those who have already received or required opioid therapy and could understand the study process, and signed the informed consent form. Exclusion criteria included the presence of psychiatric diseases or cognitive disorder, known history of substance abuse, participation in any other therapies or other study protocols that may have an impact on this study. Patients with creatinine clearance ˂15 mL/min or severe hepatic impairment (aminotransferases levels >5 times the upper limit of normal range) would be eliminated as well.
Randomization and interventions
After inclusion, basic information, including patients’ gender, age, marriage, educational level, disease diagnosis and drug usage would be collected. Patients were randomly assigned in a 1:1 ratio into the control group and experimental group using a random number table. The allocation number was concealed in opaque envelopes to guarantee the secrecy. After the assignment, pain sores, adherence and adverse events would be recorded in both groups. Participants in the experimental group would be taught to use the Mini Program beforehand and receive quality pharmaceutical care in “Yao Nin You Wo” after discharged. The home page of “Yao Nin You Wo” (Figure 2) contains Medication records, Adverse events, Medication reminder, Medication usage summary, Questionnaire, Pill counting, Report management and Health information which will push scientific articles for patients. When the pain occurs, patients can click “Medication records” (Figure 3), choose pain or breakthrough pain records, and then fill in details, including the time when the pain happens, analgesics used currently and dosage, etc. Moreover, the number of pain occurrences can be checked through the calendar and statistical charts can be generated according to patient records (Figure 4), which can help doctors, pharmacists and patients have a concise and clear understanding of pain occurrences. Pharmacists and doctors can find all the drug use of patients from the back-end, including therapeutic drugs and analgesic drugs. Furthermore, for different adverse reactions, corresponding pharmacists’ suggestions are recorded in advance (Figure 5), which can be directly sent to patients through “Yao Nin You Wo” to help patients deal with adverse reactions timely and conveniently.

Homepage of the “Yao Nin You Wo” WeChat Mini Program.

Pain record of the “Yao Nin You Wo” WeChat Mini Program.

Record summary of the “Yao Nin You Wo” WeChat Mini Program.

Back-end management of the “Yao Nin You Wo” WeChat Mini Program.
Patients in the experimental group were encouraged to use the mini program as much as possible to record their pain and the use of opioid analgesics at least once every day. Types and severity of adverse reactions should also be recorded when they occurred. They do not need to fill in the forms again and can just simply click submit if there is no change in pain scores. Pharmacists would observe and evaluate patient's pain situations, adherence and adverse events, and replied patients online in time.
Without being conducted by the mini program, the control group received conventional pharmaceutical care. Before discharged from the hospital, the patients would get detailed medication and pain education from clinical pharmacists and were requested to record pain-related problems each time it happens. Then patients were contacted through weekly conventional telephone calls to collect relevant data.
Outcomes and assessments
The primary outcome was pain scores (including pain relief rate), and the secondary outcomes were medication adherence, situation of breakthrough pain and incidence of adverse reactions.
Relief rates of pain: The intensity of pain was assessed using a numerical rating scale (NRS) during the last 24 hours, with a scale of 0 to 10, in which 0 represents no pain, 1–3 represents mild pain (pain does not affect sleep), 4–6 represents moderate pain (pain affects sleep), 7–10 represents severe pain (pain severely affects sleep). 15 If a patient's NRS has reached 4 or more than 4 and then decreased to equal or less than 3, it is defined as pain relief. Relief rates of pain are obtained by calculating the number of patients who have pain relief out of patients who have experienced pain.
Incidence and relief rates of breakthrough pain: Breakthrough pain is defined as a sudden, transient exacerbation of pain that occurs either spontaneously or triggered by a specific predictable or unpredictable factors despite relatively stable controlled background pain and adequate use of analgesics. Patients need to record NRS, daily frequency, pain duration and time to pain relief after taking analgesics. The incidence rate of breakthrough pain (NRS ≥ 4) is the percentage of cases out of the total number of patients. Breakthrough pain relief includes NRS change (decrease to ≤3), and daily incidences need to be less than three times as well. The relief rate is defined as the same as above.
Medication adherence: Patients in the experimental group need to record the prescription each time in “Yao Nin You Wo” WeChat Mini Program, helping doctors, pharmacists and patients gain more precise data. Overall adherence was assessed based on pill counting. Pill counting is calculated by comparing the physician's prescription with actual medication cycles. If patients’ actual medication cycles are consistent with the prescription, it represents good adherence. Otherwise, the adherence is poor.
Adverse events: Types and grades of analgesia-related adverse events were recorded in two groups and were evaluated based on the National Cancer Institute (NCI) Common Terminology Criteria for Adverse Event (CTCAE, version 5.0, http://evs.nci.nih.gov/ftp1/CTCAE). Grade 1 is considered as mild, grade 2 is moderate, and grade ≥3 is severe.
Statistical analysis
Statistical calculations were done by SPSS 22.0, and P value ˂.05 was considered to be statistically significant. Counting data was expressed as rate (%), Chi-squared test was used for inter-group differences. The Mann–Whitney U test or Fisher exact test was used to assess pain scores, pain relief rates, incidence and relief rates of breakthrough pain, and medication adherence between the two groups at baseline and 4 weeks later. Frequency distributions of adverse events were performed for each group.
Results
Patients characteristics
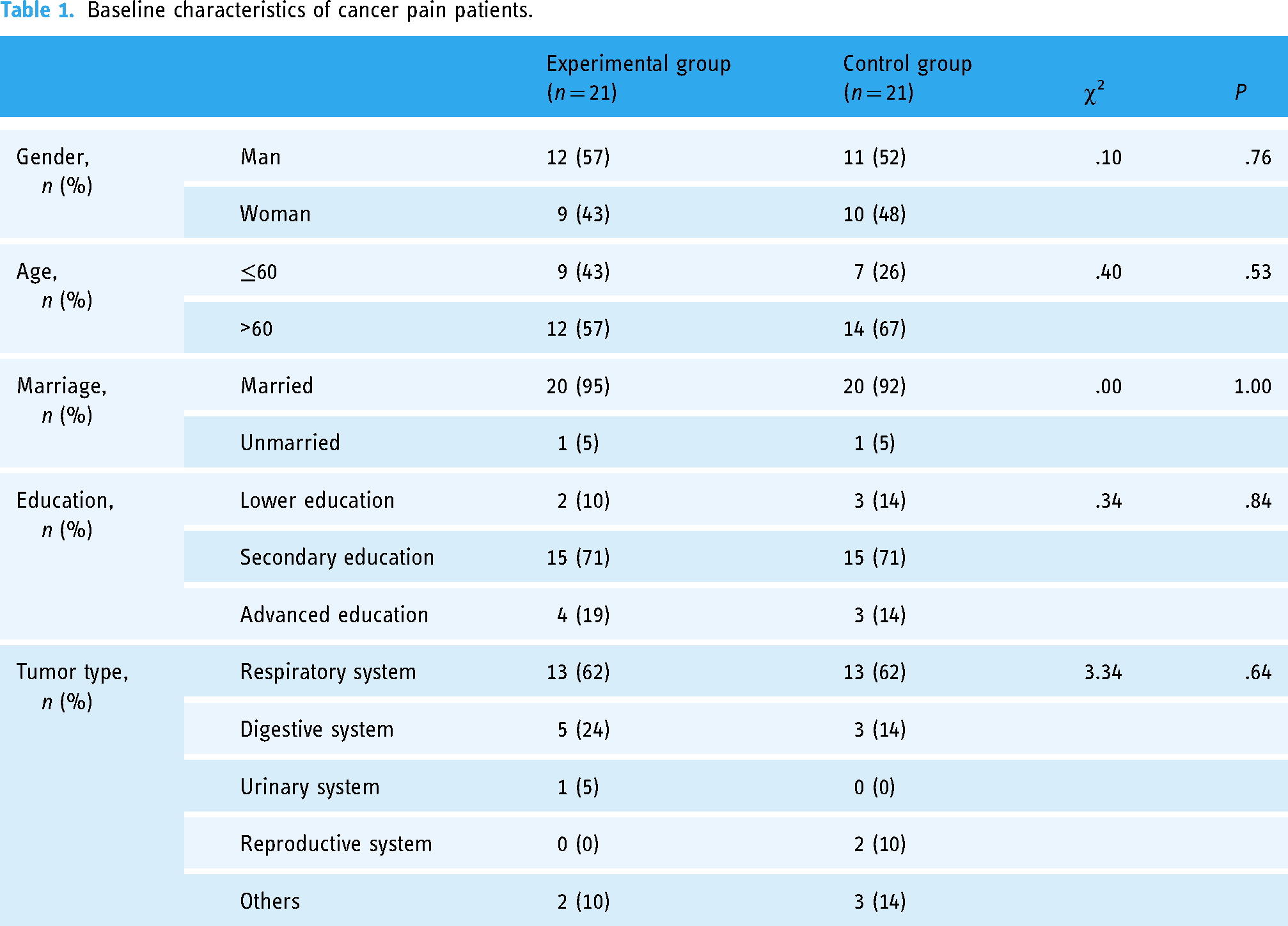
Limited by COVID-19 control,a total of 42 cancer pain patients aged from 51 to 75 were enrolled for preliminary assessment, including 23 men and 19 women, most of whom had secondary education. There were 21 cases in each group, and more than half of cases are diagnosed with lung cancer (62%). The baseline of sex, age, marriage, education and tumor types between two groups was balanced and comparable (Table 1).
Baseline characteristics of cancer pain patients.
Comparison of cancer pain
At baseline, there was no statistical difference in NRS scores (control group 3.76 ± 1.81 vs experimental group 3.57 ± 1.99, P = .54). At the end of intervention, NRS in two groups both decreased (2.76 ± 1.00 vs 1.86 ± 0.79), and the statistically significant difference was detected (P = .005) (Figure 6A). The relief rate of pain in the experimental group was higher than that in the control group, and the significant difference could be found between two groups (P = .04) (Figure 6B). During the study, nearly one-third patients in two groups experienced breakthrough pain, respectively. After the intervention, the breakthrough pain of both groups was alleviated, and the experimental group has a higher relief rate than the control group (Table 2). ns: no statistical differences, *: P ˂ .05,**: P ˂ .01.

Comparison of cancer pain between two groups.
Incidence and relief rate of breakthrough pain in both groups.

Comparison of adherence
In the initial adherence evaluation, 14 cases in the control group and 13 cases in the experimental group had poor adherence. After the intervention, the adherence was improved in both groups, and the experimental group had a significantly better adherence improvement than the control group as the sum of patients with poor adherence was reduced to 3 (P < 0.05) (Table 3).
Comparison of adherence between two groups.

Adverse events
Adverse events occurred in both groups, but serious adverse events were not observed. A patient may have more than one adverse event, so the number of adverse events are more than the number of patients. Although the number of mild adverse events (Figure 7B) in the experimental group was higher than that in the control group, the number of moderate adverse events was much lower than that in the control group. Briefly, the incidence of total adverse events in the experimental group was less than that in the control group and had statistical significance (***: P ˂ .001). Among all grades of adverse events that occurred, constipation (Figure 7A) had the most frequent occurrence, followed by nausea and vomiting and drowsiness.

Adverse events in two groups.
Discussion
Principal findings
We carried out a 4-week trial, investigated patients’ pain perception, drug perception, adherence, and influencing factors and provided professional and sustainable personalized pharmaceutical care for discharged cancer pain patients based on a WeChat Mini Program. The results confirmed that patients’ self-management ability, including medication adherence and pain relief, can be enhanced with the help of pharmacists. Occurrences of pain, breakthrough pain and adverse events can also be reduced effectively.
Consistent with previous studies, mhealth-based interventions may be a viable strategy to address inadequate pain control in patients with cancer pain. 16 Tailoring interventions to individual circumstances and dynamic pain trajectories may improve patients’ pain self-management. Our results showed significant differences existed in pain relief rates between experimental and control groups. In addition, the control rate of breakthrough pain in the experimental group was lower than that in the control group. Breakthrough pain can occur in 40% to 80% advanced cancer patients; 17 poorly controlled breakthrough pain may have a negative psychological impact, and then lead to greater pain, forming a vicious circle. Therefore, it is not surprising that the assessment of breakthrough pain is a crucial element of pain management in cancer patients. Depending on periodicity, duration, and intensity of breakthrough pain can help guide service planning and provide personalized treatment. 18 WeChat Mini Programs build a bridge for timely communication between pharmacists and doctors, and can offer doctors with real-time information about patients. Evidence suggests that innovative real-time pain assessment mechanisms and electronic reporting are more efficient in acquiring pain data.19,20 Boceta et al. also showed that App is useful for physicians to help diagnose and monitor breakthrough pain in patients with cancer. 21 Our team collected detailed information through WeChat Mini Programs, delivered personalized recommendations to patients in real time, and formed weekly reports. When patients return to the hospital, the cancer pain reports can be presented to doctors, or doctors can enter the Mini Program “Yao Nin You Wo” directly for review.
Our management team is mainly led by clinical pharmacists and consists of physicians and nurses. We have previously conducted randomized controlled trials of pharmacist-led management to ameliorate efficacy, safety, medication adherence, and quality of life in cancer pain patients,22,23 proving that pharmacists play an important role in the management of cancer pain. It was also verified that the inclusion of clinical pharmacists in the multi-disciplinary treatment team can help improve clinical outcomes for cancer patients. 13 Apart from this, medication education and guidance carried out by professional pharmacists can promote patients’ skills and knowledge and avoid pain perception and drug perception errors in order to fully implement pain self-management strategies in daily life. 24 It is reported that some patients used fentanyl transdermal patches when they had a fever or slept with an electric blanket; other patients took oxycodone sustained release tablets after grinding due to swallowing disorders. These inappropriate forms of medication can bring about opioid intoxication and poor pain control. Therefore, we use the “Yao Nin You Wo” Mini Program to regularly send relevant science articles to carry out drug education for patients, which can help them understand the pain situation and the drugs they used correctly, and ultimately avoid unreasonable use. A number of studies have shown that with pharmacists’ administration, patients’ adherence after intervention can be increased by 20% compared with that before intervention.22,25 The high compliance of patients is crucial to achieve the ideal treatment effect, and how to accurately grasp the whole treatment data of patients is the basis for improving the treatment effect of patients. Our study shows that patients’ adherence in the experimental group could be doubled after Mini Program intervention, far more than that in the control group, proving the capability of “Yao Nin You Wo” again.
With the evolution of technology, mobile devices enable to provide diverse services independently of time and place and dynamically adapt to the current usage environment and personal preferences. For now, mobile devices become well-suited platforms for Apps that can monitor and manage pre-existing health conditions more easily, provide more effective personalized care when needed, and facilitate better collaborative working between patients and healthcare providers. Several Apps and Mini Programs have been developed to manage pain and were confirmed to have positive effects. 26 Yang et al. developed an application called Pain Guard, and Zhang et al. developed a WeChat Mini Program “Medication Housekeeper” to administer the use of analgesic more thoroughly, containing self-assessment, real-time medication consultation, pain recording, etc., which are similar to “Yao Nin You Wo”.12,20 Pharmacists can address corresponding pharmacists’ recommendations based on patients’ adverse reactions and evaluate compliance by pill counting, but these are not available in Pain Guard and Medication Housekeeper. Compared with Apps, the WeChat Mini Program has the potential to assist in symptom management more conveniently and easily. 27 Besides real-time pain assessment, “Yao Nin You Wo” can also be used for adherence evaluation, adverse reaction management and information consultation. This enables pharmacists to provide timely help and effective intervention to patients, and adjust the content of intervention according to patients’ individual conditions, which is difficult to achieve in traditional pharmaceutical care. “Yao Nin You Wo” comes with a medication reminder function, and patients can set their own time to take medicine. If they miss or forget to take medicine, pharmacists will send a reminder message to patients. More importantly, when patients record adverse reactions, pharmacists can send corresponding treatment principles or measures according to the type and intensity of adverse reactions to improve patients’ self-management ability. For example, people with mild constipation are advised to eat more vegetables and fruits, drink more water, and exercise appropriately. For patients with moderate or severe constipation, medications such as lactulose and enemas are recommended.
Limitations
There are a few limitations of our study. This is a single-center, small-sample data analysis with a short intervention time, which may be biased and affect the universality of results. For example, lung cancer has a high incidence and advanced lung cancer is often accompanied by bone metastasis; advanced lung cancer patients have a high probability to experience pain and 62% of included patients in our study had advanced lung cancer. A broader participant recruitment process should be carried out to eliminate this bias. Besides, relevant data were filled by patients themselves; this means that the patient's symptoms may be underestimated and impact the completeness of the report. Also, our study did not take the psychological factors into account, such as irritability or depression caused by pain, which should be considered in the future to better manage cancer pain. Although the WeChat Mini Program is a profitable method to collect information from patients, the development time of WeChat Mini Programs is relatively short. The use of corresponding functions requires strong clinician and patient approval, and continuous clinical and patient supporters, so as to increase the overall usage rate and acceptance. The perspectives and experiences of users can be fully explored in the use of each module function, too. It is important to better understand the needs of users and upgrade the system when necessary to improve the convenience and satisfaction of patients.
Conclusion
In summary, “Yao Nin You Wo” WeChat Mini Program successfully connects pharmacists with patients to deliver personalized pharmaceutical services, thus improving patients’ self-management of cancer pain and medication adherence and reducing the incidence of opioid-related adverse events. In addition, the Mini Program is accepted easily and worthy of promotion for patients.
Footnotes
Acknowledgments
The authors acknowledge the support from Fujian Cancer Hospital.
Authors contributions
QZ, XQ and LY all contributed significantly to the conception and design in the research. JC, TC, QZ took charge of the Mini Program and communicated with patients. WL and XQ conducted data collection and statistical analysis. ZN drafted and revised the paper. RX revised the important knowledge content of the manuscript. LY reviewed and approved the final manuscript. All authors contributed to the building of the mini program and approved the submitted version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the Fujian Cancer Hospital ethical committees (K2021-022-01).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Beijing Health Alliance Charitable Foundation, Fujian Cancer Hospital Cultivation project (Grant No.WS058C; Grant No. yxky-TQ20210, Grant No. 2023YN11).
Guarantor
YL.
Trial Registration
ClinicalTrials.gov NCT05049811; https://clinicaltrials.gov/study/NCT05049811?term=FJZLYX002&rank=1
