Abstract
Objectives
We aimed to construct a theoretical model and an observer-based measurement of shared decision making (SDM) in web-based pharmaceutical care consultation.
Methods
We first constructed a theoretical model through analysis of SDM models and interviews with 24 key informants. Then, the initial measurement was formulated, and the content was evaluated by 16 key informants alone. A cross-sectional study was executed, where 300 provider-patient encounters were collected at the online platform ‘Good Doctor Website’, for reliability and validity assessment. Content validity was evaluated by appropriateness score, that is the median of 7–9 and without disagreement. Interrater reliability was evaluated by Spearman correlation coefficient using 30 out of 300 samples. Single indicator validity was tested by Pearson correlation analysis, and convergent validity was evaluated by the Multiple Indicators Multiple Causes (MIMIC) model using 300 samples.
Results
Theoretical model constructed included four elements: ‘comprehensive understanding of the patient's condition’, ‘exchanging ideas on medication options’, ‘achieving a medication plan’, and ‘providing decision support’. Measurement constructed included four dimensions and 19 items, and four dimensions were in accordance with four elements of the theoretical model. We found median scores of these dimensions and items were 7–9 and without disagreement, indicating good content validity. Spearman correlation coefficients of 19 items were greater than 0.750, suggesting good interrater reliability. Pearson coefficients between 19 items and their external variables were significant (
Conclusions
Characteristics of web-based pharmaceutical care consultation, highlighting the importance of medication history, medication regimen, and detailed medication plan, were observed in the theoretical model. Measurement constructed had good reliability and validity and may be used by health policymakers to improve the quality of pharmaceutical care consultation online.
Introduction
Irrational drug use practices, such as polypharmacy, overuse of antibiotics, and inadequacy of drug labeling, exist all over the globe, predominantly in developing countries.1–4 Severe irrational drug use has been abundantly evidenced in China. For example, overuse of injections was common, and studies indicated the percentage of injections prescribed per 100 encounters was between 21% and 60%. 5 Irrational drug use leads to prolongation of the illness, development of adverse effects, and unnecessary expense. 6
Pharmaceutical care could improve medication adherence and clinical outcomes, increase awareness of medication side effects, and reduce the number and prevalence of drug-related negative outcomes, especially for chronic disease.7–9 There is an overall low coverage of pharmaceutical care and a shortage of qualified pharmacists to meet increasing patient needs in China,10,11 so the improvement of pharmaceutical care is needed. Pharmaceutical care is the professional practice for patient-centered medication management services. 12 Effective communication is an essential tool for pharmaceutical care practice because it improves the use of medications by patients and ensures optimal therapeutic outcomes.13–16
Shared decision-making (SDM) was advocated as an ethical ideal for patient-professional relationships 17 and was hailed as the future of medical consultations. 18 SDM is an approach where clinicians and patients make decisions together using the best available evidence, and this approach respects patient autonomy and promotes patient engagement. 19 SDM aims to achieve healthcare choices that are agreed upon by patients and clinicians, and it is particularly relevant given the increasing number of healthcare choices.20,21 There is evidence that SDM can lead to better outcomes and care for people, 22 so policy documents increasingly refer to SDM as an indicator of the quality of care. 23 SDM in pharmaceutical care plays an important role in improving drug treatment adherence, treatment satisfaction, and communication efficiency,24–26 which has also become a guideline requirement. 27
The internet is rapidly gaining importance for healthcare providers and patients by enabling them to search for drug-related and other health-related information. 28 Recently, the influx of ‘ask the pharmacist’ services provided by some internet pharmacies was appeared, and patients may email questions about their drug therapies to a pharmacist who will reply with pertinent information. 29 There are many online platforms for medical communication between patients and healthcare providers from different areas and hospitals in China. For example, healthcare providers can solve patients’ drug-related problems (DRPs) through online communication on the Good Doctor Website. Remote pharmacy platform is efficient and convenient for providing continuous pharmaceutical care to patients. 30
There is an international drive to show that SDM is becoming embedded in healthcare settings, and SDM measurement, including theoretical measurement framework, is often a key part of that.31,32 There are many SDM models, such as the shared treatment decision-making model by Charles et al.,
21
integrative model of SDM,
33
three-talk model,
34
and
The development of a theoretical measurement framework should be put on the research agenda to improve the consistency of measured constructs across research groups. 31 Therefore, this study had two goals. The first goal was to develop a theoretical model for guiding measurement construction. The second goal was to develop an observer-based measurement of SDM in web-based pharmaceutical care consultation for better pharmacy research and practice.
Methods
Overview
In this article, we describe the development of an observer-based measurement of SDM in web-based pharmaceutical care consultation, which incorporated five stages of development: (1) analysis of SDM models; (2) key informant interviews with pharmaceutical care providers and patients; (3) item generation and evaluation; (4) data collection for coding (as different diseases provide varying treatment and provider-patient communication, we selected patients with hypertension or high blood pressure for our study to avoid interference from different diseases); and (5) reliability and validity assessment. We firstly constructed a theoretical model of SDM in web-based pharmaceutical care consultation through the first two stages.
Analysis of SDM models
We examined all the SDM models in three systematic reviews33,45,46 and a narrative literature review 47 before 2016. We conducted a Pubmed/Embase/Web of science/MEDLINE search to identify articles published from 1 January 2016 to 31 July 2019 using the terms ‘shared decision making’ AND ‘model’ OR ‘framework’ OR ‘theor*’. The specific research strategy is shown in Supplementary File 1. The inclusion criteria were as follows: (1) SDM model that involved the patient and healthcare provider(s); (2) providing a description of the concepts used in the model and indicating relationships between concepts; and (3) concerning the process of SDM or key features of SDM. After removing duplicates, 1345 candidate articles remained. We excluded 1263 by screening titles and abstracts and assessed 82 full-text articles. Of these 82, we excluded 45 articles and included 2 important references, and finally included 39 articles that contained 31 SDM models for analysis. The process for identifying 39 articles is shown in Supplementary File 2 and 31 SDM models in Supplementary File 3. Based on the theory analysis elements, 20 such as meaning, development process, testability, clinical context, and generalizability, we examined these models.
Key informant interviews
Key informants were pharmaceutical care providers and patients who have participated in or were familiar with web-based pharmaceutical care consultation. Pharmaceutical care providers and patients were recruited through convenience sampling and snowball sampling. To better identify and approach interviewees, the hospital administrator helps us contact the pharmaceutical care providers (clinical pharmacists and clinicians) and schedule the time and place of the interviews. The criterion for determining the number of participants was saturation of information. We approached 20 pharmaceutical care providers and 11 patients initially, but five providers and two patients were not interviewed for being busy or unfamiliar with web-based pharmaceutical care consultation. We used semistructured interviews and interviewed them with the following questions: the characteristics and differences of online and offline pharmaceutical care consultation, the differences between pharmaceutical care consultation and clinician–patient communication, and how to achieve SDM in web-based pharmaceutical care consultation (what should clinicians/pharmacists and patients do). Each interview was conducted by two researchers, and the interview process was audio-recorded after receiving consent from the pharmaceutical care providers or patients. We transcribed the audio or arranged the written documents after the interviews. The education level of the two interviewers who had PhD degrees was often higher than that of some participants, which may have created a power imbalance. To overcome this potential issue, the interviewers stressed the importance of hearing what the participants thought and that there were no wrong or right answers. 48 We extracted and analyzed the interview data using the ‘Implement-SDM model’ as the guidance framework. When we were presenting data from the interview analysis, this was anonymized. We use PH1-PH6 to present six clinical pharmacists, CL1–CL9 to present nine clinicians, PA1-PA9 to present nine patients.
Item generation and evaluation
Initial dimension and item formulations were mainly based on core aspects of the ‘Implement-SDM model’ and an analysis of interview data. We then assessed the appropriateness of dimensions and items from the key informant's perspective. Key informants were pharmaceutical care providers and patients who have participated in or were familiar with web-based pharmaceutical care consultation. Pharmaceutical care providers and patients were recruited through convenience sampling and snowball sampling. The participants (11 pharmaceutical care providers and 5 patients) rated the dimensions and items alone. We sent the evaluation material to participants and explained the evaluation goals before evaluation. Participants can also easily contact us if there are any questions. Participants were asked to rate the appropriateness of each dimension or item on a nine-point Likert scale, ranging from 1 (most inappropriate) to 9 (most appropriate). Our study mainly adopted the RAND/UCLA appropriateness method, 49 that is median of 7–9 and without disagreement (DI < 1). DI = IPR / IPRAS; IPR is interpercentile range, and IPRAS is the interpercentile range adjusted for symmetry required for disagreement.
Data collection for coding
Data were collected from a retrospective review of the online platform ‘Good Doctor Website (https://www.haodf.com/)’ for provider–patient communication. Good Doctor Website was founded in 2006, and it is one of the largest websites serving for health consultations. 50 Currently, over 7500 hospitals and more than 500,000 physicians are active on the website. 51 According to Good Doctor, online physicians are divided into 28 groups and more than 100 departments, 51 and increasing pharmacists join the Good Doctor Website to provide pharmaceutical care consultation. Web-based pharmaceutical care consultation is mainly asynchronous, which is not suitable in urgent situations because there is a time gap in communication. 52 Chronic diseases are more suitable for web-based pharmaceutical care consultation than acute diseases, and hypertension is one of the common chronic diseases. Therefore, when we collected data for measurement assessment, we selected patients with hypertension or high blood. The inclusion criteria of the consultations were: (1) patients with hypertension or high blood: Due to patients’ incomplete reporting online, we did not exclude patients if they had hypertension as well as other comorbidities, and there were not any exclusions based on age or other characteristics; (2) consulting DRPs; (3) not during the period of pregnancy or breastfeeding; and (4) including patient questions and provider responses. The data of the provider–patient exchanges in this study is reported on the website in public while the content is anonymous for health information sharing. 53
In China, pharmaceutical services are mainly provided by clinicians. Although services provided by pharmacists have been increasing recently, pharmacist–patient encounter samples online are still limited. Therefore, we included both clinician–patient encounters and pharmacist–patient encounters for coding. First, we selected 30 provider-patient encounters, including 15 clinician–patient and 15 pharmacist–patient encounters, for pre-survey and excluded the items that were not observed. Then, we selected 300 provider–patient encounters, including 150 clinician–patient and 150 pharmacist–patient encounters, for formal coding. At the classification of hypertension, 15 provinces/municipalities (eastern China: Beijing, Tianjin, Hebei, Shanghai, and Jiangsu; central China: Shanxi, Anhui, Jiangxi, Hubei, and Henan; western China: Chongqing, Sichuan, Shanxi, Gansu, and Guizhou) were selected, and we selected 10 clinicians from each province. There were plenty of clinicians online, and many of them without any communication records or with communication records which were not meet the inclusion criteria. Therefore, we checked the clinicians in the order they appear on the webpage. If a clinician had communication records that meet the inclusion criteria, we would select this clinician until 10 clinicians were included. On 31 December 2019, we included the latest clinician–patient encounter according to the inclusion criteria for each clinician. Pharmacist–patient encounter samples online are limited, and there were not enough pharmacists with communication records that meet the inclusion criteria. Therefore, the process of selecting 150 pharmacist–patient encounters was as follows. At the classification of the pharmacy department, we checked pharmacists in the order they appear on the webpage of each province. If a pharmacist had communication records that meet the inclusion criteria, we would include this pharmacist. On 31 December 2019, we included all the pharmacist–patient encounters according to the inclusion criteria for every pharmacist until 150 pharmacist–patient encounters were included. Coding for items used a magnitude instead of an attitude scale. The score ‘0’ was allocated to the situation where the item content described was not observed. Other scores (such as 1–4) were allocated to increasing levels of the item content described.
Reliability and validity assessment
Before reliability and validity assessment, we used the Harman single-factor test to evaluate common method biases of the coding data. Common method biases are not serious and will not significantly impact the study results when the largest eigenvalue is less than the critical standard of 40%. 54
When undertaking reliability and validity studies of questionnaire scales, an initial issue is whether the scales in question are indeed reflective scales or formative scales,55,56 and the initial measurement (Supplementary File 4) in our study had the characteristics of formative scale
In order to evaluate the validity of the formative scale, Diamantopoulos recommended that each item could be correlated with another variable (external to the index), and only those items that are significantly correlated with the variable of interest would be retained.
58
One possibility is to use as an external criterion a global item that summarizes the essence of the construct that the index purports to measure.
58
In our study, we developed two global items to reflect the core content of every dimension, and items were scored from 1 to 5 ranging from strongly disagree to strongly agree. The score of an external variable in a dimension is the average score of the two global items of this dimension. Pearson correlation analysis was used to examine the correlation between each item and the external variable to test the single indicator validity. A more satisfactory approach to validation is convergent validity, that is the extent to which a measure correlates positively with other (e.g. reflective) measures of the same construct using different indicators.
59
Multiple Indicators Multiple Causes (MIMIC) model was used to evaluate convergent validity. If the overall model fit proves acceptable, this can be taken as supporting evidence for the set of indicators forming the index (
Results
Analysis of SDM models
By examining each model in turn, we noted the overlap of elements across models, such as information gathering,32,47 choice introduction/recognizing alternatives,32,34,35,60,61 providing options and their pros/cons,32,34,35,60,61 patient preferences,32,34,35,60,61 and make or defer decision,32,34,35,47,61 but the same elements may be classified into different dimensions in different models. Compared with other models, ‘Implement-SDM model’ 32 was constructed by consultations between clinicians who had received SDM skills training and patients with chronic kidney disease or early-stage breast cancer, and put forward some new elements based on the ‘three talk model’. Therefore, the ‘implement-SDM model’ is constructed with a clear development process and derived from empirical data. This model probably has good applicability in different diseases and could be a basis for the measurement of SDM in routine care.
Participants of interviews
A total of 24 participants (Table 1), including 15 pharmaceutical care providers (nine clinicians and six clinical pharmacists) and nine patients, were interviewed in stage 2. The response rates of pharmaceutical care providers and patients were 75% (15 out of 20) and 82% (9 out of 11), respectively. Of the 15 pharmaceutical care providers, the largest proportions of providers were female (67%), 30–39 years old (73%), and cardiologists (47%), and the largest proportions possessed master degrees (53%), working experience 6–10 years (47%), and intermediate professional title (73%). Of the nine patients, the largest proportions were female (67%) and 20–29 years old (44%).
Participant characteristics in stage 2.
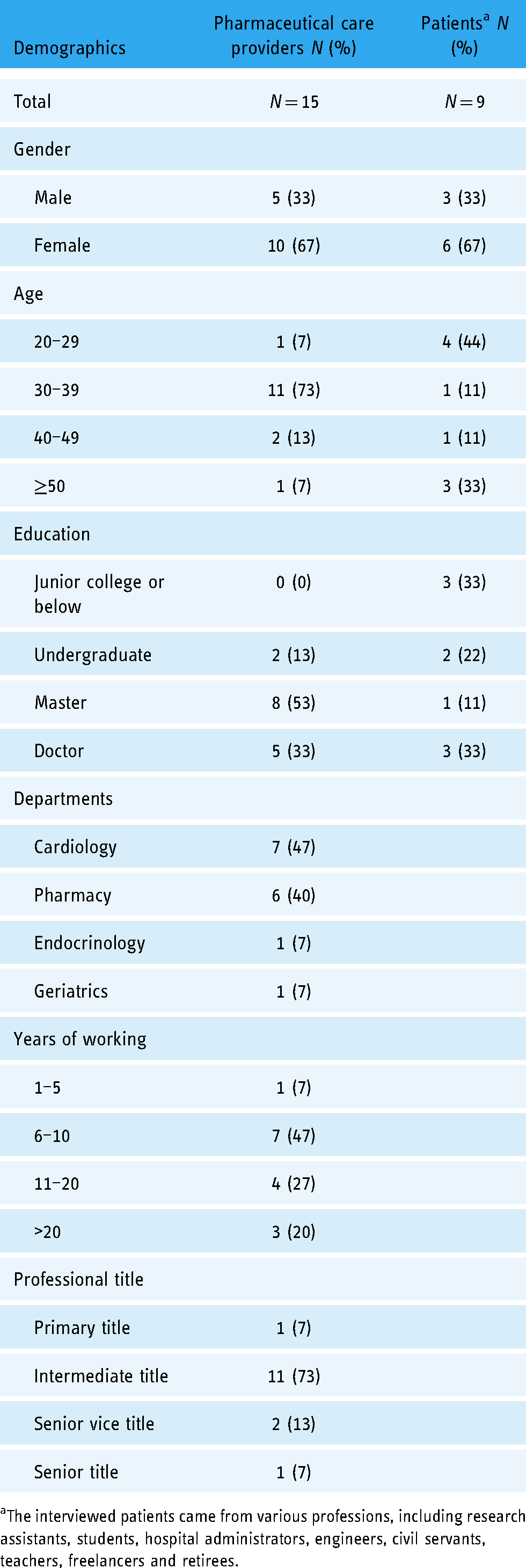
The interviewed patients came from various professions, including research assistants, students, hospital administrators, engineers, civil servants, teachers, freelancers and retirees.
Theoretical model
Based on core aspects of the ‘Implement-SDM model’ and interview data, five core constructs of SDM were identified, that is ‘comprehensive understanding of the patient's condition’, ‘exchanging ideas on medication options’, ‘achieving a medication plan’, ‘providing decision support’, and ‘drug tracking and adjustment’ (Supplementary File 5), and then we constructed a theoretical model of SDM in web-based pharmaceutical care consultation (Figure 1).

Theoretical model of shared decision-making in web-based pharmaceutical care consultation. (a) Patients with critical situation were not applicable. (b) The form of medication plan is mainly suggestions.
Initial items of measurement and content evaluation
Based on the interview data (Supplementary File 5), we found that participants believed it is essential for the patients to provide detailed medication history and detailed medication regimen in the web-based pharmaceutical care consultation. Although participants have mentioned a lot of information that should be exchanged on medication online, such as drug name, classification, indications, specifications, dosage, medication monitoring, time to use, duration of treatment, expected effects, observed effects, adverse effects and their solution, drug allergy, interaction, medication compliance, and consequences and solution of non-compliance. In order to comprehensively evaluate the information which should be exchanged on medication online, SDM strategy tools 62 and MEDICODE 63 are also referred to generate the items. Participants believed that other medical history, such as symptoms, previous medical history, economic conditions, work nature, and living habits, is also essential in the web-based pharmaceutical care consultation. In order to comprehensively evaluate the information about other medical history, Textbook of Diagnostics (9th edition) 64 also referred to generate the items. Therefore, according to the theoretical model in our study, interview data, and relevant literature or materials, we formed the initial measurement which includes four dimensions and 24 items (Table 2). Four dimensions were ‘comprehensive understanding of the patient's condition’, ‘exchanging ideas on medication options’, ‘achieving a medication plan’, and ‘providing decision support’. The detailed descriptions and coding scheme of measurement items were shown in the supplementary file (Supplementary File 4).
Measurement construction process.

‘patient provided/expressed…’ means that patient/patient’s family proactively provided/expressed information, or patient/patient’s family provided/express information under the guidance of provider. Similarly, ‘provider described/provided…’ means that provider proactively described/provided information, or provider described/provided information after the inquiry/request from the patient/patient’s family.
In stage 5, we developed two global items in each dimension to reflect the core content for validity assessment, and these global items were: (1) ‘patient provided diagnostic data, medication history and other medical history’ and ‘provider comprehensively grasped the patient’s condition’ in dimension 1; (2) ‘two sides expressed the ideas on medication’ and ‘two sides fully knew the ideas of each other on medication” in dimension 2; (3) ‘provider considered the patient’s ideas and provided patients with detailed medication regimen’ and ‘patient fully accepted the medication regimen’ in dimension 3; (4) ‘provider provided decision support well’ and ‘patient received decision support well’ in dimension 4.
Drugs include these are currently taking or had taken in the past.
Sixteen key informants evaluated the appropriateness of the dimensions and items of the measurement. We found that except for the item ‘patient provided the previous medical history’, the median scores of four dimensions and 23 items were 7–9 and DI < 1 (Supplementary File 6). Therefore, we deleted the item ‘patient provided the previous medical history’ (Table 2).
Reliability and validity results
In the pre-survey, we deleted two items ‘patient provided the compliance of drugs’ and ‘in the medication plan, provider provided compliance information of drugs’, which were not observed in actual encounters (Table 2). In the formal survey, 150 clinician–patient encounters were provided by 150 clinicians from 15 provinces/municipalities (50 clinicians from eastern China, 50 from central China, and 50 from western China), and 150 pharmacist–patient encounters were provided by 27 pharmacists from 14 provinces/municipalities (19 pharmacists from eastern China, 4 from central China, and 4 from western China). Finally, 300 provider–patient encounters were included for analysis, and we found that the scores of these items were generally not high (Table 3). Among them, there were three items with an average score >1, which were ‘patient provided the overview of drugs’, ‘patient provided the main symptoms’, and ‘in the medication plan, provider considered the patient's expectations and concerns’. Harman single factor test results found that the cumulative percentage of the first factor was 16.648% by exploratory factor analysis
Interrater reliability and single indicator validity of the measurement.
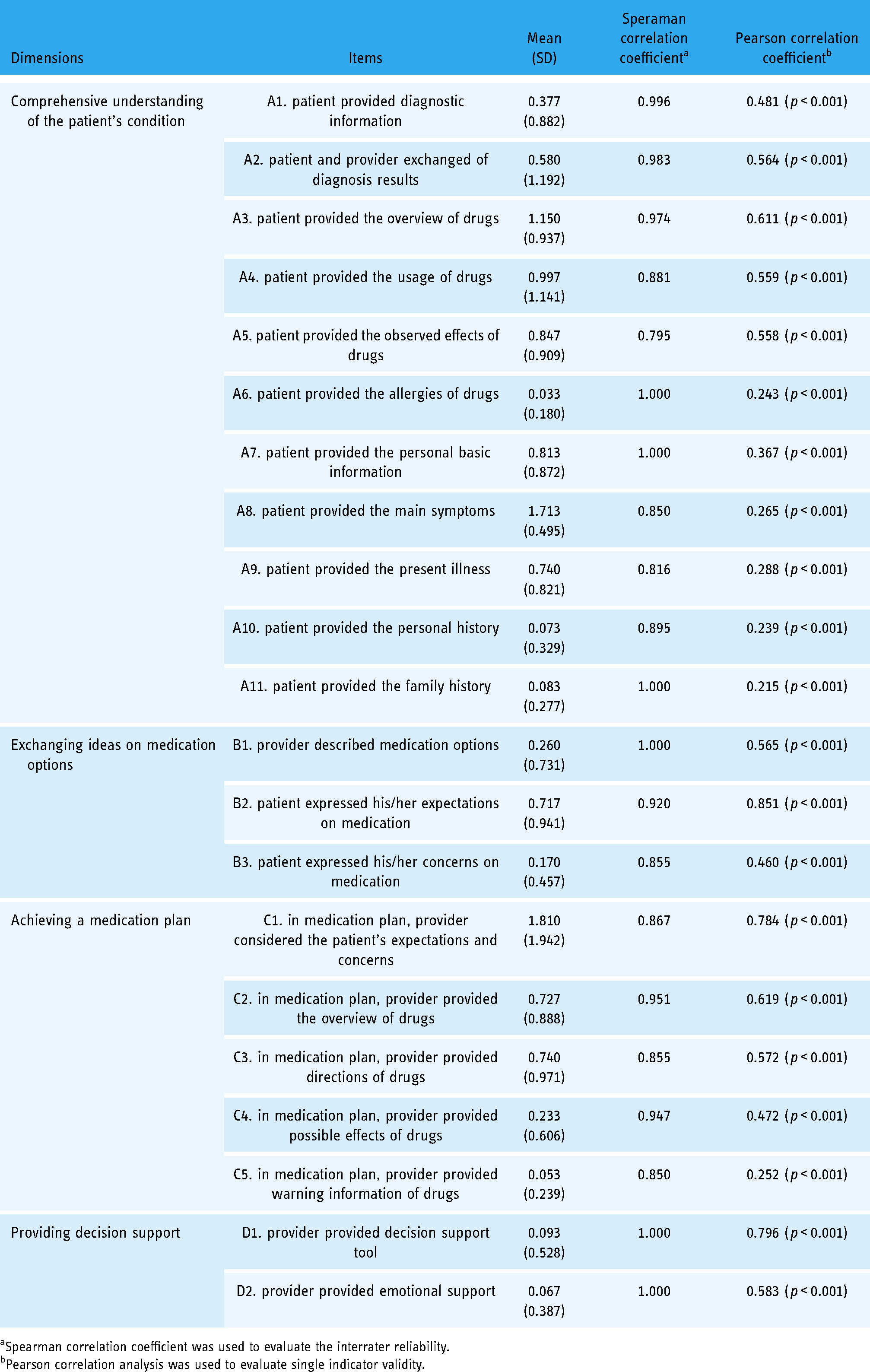
Spearman correlation coefficient was used to evaluate the interrater reliability.
Pearson correlation analysis was used to evaluate single indicator validity.
Interrater reliability and single indicator validity are summarized in Table 3. Spearman correlation coefficients of all the items and eight global items were greater than 0.750, suggesting good interrater reliability. Pearson coefficients between 21 items and their external variables were significant (
MIMIC model results of dimensions 1–4 are summarized in Figure 2. The
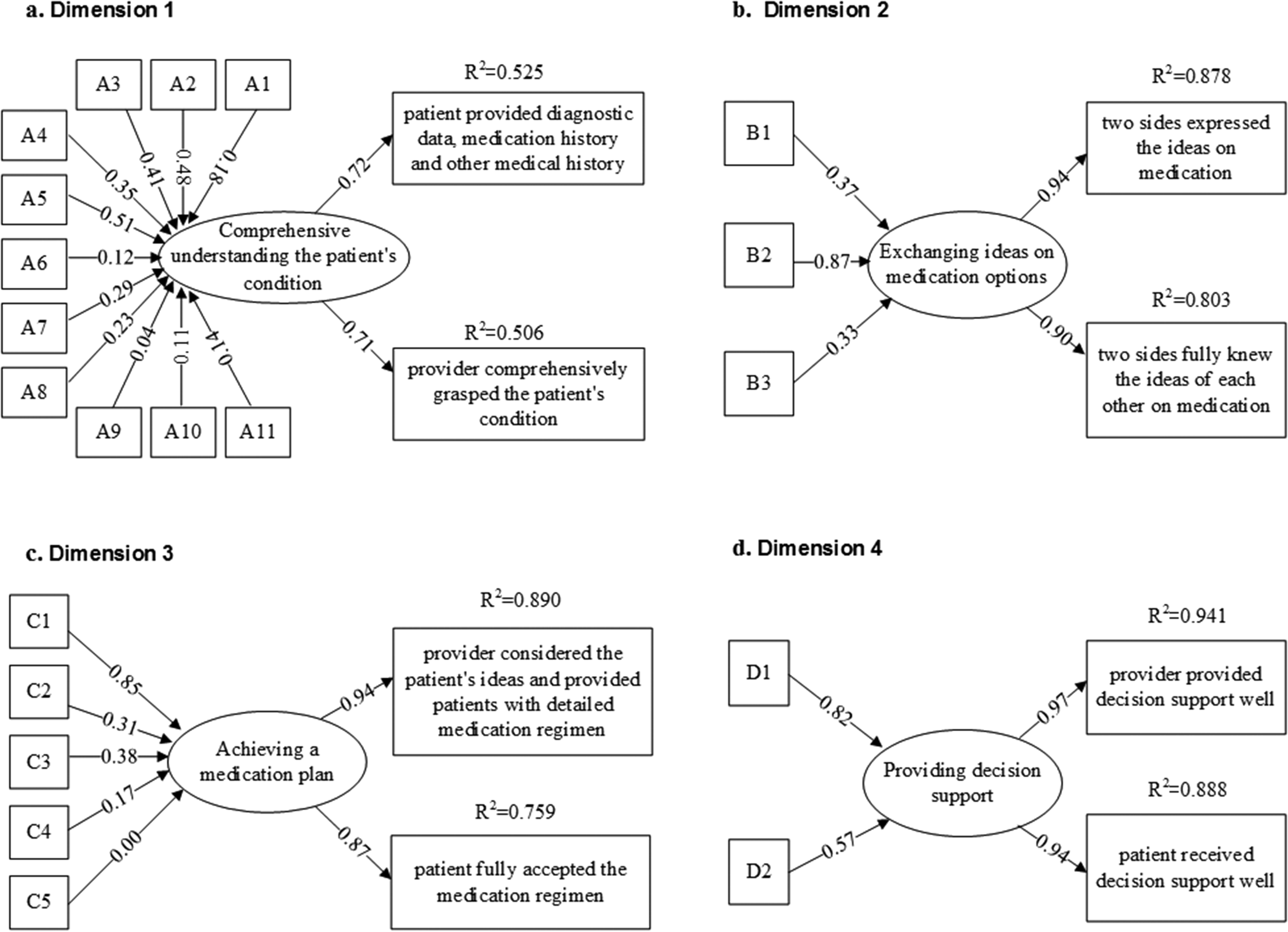
Multiple indicators multiple causes (MIMIC) model of dimension 1–4.
Discussion
Principal findings
In this study, we first proposed a theoretical model that contains four elements as a foundation for SDM measurement in web-based pharmaceutical care consultation, which enriches SDM models. The four elements describe a series of communicative efforts of both sides, namely: (1) comprehensive understanding of the patient's condition, (2) exchanging ideas on medication options, (3) achieving a medication plan, and (4) providing decision support. Then, we constructed an observer-based measurement of SDM in web-based pharmaceutical care consultation with four dimensions and 19 items, which had good reliability and validity. Four dimensions of this measurement were in accordance with four elements of the theoretical model.
Characteristics of online consultation were observed in the theoretical model or the process of construction of the theoretical model in our study. The elements of our model seem to be similar as the face-to-face SDM model, but the range and form of some elements were different. Due to asynchronous communication, 52 the range of ‘patients’ in the element ‘comprehensive understanding of the patient's condition is limited, and patients with critical situations were not applicable online. The form of ‘medication plan’ in the element ‘achieving a medication plan’ is mainly suggestions, and pharmaceutical care providers don't prescribe online usually. In China, the clinicians’ prescription rights are limited online, 65 and the exploration of pharmacists’ prescription rights is still in its infancy, 66 particularly on the internet. ‘Providing decision support’ also reflects the characteristics of online communication, such as asynchronous communication and education function. Healthcare providers were busy and spent less time in face-to-face communication in China, 67 so there was often no time to give patients to read decision support materials. However, due to asynchronous communication online, providers have more time to seek relevant information and give patients to read these materials. Meanwhile, it is more convenient to provide approved educational materials online so as to improve patients’ health literacy and communication efficiency. Although ‘providing decision support’ was of importance in previous SDM models,32,34 the internet provides a facility to web-based forms of decision support, such as web links, and some web-based decision-making tools were developed nowadays.68,69
Characteristics of pharmaceutical care consultation were observed in the theoretical model in our study compared with previous SDM models. First, our model highlighted the importance of exchanging medication history (e.g. drug name, dosage, observed effects, and adverse effects) and detailed medication plan (e.g. drug name, classification, duration of treatment, and expected effects), in accordance with the instrument to describe and evaluate exchanges on medications during medical consultations. 63 Enough information sharing is also a prerequisite to SDM. 70 Second, the conception of medication options in our model is broader, including the selection of different drug types/combinations, dosage forms, or domestic/imported drugs.
Some SDM models, including three talk model,
Then, based on the theoretical model, we constructed the measurement of SDM in web-based pharmaceutical care consultation, including four dimensions and 19 items. We found good content validity, inter-rater reliability, single-item validity, and convergent validity of this measurement by appropriateness score, Spearman correlation coefficient, Pearson correlation coefficient, and MIMIC model. This is probably beneficial from our efforts in the process of measurement development. For example, we first constructed a theoretical model of SDM in web-based pharmaceutical care consultation as a foundation for measurement; we evaluated the measurement content before further validation; and we provided more detailed descriptions of items, which is the way to improve inter-rater reliability. 41 Our efforts were consistent with the recommendations for the development of SDM measurement by previous studies.31,41 Some items of the measurement, such as ‘provider described medication options’, ‘patient expressed his/her expectations on medication’, and ‘patient expressed his/her concerns on medication’, were consistent with the items of the existing observational instrument (OPTION scale. 39 )
Limitations
There were some limitations to this study. First, when we collected data for measurement assessment, we were mainly concerned the patients with hypertension or high blood pressure. Therefore, the measurement needs to be verified in other diseases. Second, there was limited empirical data about the pharmacist–patient encounters on the internet, and the pharmacists mainly came from the eastern region in China. Therefore, the measurement needs to be verified in other regions. Third, our theoretical model was proposed based on the ‘implement-SDM model’ and interview data, but both the existing model and interviewees may have problems to oversee the whole concept of SDM. Therefore, there remains room for improvement of this model.
Implications
Although four elements of the theoretical model seem to be common across SDM models and literature, the model and measurement in our study provide several new insights, which integrate the characteristics of online environment (e.g. asynchronous communication and education function) with SDM and highlight the importance of exchange medication history and detailed medication plan. From a research perspective, we hope that this allows researchers to explore these new insights we identified, such as characteristics of online environment and pharmaceutical care consultation in SDM study. From a clinical perspective, we hope that an increased focus on these new insights will facilitate awareness among pharmaceutical care providers of their communication during decision-making online, such as exploiting the advantages of the internet for better decision support, obtaining sufficient medication history, and providing detailed medication plan.
Conclusions
Characteristics of web-based pharmaceutical care consultation, highlighting the importance of medication history, medication regimen, and detailed medication plan, were observed in the theoretical model in this study compared with previous SDM models. Measurement of SDM in web-based pharmaceutical care consultation in this study had good reliability and validity and may be used by health policymakers to improve the quality of pharmaceutical care consultation online.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076221089794 - Supplemental material for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation
Supplemental material, sj-docx-1-dhj-10.1177_20552076221089794 for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation by Haihong Chen, Yuqi Xiong, Zinan Zhang, Qian Zhou, Dan Wang, Xuemei Wang and Xinping Zhang in Digital Health
Supplemental Material
sj-docx-2-dhj-10.1177_20552076221089794 - Supplemental material for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation
Supplemental material, sj-docx-2-dhj-10.1177_20552076221089794 for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation by Haihong Chen, Yuqi Xiong, Zinan Zhang, Qian Zhou, Dan Wang, Xuemei Wang and Xinping Zhang in Digital Health
Supplemental Material
sj-doc-3-dhj-10.1177_20552076221089794 - Supplemental material for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation
Supplemental material, sj-doc-3-dhj-10.1177_20552076221089794 for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation by Haihong Chen, Yuqi Xiong, Zinan Zhang, Qian Zhou, Dan Wang, Xuemei Wang and Xinping Zhang in Digital Health
Supplemental Material
sj-doc-4-dhj-10.1177_20552076221089794 - Supplemental material for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation
Supplemental material, sj-doc-4-dhj-10.1177_20552076221089794 for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation by Haihong Chen, Yuqi Xiong, Zinan Zhang, Qian Zhou, Dan Wang, Xuemei Wang and Xinping Zhang in Digital Health
Supplemental Material
sj-doc-5-dhj-10.1177_20552076221089794 - Supplemental material for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation
Supplemental material, sj-doc-5-dhj-10.1177_20552076221089794 for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation by Haihong Chen, Yuqi Xiong, Zinan Zhang, Qian Zhou, Dan Wang, Xuemei Wang and Xinping Zhang in Digital Health
Supplemental Material
sj-doc-6-dhj-10.1177_20552076221089794 - Supplemental material for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation
Supplemental material, sj-doc-6-dhj-10.1177_20552076221089794 for Theoretical model and measurement of shared decision making in web-based pharmaceutical care consultation by Haihong Chen, Yuqi Xiong, Zinan Zhang, Qian Zhou, Dan Wang, Xuemei Wang and Xinping Zhang in Digital Health
Footnotes
Acknowledgements
The authors would like to thank Shuangjiang Zheng for his assistance in promoting this study and his help in contacting the investigated healthcare providers, and acknowledge the key informants who participated in this study.
Conflict of interest
The authors have no conflict of interest to declare.
Contributorship
CH contributed to the conception and design, data collection, analysis and interpretation, and manuscript writing and revising. XY contributed to the conception and design, data collection and interpretation, and revising the manuscript. ZZ, ZQ, WD, WX and ZX contributed to the conception and design, data interpretation and manuscript revising. All authors reviewed and approved the final version of the manuscript.
Ethical approval
The Ethics Committee of Tongji Medical College, Huazhong University of Science and Technology approved this study (IORG: IORG 0003571).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by National Natural Science Foundation of China [Grant No. 71774059], General Project of Philosophy and Social Science Research for Jiangsu University [Grant No. 2021SJA0301] and The Excellent Innovation Team of the Philosophy and Social Sciences in the Universities and Colleges of Jiangsu Province “The Public Health Policy and Management Innovation Research Team”.
Guarantor
ZX.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
