Abstract
Objective
The objective of this study was to assess the feasibility of monitoring and diagnosing compromised walking motion in the frontal plane, particularly in persons living with the chronic effects of stroke (PwCS). The study aimed to determine whether active control of walking in the frontal plane could be monitored and provide diagnostic insights into compensations made by PwCS during community living.
Methods
The study recruited PwCS with noticeable walking asymmetries and employed a monitoring method to assess frontal plane motion. Monitoring was conducted both within a single assessment and between assessments. The study aimed to uncover baseline data and diagnostic information about active control in chronic stroke survivors. Data were collected using sensors during 6 minutes of walking and compared between the paretic and non-paretic legs.
Results
The study demonstrated the feasibility of monitoring frontal plane motion and diagnosing disturbed endpoint control (
Conclusions
This study's findings indicate that monitoring frontal plane motion is a feasible approach for diagnosing compromised walking motion. The results suggest that individuals with walking asymmetries, exhibit differences in endpoint control and variability between their paretic and non-paretic legs. These insights could contribute to more effective rehabilitation strategies and highlight the potential for monitoring compensations during various activities of daily living.
Introduction
Remote monitoring has provided essential safety feedback to medical staff and patients. 1 Given that curtailing walking and other essential activities of daily living (ADL) is detrimental to physical and mental health,2–5 it is critical to remotely monitor events that can impede activities of daily living (ADL). Such monitoring would give essential feedback necessary for assessments and diagnosis.6,7
Monitoring frontal plane compensations is essential for safe walking given that persons living with the chronic effects of stroke (PwCS) have compromised voluntary regulation of their movements. 8 In fact, the need for such work was called for in a recent study. 9 Monitoring step count can improve walking in PwCS. 10 This monitoring is critical because PwCS have impaired coordination, balance, posture, and voluntary movement.11–19 Specifically, their affected lower limb often remains severely impaired hampering walking, heightening falling risk, and reducing daily functioning at home and in the community. 20 In addition, poor control of their foot in space, or endpoint control, causes greater kinematic variability on the affected side which is heightened when individuals perform tracking and targeting tasks while walking.21,22
Inadequate endpoint control in the frontal plane can have dire implications for fall risk.23,24 Continuous monitoring of mediolateral (ML) displacement (MLD) at the foot (the endpoint) during the swing, in conjunction with simultaneous pelvic compensations, can serve as a valuable diagnostic tool given that ML motion is representative of dynamic balance, 23 that impaired ML balance control has been identified as a common cause of falls, 24 and that hip fractures are strongly associated with falls to the side occurring mostly during locomotion.25,26 An MLD asymmetry would be a diagnostic indicator of paretic leg deterioration in endpoint control. In addition, asymmetric pelvic frontal plane motion requires greater active balance control studied by others.27,28 Indeed, pelvic displacement can provide greater insights for diagnostic and treatment purposes than classifications based solely on speed. 29 It is worth noting that while there is evidence of exaggerated pelvic motion in PwCS,30–32 limited knowledge exists concerning the intricate coupling of pelvic mechanics with mechanical compensations distal to the pelvis. 29 Furthermore, these pelvic motion abnormalities have shown a negative correlation with favorable clinical outcomes on assessments such as the Fugl-Meyer Assessment, Berg Balance Scale, and Timed Up and Go. 33 Given this negative correlation, continuous monitoring of MLD and accompanying pelvic kinematics would significantly inform walking performance deterioration, an undesirable change in performance during a defined task and related to fatigability. 34 Moreover, diagnosing compensation deterioration is critical because PwCS would then need enhanced active control to keep falling risk stable. This practice would also ensure safety during telerehabilitation.35,36
By monitoring MLD and the pelvis continuously, clinicians could modify therapeutic strategies based on a real-time abnormality or deviation from the long-term goal in order to get a better outcome, such as maximizing active control. This could be used for diagnostic purposes in telerehabilitation or virtual reality (VR) applications while adopting strategies to prevent adverse events as others have advised. 36 Telerehabilitation has proven to be an effective tool for stroke survivors to perform rehabilitation tasks with remote supervision,37,38 preventing the disruption of the rehabilitation process, allowing clinicians to monitor patients without physical limitations, especially when delivering therapy to underserved areas and during a pandemic.38,39 Moreover, when comparing face-to-face rehabilitation with telerehabilitation the results are reportedly similar. 40 Our aim was to determine whether it was feasible to monitor and diagnose compromised motion during in the frontal plane, which has been little monitored. 41 This would potentially benefit many individuals, whether advanced in age, living with a neurodegenerative disease, or recovering from an injury given that remote monitoring during telerehabilitation can target improvements in active control. A critical step in this process is monitoring those who have notable walking asymmetries and severe limitations on their walking speed, PwCS. We tested the feasibility of monitoring one walking exercise bout during two assessments in an exploratory study, aiming to uncover baseline data and diagnose disturbances in the active control of PwCS. Our hypotheses were that monitoring would: (1) demonstrate greater MLD magnitude and variability in the paretic limb than the non-paretic limb, (2) show performance deterioration during the six-minute walking test assessment and between assessments, and (3) establish that a stronger coupling of pelvic sway exists with the paretic leg in comparison to the non-paretic leg.
Methods
After approval by the Institutional Review Board of the Northeast Ohio Veteran's Affairs Medical Center, 10 PwCS gave written consent and were enrolled in this study. The inclusion criteria (Appendix 2) included years of age > 18 years, stroke onset > 6 months prior, medically, and psychologically stable, cognitively intact, sufficient endurance to participate in the study, Fugl-Meyer for lower extremity (FM-LE) score > 15, and active paretic ankle dorsiflexion in synergy (the ability to dorsiflex the foot during coupled joint movements in the lower extremity). PwCS participated in two treadmill walking assessments; during each assessment they were continuously monitored for 6 minutes at their preferred speed, 41 following one minute of warm-up walking. Data were obtained from six 14 mm sensors placed over four pelvic landmarks (anterior and posterior superior iliac spines) and the feet (lateral malleoli). For exploring the feasibility of monitoring, we began with monitoring via the gold standard sensors for motion capture (100 Hz, Vicon, Oxford, UK). The mediolateral motion of the feet and pelvis were monitored. The extrapolated center of mass (XCoM) was calculated using previously accepted methods (Appendix 1).42–44 XCoM is commonly used to investigate dynamic balance and stability in the frontal plane.45–47 Cross-correlation analysis was performed for the feet and XCoM synchronized in time, resulting in the maximum absolute cross-correlation value (xCorr) and corresponding time lag in seconds between the feet and XCoM (tLag) (MATLAB, Natick, MA; Appendix 1).
The maximum MLD, MLD standard deviation (SDMLD), xCorr, and tLag values for each step were calculated for each leg over the 6 minutes of walking with SDMLD representing variability for each 30-second window. A second monitoring assessment was performed 6 to 12 months following the first monitoring assessment. A three-factor repeated measures analysis of variance (ANOVA) was completed for MLD, SDMLD, xCorr, and tLag values (Leg*Time*Assessment) with significance set at
Results
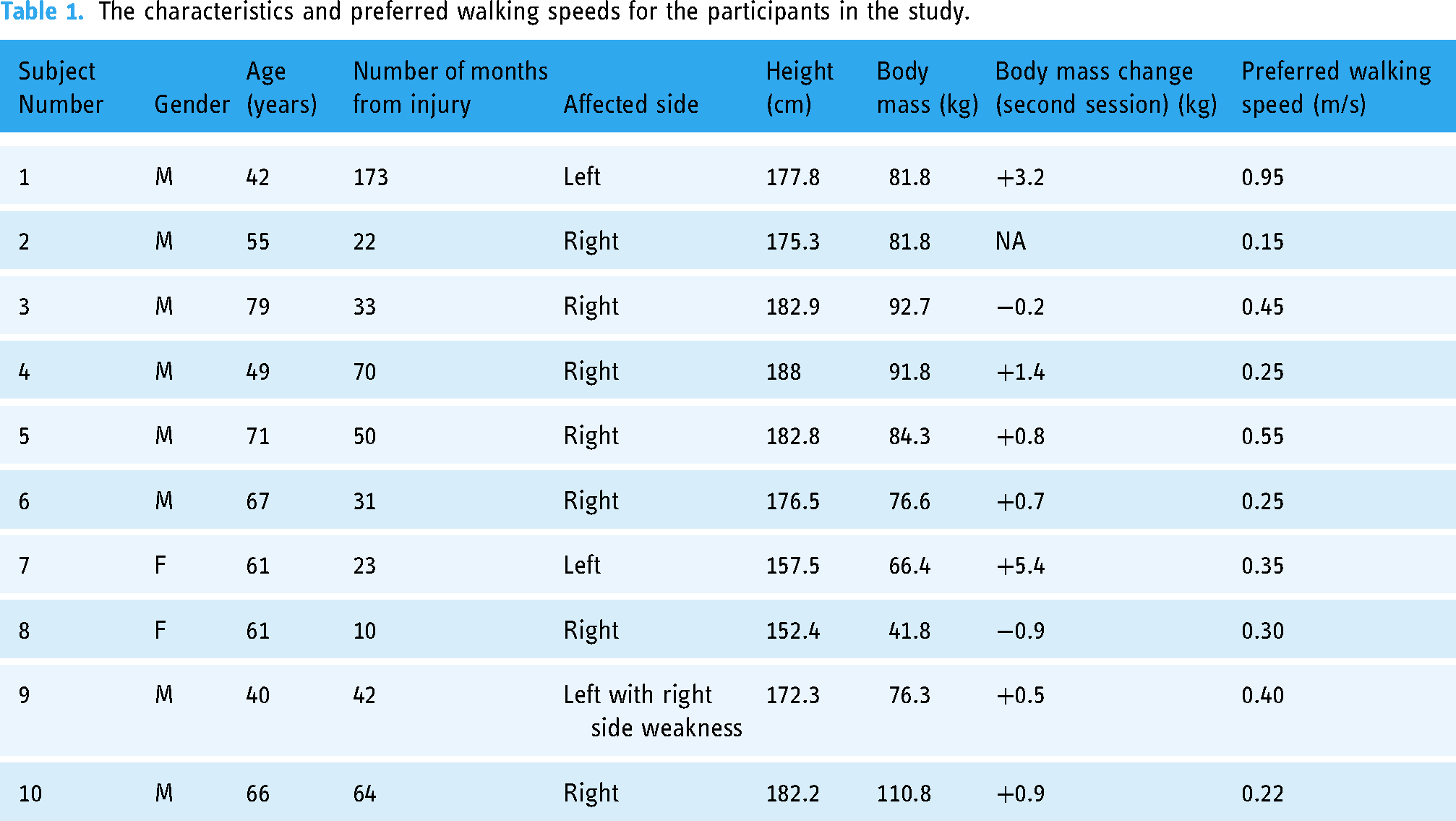
Ten stroke survivors > 6 months post-stroke completed the study (Table 1). Walking speeds ranged from 0.15 to 0.95 m/s with an average walking speed for the group of 0.56 + 0.292 m/s (Table 1). MLD compensations displayed a non-normal distribution, thus prior to the ANOVA data were transformed to the square root, satisfying the normal distribution requirement. The repeated measures ANOVA showed significant differences in MLD between the affected and unaffected legs (Leg,
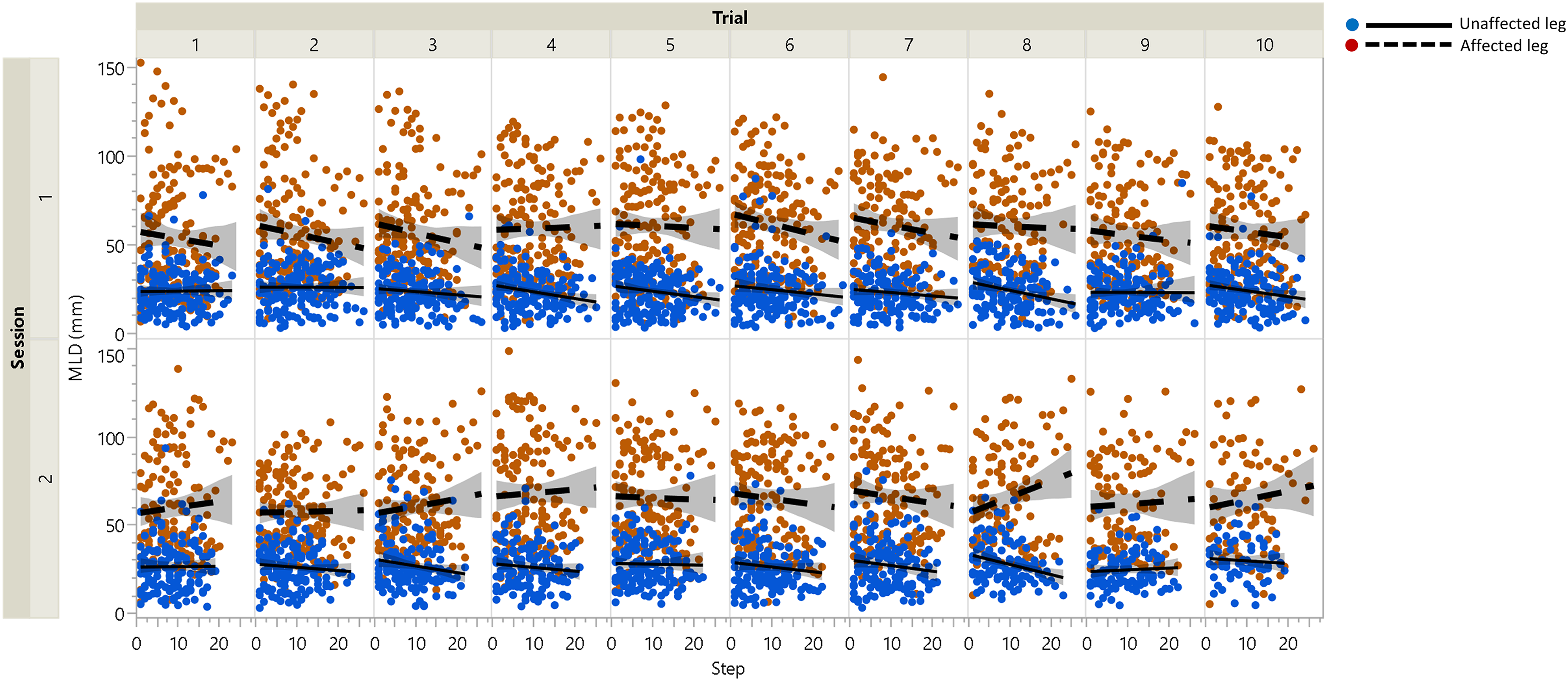
Mediolateral displacement (MLD) results were displayed for both legs displaying the results over time with each panel containing the data for each successive 30-second time period over the six-minute walk. MLD was greater in the affected leg (red) compared to the unaffected leg (blue). The shaded portions of the fitted lines show the confidence intervals.

The mediolateral displacement standard deviation (SDMLD) results were displayed for both legs (left) with the subjects’ data binned and arranged in order of slower to faster walking speeds. SDMLD was greater in the affected leg (red) compared to the unaffected leg (blue). The affected leg significantly increased its displacement with increasing speed (right), while the unaffected leg had negligible increases with increasing speed. The shaded portion of the fitted lines shows the confidence regions.

The top panel displays the cross-correlation (xCorr) results over time for each successive 30-second time section over the six-minute walk, and the bottom panel displays the xCorr results. xCorr was stronger on the affected side (red, dashed line) while tLag was shorter for the affected side. The shaded portion of the fitted lines shows the confidence intervals.
The characteristics and preferred walking speeds for the participants in the study.
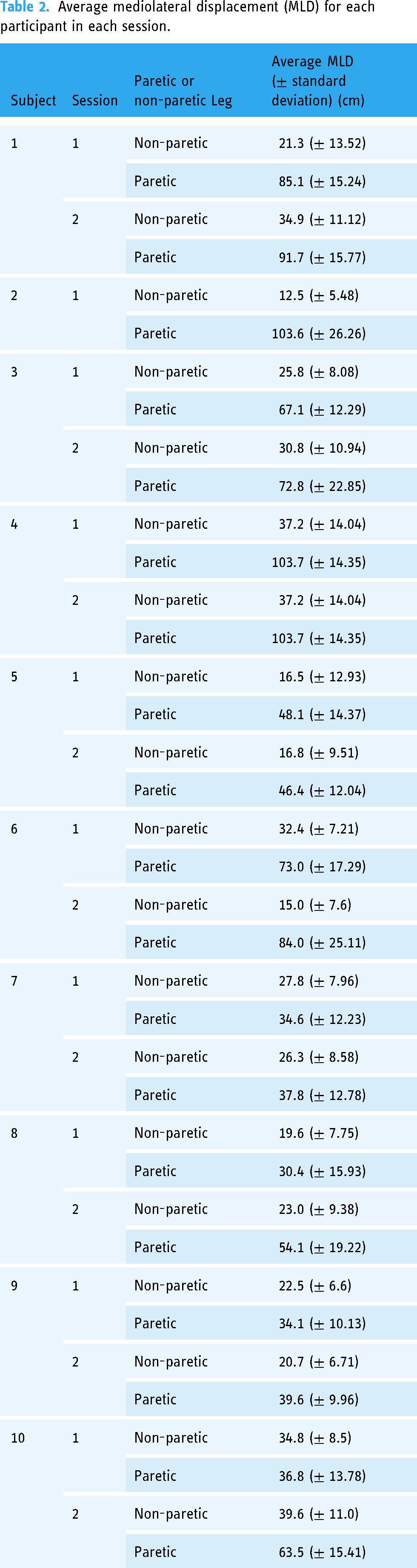
Average mediolateral displacement (MLD) for each participant in each session.
Discussion
Lower extremity endpoint control deficits were diagnosed via monitoring mediolateral endpoint displacement (MLD) in PwCS. Monitoring exposed the affected leg over 6 minutes of walking with increased endpoint control variability. This supports our first hypothesis, that monitoring would find greater MLD magnitude and variability in the affected limb. This observation also draws parallels with previous research on upper extremities, where mediolateral movement differences were simultaneously observed in the affected shoulder and the pelvis. 48 Our results did not support our second hypothesis which was that remote monitoring would uncover performance deterioration during the six-minute walking assessment and between assessments. MLD did not change during the six minutes of walking or between assessments. However, monitoring validated that the affected leg was more strongly coupled to pelvic sway than the unaffected leg, supporting our third hypothesis. The same walking speed was used between the two assessments, thus, walking speed was not a confounder in the results which can happen during the clinical six-minute walk test where walking speed can fluctuate.
Monitoring uncovered no progressive ML endpoint compensations either during the 6 minutes of walking or between assessments. There is a dearth of literature as to whether compensations such as these stagnate or deteriorate during the chronic phase. Others have found compensations in the affected limb compared to non-disabled controls, 11 but they only assessed five steps from each leg and only during one walking assessment. Our study, however, implemented a comprehensive monitoring approach that encompassed over 100 steps per assessment. This extensive coverage of steps provides compelling evidence for the reliability and robustness of our monitoring methodology, elevating its potential as a diagnostic tool. It is worth noting that the average number of steps taken by stroke patients during a typical six-minute walk ranges from 200 to 400 steps. 49 Despite this, our study successfully captured meaningful endpoint control data with just 100 steps per assessment and showed no diagnosis of performance fatigue, corresponding to no debilitation of active control. This reduction in the required number of steps represents a substantial leap forward in efficiency and feasibility, making our monitoring strategy an attractive alternative to traditional methods. The finding that monitoring endpoint control did not change between assessments might have been expected considering the participants weren’t engaged in physical therapy which is generally the case for stroke survivors in the chronic phase (> 6 months post-stroke) and was confirmed for these participants. Once PwCS are placed in a challenging walking environment, however, this measure might be a marker for performance fatigue. A monitoring strategy such as this would inform the debilitation of active control in PwCS given that progressive differences in ML endpoint displacement would require enhanced active control to keep falling risk stable, considering frontal plane components require greater active balance control as discovered in others.27,28
Monitoring lateral pelvic displacement would provide one integral walking diagnostic feature, 50 hip abductor control,22,51,52 providing mediolateral endpoint trajectory during walking, and actively stabilizing foot placement. 53 PwCS have compromised hip control and abduction torque.21,54,55 Thus, when monitoring both endpoint compensations and pelvic coupling, one could diagnose mechanically inefficient and energetically costly walking. 27 Coupling can cause greater MLD, and, moreover, be diagnostic of undesirable neurological synergies, as well as be a common cause of falls. 24 Thus, diagnosing impaired ML balance control is critical for safety. These findings about modifications to achieve dynamic balance may be due to increased pelvic coupling in the affected leg's swing phase, but this has not been investigated until now. This asymmetric coupling could also be a critical reason for step length asymmetry, even seen in able-bodied individuals. 56
Ten subjects were used for this exploratory study as a convenience sample, and the finding of a significant difference between legs indicates our sample size was large enough. A post-hoc power calculation, however, revealed that this study was adequately powered to find differences between the affected and unaffected legs with ten participants. Given this convenience sample size, differences during the walking time and between sessions could have been found, but with a larger sample size, the differences could have been so small that they would not have been clinically meaningful which is generally < 20%.
Our study highlights the critical role of sensors in monitoring and assessing stroke survivors’ walking ability. The inclusion of a single position sensor on each leg proved feasible in differentiating between the affected and unaffected legs, even at various walking speeds. This emphasizes the practicality and versatility of sensor-based approaches in stroke rehabilitation. Recently, the importance of sensors used in telerehabilitation for neurological pathologies was highlighted. 57 Our study contributes to this growing body of knowledge by demonstrating the potential application of sensors for remote frontal plane monitoring and feedback in stroke rehabilitation. For instance, the motion data collected by these sensors can serve as valuable feedforward signals, enabling therapists to remotely monitor patients’ progress and provide real-time feedback. This concept aligns with the work of Chen et al., 58 who successfully implemented interactive virtual telerehabilitation using motion data as a basis for therapeutic interventions. By leveraging the capabilities of wearable sensors, we can enhance the effectiveness and accessibility of stroke rehabilitation programs.59,60 The integration of wearable sensor technology into telerehabilitation platforms holds promise for expanding the reach of rehabilitation therapy, enabling continuous monitoring, and facilitating personalized interventions.61–64
There were limitations with this work. First, the method of computing the center of mass did not include the arms, but others have found that it produces very similar mediolateral center of mass motion and timing. 65 Our method captured pelvic segment mediolateral kinematics more accurately than only using a sacral sensor as used in previous work. 12 Second, we used XCoM because it is directly related to whole body angular momentum, a sensitive assessment for determining dynamic balance control. 66 Additionally, preferred walking speed was determined during the first session and may have changed when data were collected for the second session, however keeping speed consistent between sessions was critical to eliminate the confounding effect of mechanical differences due to speed alone. Anecdotally, participants did not report feeling challenged to complete the walking data collection period for each session. Also, these data were collected during treadmill walking and may not generalize to overground walking. A final limitation is the potential lack of direct applicability to telehealth and rehabilitation. Further research will enhance the relevance of these results by investigating the use of wearable devices for remote data capture, offering a more practical and transferable approach for monitoring and intervention in these settings.
In the future, therapists might target improved endpoint control by reducing the affected leg's MLD in the acute phase, however such efforts to normalize kinematics may help with active balance control while having less long-term effects on walking speed, as found by others. 67 Therapies to reduce abnormal coupling are feasible in the upper extremity with the shoulder and elbow muscles. 68 Thus, therapies that target motor control to reduce endpoint and pelvic coupling in the lower extremity may also be possible, and benefit dynamic balance performance.
Conclusions and clinical messages
Monitoring persons with chronic stroke for decreased endpoint control was feasible; diagnosing decreased affected leg control was possible.
Monitoring uncovered that one could diagnose stronger coupling of the pelvis and affected leg.
There was no performance fatigue during monitoring either over six minutes of preferred speed walking or between two assessments. This may be reflective of undesired neurological synergies stagnating.
Footnotes
Acknowledgements
The authors thank the participants in this study.
Contributorship
JS and EH researched the literature and conceived the study. EH was involved in protocol development, gaining ethical approval, and patient recruitment. JS and EH were involved with data analysis. JS wrote the first draft of the manuscript. JS and EH reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The Institutional Review Board of the Northeast Ohio Veteran's Affairs Medical Center approved this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by an award from the Rehabilitation Research and Development Service of the U.S. Department of Veterans Affairs (grant number: B4668W).
Guarantor
EH.
Appendix 1
Pelvic mechanics (XcoM) were calculated using previously accepted methods42,44:
Cross-correlation analysis was performed using the lateral malleoli's mediolateral axis coordinates and the extrapolated center of mass data synchronized in time (MATLAB, Natick, MA). We assumed the time-domain vector for the mediolateral axis coordinates and extrapolated center of mass are
Appendix 2
Inclusion criteria:
Participants will be stroke survivors who are older than 18 years old and who are beyond 180 days from their first clinical hemorrhagic or non-hemorrhagic stroke. Participants will be stroke survivors with unilateral hemiparesis who have sufficient endurance and motor ability to ambulate at least 30 feet continuously with minimal assistance without the use of an AFO. Participants will have a Berg Balance Scale score of 23 or greater without any assistive devices. Participants will have ankle dorsiflexion strength of no greater than 4/5 while standing and demonstrate foot drop during ambulation such that the participant exhibits gait instability or inefficient gait patterns. Participants will have adequate social support and stability and will be medically stable with intact skin. Participants will have full coverage of the acetabulum and minimal knee and ankle laxity.
Gait instability will be defined as the need for supervision, physical assistance, or an assistive device (cane, walker) to maintain balance or prevent falls. An inefficient gait pattern will be defined as a gait pattern manifesting “dragging” or “catching” of the affected toes during the swing phase of gait, or the use of compensatory strategies such as circumducting the affected limb, vaulting with the unaffected limb or hiking the affected hip to clear the toes.
Candidates may be referred for neuropsychological and/or psychosocial evaluation. If this evaluation is necessary, candidates will be referred to their primary care physician with a request for a psychological evaluation to determine mental stability, presence of acute depression, acute anxiety, bipolar issues, or inability to adjust to circumstances associated with the research study such as waiting for appointments and delays during data collection. Referral for neuropsychological testing or psychosocial assessment will be determined by the state of the subject's cognitive, motor, behavioral, language, executive, and psychosocial functioning during the screening process. Referral necessity will be determined by the physical therapist and/or research nurse during the screening process. The responsible investigator associated with the study could also refer a subject for neuropsychological evaluation based on findings during contact with the subject. In addition, a psychosocial evaluation will be conducted if members of the research team believe it warranted after evaluation and review of the candidate's medical history. If any evaluation is necessary, candidates will be referred to their primary care physician with a request for a neuropsychological and/or psychosocial evaluation. The tools used for said examinations are left to the discretion of the psychologist/psychiatrist. Based on these findings, subjects could be either recommended for inclusion or exclusion by their primary care physician.
